Abstract
Opacification of hydrophilic intraocular lenses (IOLs) is an uncommon complication, that can appear after uneventful cataract surgery. We report a case of opacified Hydroview® IOL in a 76-year-old woman, with a previous history of pars plana vitrectomy with silicon oil tamponade in her right eye for a proliferative diabetic retinopathy, who developed an opacification of the IOL after over two years from a silicon oil/BSS exchange in combination with an uneventful phacoemulsification. The patient complained about a progressive decrease in her visual acuity. The slit-lamp examination confirmed the opacification of the IOL. Therefore, because of blurred vision, a combined procedure of explant and exchange of the IOL was performed in the same eye. Qualitative (Optic microscope, X-Ray powder Diffraction (XRD), Scanning Electron Microscopy (SEM)) and quantitative (Instrumental Neutron Activation Analysis (INAA)) analysis of the IOL material were performed. Here, our aim is to report the acquired data of the explanted Hydroview® H60M IOL.
Keywords
Introduction
Since the 1990s, postoperative optic opacification of different hydrophilic intraocular lenses (IOLs) has been reported and continuous ameliorations in IOL manufacturing and materials have minimized incidence of hydrophilic IOL opacification.1,2 Several causes may induce clinically significant IOL opacification, which leads to IOL explantation in most cases. 3 Although it is rare, intraocular lens opacification has been observed when using biomaterials, such as hydrophilic acrylic, hydrophobic acrylic, and silicone. This process is usually attributed to specific defects of IOL materials, use of adjuvants, surgical technique, other associated medical conditions, or a combination of these.4–7 Primary opacification has been observed in four different IOL brands: Hydroview® (Bausch and Lomb, Rochester, N.Y., USA), Memorylens (Ciba Vision, Duluth, Ga., USA), SC60B-OUV (Medical Developmental Research, Clearwater, Fla., USA) and Aqua-Sense (Ophthalmic Innovations International, Ontario, Calif., USA).8–9 Conversely, secondary opacification has been described in association with air/gas injection during Descemet's stripping endothelial keratoplasty or with surgical procedure for Descemets’ membrane detachment after cataract surgery.10–13
The Hydroview® IOL model H60M is a hydrophilic one-piece lens with a 6 mm optic and two C–loop haptics. The optic was made from a copolymer of 2–hydroxyethylmethacrylate and 6–hydroxyhexylmethacrylate plus a small quantity of 1,6–hexanediol and an ultraviolet blocker. 14 The haptics were made from a polymethylmethacrylate (PMMA) cross linked with ethylene glycol dimethacrylate and coloured blue. 2 The percentage of water is 18%, and the refractive index is 1.474. H60M lenses were designed to be folded and implanted into the capsular bag through a 3.5–4 mm corneal incision. 15 To reduce the risk of opacification, the manufacturer developed silicone-free, sure-hold packaging for the Hydroview IOL. 16
To our knowledge, while several studies have reported late post-operative calcification, especially in hydrophilic acrylic IOLs,17–19 to date, there is no available literature reporting clinical, ultrastructural and chemical analysis with a quantification of opacified Hydroview® IOL material.
Our aim is to present qualitative and quantitative analysis performed on a Hydroview® H60M opacified IOL, explanted from a diabetic patient with previous history of pars plana vitrectomy (PPV) with silicon oil tamponade for proliferative diabetic retinopathy. The lens was removed two years after cataract surgery.
Case presentation
A 76-year-old woman was referred to our hospital in February 2002 with a history of blurred vision in her right eye. She had been affected by diabetes type II since 1978. In July 1999, due to proliferative diabetic retinopathy, she underwent right eye PPV with silicon oil tamponade in another hospital. In November 1999, a silicon oil/BSS exchange in combination with an uneventful phacoemulsification was performed in the same eye. The Hydroview® HM60 IOL was implanted in the capsular bag (power + 24.5) and the Viscoat® was the ophthalmic viscosurgical device used during that procedure. Postoperative medication included Diclofenac 0.1% eyedrops (Voltaren Ofta) and Ofloxacin 0.3% eyedrops (Exocin), 1 drop 4 times daily for 4 weeks. She did not report any other pathologies or risk factors.
