Abstract
Background
The association between Autism spectrum disorders (ASD) and visual impairment has been mentioned in the literature. The aim of our study was to investigate the prevalence of autism among children with albinism compared to the prevalence of ASD in children with visual impairment secondary to other causes.
Methods
Retrospective study of children with albinism from January 2015 to December 2020. A control group was created with children with early onset visual impairment of similar visual range and age, secondary to diagnosis other than albinism. Patients with associated Autism were identified in both groups.
Results
Seven hundred and eight children aged 1–18 years with visual impairment were included in the study. 401 children had a diagnosis of albinism, of whom 14 were also diagnosed with ASD. In the control group, composed of 307 patients, only 3 had ASD (p: 0·03).
Conclusions
The prevalence of ASD in patients with albinism was 1 in 28, while in children with visual impairment from other causes was 1 in 102. We aim to raise awareness of the higher prevalence of autism in children diagnosed with albinism in order to reach earlier diagnosis and support.
Keywords
Introduction
Oculo-cutaneous albinism (OCA) is an inherited autosomal recessive disorder, characterized by a partial or complete deficiency of melanin pigment in the skin, hair, and eyes. 1 Clinical manifestations include iris and retinal pigment deficiency, foveal hypoplasia, photophobia, nystagmus, strabismus, decreased visual acuity, significant refractive errors, and misrouting of the optic nerve to the visual cortex. 1 OCA prevalence varies among different populations; reaching approximately 1:14,000 worldwide, 2 and at least 1:10,000 in Israel. 3 To date, 8 genes have been identified which cause OCA 1–8 respectively, with OCA1 representing the most severe form of OCA and is characterized by bi-allelic pathogenetic variants in the Tyrosinase gene (TYR).1,4 The prevalence of OCA subtypes varies according to ethnicity; OCA1 accounts for about 50% of cases worldwide and is the most common subtype in Caucasians. OCA2 accounts for 30% of cases worldwide and is the most common subtype in Africa, while OCA4 accounts for 17% of cases worldwide, and is the most common subtype in Japan but is rare among Caucasian and African populations. 5
Autism spectrum disorder (ASD) is a developmental disorder characterized by impaired communication and social interaction and by repetitive behaviors. 6 Several lines of evidence indicate that genetic and environmental factors may play a role in its pathogenesis. 6 The sex ratio for disease occurrence is estimated at 4:1 male to female, and the prevalence has increased to 1:100 in past decades, plausibly because of the broadening of the diagnostic criteria expanding to a ‘spectrum’ of clinical features. 7 This increase was seen in the Israeli population as well, with ASD incidence rate rising from 0.46% to 1.3% in a decade. 8
Genetic studies have been conducted on ASD patients and their families, with dozens of associated genes identified. 6
The etiology of ASD is multigenic, highly heterogeneous and often co-occurs with other conditions. 6 Only a handful of ASD-related diseases have monogenic causes, and rates of ASD co-occurrence in these diseases vary. 6 These include fragile X syndrome (ranged from 5 to 60%), tuberous sclerosis (ranged from 0.4 to 14%), neurofibromatosis (ranged from 0.2 to 14%) and Rett syndrome. 9
Co-occurrence of OCA and ASD was reported in several case reports. The first one from 1978 describes two tyrosinase-positive (not affected by OCA1) albino boys with moderate mental retardation and autistic behavior. 10 Other reports include a 3-year-old boy with OCA and ASD, 11 a 13-year-old boy with OCA and ASD, 12 and two siblings aged 11 and 13 diagnosed with OCA, ASD and Attention deficit hyperactivity disorder (ADHD). 13 Fazzi et. al described 12 patients with visual impairment and among them, 1 patient had OCA1 albinism and two had ocular albinism (OA). 14 Delong et al. described one or two members of 4 families with a paternal origin of ASD and mild albinism, possibly OCA2. 15
The association between ASD and visual impairment has been mentioned in the literature, 16 and ASD prevalence among children with visual impairment was found to be between 7.3% and 19%.14,17 A higher incidence of ASD was found among children with peripheral visual impairment such as ocular blindness, compared to children with central (cerebral) visual impairment. 14 In addition, children with visual impairment often have symptoms associated with both ASD and visual impairment, such as social isolation and difficulties with communication, making ASD difficult to diagnose. 14
The purpose of this study was to evaluate the co-occurrence of OCA and ASD in Israeli patients. We also aim to examine whether ASD cases among OCA patients could be due to visual impairment alone, or due to an underlying cause related to genetics or hypopigmentation.
