Abstract
Purpose
To examine structural and clinical changes in previously treated type 1 macular neovascularization (MNV) in non-responder age-related macular degeneration (nAMD) eyes switched to brolucizumab. Subretinal hyper-reflective material (SHRM), intraretinal (IRF) and subretinal fluid (SRF) presence, fibrovascular-pigment epithelium detachment (PED) height and central macular thickness (CMT) variation were analyzed using optical coherence tomography (OCT).
Methods
In this prospective study all patients underwent a complete ophthalmological evaluation including structural OCT at baseline (T0), one month (T1), three (T2), four (T3) and six months after switching to brolucizumab treatment (T4). Non-responder criterion was the persistence of IRF and SRF. Moreover, CMT and BCVA had shown worsening or no improvement before switching to brolucizumab. Clinical function and structural activity biomarkers were measured at each visit and changes were analyzed. P value <0.05 was considered statistically significant.
Results
Twenty eyes of twenty patients were enrolled. All the structural variables examined during the follow-up showed significant reductions. Decreases in IRF, SRF and PED were already significant at T1 (p < 0.05). SHRM was significantly reduced at T2 (p < 0.05). Structural biomarkers were absent at T3. At T4, all biomarkers remained stable while SHRM was no longer detectable in 18 patients. Changes in visual acuity from baseline to T4 were not significant.
Conclusion
This short-term experience highlights that brolucizumab might be considered an effective treatment option in nAMD with type 1 MNV, as it can promote a reduction of structural activity biomarkers.
Keywords
Introduction
The main treatment for exudative age-related macular degeneration (AMD) is currently represented by anti-vascular endothelial growth factor (VEGF) agents, injected intravitreally. 1 However, responses after intravitreal injections of anti-VEGF usually show some inter-individual differences. 2 Several studies have reported a lack of anatomical and functional response in about 25%.3,4 In particular, patients could be defined as “non-responders” if intraretinal (IRF) and/or subretinal (SRF) fluid persisted and/or increased, central macular thickness (CMT) increased and best-corrected visual acuity (BCVA) worsened by more than five letters. 2
In this scenario, brolucizumab, a new anti-VEGF drug recently approved by the FDA for the treatment of exudative AMD, may play a constructive role in non-responder (nAMD) patients. 5 This new humanized monoclonal single-chain Fv antibody fragment links the isoforms of VEGF-A preventing them binding VEGF receptor-1 and −2. The Phase 3 HAWK and HARRIER trials showed sustained, longer dryness in patients treated with brolucizumab compared to those receiving aflibercept, with fewer injections required. 5
Nowadays multimodal imaging plays a pivotal role in the management of retinal diseases. Optical coherence tomography (OCT) is now the most widely applied method for analyzing the progression and activity of AMD. Providing detailed in-vivo images of retinal and choroidal structures, OCT can identify characteristic activity biomarkers of MNV such as IRF, SRF and subretinal hyper-reflective material (SHRM).6–9 These biomarkers reflect the activity of neovascular AMD, and help the physician decide the optimal management.10,11
The aim of this study was to analyze structural and clinical changes in six months, focusing on the retinal activity biomarkers IRF, SRF and SHRM with OCT after brolucizumab injections in type 1 MNV nAMD patients, already treated unsatisfactorily with other anti-VEGF agents. The secondary endpoint was to evaluate changes in BCVA during the treatment.
Materials and methods
Study population
In this prospective observational study, we recruited non-responder patients with type 1 MNV in nAMD, previously treated with several anti-VEGF agents and then switched to brolucizumab. All patients were examined at the Melegnano Hospital Department of Ophthalmology between September 2021 and April 2022. In the same clinic they received brolucizumab 6 mg/0.05 ml (Beovu, Novartis, Basel, Switzerland) intravitreal injections on a pro re nata (PNR) anti-VEGF regimen, in the surgery room.
