Abstract
Purpose
To describe multimodal imaging of two cases of bilateral non-vascularized pigment epithelial detachments (PED) in young patients with a long-term follow-up.
Methods
A complete ophthalmological examination was performed at each follow-up visit including best corrected visual acuity (BCVA), intraocular pressure, slit lamp examination, spectral domain optical coherence tomography (SD-OCT), fluorescein and indocyanine green angiography, OCT angiography.
Results
Multimodal imaging of two women presenting avascular PED, aged 43 and 57, respectively, was described. In both patients, SD-OCT revealed a high central macular hyporeflective elevation corresponding with PED. Both patients showed a choroidal layer thicker than 420 μm. Fluorescein and indocyanine green angiography didn’t show any choroidal neovascularization either at early or late frames. Cross-sectional and en face optical coherence tomography angiography (OCTA) didn’t show any flow beneath the PED. During the follow up period one eye showed a retinal pigment epithelium tear and all eyes showed the presence of apical sub-retinal fluid and hyperreflective material on the top of the PED. None of the two patients showed any sign of atrophy during the follow-up period.
Conclusion
The peculiar characteristics of the presented cases suggest that specific pathogenetic mechanisms, not necessarily related to age related macular degeneration, may play a key role in the development of these lesions. Whether early onset of such drusenoid PED is a specific entity resulting from a genetic deficit of lipid transporters in the RPE is unknown. Further genetic and metabolic studies should be conducted.
Keywords
Introduction
Pigment epithelial detachment (PED) is defined as the anatomical separation between the basal lamina of the retinal pigment epithelium (RPE) and the underlying Bruch's membrane and is a well-known feature of many chorioretinal disease processes, the most prevalent of which is age-related macular degeneration (AMD). 1 Pigment epithelial detachments are classified as vascularized and non-vascularized. 2 Non-vascularized PEDs are usually associated with age-related non-neovascular macular degeneration and central serous chorioretinopathy. These lesions typically present as round areas of hypofluorescence in the last frames of the Indocyanine green angiography. 3 Zayit-Soudry and collaborators classified PEDs into four categories according to their etiology, in particular: inflammatory, ischemic, idiopathic, and degenerative. 4 They further classified degenerative PED into three main groups according to multimodal imaging findings: (i) serous, (ii) drusenoid and (iii) fibrovascular.2,4 On SD-OCT, Serous PEDs are defined as localized, sharply demarcated, abrupt dome-shaped RPE elevations, with a homogenously hyporeflective sub-RPE space, secondary to a collection of fluid between the RPE and Bruch's membrane.2,4 Drusenoid PEDs are defined on SD-OCT imaging as lobular detachments of the RPE with a homogenous, moderately hyperreflective content, affecting the central macula. 2 Secondary neovascularization is a possible evolution of non-vascularized PED. Interestingly, Elman et al. showed that this complication occurs in 32% of patients with serous PED. 5 Drusenoid PEDs are associated with a lower risk of developing CNV, which was observed in 13% of patients in a retrospective study by Roquet et al. 6 Their phenotypic characteristics and associated features have been described on multimodal imaging but the pathophysiology of these lesions remains unclear. 7
In this study, we present multimodal imaging of two cases of bilateral non-vascularized PED in patients younger than 60 years with a long-term follow-up. Their peculiar characteristics suggest that specific pathogenetic mechanisms, not necessarily related to AMD, may play a key role in the development of these lesions.
Cases description
Case 1
A 43-year-old woman presented to the emergency room with a recently decreased vision in the left eye. The patient was phakic and refractive error was +0.25 in both eyes. She had a family history of early-onset coronaropathy and she did not take any medications. Best corrected visual acuity was 20/20 in the right eye and 20/30 in the left eye. Spectral-Domain OCT revealed in both eyes a high central macular hyporeflective elevation of the retinal pigmented epithelium qualified as a serous PED (Figure 1) associated in the right eye with smaller macular drusenoid PEDs and in the left eye with serous retinal detachment at the top of the PED. Choroidal retrofoveolar thickness was 428 μm in the right eye and 482 μm in the left eye. Both eyes presented peripheral retinal drusen. Both fluorescein and indocyanine green angiography didn’t show any choroidal neovascularization either at early or late frames (Figures 2 and 3). The patient received oral spironolactone 25 mg/day for 4 months with no significant effect on PED. Six months later, the PED height increased in both eyes and sub-retinal fluid was still present at the top of the PED in the left eye (Figure 1, Part c-d). On the left eye, the sub RPE fluid appeared in an excavated area above the choriocapillaris layer (Figure 1, Part d).