Methods
A retrospective cohort study design was used to interpret the association between OCA and ASD. Data were collected from medical records of patients seen at the Michaelson Institute for low vision, Department of Ophthalmology, Hadassah Medical Center, Jerusalem, Israel. About 1200 children from all over the country are examined annually in our low vision institute, which is a national center for visually impaired children in Israel. Evaluation is carried out by a multidisciplinary team including optometrists, social workers, genetic counselors, and ophthalmologists. Relevant patients are referred to molecular genetic testing.
Our study group included children aged 1–18 diagnosed with albinism, who were seen at our clinic between January 2015 and December 2020. Diagnosis was done based on clinical characteristics; confirmation by molecular testing was obtained in some cases.
A control group included patients with similar visual acuity and age, diagnosed with visual impairment from their first year of life secondary to early onset retinal dystrophies (EORD) or aniridia. Diagnosis was done based in clinical characteristics, electrophysiology tests and/or molecular diagnosis. Children with best-corrected visual acuity (BCVA) better than 6/12 and children with diagnosis of typical Retinitis pigmentosa (RP) due to general conserved central vision during early childhood were excluded from the control group.
ASD diagnose was done following the American psychiatric association's “Diagnostic and Statistical Manual, fifth edition” (DSM-5) which bases diagnosis on at least three persistent deficits in social communication and interaction, and at least two restricted, repetitive behaviors to meet the criteria.
Medical records for subjects in both groups were reviewed for age, visual acuity (BCVA), phenotype and associated ASD. Furthermore, OCA patients in our study group were reviewed for albinism sub-types according to phenotype or/and genotype when available.
Molecular testing included whole exome sequencing (WES), panel for albinism or inherited retinal diseases and Sanger sequencing of known gene variants. Only pathogenic and likely pathogenic variants were considered as a positive genetic result.
Statistical analysis
To examine the relationship between two categorical variables, Chi-square (χ2) and Fisher's exact test were used. Comparison of quantitative variables between the two groups was done using a T-test or the asymmetric Mann-Whitney test.
Statistical analyses were performed using IBM SPSS software. Significance levels were set as p < 0·05.
Results
The study group consisted of 401 OCA patients. Full genotype data were available for 115 patients, and included variants in the TYR gene causing OCA1, the OCA2 gene, the SLC45A2 gene causing OCA4 and and the HPS3 gene causing Hermansky-Pudlak syndrome type 3. The control group consisted of 308 patients with early onset visual impairment. Figure 1.
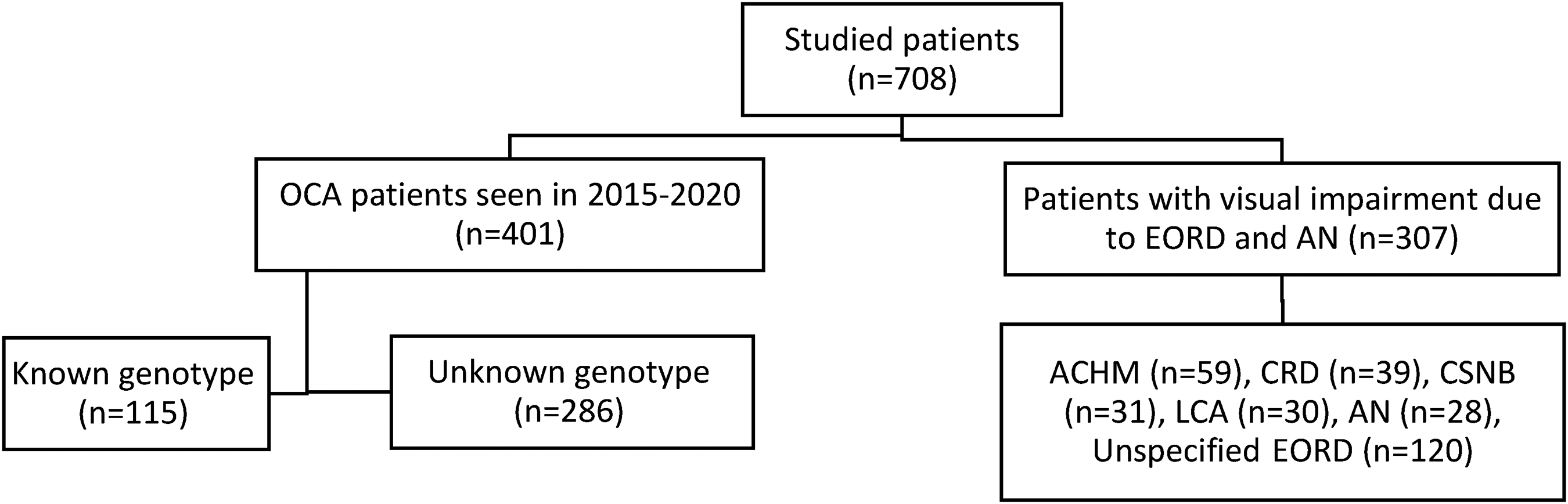
Distribution of OCA and control groups.
