Abstract
Purpose
Ghost drusen (GD) are pyramidal or dome-shaped retinal pigment epithelium elevations observed in some geographic atrophy (GA) areas in the context of age-related macular degeneration (AMD). The purpose was to investigate the first morphologic features preceding GD on spectral-domain optical coherence tomography (SD-OCT) on patients with GA associated with AMD.
Methods
A retrospective observational study was performed on a series of patients with GA that had at least 3 years of follow-up. Using the follow-up tool of SD-OCT, we tracked the initial lesions that could lead to GD.
Results
Among 442 patients with GA, 37 had well defined GD (8%). We included the 17/37 patients (31 eyes) with at least 3 years of follow-up for analysis, which led to a total of 582 counted GD. Most GD were already present at the first visit, and remained stable. However, on 13 of the 582 analyzed GD (2.2%), soft drusen were shown as the initial lesion, which progressively turned into GD.
Conclusions
GD were observed in less than 10% of eyes with GA. None of the ghost drusen turned into another shaped lesion, suggesting that GD is a possible final stage of evolution. In a few cases, large drusen were shown as the primary lesion that progressed into GD.
Introduction
Age-related macular degeneration (AMD) remains a leading cause of irreversible visual loss in people over 50 years old in the developed world (1). The clinical hallmark of early AMD is termed “drusen” as a focal deposition of acellular polymorphous material-extracellular debris. Drusen are located between the retinal pigment epithelium (RPE) basal lamina and the inner collagenous layer of the Bruch membrane (2). They have been classically characterized as hard or soft, and small (<63 microns), intermediate (>63 microns but <125 microns), or large (>125 microns) (3-4-5). Reticular pseudodrusen are described as a distinct type of drusen with small and pyramidal elevations located internal to the RPE (6).
Ghost drusen (GD) have been described in patients with geographic atrophy (GA) in 2014 (7). They were reported as pyramidal or cone-shaped structures in atrophic areas below a disorganized RPE associated with posterior shadowing. In most cases, they appeared on infrared reflectance (IR) images as hypo-reflective lesions capped by a hypo-reflective halo with or without hyper-reflective spots inside. On fundus autofluorescence (FAF) imaging, GD are either hypo-autofluorescent (59%) (7) or mildly hyper-autofluorescent. On spectral-domain optical coherence tomography (SD-OCT), the surface of this pyramidal structure is always hyper-reflective, whereas the core appears mainly hypo-reflective. Drusen have already been described in several studies as biomarkers of inflammatory process in AMD (8-9-10-11-12-13). However, the physiopathological mechanism of GD formation remains unclear.
The present study was an attempt to detect the initial morphologic findings preceding GD, and then understand their origin. We performed a retrospective analysis of images over 3 years in GA patients associated with GD on the latest SD-OCT examination. SD-OCT features were analyzed retrospectively in order to find and describe a primitive lesion that leads to GD.
Methods
Setting and design
This was a monocentric, retrospective, and observational study performed at the University Eye Clinic of Creteil, a tertiary care retina unit in France.
Study population
We reviewed charts of all patients presenting GD on SD-OCT in GA, from June 2014 to February 2015 who had been previously examined on a regular basis in the Eye Clinic for at least 3 years. In all cases, we went back to the first imaging examination recorded in the Spectralis device in our department. The criterion for inclusion was the presence of at least 1 GD associated with GA on the latest examination. For each patient, depending on the presence (or not) of bilateral GD, we could include 1 or 2 eyes. All patients over 50 years with GA and GD were included. GA was defined as any sharply demarcated area of apparent absence of RPE (larger than 500 microns), with visible choroidal vessels, without choroidal neovascularization activity, and no intraretinal or subretinal fluid. GD was identified as pyramid- or dome-shaped elevations of RPE in atrophic areas with hyper-reflective borders and a hypo-reflective center on SD-OCT.
All patients underwent complete ophthalmologic examination, including confocal scanning laser ophthalmoscopy imaging and eye-tracked Spectralis SD-OCT scans, as a part of the routine clinical work-up. For each patient, all SD-OCT scans were analyzed from the oldest (also called “first visit”) to the most recent exam.
Patients with neovascular AMD were excluded from the analysis.