She started complaining a progressive significant visual acuity decrease in her right eye during late postoperative period. Best corrected visual acuity (BCVA) was 20/640 on Snellen Chart with −1.25 sfere −2 cylinder axis15°.
A slit-lamp examination of the right eye showed anterior fibrosis of the rexis associated with a thin membrane placed on the anterior IOL surface and a wide opacification of IOL with a grey-whitish haze involving the entire optic. This made fundus examination difficult to perform (Figure 1). No tyndall was noticed in the anterior chamber. Intraocular pressure (Goldmann tonometry) was 16 mmHg in the study eye.
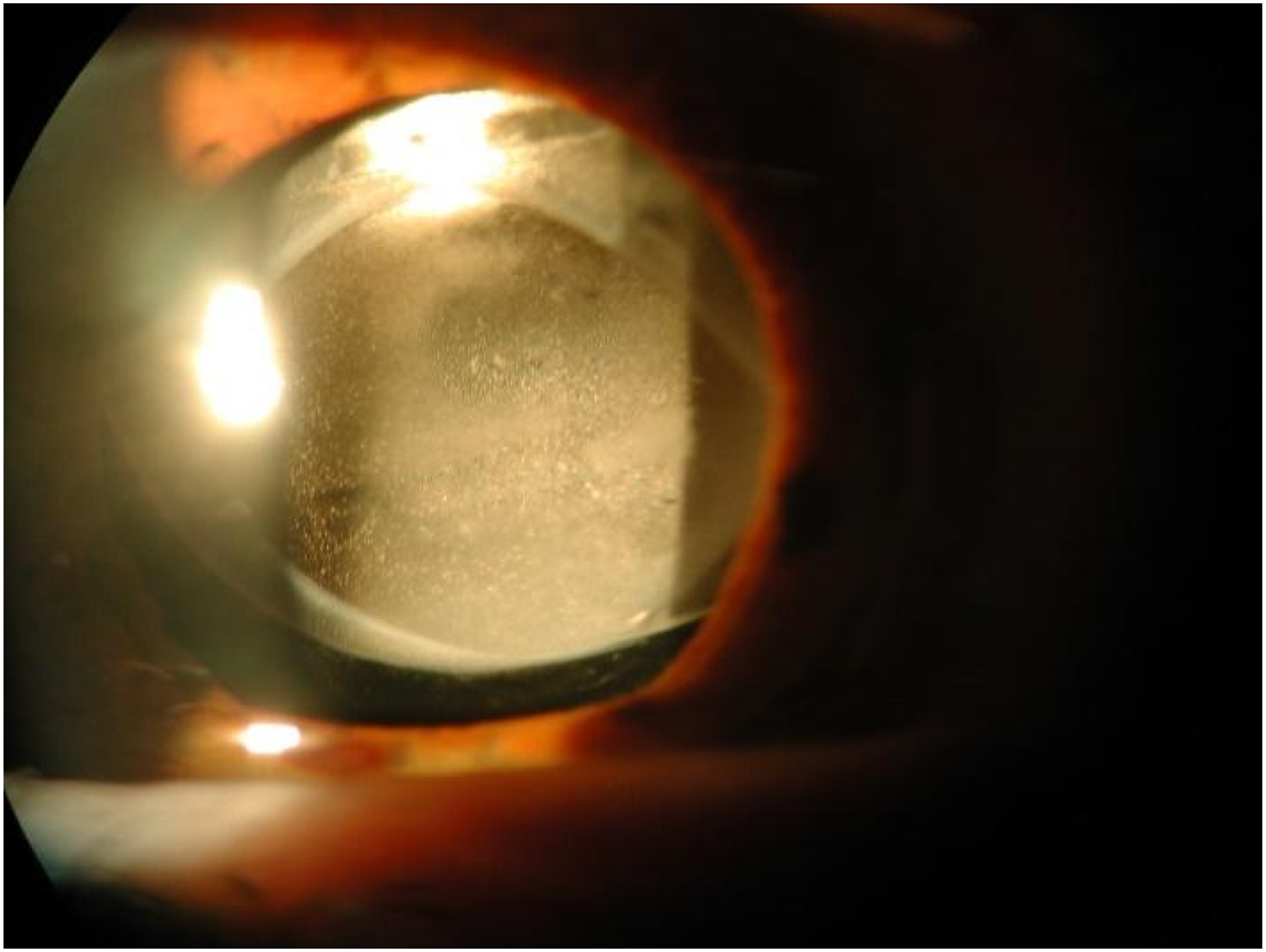
Slit lamp photograph shows anterior fibrosis of the right eye capsular bag, with a grey-whitish haze involving the entire optic with deposits inside both surfaces of the IOL.
