Abstract
Purpose
Herein we describe the change in the amount of macular oedema in one eye after contralateral intravitreal brolucizumab injections in a patient with neovascular age-related macular degeneration (nAMD).
Case Report
A patient with bilateral nAMD underwent intravitreal bevacizumab injections in both eyes with little improvement in the best-corrected visual acuity (BCVA) and central macular exudation. The treatment was switched to aflibercept, but there was incomplete drying of the macula in both eyes. After uneventful cataract extraction, the central macular thickness (CMT) increased markedly in the operated left eye (LE), which was unresponsive to subtenon triamcinolone and further intravitreal aflibercept. Cataract surgery was also performed in the right eye (RE) combined with an intravitreal sustained-release dexamethasone implant. Nevertheless, the CMT increased. Intravitreal brolucizumab injections were performed in the RE with almost complete disappearance of the oedema in the treated eye. Concurrently, the contralateral uninjected eye showed a remarkable decrease in CMT. Five months after the first brolucizumab injection, the macular exudation increased again in both eyes. A second brolucizumab injection was performed in the RE only, and was followed by a prompt reduction in CMT in both the injected RE and uninjected LE.
Conclusions
Although contralateral retinal changes have been described for many other vascular endothelial growth factor inhibitors, there is little evidence for brolucizumab. We describe a repeated dose- and time-related effect on the uninjected eye in a case of nAMD.
Keywords
Introduction
The use of anti-vascular endothelial growth factor (VEGF) inhibitors has become the standard treatment for eyes affected by exudative macular disorders, such as neovascular age-related macular degeneration (nAMD) and diabetic macular oedema (DME).1,2 Several anti-VEGF drugs are currently available in Europe to slow and eventually stop the progression of these pathologies. Bevacizumab is a recombinant humanised monoclonal IgG1 antibody that binds to and inhibits VEGF; ranibizumab (Lucentis®; Genentech, San Francisco, CA, USA) is a recombinant humanised IgG1 monoclonal antibody fragment that binds to and inhibits VEGF-A; aflibercept (Eylea®; Regeneron, Tarrytown, NY, USA) is a soluble decoy receptor that binds VEGF-A, VEGF-B, and placental growth factor (PIGF) with greater affinity than the body's native receptors; and brolucizumab (Beovu®; Novartis, Basel, Switzerland) is a humanised monoclonal single-chain variable fragment (scFv) that binds and inhibits VEGF-A. Brolucizumab is the latest anti-VEGF drug to be approved by the European Medicines Agency for DME and nAMD.
Various studies have reported bilateral retinal changes following unilateral injections of bevacizumab, ranibizumab, or aflibercept.3–6 Only one report describes the occurrence of changes in both eyes after unilateral brolucizumab injection. 7
Here, we present a patient with bilateral nAMD in whom we repeatedly documented bilateral improvement in macular oedema after brolucizumab injections in a single eye.
Case report
An 85-year-old Caucasian male was referred to our ophthalmology clinic (University Hospital of Parma, Parma, Italy) to start intravitreal anti-VEGF treatment for naïve bilateral nAMD. At first evaluation, the best-corrected visual acuity (BCVA) tested with an Early Treatment Diabetic Retinopathy Study chart at 4 m was 0.4 logMAR in the right eye (RE) and 0.3 logMAR in the left eye (LE). Examination of the anterior segment noted mild nuclear cataracts in both eyes, while the presence of active nAMD was confirmed by mydriatic fundoscopy, spectral domain optical coherence tomography (SD-OCT), and fluorescein and indocyanine angiography. The patient underwent a standard pro re nata treatment protocol, which consisted of four bevacizumab injections in the RE and six in the LE over the following 6 months.
Since the BCVA did not improve significantly, with only a moderate reduction in the macular exudation, we changed the treatment to ranibizumab, but after a loading dose of 1 intravitreal injection per month for 3 months we observed a limited response also to this new treatment; therefore, the decision to switch to aflibercept. One bilateral administration of aflibercept stabilised the amount and extent of the intraretinal fluid, but did not dry the macula in either eye. During follow-up, the cataract in the LE became severe and phacoemulsification with a monofocal intraocular lens implant was performed uneventfully. One month postoperatively, the patient's macular oedema in the operated eye increased acutely, which we attribute to Irvine–Gass syndrome (BCVA 0.5 logMAR). Three triamcinolone acetonide subtenon injections were performed monthly in the LE, combined with an additional bilateral aflibercept injection to control the underlying nAMD activity in both eyes. Despite this therapy, the BCVA of the operated eye fell further to 1.1 logMAR.