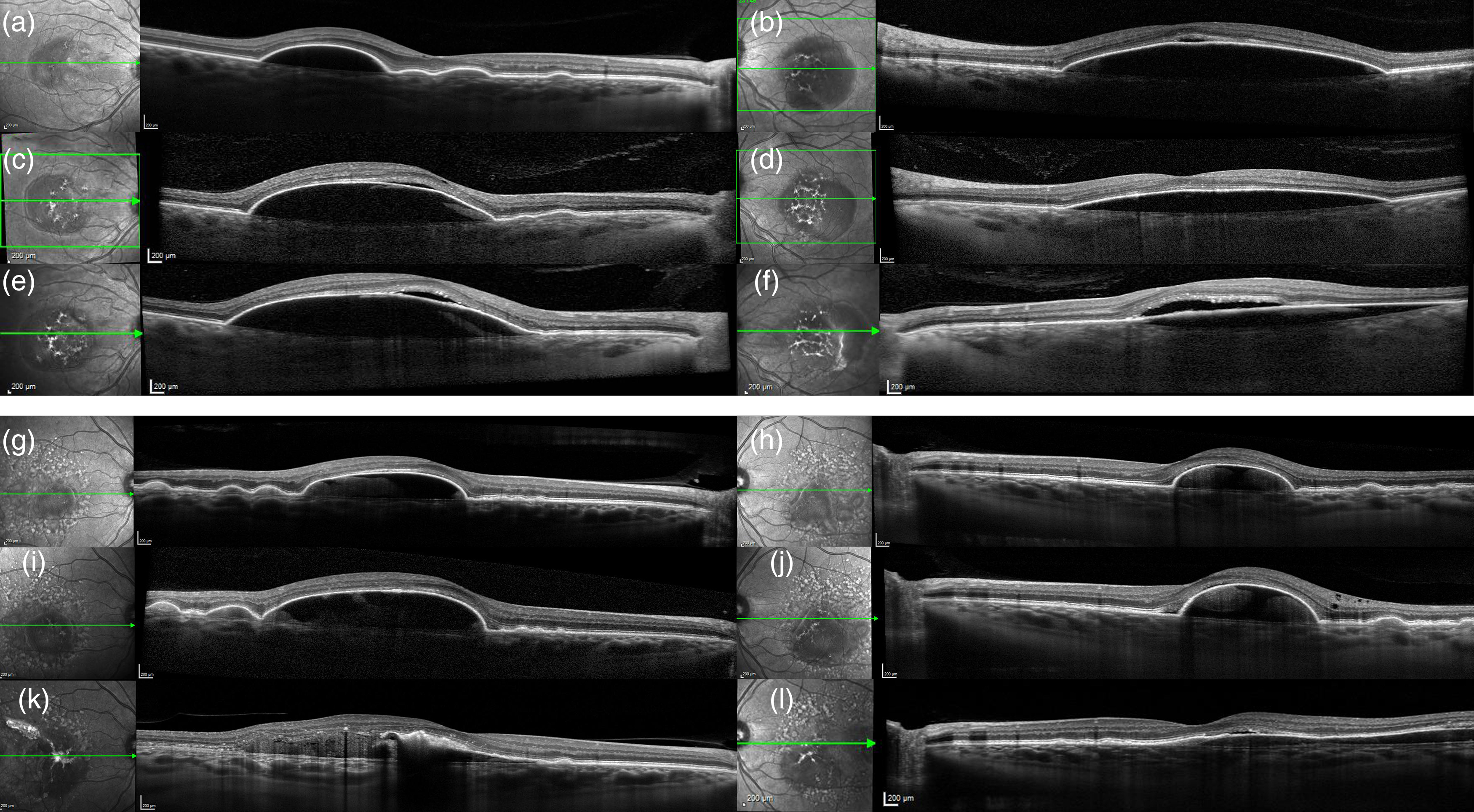
Long term follow-up on spectral domain optical coherence tomography (SD-OCT) of case 1 and 2.