At that time, a neodymium:yttrium-aluminum-garnet (Nd:YAG) laser (VISULAS® YAG III, Carl Zeiss Meditec, USA) posterior capsulotomy was performed, with no increase in BCVA. Therefore, we decided to remove the opacified IOL through a corneal incision and to replace it. It was done approximately two and half years after the original surgical procedure. A local anesthesia (standard retrobulbar injection) was performed. We used a 25-gauge needle to separate the lens from the capsule bag. After that, viscoelastic was injected between the IOL and the posterior capsule to pull the IOL out of the capsular bag through a viscodissection. Because of adherence between them, it was necessary to cut the haptics before removal. The lens was brought in the anterior chamber and removed through a 6.0 mm corneal incision. A three pieces IOL, Sensar AR40 [Abbott Medical Optics, Inc. (AMO)], was put in the sulcus. The corneal wound was then sutured with interrupted 10/0 nylon stitches. The patient's vision improved to 20/100 Snellen with +0.75 sfere −1.5 cylinder axis 20 ° on the first post-operative week and continued to be stable during regular follow-up visits. She did not report any adverse events and she referred a better quality of vision.
Examination of the explanted IOL showed a homogeneous, white rounded opacification localized in the centre, saving the optic edges and haptics. The lens was involved in its thickness and not on its surface. After the explantation, the IOL was placed in a sterile container with a balanced salt solution (BSS) and it was stored at a room temperature. After that, it was immediately sent to the laboratories. The analysis of the material performed were qualitative [Optic microscope, X-Ray powder Diffraction (XRD), Scanning Electron Microscopy (SEM)] and quantitative [Instrumental Neutron Activation Analysis (INAA)]. The optic microscope revealed a homogeneous opacification inside the IOL with scattered, small round granules (Figure 2(a)) and paired and linear marks (Figure 2(b)) in the central part of the IOL, probably corresponding to forceps marks. On the contrary, the haptic parts were not affected by granules. The deposits on anterior and posterior surface of the IOL showed a similar distribution.

Optic Microscope images of the opacified IOL. (a) Diffuse opacification of the optic with scattered, small round granules. (b) Part of the IOL with paired and linear marks. Optical scale: 1mm.
The SEM (Figure 3) permitted an overview and revealed diffuse granular deposits of approximately 3 to 5 μm diameter covering the optic surfaces. On the contrary, the lens haptics were not involved. No cellular reaction was reported. The SEM demonstrated these deposits were different in shape and size, unless the majority of them tended to be roundish. Another characteristic noticed was that the lesions were confluent in the central part of the IOL with dense aggregates, but they became thinned out in the area close to the edges.

SEM showing the optical surface of the lens with rounded granules with irregular distribution and different size. Optical scale: 200 µm.
The XRD identified several elements inside the opacified IOL (Figure 4).
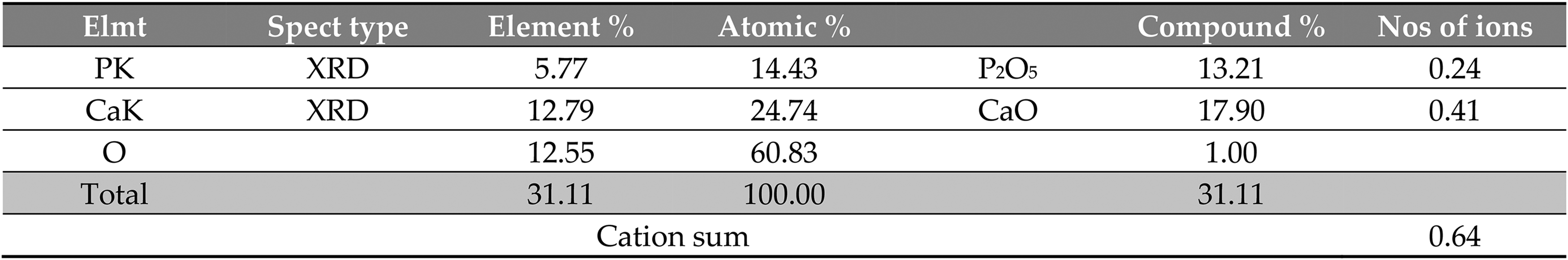
Elements inside the opacified IOL characterized by XRD.