Control group characteristics and mean visual acuity are presented in Table 1.
Control group characteristics.
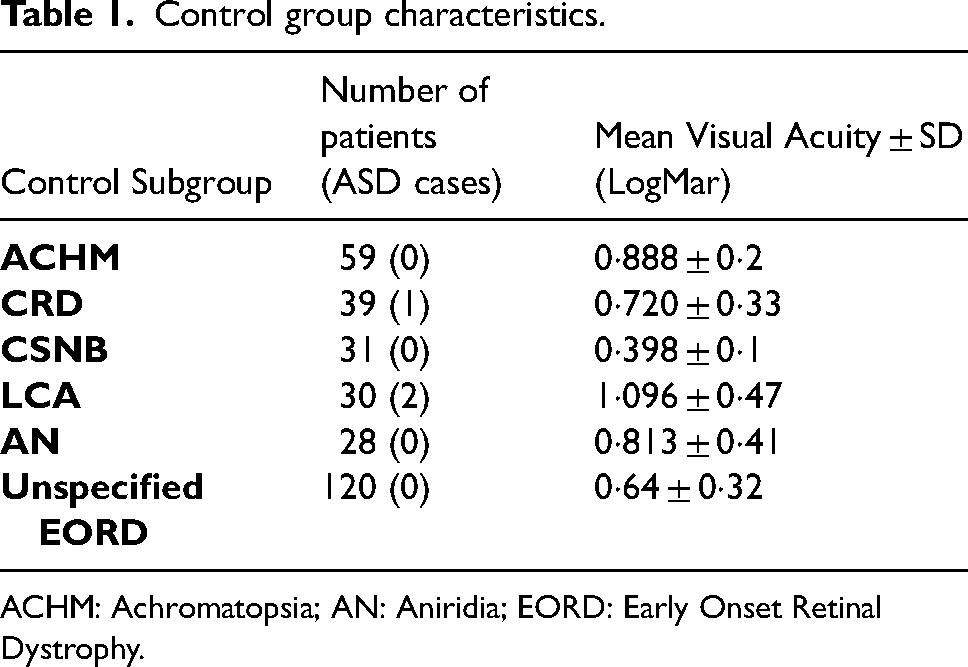
ACHM: Achromatopsia; AN: Aniridia; EORD: Early Onset Retinal Dystrophy.