All the subjects received a complete ophthalmological examination including BCVA according to the Early Treatment of Diabetic Retinopathy Study (ETDRS), slit-lamp biomicroscopy, fundus examination, fluorescein angiography (FA), indocyanine green angiography (ICGA) (Spectralis, Heidelberg Engineering, Heidelberg, Germany) and spectral-domain (SD)-optical coherence tomography (OCT) (CIRRUSTM HD-OCT, Carl Zeiss Meditec, Inc). Multimodal imaging was useful to establish the type of MNV in all patients and to ensure correct enrollment. Type 1 MNV were detected with FA, observing ill-defined regions of stippled hyperfluorescence, mostly evident in the later phases of the angiography; these correspond topographically to an irregular area of elevated RPE, detected with OCT. ICGA helps in identifying the vascular network of type 1 MNV in the late phases. All the examinations except FA and ICGA were performed at baseline (T0) and repeated one month (T1), three (T2), four (T3) and six months (T4) after treatment.
Inclusion criteria were nAMD patients complicated with type 1 MNV, already treated with other anti-VEGFs. Non-responders had IRF and/or SRF associated with SHRM. Moreover, CMT and BCVA had shown worsening or no improvement before switching to brolucizumab after at least 3 additional consecutive (loading dose) injections of other anti-VEGF agents.
Outcomes measured included OCT structural activity biomarkers such as IRF and SRF variations. Moreover, we collected presence/absence of SHRM and subfoveal fibrovascular PED height variations. Presence of PED alone was not considered as activity sign and consequently patients with PED alone were not included in the inclusion criteria.
We also recorded CMT at the times selected. Clinical function, as BCVA, was measured at baseline and at T1, T2, T3 and T4.
Exclusion criteria were geographic atrophy, type 1 MNV secondary to other causes, retinal angiomatous proliferation (RAP), type 2 MNV, polypoidal vasculopathy, retinal vascular diseases, history of previous uveitis and of severe ocular inflammation, serous and drusenoid PED in OCT scans, absence of SHRM, myopia greater than six diopters, history of ocular surgery, and significant lens opacity.
The study was approved by the Institutional Review Board and was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients.
Assessment of structural activity biomarkers at optical coherence tomography
Spectral domain (SD) OCT images were obtained with the CIRRUSTM HD-OCT, using the 512 × 128 scan pattern (Macular Cube protocol) where a 6 × 6 mm area on the retina was scanned with 128 horizontal lines, each consisting of 512 A-scans per line in a scan time of 2.4 s, as previously described. 12 A 6 × 6 mm macular scan, centered on the fovea, was done to visualize the macular area. Structural activity biomarkers in nAMD patients - SHRM, IRF and SRF – and fibrovascular-PED were assessed examining each horizontal and vertical scan of the Macular Cube protocol.
As reported in the literature, SHRM was considered an OCT activity sign in exudative AMD and was qualitatively assessed as present or absent. 13
Two expert retinologists, in consensus, record the presence or absence of SRF in the subfoveal and extrafoveal retina in all the b-scans. Subfoveal fibrovascular-PED changes were evaluated in all the lines, measuring the highest fibrovascular PED using the in-built measurement tool in the Cirrus software.
We excluded from the analysis any images with a signal strength index less than 60 and residual motion artefacts, incorrect segmentation, low centration or poor focus.
Statistical analysis
Statistical analysis was done with SPSS 21.0 statistical software (Chicago, IL, USA). Continuous data were described with mean and standard deviation (SD) or median and range, categorical data as percentages. Friedman test was used to establish difference along the time. Post hoc analysis was done with a paired non-parametric test (Wilcoxon) for continuous data. The McNemar test was used to assess changes in patients’ status over time. A P value <0.05 was considerate significant, and the Bonferroni correction for multiple comparisons was applied.