Confocal scanning laser ophthalmoscopy and SD-OCT image acquisition
All included eyes underwent confocal scanning laser ophthalmoscopy imaging as IR-fundus illumination (l = 830 nm), FAF (excitation, l = 488 nm; barrier filter, l = 500 nm), and eye-tracked Spectralis SD-OCT (Spectralis HRA + SD-OCT; Heidelberg Engineering) with a minimum resolution of 768 × 768 pixels and a 30° × 30° field centered on the fovea. Each SD-OCT line acquisition follow-up was matched with IR by the eye tracker.
Confocal scanning laser ophthalmoscope SD-OCT image analysis
For each SD-OCT image, we counted GD within the GA area on each eye of the latest and the earliest visit examination. For each patient, 2 independent readers (C.B., O.Z.) analyzed the shape changes of GD throughout the retrospective follow-up period on SD-OCT lines matched with IR in follow-up mode. In order to confirm that our follow-up OCT was performed exactly on the same lines as each previous examination, we focused on the presence of a “landmark,” such as soft drusen or retinal vessels, located at the same place. This allowed us to compare lines from the same patient at different times and to be sure that they were actually located at the same place in the retina for each examination line.
Results
A total of 442 patients with GA were analyzed. Among them, 37 patients (8%) presented, on at least 1 eye, well-identified GD with fine reproducibility of lines on the SD-OCT at the latest visit. Within this group, we included the 17 patients with a minimum of 3 years follow-up (mean follow-up 52 months; mean age 79 years old). We excluded 3 eyes from the study because of poor quality imaging during the follow-up. A total of 31 eyes with a total of 582 counted GD were finally included in this population.
Most of the GD (420/582, 72%) were already present at the first visit and were not modified during the follow-up period (Fig. 1).
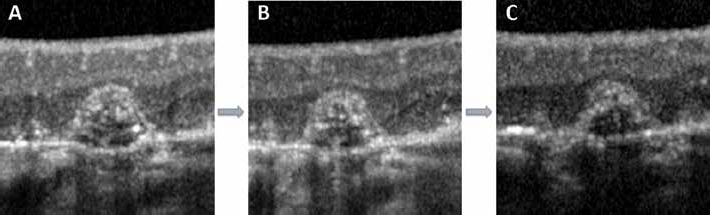
Stable ghost drusen (GD) imaging with spectral domain optical coherence tomography (SD-OCT). These are follow-up scans over 3 years (December 2011 (
However, in some cases, an initial soft drusen that seemed to become hypo-reflective throughout the follow-up was observed. Indeed, the presence of an initial soft drusen preceding GD was ascertained in 13/582 (2.2%) GD. These images were issued from 2 different patients (Figs. 2 and 3). These drusen appeared primarily hyper-reflective, then became heterogeneous with some hyper-reflective spots inside, and finally became mostly hypo-reflective at the GD stage. The dome-shape of the soft drusen simultaneously evolved to a progressive hyper-reflective pyramidal or conic shape.
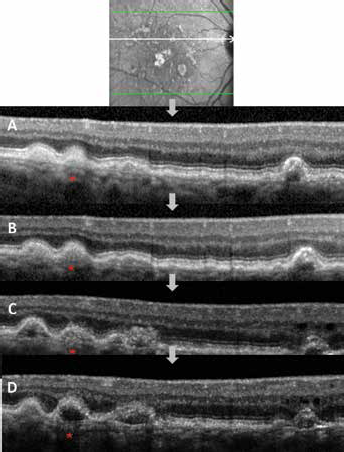
Progression of initial soft drusen to ghost drusen (GD) over time, imaged with spectral domain optical coherence tomography (SD-OCT). Follow-up over 4 years (March 10, 2010 (
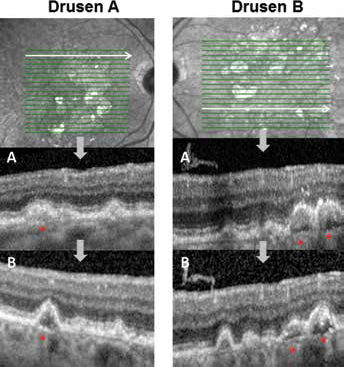
Two cases of progression from initial soft drusen to last ghost drusen (GD) of the same patient with geographic atrophy imaging with spectral domain optical coherence tomography (SD-OCT).