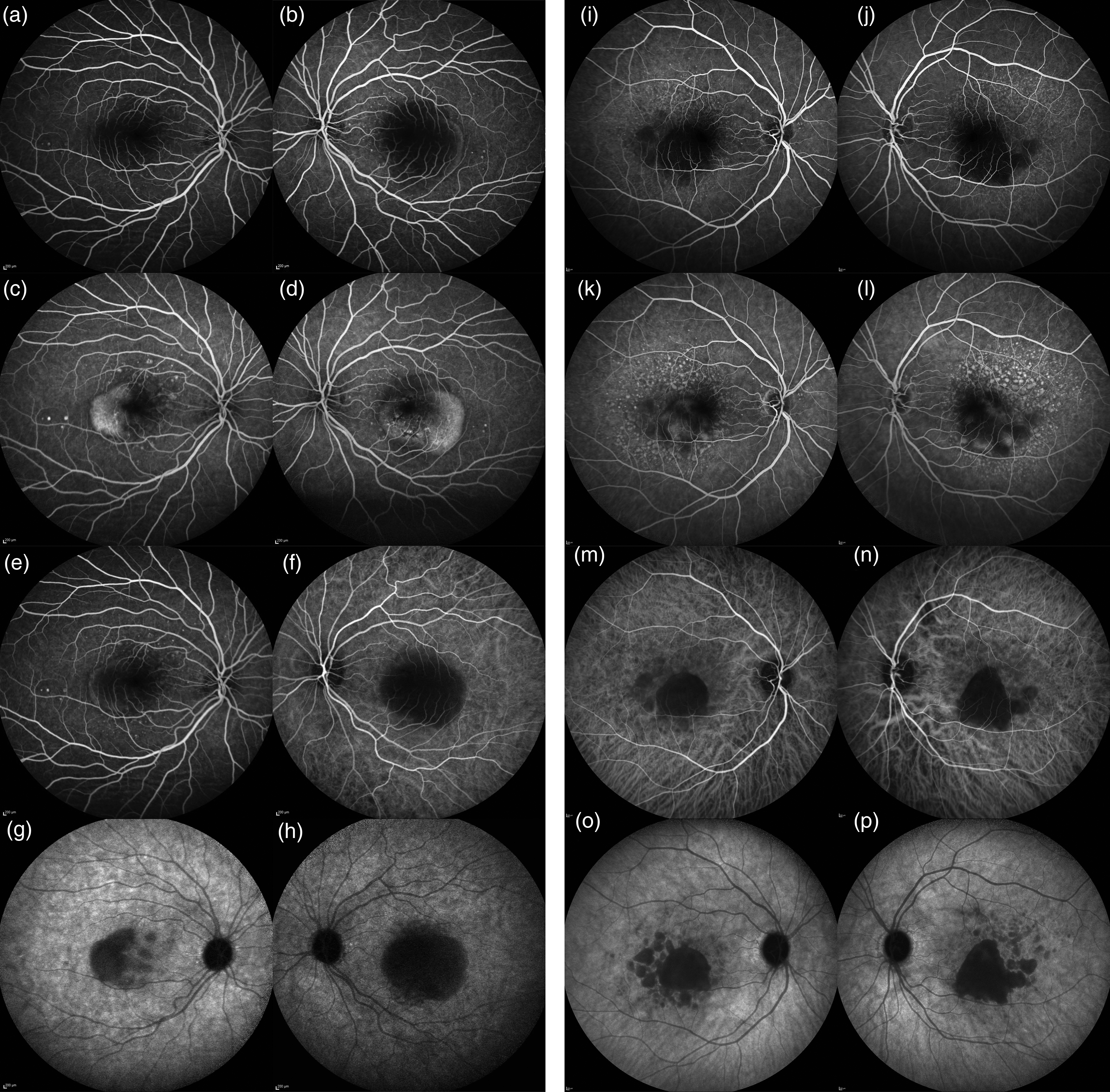
Fluorescein and indocyanine angiography of case 1 and 2

High-definition SD-OCT B-scan of case 1
Case 2
A 57-year-old woman presented to the emergency room for bilateral decreased vision 1 months earlier. She had a family history of AMD. She suffered of recurring vasovagal episodes with arterial hypotension and migraine. She was hyperopic (+6.50 in the right eye and + 7.50 in the left eye) and was treated for angle closure related ocular hypertension with bilateral iridotomy. Best corrected visual acuity was 20/35 in both eyes. Spectral-Domain OCT revealed bilateral high central macular hyperreflective elevations of the retinal pigmented epithelium qualified as a drusenoid PEDs (Figure 1). These lesions were associated with numerous serous drusen in the macula. Choroidal retrofoveolar thickness was 433 μm in the right eye and 475 μm in the left eye. Both fluorescein and ICG angiography didn’t show any signs of choroidal neovascularization at early and late frames (Figure 2). Eight months later, the PED height increased in both eyes and the subretinal fluid was more visible laterally and at the top of the PED in the left eye (Figure 1, Part i-i). Four years later, a RPE tear on the right eye appeared and hyperreflective material on the top of the PED increased (Figure 1, Part m).