Results
From September 2021 to April 2022, 22 eyes of 22 patients were treated with brolucizumab in our practice. Two eyes were excluded due to missing follow-up. Thus, 20 eyes of 20 patients (mean age 77.4 ± 8.45; 13 males, 7 females) were enrolled in this prospective study. All twenty patients had a six-months follow-up.
The mean number of previous anti-VEGF treatments was 14.8 ± 6, received by patients in a mean time of 26 months. All eyes had already been treated with other anti-VEGF: 8 eyes with bevacizumab (40%), 6 with both aflibercept and bevacizumab (30%), 3 with both ranibizumab and bevacizumab (15%), and 3 with aflibercept (15%) on a PRN regimen (Table 1).
Demographic and clinical characteristics of patients enrolled at baseline.

Data are expressed as mean ± standard deviation.
Patients received an average of two intravitreal brolucizumab injections (range 1–4) with a mean interval of five weeks. All patients completed the six-months follow-up. No side effects were detected during treatment.
At T0, 20/20 eyes showed SHRM, 14/20 presented IRF, 17/20 had persistent SRF; 17 (85%) had persistent fibrovascular PED at baseline with a mean height of 203.45 µm (highest 515 µm; lowest 66 µm). Median CMT was 313.5 µm at baseline (200 µm lowest, 493 µm highest).