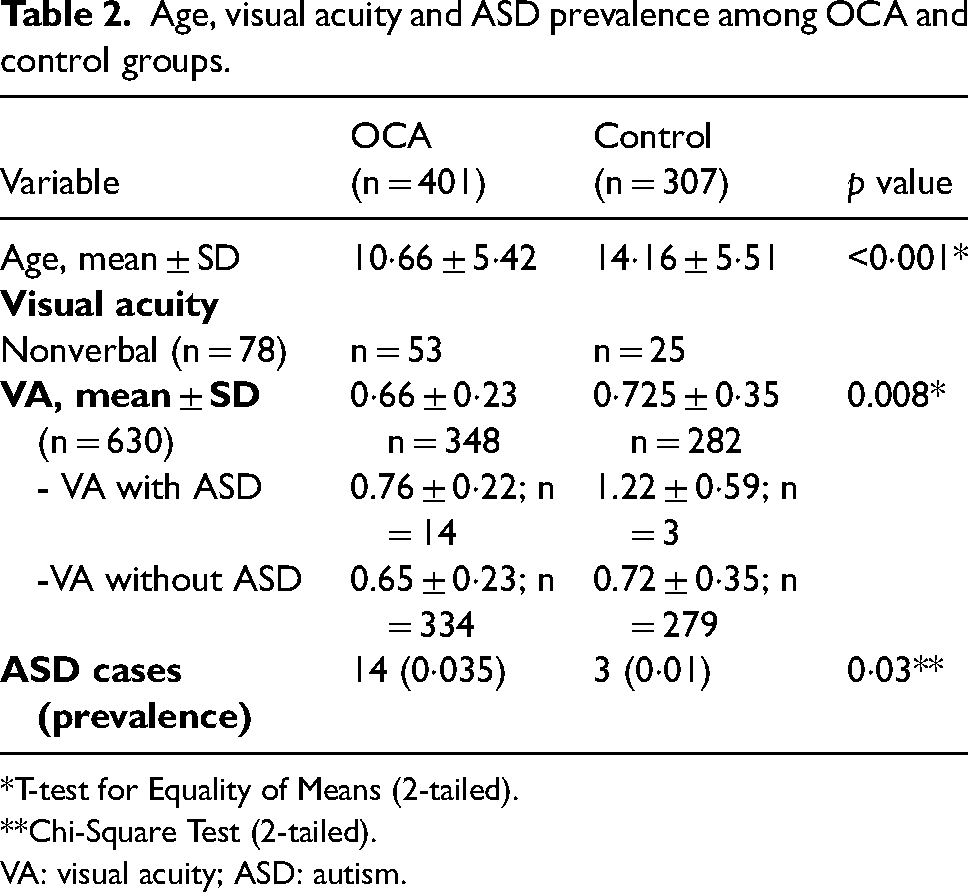
Comparison of age and visual acuity between the studied groups
Visual acuity was recorded as BCVA in Snellen chart and converted to logMAR scale for statistical purposes. Mean age among the OCA patient group was 10.66 ± 5·42, compared to 14.18 ± 5·51 in the control group (Table 2). Verbal visual acuity was obtained in 348/401 participants from the study group and in 282/307 from the control group.
Age, visual acuity and ASD prevalence among OCA and control groups.
*T-test for Equality of Means (2-tailed).
**Chi-Square Test (2-tailed).
VA: visual acuity; ASD: autism.
For the specific analysis of visual acuity in both groups, very young patients (non-cooperative for verbal visual acuities) and blind patients with finger counting, hand movement or light perception vision, (n = 77) were not included in testing for average visual acuity.
Mean visual acuity was 6/27 (LogMar 0·66 ± 0·23) among OCA patients compared to 6/32 (LogMar 0·72 ± 0·35) among the control group (Table 2).
Prevalence of ASD in the OCA group and the control group
Among the 401 children affected with OCA, 14 had a co-occurring ASD, compared to 3 ASD cases among 308 patients in the control group. (p = 0·03) (Table 2).
Analysis of visual acuity among patients diagnosed with ASD showed that 15/17had a numeric visual acuity measurement. Mean visual acuity was 6/43 (LogMar 0·86 ± 0·35) among ASD patients compared to 6/29 (LogMar 0·68 ± 0·29) among patients without ASD (p = 0·026). Among OCA patients, mean visual acuity was 6/35 (LogMar 0·764 ± 0·22) for ASD patients, compared to 6/27 (LogMar 0·656 ± 0·23) for patients without ASD (p = 0·1). Among control patients, mean visual acuity was 6/101 (LogMar 1·226 ± 0·59) for ASD patients, compared to 6/31 (LogMar 0·720 ± 0·35) for patients without ASD (p = 0·084). Table 2
Genotype in the OCA study group
Overall, 115 OCA patients had a known genotype. The disease-causing variants were identified in 87 patients in the TYR gene causing OCA1; in 22 patients in the OCA2 gene; in 1 patient in the SLC45A2 gene causing OCA4 and in 5 patients in the HPS3 gene, causing Hermansky-Pudlak syndrome type 3.