In particular, this analysis showed a significant calcium peak between 3.5 and 4 Kev and phosphorus peak at about 2 Kev from the lens surface deposits (Figure 5). The areas close to the edges, which were not involved by deposits, were also tested as controls. Among these, the XRD showed only peaks of carbon and oxygen.
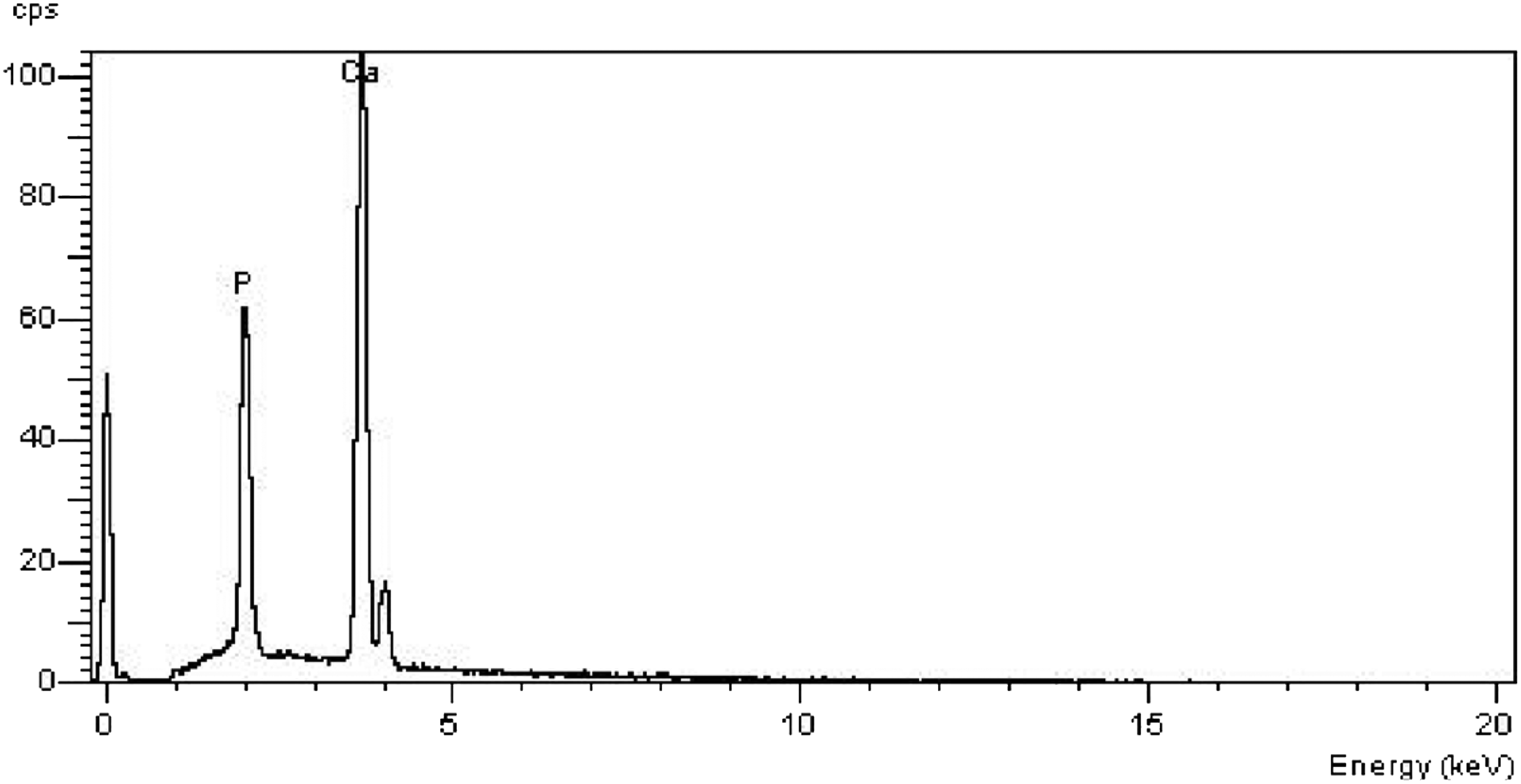
XRD analysis of the deposits shows peaks of calcium and phosphorus.