All variables showed significant reductions (p < 0.05) during the six-month follow-up (Figure 1 and Figure 2) (Table 2). The decrease was already significant after the first month (T1) for IRF (p = 0.001), SRF (p = 0.0001) and fibrovascular-PED (p = 0.0017) but was not evident for SHRM (p = 0.07).
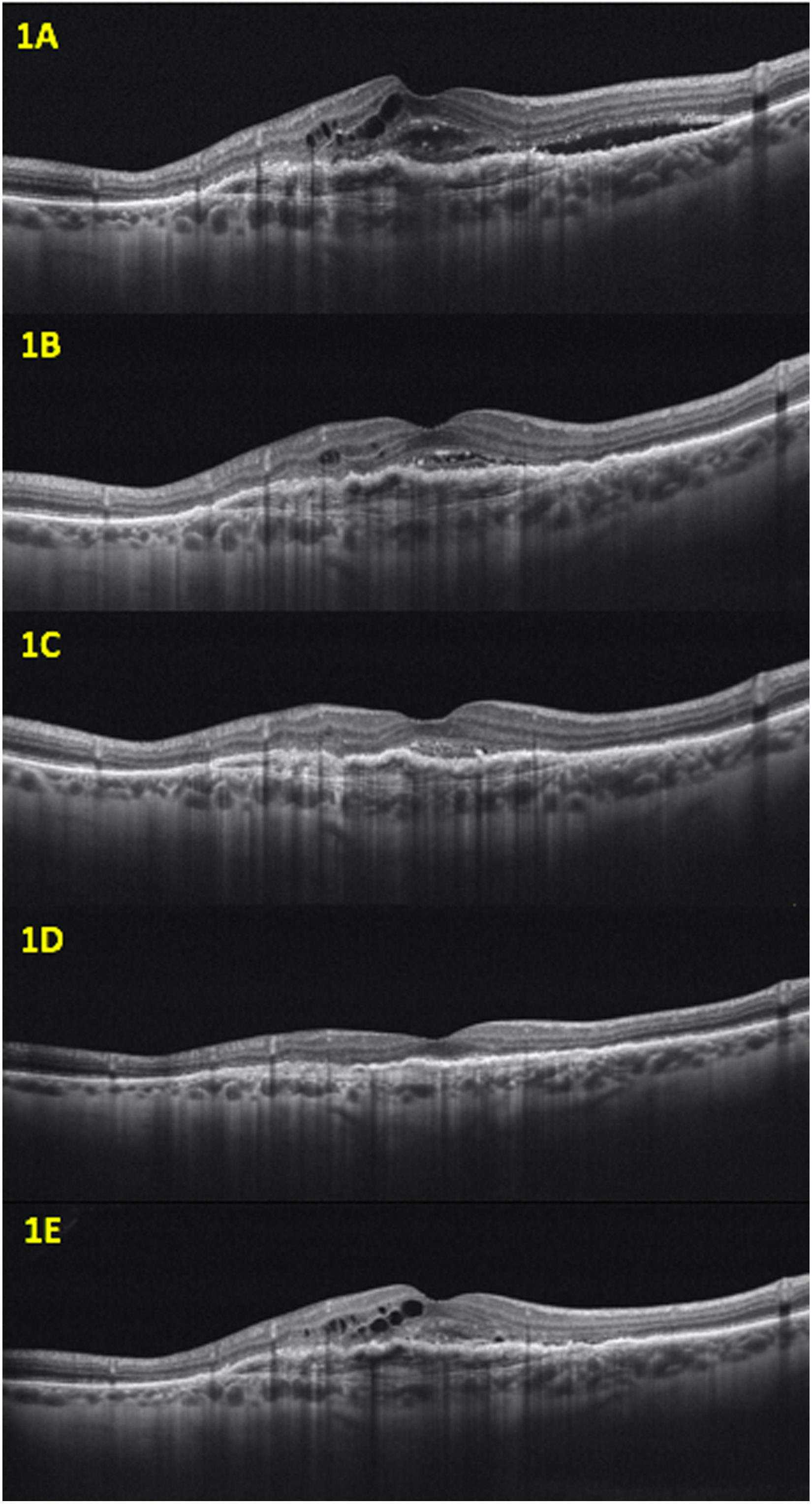
Right eye of 73-year-old non-responder age-related macular degeneration (nAMD) man: optical coherence tomography (OCT) showed intraretinal (IRF) and subretinal fluid (SRF) associated with subretinal hyperreflective material (SHRM) and fibrovascular pigment epithelium detachment (PED) in macular region at baseline (T0) (1A). At T1 there was absence of SRF and decrease in IRF and SHRM. PED height remains stable (1B). After two intravitreal injection, OCT scan showed neither IR nor SRF at T2. SHRM and PED height were reduced (1C). No activity biomarkers were observed at T3. PED height decreased compared to baseline (1D). At T4 PED height increased associated with recurrence of IRF, SRF and SHRM.
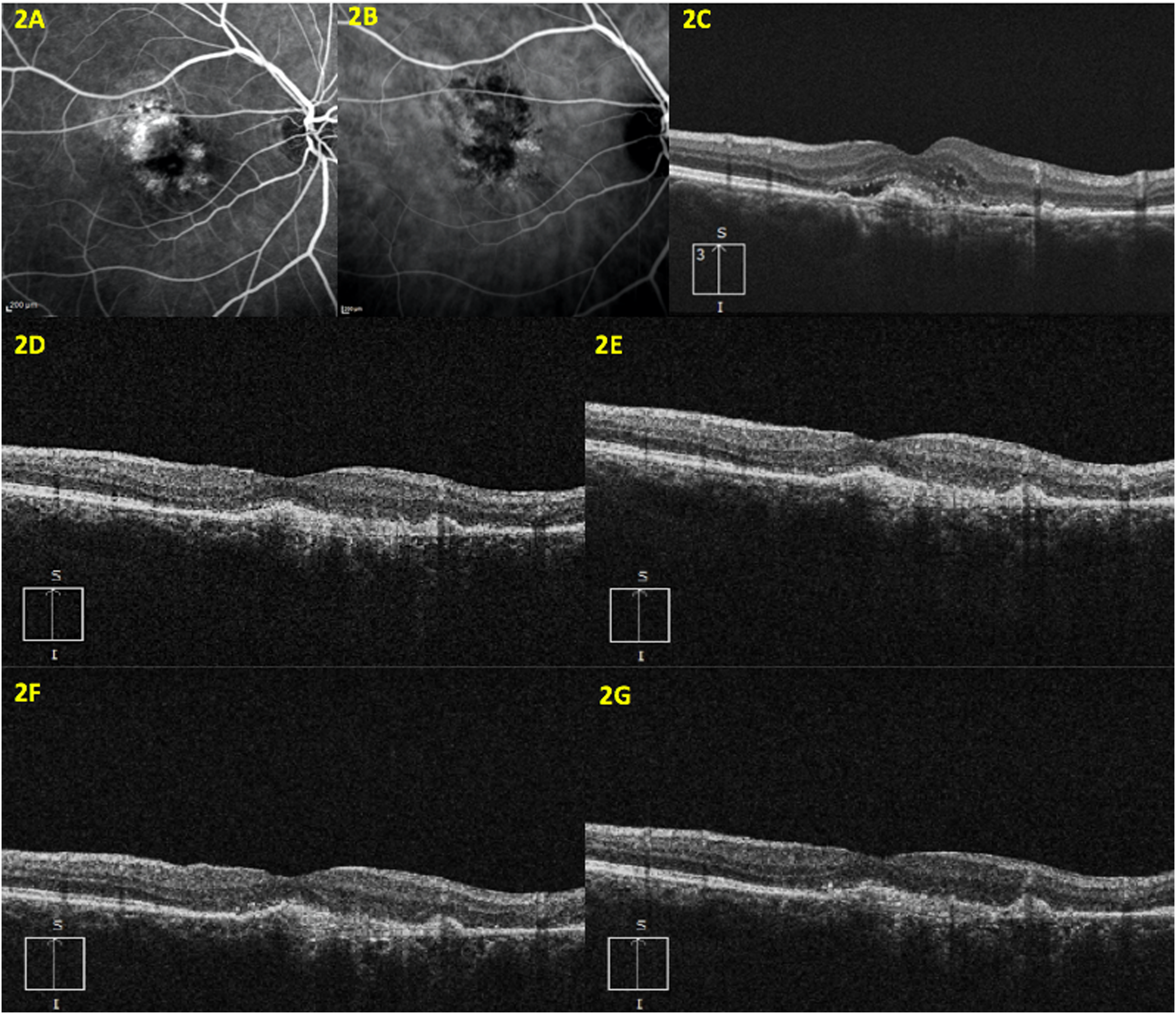
Right eye of 81-year-old nAMD woman: fluorescein and indocyanine angiography showed type 1 macular neovascularization in a nAMD patient (2A, 2B). OCT performed at baseline (T0) detected SHRM, IRF, SRF and fibrovascular pigment epithelium detachment (PED) (2C). One month after the first Brolucizumab injection (T1), no activity biomarkers were present (2D). Similarly, at T2, T3 and T4 there were no SHRM, IRF and SRF (2E-2G).
Structural biomarkers variations and visual acuity changes over the six months follow-up.

SHRM: subretinal hyperreflective material; SRF: subretinal fluid; IRF: intraretinal fluid; PED: pigment epithelium detachment; CMT: central macular thickness. * data expressed as median (range).
At T1, SRF and IRF was absent in 17 eyes (85%), with no SHRM in 10 patients (50%). Out of 17 eyes with subfoveal PED, this biomarker decreased in 13 cases (76%), remained unchanged in 2 (12%) and increased in other 2 patients (12%). Median CMT was 246.5 µm at T1 (range 177–359 µm). Ten patients had SHRM and two of them also had SRF and IRF. One patient had only SRF. So, they received the second dose.
At T2 SHRM was significantly reduced (p = 0.039), while the other variables showed no significant reduction between T1 and T2. The median height of fibrovascular PED was 149 µm (range 47- 402 µm). One patient had SHRM, SRF and IRF while three patients presented only SHRM and one had SRF and IRF with no SHRM at this time. As they presented persistent signs of activity these five patients received a further intravitreal injection of brolucizumab in a PNR regimen. Median CMT was 228 µm at T2 (range 156–367 µm).