When analyzing the morphological aspect of the initial soft drusen on SD-OCT, it was noticed that the integrity of the ellipsoid zone (EZ) was maintained in the early soft drusen formation, but was then disrupted when GD appeared (Fig. 2D). These changes were associated with a thinning of the outer nuclear layer and an abnormal continuity between GD and the outer plexiform layer. It is notable that, when constituted, GD morphology remained stable and none disappeared in this 3-year analysis. We did not identify any primary lesions for other GD that appeared during follow-up (149/582).
Discussion
In this present retrospective study, in the vast majority of cases (420/582), GD were already present 3 years before the last SD-OCT examination. In 2.2% of cases, the initial lesions preceding GD could be ascertained as typical soft drusen. Although drusen were first described nearly 150 years ago as a risk factor for exudative AMD, the cellular and molecular events involved in their formation have not been fully elucidated (14, 15). Molecular composition of drusen comes from both sources: RPE (and perhaps other local cell types) (11, 16-17-18-19-20-21) and molecules that are extravasated from the choroidal vascularization (17, 22-23-24-25-26). Thompson et al (27) proposed a mechanism in which hydroxyapatite spherules formation around lipids in the sub-RPE space may promote further protein binding, leading to the growth of sub-RPE deposits.
However, drusen seem to be not only waste, which can no longer eliminated, but also active systemic immune-related mechanisms triggered by the failure to eliminate trapped material. The observation of plasma proteins in drusen has led to the hypothesis that the immune response could be implied in their formation (11, 17, 28-29-30). Indeed, the RPE layer is part of the outer blood-retinal barrier that prevents nonspecific diffusion from the choroid, and dysfunction of RPE cells in AMD may allow excessive inflammatory activation in the retina (31). Because of the relative loss of its original components inside the GA area, due to loss of RPE and the presence of an abnormal choroidal layer, our hypothesis is that soft drusen dislocation could lead to GD (32).
In the present series, GD were present in only 37 of 442 (8%) patients with GA. In the included eyes, the number of GD increased systematically over the total follow-up. In 2008, Flenckenstein et al (33) described several SD-OCT alterations of the outer retinal layer in GA and which ones were crown-like elevations of the RPE/Bruch membrane complex with “debris” beneath. It has been postulated that these alterations may represent various stages of regressing drusen and may remain for many years.
In the present analysis, GD were associated with abnormal continuity with an outer plexiform layer and disrupted EZ. A recent study compared SD-OCT imaging with the histological features of remaining hyper-reflective undulating deposits contiguous to the outer portion of RPE (34). These lesions were described in advanced AMD, within an atrophic lesion (secondary to GA or choroidal neovascularization), and were named outer retinal corrugations. This analysis suggested that these curvilinear hyper-reflective lines could represent persistent basal laminar deposits despite the loss of the RPE and the photoreceptors. Concavities created by undulating deposits are described as a relative hypo-reflective space with cells of retinal origin and fragments of calcified drusen. We may hypothesize that GD with pyramidal or conic hyper-reflective shape below the RPE were consistent with persistent basal laminar deposits despite the loss of the RPE and the photoreceptors.
Suzuki et al (35) localized refractile drusen from non-neovascularized AMD in four concentric zones across the posterior pole. They found that the centrifugal evolution of the calcification process on color and SD-OCT imaging correlated with macular atrophy on FAF imaging. They also identified calcific nodules from histologic macular sections in geographic atrophy that remained remarkable over RPE atrophy. Refractile drusen have already been associated with the progression of geographic atrophy in several studies (36, 37).
In our study, most of the GD were already present at least 3 years ago. However, initial lesions preceding GD were detected in only 2.2% of cases, probably because most of the patients were not imaged soon enough during the AMD evolution. It is notable that no other kind of precursor lesion could be identified. However, the reason for this relatively rare evolution remains unsolved. A prospective follow-up of soft drusen would be of major interest to further understand the physiopathology and prognosis of GD.
Conclusion
GD were observed in less than 10% of eyes with GA. We could detect soft drusen as initial lesions preceding GD in a small number of cases (2.2%). Once constituted, GD remain remarkably stable; thus, they could be considered as a possible final stage of the progression of soft drusen.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has financial interest related to this study to disclose.