Finally, the Instrumental Neutron Activation Analysis (INAA) quantified the calcium quantity due to a detection system, an analysis system and an elaboration and output data system. This method showed that in our opacified IOL the calcium quantity was 144 µg.
Therefore, the previous analysis confirmed the deposits on the lens surface were composed of calcium and phosphorus, both of which are characteristically absent in normal IOLs of this type. The INAA quantified the exact amount of Calcium on the IOL.
Discussion
Opacification of IOLs is an uncommon event that can affect visual function. The Hydroview® IOL opacification was firstly described by Chang and co–authors in 1999, and since then several cases of late opacification of hydrophilic IOLs have been reported in the literature.14,20–22 Patients usually become symptomatic during the second year or later, 23 as our case. The opacification of these lenses is resistant to treatment with YAG laser. 24 In fact, with our patient we tried to perform it, but it was unsuccessful.
Optic opacification is described as primary or secondary complication. The first one is due to a problem with the IOL itself without pre-existing diseases, the second one is a consequence of the intraocular environment (for example uveitis) or conditions that disrupt the blood-aqueous barrier. 25 Our patient was diabetic, and she had undergone PPV with silicon oil tamponade before the cataract surgery.
Several studies have shown a higher percentage of associated conditions (e.g., glaucoma and diabetes) in patients with hydrogel IOL opacification.14,26–28 In particular, Yu et al. 26 showed that diabetic patients tended to have worse visual loss caused by opaque Hydroview® IOL. Moreover, authors reported a prevalence of 93% of generalized IOL opacification. We observed that our patient's explanted IOL had a total opacification with localized paired and linear opacifications, corresponding to forceps marks, as suggested from the previous study.
On the contrary, some studies did not find a correlation between opacification and systemic diseases (diabetes, hypertension, autoimmune pathologies), rather they found hypertension to be protective for IOL opacification.24,29 Other authors reported that hydrophilic IOL calcification can occur in traumatized eyes, and it may be associated with a history of gas or other intraocular substances used.13,30,31
Altaie et al. 24 associated the high prevalence of opacified Hydroview® IOL with the use of Viscoat® during the primary cataract surgery in their study-cohort of 103 patients. The proposed mechanism was the facilitation of hydroxyapatitie formation due to Viscoat® high phosphate content. Their hypothesis is also consistent with the area of relative transparency on the optic, left by the forceps, as this part of the IOL will be partially spared to Viscoat® exposure. The same viscoelastic product was used during the cataract surgery of our patient.
Not all the IOL opacifications have an impact on patient vision. An important factor could be the size of the opacities which determines the amount of light reaching the retina. 32 Contrast sensitivity was noted to be reduced in case of opacification 24 ; some patients complained of glare and of mistiness. 29 Other reported symptoms included blurred vision, seeing star shapes around lights, seeing haloes around lights, and colours seeming dull. 32 In fact, our patients complained about a progressive significant decrease in her visual acuity. When vision is affected, IOL exchange is the unique solution. Although it is uneventful in most of the cases, anterior vitrectomy is performed in almost one third of the eyes, and in most of the patients (more than 60%), the new IOL is implanted in the ciliary sulcus. 32 The technique is usually challenging, especially after a long time since the cataract surgery. Often, the haptics must be cut before removal of the IOL, because of the adherence between the IOL and the capsular bag. Several complications are reported in literature, such as zonular dehiscence, rupture of the posterior capsule or corneal decompensation.33–34 In our case, we implanted a three pieces IOL in the sulcus after cutting the haptics of the previous opacified IOL.