At T3 none of the patients showed any signs of activity. Median CMT was 230 µm (159 µm lowest, 370 µm highest). No patient was re-treated.
At T4, SRF and IRF remained stable while the decrease in PED persisted in 9 eyes (45%). SHRM was absent in 18 patients (90%). Median CMT was 232 µm (174 µm lowest, 398 µm highest). Four patients were re-treated because they showed persistent signs of activity: two presented SRF + IRF + SHRM, one had SRF + IRF, another had only SRF).
CMT showed substantial decreases over time (p < 0.05). It was significant at T1 (p = 0.0001), but the reduction from T1 to T4 was not significant (p > 0.05).
At T0 logMAR BCVA was 0.1 (20/200 Snellen) in 12 patients, 0.25 (20/80 Snellen) and 0.32 (20/63 Snellen) respectively in 6 and 2 eyes. At T1 and T2 12 out of 20 eyes showed visual acuity of 0.1 logMAR, 7 out of 20 presented 0.25 logMAR and only one eye had 0.32 logMAR. At T3 logMAR BCVA was 0.1 in 13 eyes, 0.25 in six eyes and 0.32 in one eye. At T4 14 eyes showed BCVA of 0.1 logMAR, while 4 and 2 eyes had respectively 0.25 logMAR and 0.32 logMAR. The differences in vision between T0, T1, T2, T3 and T4 were not significant (p = 0.78).
Discussion
By 2040 the number of patients with AMD is predicted to be around 288 million. 14 In the light of this growing incidence and prevalence, in the last decade there has been increasing interest in biological molecules such as VEGF inhibitors. These drugs are currently the treatment of choice for exudative AMD, as they limit the activity of the disease, slowing its progression and stabilizing the visual acuity. 15
Several trials have been described in the last twenty years, indicating the efficacy of different anti-VEGF agents such as bevacizumab, ranibizumab and aflibercept in reducing the retinal fluid and therefore limiting the development of the MNV.16–18 However, it should be considered that in clinical practice patients often do not show complete fluid resolution, with a plummet in visual acuity and persistence of activity biomarkers. Furthermore, the response may show several ups and downs after intravitreal injections, usually due to inter-individual differences and the limited half-life of the antiVEGF. 2 Several studies have reported a lack of anatomical and functional response after intravitreal injections in about 25% of exudative AMD patients.3,4 Heier et al. found that approximately 30% of patients receiving intravitreal Aflibercept injections every 8 weeks still had persistent IRF and/or SRF 52 weeks after the first dose; these are the “non-responders”. 18
Intraretinal fluid is the most important negative prognostic biomarker in AMD and is strongly correlated with a higher risk of vision loss.7,10 In contrast, SRF, described as fluid between the neuroretina and inner border of the retinal pigment epithelium (RPE), is often associated with better visual outcomes, regardless of the suitability of the intravitreal injection.10,11 Amoaku et al. in 2015 recommended switching to another type of anti-VEGF in case of persistent or increasing IRF and SRF, increases in CMT, and BCVA worsening by more than five letters. 2 In line with these criteria, in our study we analyzed non-responder patients, adding the persistence of fibrovascular PED and SHRM changes as outcomes to be analyzed.