From the 14 children diagnosed with ASD among the OCA group, 10 had a confirmed genetic diagnosis: 8 were OCA1 patients (among them a pair of twin brothers), one OCA2 and another one OCA4. Four children had classical phenotype for OCA1A including white hair and skin, severe lack of pigment in iris and retina, foveal hypoplasia and severely reduced vision. Vision among albino sub-groups was similar (Table 3).
Genetically confirmed albinism sub-groups and visual acuity.

HPS: Hermansky-Pudlak syndrome.
Evaluation of causing variants for OCA1 revealed that the most common identified TYR variants were IVS2-7T > A (in Ashkenazi Jews) and M1 V. Gender distribution among children with ASD was 50% female and 50% male. (Table 4).
Genetics and ethnic characteristics of children with albinism and associated ASD.
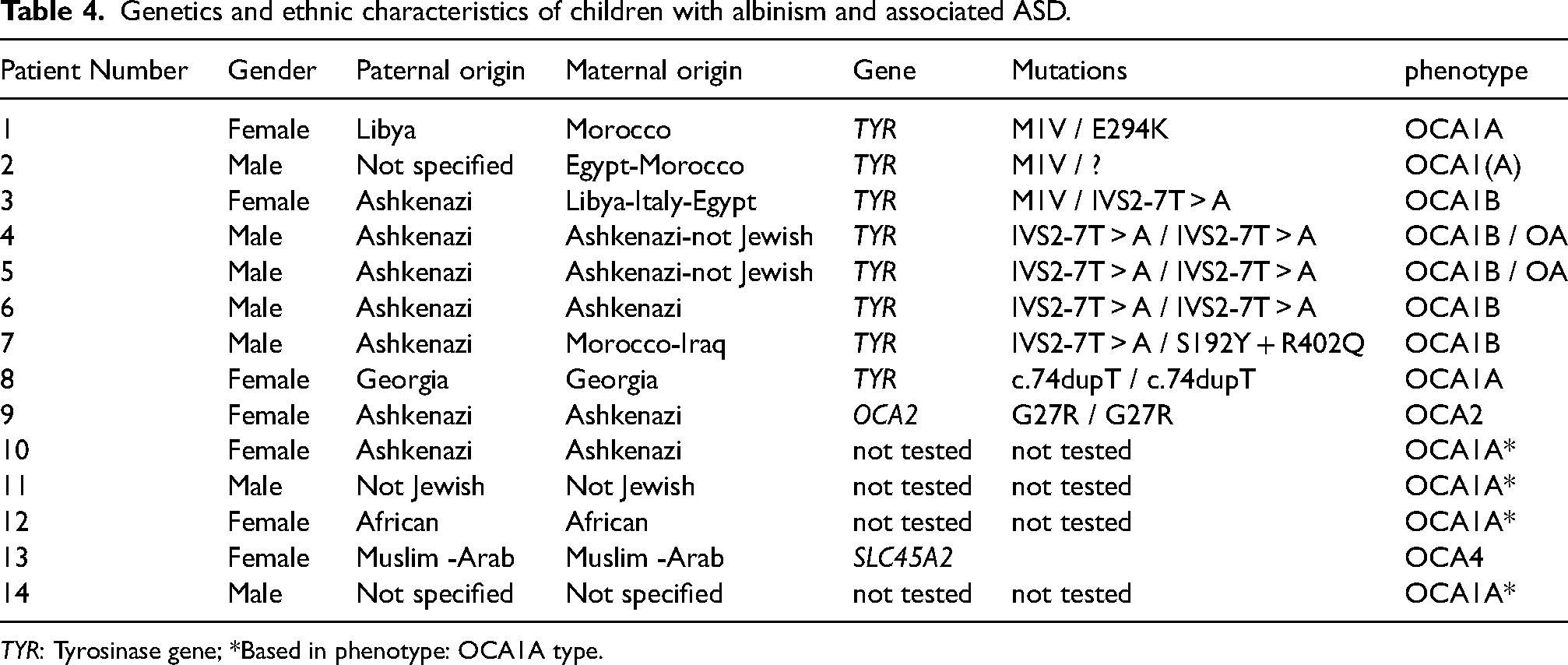
TYR: Tyrosinase gene; *Based in phenotype: OCA1A type.