Conclusions
We reported herein the multimodal retinal imaging of two early-onset bilateral avascular PED cases with long-term follow-up. During follow-up, in both cases we observed an increase in the height of the PED and the presence of various associated complications such as SRF and RPE tear. Although such PED and associated complications can occur in cases of AMD, the two cases presented here show atypical features in the context of AMD. The large and high, bilateral, and symmetric PEDs affect women, younger than expected for AMD. In addition, in both cases, numerous peripheral drusen are observed and both patients had a thick choroid which is a well-known characteristic peculiar disease processes other than AMD, in particular pachychoroid spectrum disease. Usually, pachychoroid associated PED are narrow-peaked serous PEDs, chronic flat irregular PEDs or polypoidal shaped in case of polypoidal vasculopathy. 4 They are typically associated to a thick choroid and can be associated to retinochoroidal folds in case of peripapillary pachychoroid syndrome. PED can also be similar to those in AMD in case of secondary neovascularization. In AMD, PEDs have different types: non-neovascular serous or drusenoid PEDs, and fibrovascular PEDs in case of choroidal neovascularization. Serous non-neovascular PED's description can be similar to our patient's lesions but it appears usually in older patients with a thin choroid.
Family members of these two patients (parents, brother and sisters and children) had multimodal retinal imaging and none of the examined cases (older and younger than the patient) showed any abnormality.
To date, multimodal imaging does not allow us to define the underlying cause of these lesions. However, these findings may suggest a separate entity from AMD with a possible genetic substrate and high-definition scans may help to better understand the pathophysiology of these lesions.
Several studies conducted on the pathophysiology of serous drusen in AMD suggested that biochemical changes in Bruch's membrane hamper the normal flow of fluid through it. Age-related deposition of lipids, such as cholesterol esters, triglycerides, and fatty acids, in the Bruch's membrane may modify its permeability and alter retinochoroidal flow. 8 In particular, it has been hypothesized that in the development of drusenoid PEDs the progressive accumulation of lipids at the level of Bruch's membrane would make it more and more hydrophobic.
Otherwise, it is possible that the initial dysfunction may be at the level of the RPE. In particular, a stressed RPE pump could become unable to adequately move fluid and debris across the Bruch's membrane, leading to their accumulation in the sub-RPE space and enlargement of drusen. 9 In fact, the subretinal fluid is a frequent feature of high avascular PED and can be considered as a sign of deficient RPE pump, probably resulting from the mechanical stress at the basolateral side of the RPE, located at the top of the hill. Interestingly, Tan et al. showed that intraretinal and subretinal hyperreflective foci on SD-OCT scans could correspond to displaced RPE cells on histologic examination. 3
In our patients the whole ICG angiography sequence didn’t show any diffusion of the dye at the level of the sub-RPE space, although a partial filling with fluorescein followed by a late wash out is observed. This dye behaviour is suggestive of a dysfunction of the RPE with increased permeability to water soluble and small molecules but also of a lipidic content of the sub RPE deposits.
Concerning the treatment for non-neovascular PEDs, to date, there is no consensus on the best therapeutic option. Half-dose PDT has been shown to be potentially effective, however further prospective studies are needed to investigate this issue. 10
In conclusion, whether early onset of such drusenoid PED is a subtype of early AMD or whether it is a specific entity resulting from a specific and possible genetic deficit of lipid transporters in the RPE is unknown. Genetic and metabolic studies should be undertaken in patients presenting this phenotype to identify potential therapeutic targets for these young and active patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Patient consent
Written informed consent was obtained from all patients.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