Twelve months after the surgery in the LE, the RE developed severe lens opacification with a BCVA of 0.8 logMAR. The patient requested cataract extraction knowing that the macular oedema could increase significantly in this eye. Uneventful cataract surgery was combined with a concomitant sustained-release dexamethasone intravitreal implant, but the macular oedema increased, although it was less severe than that previously recorded in the LE.
Six months after this second cataract surgery, when the effect of the intravitreal dexamethasone implant should have disappeared, the BCVA of the RE was 0.7 logMAR, with active signs of nAMD in the macula (Figure 1a). The RE central macular thickness (CMT) was 640 μm, while the BCVA in the LE was stable at 1.1 logMAR, with a CMT of 739 μm. Two brolucizumab intravitreal injections were performed 8 weeks apart in the RE. One month after the second injection, no adverse reaction was recorded. In the treated eye, the CMT decreased to 177 μm and the BCVA improved to 0.5 logMAR. Surprisingly, the patient also reported subjective functional improvement in the LE, with better contrast perception and a broader visual field. Although the BCVA had not changed (1.1 logMAR), SD-OCT showed a notable reduction in the CMT to 228 μm in this uninjected eye (Figure 1b). We chose not to complete the standard protocol because we feared just the possible inflammatory side effects. Dealing with some Irvine–Gass syndrome manifestations, we limited the administrations to the first clinical response.
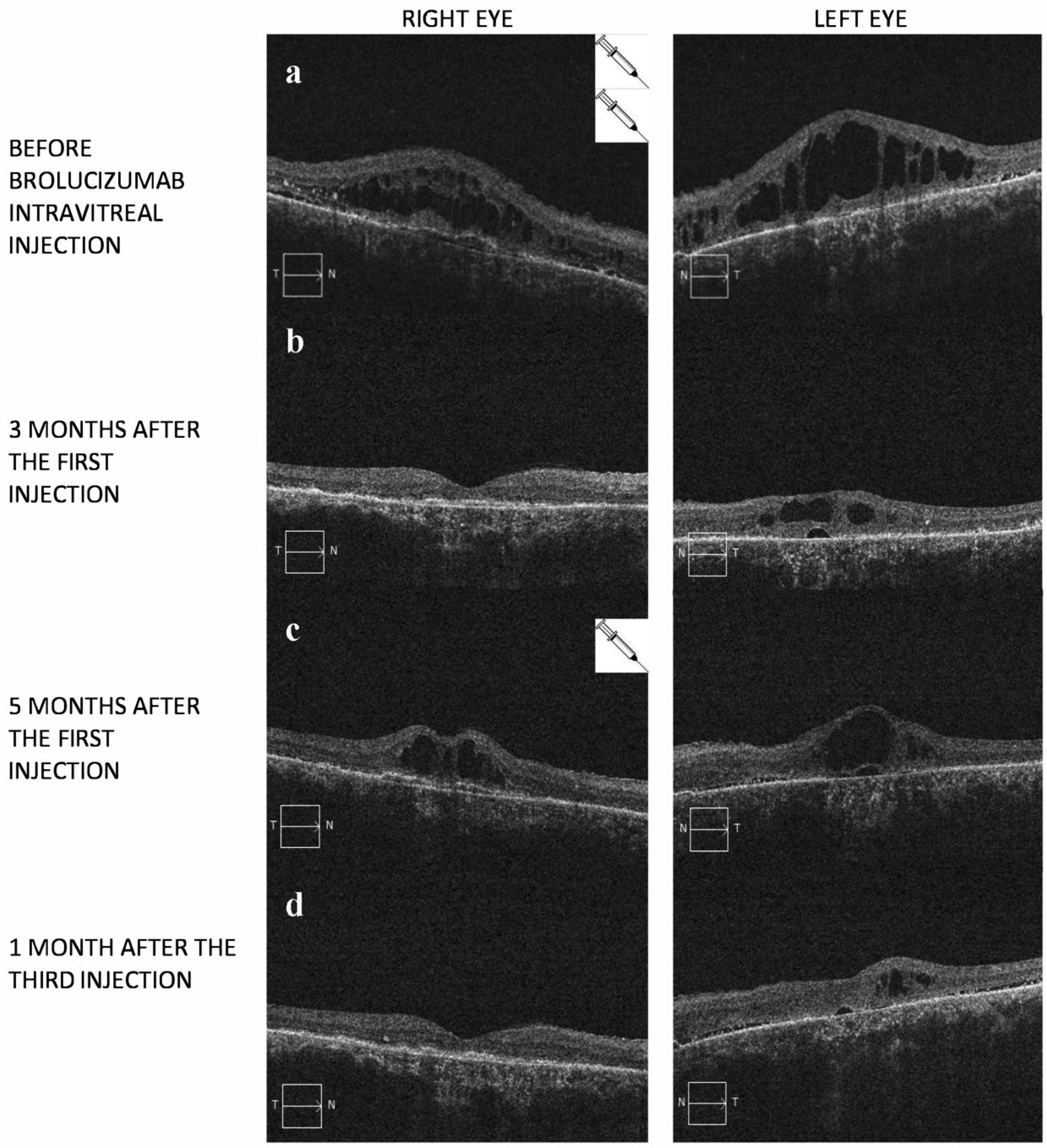
(a-d): composite of optical coherence tomography (OCT) findings showing the macular aspect of the right eye (left column) and left eye (right column) of patient during the follow up. a) Before the Brolucizumab intravitreal injections in the right eye an important macular edema is evident in both eyes. b): the OCT scans taken 3 months after the first Brolucizumab injection in the right eye report an important reduction of the macular thickness in both treated and untreated eyes. 5 months after the treatment, the macular thickness increased again, as showed in c), but again a third Brolucizumab intravitreal injection in the right eye was followed by a bilateral improvement, as reported in d).
Five months after the first brolucizumab injection, the intraretinal fluid increased in the RE and in the untreated LE (Figure 1c). A third injection was performed in the RE. One month after this injection, the CMT decreased in both the treated (from 390 to 176 μm) and untreated (from 429 to 313 μm) eyes, as shown in Figure 1d. At the end of follow-up, the BCVA was 0.4 logMAR in the RE and 1.1 logMAR in the LE.
Discussion