Discussion
To the best of our knowledge, this is the first study to explore the rate of ASD in a large cohort of patients diagnosed with albinism. ASD prevalence was found to be 3.5% among patients with albinism, compared to 1% among patients with visual impairment secondary to various retinal dystrophies, and 5.3 times higher than ASD prevalence in the general Israeli population. These results suggest an increased prevalence of ASD among OCA patients (p = 0.03).
While a possible connection between visual impairment and development of ASD was proposed by several studies, most lack stratification for the severity of visual impairment, or the underlying cause for visual impairment.
The high incidence of ASD in the OCA group in this study cannot be explained by visual impairment alone, as visual acuity was similar or even slightly worse in the control group. In addition, the majority of children with ASD were found in the OCA1 group but there was no major difference in vision among albino sub-groups.
These results further strengthen our suspicion of an underlying cause other than visual impairment for the development of ASD among OCA patients.
Focusing on the molecular aspect, 12/87 children with OCA1 had ASD. Interstitial deletions involving the TYR gene, were previously described in patients with different types of learning difficulties, mental retardation and ADHD among other, but no albinism. 18
In contrast, only one of the 22 children with OCA2 in our study had ASD, significantly decreasing the possibility of a previously proposed connection between ASD and OCA2, based on the proximity of the OCA2 gene to a cluster of three GABA receptor subunit genes – GABRA5, GABRB3, and GABRG3. Allelic variants of these genes have been associated with increased risk of ASD. 19
Further research is needed to explain the co-occurrence of OCA and ASD.
Several hypomelanotic skin disorders, including oculocutaneous albinism, have been reported to co-occur with ASD, and susceptibility genes for several of these hypomelanotic skin disorders have chromosomal loci that lie near the loci for several major susceptibility genes for ASD. Examples of these diseases include tuberous sclerosis, hypomelanosis of Ito and Angelman syndrome.15,20 ASD occurrence ranges between less than 1% and up to 60% among individuals suffering from these disorders. 20
Abnormal neural migration and impaired neural connectivity have been shown to cause development of ASD.21,22 This same pathophysiological process has been proposed for the co-morbidity of ASD in other hypomelanotic disorders such as tuberous sclerosis. 21 OCA is characterized by impaired neuronal migration in the visual pathway, 23 and TYR gene expression was observed in embryonic neuroectoderm and migrating neural crest cells in mice model. 24 This anomaly in neuronal migration might explain the co-morbidity of ASD and OCA, and more specifically OCA1 patients, with impaired or absent TYR gene activity. Further evaluation of OCA1 specific variants might lead to better understanding of the genetic mechanism and point to genetic variants associated with increased risk for ASD.
In our study, the most common TYR gene variant identified was IVS2-7T > A that is considered a founder variant among Ashkenazi Jews with an incidence of 3.18% (http://server.goldenhelix.org/israeli, last checked in November 10th 2022). Among our cohort of children with ASD and Albinism, 10/14 do have an identified genotype including 6/10 from Ashkenazi origin (at least from one parent) that would explain the high occurrence of the IVS2-7T > A variant.
We have identified several limitations of our study design. First, age of ASD diagnosis was not recorded. We also suspect that there might be under diagnosis of ASD in both groups, partially due to patients’ young age, as 9.3% of the study patients are 4 years old or younger, and partially due to overlapping of ASD and visual impairment similar behaviors. Due to the relative scarcity of OCA and ASD, a larger cohort might have yielded more statistically significant results.
Visually impaired and autistic children often express similar behaviors, such as impaired communication, impaired social interactions, and lack of eye contact. 14 This, combined with the relative scarcity of ASD and OCA co-occurrence reports leads us to believe that there might be an underdiagnosis of ASD in OCA patients.
In Conclusion, the prevalence of ASD was higher in patients with OCA compared to the early onset visual impairment control group, which implies that this association is not explained by visual impairment alone. OCA1 was the main type of albinism associated with ASD. Further research is needed to understand the possible connection of ASD and OCA co-occurrence. Raised awareness of the extent to which ASD may be a co-occurring diagnosis in children with albinism can guide professional practice ensuring that individuals with albinism and suspected ASD receive early diagnosis and possible better outcome over time.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