Previous studies have investigated the explanted opaque Hydroview® IOLs by x-ray diffraction, optic and electron microscopy. Some authors have reported an opacification due to formation of calcium hydroxyapatite {[Ca3 (PO4)2] 3 Ca (OH)2} on the optic of the implanted IOL.24,26,35 In our case report as well, the microscopic and chemical studies of the explanted IOL are consistent with the findings of the previous literature.
Hydrophilic acrylic IOLs can be inserted through a small incision due to their flexibility, thanks to a high-water level. Although the exact mechanism of IOL opacification is not completely understood, Gartaganis et al. hypothesized that the IOL hydroxyl groups could be ionized when exposed to the physiological aqueous humour (AH) pH. This could lead to the precipitation of calcium phosphate crystals on the IOL surface. 36
Izac et al. 20 proposed that various patterns of calcium precipitation might be caused by differences in the water content of the materials used in the manufacturing process of hydrophilic IOLs. Moreover, regarding the Hydroview® IOL, it has been hypothesized that the lens optic could have been contaminated by the silicone inside the lens packaging. This would have acted as a nidus for calcium precipitation. 37 On 29 July 2003 the manufacturing company itself stated that a change in packaging of the Hydroview® IOL was linked with the opacifications. 37
Additionally, studies on IOL surfaces treated with viscoelastic substances suggested that hydrophobic silicones adsorbed by the IOL surfaces interacted with fatty acids and that this reaction could induce active calcification. 38
The x-ray diffraction performed on the explanted IOL of our patient demonstrated the presence of calcium and phosphates within the deposits. Similarly, previous studies have shown calcium and phosphate as major components.9,2,15,20,39,5 However, X-ray fluorescent spectrum (XRF) has also detected peaks of sodium, magnesium, chloride, fluorine and silicon.2,8,15,5,40–42
Taboada-Esteve et al. 39 analyzed 13 explanted Hydroview® by XRF and they found uncommon elements in the IOL deposits: magnesium, aluminum and iron in high concentration. Zinc, strontium, yttrium and iodine were also detected as minor elements.
According to the literature, the metallic elements could derive from the injectors, forceps or other components used intraoperatively. 41 Another hypothesis was that intraocular inflammatory reaction might have caused an increase in iron. This could be caused by the plasma Fe-binding protein (transferrin) entry into the AH, through the disrupted blood-aqueous barrier (BAB). 39 In case of predisposing conditions, infiltration of proteins and cell aggregates might serve as a substrate for calcification.25,39
According to the analysis by Katsimpris et al., another pathogenetic factor able to enhance the risk of Hydroview® IOL opacification may be the depletion of the ultraviolet light (UV) absorber. 43 This usually causes the disintegration of the polymer and an alteration of its physical properties.43,44 Therefore, mechanical stress during surgery may serve as the initiating factor for a sequence of events where elements such as oxygen, calcium, silicon, iron, and/or UV-related alterations lead to gradual alteration of the polymer network and its opacification. 43
In summary, several risk factors were included in the pathogenesis of IOL opacification, suggesting that the mechanism is complex and may include different pathways. According to previous studies, our case report shows that intraocular inflammation due to previous surgeries, diabetes or some viscoelastic substances, might promote Hydroview® IOL opacification and calcification.
Considering potential complications such as late opacification of Hydrophilic IOLs, this must be considered as an option in selected patients during preoperative screening. Further studies on larger samples and a longer follow-up are needed to clarify this complication and its pathogenesis.
Conclusion
Opacification of hydrophilic IOL is a late postoperative complication of unknown etiology and may be clinically diagnosed using the slit-lamp. This condition may mimic the posterior capsule opacification, but the YAG laser posterior capsulotomy is unsuccessful. Surgical explant is the only solution when the opacification is symptomatic. Clinical pathological correlation, as highlighted in this case report, is an important factor for an accurate diagnosis.
Since the incidence of lens opacification is more frequent among diabetic patients, these should be followed with frequent and accurate visits, especially if a IOL, with documented opacification in literature, has been implanted. On the other hand, the use of these IOLs should be avoided in young patients with known risk factors. Indeed, these eyes are more prone to BAB rupture, with increased postoperative inflammation and secondary calcium deposits formation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for publication of this manuscript.