PED, defined as separation between the RPE and Bruch's membrane, are classified as fibrovascular, drusenoid, and serous. Fibrovascular PED, detected at OCT as hyper-reflective content with an irregular lifting edge, is believed to be associated with occult choroidal neovascularization.7,19 Thus, though its pathogenesis is still incompletely unknown, its persistence in the OCT scan, associated with IRF, SRF and SHRM, is believed to be signs of potential active disease. 20
SHRM, detected by OCT as hyper-reflective material located externally to the neuroretina and internally to the RPE, serves as a biomarker of activity for MNV. 9 This biomarker is associated with worse BCVA than in eyes without it. 9 The rapid loss of thickness of the SHRM is believed to be the consequence of fluid reduction induced by the VEGF inhibitors. 9 However, this activity biomarker can persist despite treatment, constituting a risk factor for the development of a scar. 21
Nowadays, aiming to understand the pathogenesis of exudative AMD better, the spotlights tend to be focused on active biomarkers of disease detected by OCT and OCT angiography.22–24
Brolucizumab, recently approved by the FDA, may play a vital role in AMD non-responders. 5 To the best of our knowledge this is the first prospective study analyzing SHRM and other structural activity biomarkers in type 1 nAMD after brolucizumab injections. We observed significant declines over time for all the structural activity biomarkers examined. IRF, SRF and fibrovascular-PED were significantly reduced one month after the first injection. SHRM was clearly reduced at T2. Interestingly, six months after the first dose of brolucizumab, SHRM was not present in 18 out of 20 eyes, showing a slower response than the other variables. It has been reported that after several anti-VEGF injections the fluid component of SHRM may decrease while the fibrotic one increases, rendering the therapy less effective in eliminating or reducing SHRM.25,26 Anti-VEGF inhibitors do favor remodeling of the MNV complex towards an organized tissue, with an increase in hyper-reflective material. 27 As the molecular size of brolucizumab is 26kDa, it is smaller than the other anti-VEGF agents, so better tissue penetration and a higher dose at each injection might help explain our findings. Hypothetically, SHRM may change the percentages of its components, increasing the fibrotic one and then declining itself, with the advantage of brolucizumab longer duration of action than ranibizumab, aflibercept and bevacizumab.
The reduction of fibrovascular PED in our study was significant at T1 and remained stable in the subsequent timepoints. Similar findings were described after only one brolucizumab injection by Rispoli et al. 28 Those authors analyzed 12 eyes of 12 non-responders, recording the PED-horizontal maximum diameter (PED-HMD) and PED-maximum high (PED-MH) with OCT. Only PED-MH showed a significant reduction in the study group during the month after the injection. 28 We can confirm their findings, narrowing the field of study to persistent fibrovascular PED and extending the observation period to six months.
We also noted a significant reduction of CMT. Comparable results were described by Ota et al, after switching from aflibercept to brolucizumab for refractive neovascular AMD. 29 They reported that the mean CMT decreased significantly, by 57.0 µm one month after the switch, in agreement with data already published. 29 Here we confirmed the results described by Hussain RM et al who, reporting their findings after brolucizumab injection in nAMD patients, saw a larger reduction in CMT in eyes previously treated with bevacizumab than those treated with aflibercept. 30 However, in contrast with our results, during the second and third visits after the injection the authors reported a slight increase in CMT, which they explained as a consequence of the treat-and-extend (T&E) regimen employed in their study.
The present study found no significant differences in BCVA after switching from some other anti-VEGF to brolucizumab, as reported in the literature and similarly to what has been described after the switch from ranibizumab to aflibercept.28–30
In the phase 3 Hawk and Harrier trials naïve AMD patients gained a mean of 4 letters at 4 weeks after the injection. 5 However, our findings might be the consequences of the pre-existing photoreceptor damage caused by the chronic presence of retinal fluid. 30
The major limitations of this study were the limited sample size and the short follow-up. Moreover, this is a pilot investigation based on early changes, therefore, long-term experience is necessary to boost the value of this new anti-VEGF for treatment of non-responder AMD patients.
In conclusion, brolucizumab showed good pharmacological efficacy and reduced all the structural activity biomarkers. IRF and SRF reabsorption and reduction of fibrovascular PED were already evident after the first dose and remained stable at six months. The reduction of SHRM became significant later, three months after the treatment, and remained stable at six months. Thus, brolucizumab may offer an effective treatment option, promoting the reduction of the biomarkers of structural activity in nAMD complicated with type 1 MNV.
Footnotes
Acknowledgement
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent for publication
Written informed consent was obtained from all the patients enrolled for the preparation of this work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