Bilateral effects following unilateral anti-VEGF injections of bevacizumab, ranibizumab, and aflibercept have been reported in patients with macular oedema.3–6 Although the exact mechanism is unclear, the most generally accepted hypothesis is the “molecular escape” of anti-VEGF via the systemic bloodstream. In 2014, Avery et al. 8 demonstrated the influence of the molecular size of anti-VEGF on this. Since brolucizumab was not available at that time, it was not evaluated. More recently, Chakraborty et al. 7 reported a bilateral effect after unilateral brolucizumab administration in a case of DME. According to the above-mentioned hypothesis, the low molecular weight of brolucizumab compared with the other anti-VEGFs (i.e., brolucizumab 26 kDa, ranibizumab 48 kDa, aflibercept 110 kDa, and bevacizumab 149 kDa) supports its eased passage through the haemato–ocular barrier to the other eye. Chakraborty et al. 7 addressed the use of intravitreal anti-VEGF to treat local complications of a systemic metabolic disease, such as diabetes. An improvement in some extraocular conditions, such as the general control of the disease due to the natural history of the pathology, the patient's diet, and ongoing hypoglycaemic treatment, might have influenced the status of the contralateral uninjected eye regardless of the effect of the anti-VEGF itself. This contingency does not apply to nAMD, as in our report.
We are unable to ascertain whether the repeated changes in the macular oedema of the contralateral eye in our patient were actually related to the brolucizumab injections in the contralateral eye. However, the repetition of this finding and the time- and dose-dependent relationships with the injections support this hypothesis.
One limitation of our report is that it is based on a single patient with a history of previous different bilateral anti-VEGF injections and a probable post-surgical inflammatory component to the neovascular oedema. This could have altered the haemato–ocular retinal barrier, allowing a greater-than-normal amount of brolucizumab to enter the systemic circulation. Nevertheless, we believe that this evidence is worthy of mention and further investigation.
Footnotes
Acknowledgements:
The English in this document has been checked by at least two professional editors, both native speakers of English. For a certificate, please see: http://www.textcheck.com/certificate/Jk0X56
Availability of data and material
all data and material are available from the corresponding author.
Consent for publication
The Local Ethical Committee “Area Vasta Emilia Nord (AVEN)” does not require official approval for the publication of single case reports. Written informed consent to publish information and images included in this report was obtained from the patient.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
