Abstract
Introduction
Torpedo maculopathy (TM) is a rare, typically benign and congenital anomaly of the retinal pigment epithelium (RPE) characterized by a torpedo-shaped lesion in the macula. DeSanto-Shinawi syndrome (DESS) is an equally rare genetic disorder caused by mutations in the WAC gene, presenting with intellectual disability, dysmorphic features, and growth retardation.
Case description
This report illustrates the case of monolateral TM in a patient with DESS. The genetic and phenotypic characteristic were described and correlated with retinal features, assessed by means of retinography, spectral domain OCT (SD-OCT) and fluorescein angiography (FA).
Conclusion
This case report discusses the unusual co-occurrence of TM in a patient diagnosed with DESS, contributing to the sparse literature on this association and expanding the phenotypic spectrum of both conditions.
Keywords
Introduction
Torpedo maculopathy (TM) is a retinal anomaly first described by Roseman and Gass in 1992. 1 It typically presents as a solitary, hypopigmented lesion with a pointed end directed towards the fovea, often located temporally within the macula. The etiology of this condition remains unclear, though it is considered a non-progressive and benign anomaly with minimal impact on visual function.1–3 The differential diagnosis should consider several other congenital lesions of the retinal pigment epithelium (RPE), such as combined hamartoma of the retina and RPE, congenital simple hamartoma of the RPE, and especially congenital hypertrophy of the RPE (CHRPE). CHRPE is associated with Gardner's syndrome, a dominantly inherited familial cancer syndrome that includes familial adenomatous polyposis and various soft tissue tumors.1–3
Even if a case of TM associated with a novel de novo NEXMIF mutation was reported, 4 a retrospective review of 17 patients with TM concluded that no pathogenic variant of NEXMIF gene was observed. 5
DeSanto-Shinawi syndrome (DESS) is a recently identified genetic disorder caused by heterozygous mutations or deletions in the WAC gene, which is located on chromosome 10p12.1, with fewer than 50 cases reported worldwide.6–8
The syndrome is characterized by facial dysmorphia, hypotonia, and cognitive alterations, including attention deficit hyperactivity disorder and autism. While ocular abnormalities have been reported in DESS (strabismus and amblyopia), the presence of TM has not been previously documented.
Despite their distinct presentations, there may be underlying genetic and developmental connections between these two conditions. This paper explores the possible etiologic links between TM and DESS.
Case presentation
The patient is a 4-year-old female with a confirmed diagnosis of DeSanto-Shinawi syndrome, identified through whole-exome sequencing (eterozygosys in WAC c.97A > T; pLys33Ter). She presented with a history of developmental delays, mild intellectual disability, and dysmorphic features including a broad forehead, hypertelorism, and downslanting palpebral fissures. Brain MRI highlights hypoplasia of the corpus callosum. There was no significant family history of ocular or genetic disorders. Informed written consent were obtained from child's parents. The patient underwent a routine ophthalmic examination as part of her comprehensive medical evaluation. Slit-lamp examination revealed no abnormalities in the anterior segment, and intraocular pressures were within normal limits. Fundus examination of the left eye revealed a solitary, well-demarcated, hypopigmented lesion located temporal to the fovea, with a characteristic torpedo shape, consistent with TM (Figure 1(b)). The lesion measured approximately 1.5 disc diameters in length, with the pointed end directed towards the fovea. No other retinal abnormalities were noted in either eye. Fluorescein angiography (FA) showed no leakage or abnormal vascularization associated with the lesion (Figure 1(d)). Spectral-domain Optical Coherence Tomography (OCT) (SD-OCT) (Spectralis®, wavelength: 870 nm; Heidelberg Engineering Co, Heidelberg, Germany) of the macula confirmed the presence of a hyporeflective area corresponding to the lesion with focal disruption or thinning of the retinal pigment epithelium (RPE) at the site of the lesion. The outer retinal layers, including the ellipsoid zone and the interdigitation zone, show irregularities of reflectivity overlying the area of RPE alteration. The choroid beneath the lesion shows signs of hyperreflectivity (“window effect”) (Figure 1(f) and (h)). Retinography (Figure 1(a)), FA (Figure 1(c)) and OCT (Figure 1(e) and (g)) of the fellow eye were unremarkable.
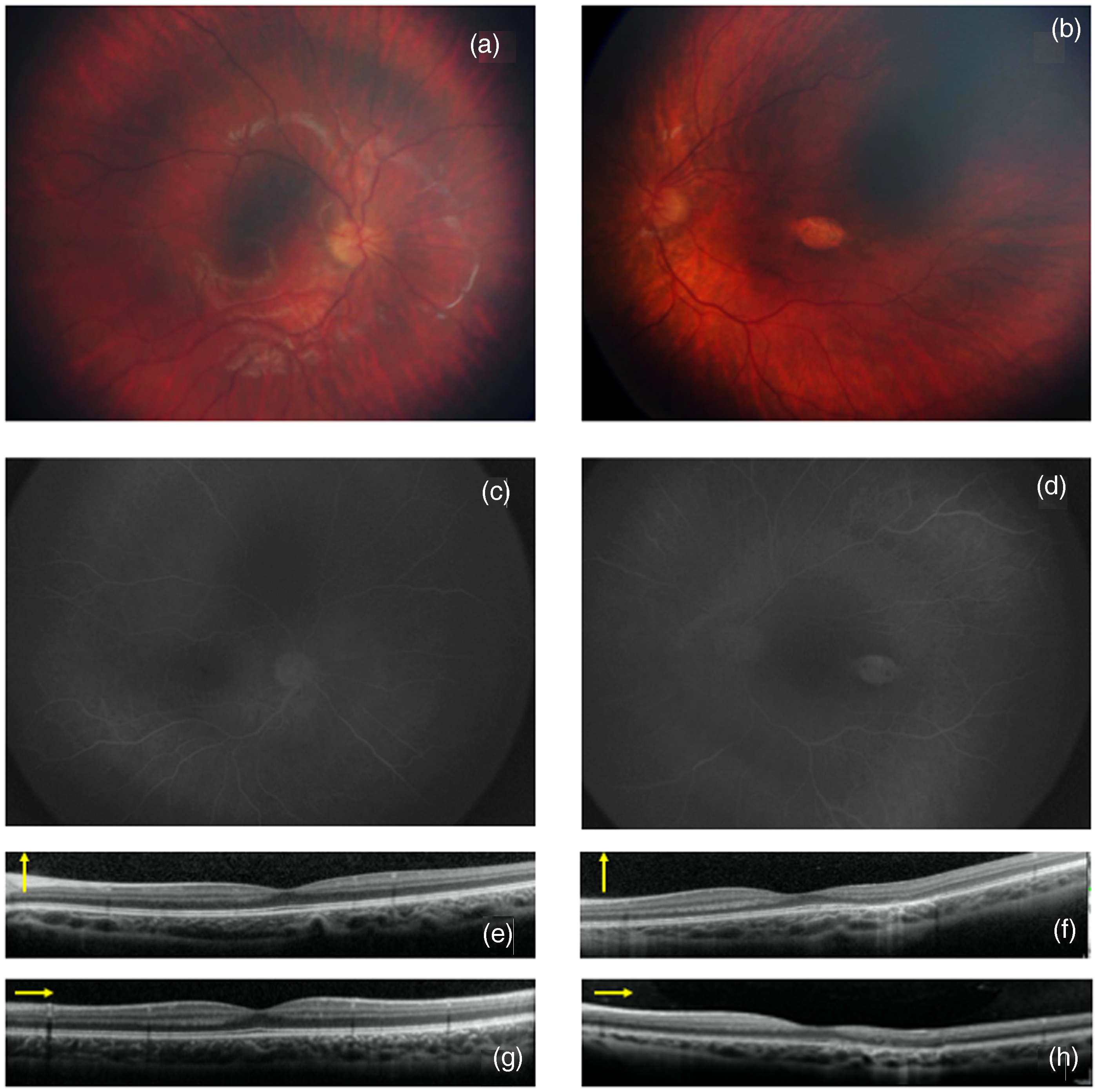
Color fundus photography, fluorangiography (FAG) and optical coherence tomography (OCT) in a patient with DeSanto-Shinawi Syndrome with Torpedo Maculopathy (description in the text).
Discussion
This case represents the first documented occurrence of Torpedo Maculopathy (TM) in a patient with DeSanto-Shinawi Syndrome (DESS). Although TM is typically regarded as an incidental finding with no known systemic associations, its presence in a patient with DESS suggests a potential underlying connection that warrants further exploration. DESS is associated with mutation in WAC gene.6–8 The WAC gene encodes a protein involved in transcriptional regulation and chromatin organization, essential processes for gene expression and normal cellular function during development. The WAC protein is critical for various cellular activities, including regulation of transcription (ensuring that the correct genes are expressed at the right time) and chromatin remodeling (altering the structure of chromatin to allow or restrict access to DNA, which is crucial for gene expression and DNA replication). Both the retina and the central nervous system originate from the neuroectoderm during embryogenesis. Disruption of WAC function due to mutations could potentially impact the neuroectodermal derivatives, leading to concurrent developmental anomalies in the brain (manifesting as intellectual disability and other neurological features in DeSanto Syndrome) and the retina (potentially contributing to the development of TM). The WAC gene's role in chromatin remodeling and transcription regulation might influence the formation and maintenance of the RPE, a layer crucial for retinal health and function. Abnormalities in RPE development could result in the distinct lesions seen in TM. Since TM is thought to stem from a defect in the RPE, a WAC mutation could feasibly disrupt the normal developmental processes of the RPE, leading to the formation of the torpedo-shaped lesion characteristic of TM. The WAC protein is also implicated in pathways that regulate cytoskeletal organization and signal transduction. These processes are essential not only for the structural integrity of cells but also for intercellular communication during development. A disruption in these pathways could impair the development of retinal cells, including those in the RPE and choroid, potentially leading to TM. Although the co-occurrence of DESS and TM has not been widely reported, the identification of both conditions in a single patient could suggest a shared developmental origin or a common underlying genetic disruption. Further genetic studies focusing on patients with WAC mutations who also present with TM could help clarify whether there is a statistically significant correlation between these conditions. A higher prevalence of TM in patients with DESS would support the hypothesis of a pathogenic link. While direct etiopathological correlations between WAC mutations in DESS and TM remain speculative, the potential shared developmental pathways offer a plausible explanation for their co-occurrence. The WAC gene's role in transcription regulation, chromatin remodeling, and cytoskeletal organization could impact both brain and retinal development, leading to the neurological features of DESS and ocular anomalies like TM. Further research, including detailed genetic and clinical studies, is needed to explore these connections and to determine whether mutations in WAC could be a contributing factor to the development of TM in affected individuals.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This work was supported also by the Italian Ministry of Health with “Current Research funds”.
