Abstract
Purpose:
To assess the efficacy and safety of intracameral mydriatic and anesthetic combination for pupillary dilation in pediatric cataract surgery
Methods:
This prospective series included children <12 years, with visually significant unilateral or bilateral cataracts planned for cataract surgery with/without intraocular lens implantation. At the beginning of surgery, 0.025 ml of a combination of phenylephrine hydrochloride (0.31%), tropicamide (0.02%), and lidocaine hydrochloride (1%) was injected intracamerally. The efficacy of the combination was tested by achieving capsulorhexis and intraocular lens implantation without additional mydriatics.
Results:
We recruited 13 patients (16 eyes) with a mean age of 4.1 ± 3.9 years. The mean pupillary diameter changed increased from 1.92 to 5.68 mm after injection of one unit (0.025 ml) of drug (p < 0.0001). There was a strong positive correlation of the pupillary dilation with axial length (R = 0.86) and horizontal corneal diameter (R = 0.81). Seventy-five percent patients had a pupillary diameter >6 mm and surgery could be completed successfully in all cases without additional mydriatics. In all cases, pupil dilated as the surgery progressed. No adverse event to the drug was noted.
Conclusions:
Intracameral mydriatic-anesthetic combination is an effective and safe way to obtain stable mydriasis in pediatric cataract surgery.
Keywords
Introduction
The success of pediatric cataract surgery largely depends on the adequacy of intraoperative pupillary dilatation and stable mydriasis. Topical dilators used before cataract surgery are not weight-adjusted and may be absorbed to a substantial degree with the potential to cause serious systemic side effects.1–5 Also, the mydriatic effect has been shown to wane off leading to intraoperative miosis, adding to the complexity of surgery.6–8
Intracameral combination of anesthetic and mydriatic has been used in adult cataract surgery with good success and safety since a few years.7–12 The objective of the present study was to evaluate the efficacy and safety of intracameral mydriatic, anesthetic combination of phenylephrine (0.31%), tropicamide (0.02%), and lidocaine (1%) (Phenocaine Plus 1 ml, Entod Pharma, Gujarat, India) in dilating pupil during pediatric cataract surgery.
Methods
This pilot study enrolled pediatric patients scheduled for cataract surgery where intracameral combination of mydriatic anesthetic was planned to be used. It was carried out in the pediatric ophthalmology unit of the eye department at the Post Graduate Institute of Medical Education and Research, Chandigarh, India. Institutional ethical board approval was obtained, and the study conferred to the Declaration of Helsinki. After informed consent from parents, consecutive patients <12 years, with visually significant unilateral or bilateral cataract planned for cataract surgery with/without intraocular lens implantation were prospectively enrolled for the study. Patients in whom the pupil dilated with topical drops during clinic examination were only included. Those with known hypersensitivity to the active substances (tropicamide, phenylephrine hydrochloride, and lidocaine hydrochloride) or experiencing an allergic reaction to topical formulation of these drops during examination before surgery as well as those with pupillary abnormalities, synechia formation or glaucoma were excluded. Baseline characteristics including age, gender, laterality, etiology of cataract, and ocular biometry were noted.
All children underwent routine preoperative assessment for cataract a few days before surgery. None of the patients received any preoperative dilating drops. Intraoperatively the baseline horizontal corneal diameter was measured before starting surgery with a Castroviejo caliper. After that a clear corneal incision of 1.5 mm was made followed by paracentesis and injection of 0.025 ml Phenocaine (PC) via a tuberculin syringe into the anterior chamber. After waiting for 60 s, if the pupillary dilation was considered inadequate, double dose (0.05 ml) was injected again. We waited for another 60 s to document the maximum pupillary dilation. The time taken for adequate dilation was recorded. The rest of the surgical steps were the same as the standard procedure in pediatric cataract surgery. This included staining of the anterior capsule, viscoelastic injection, anterior continuous curvilinear capsulorhexis followed by aspiration of lens material. The same viscoelastic was used in all cases. A primary posterior capsulorhexis was done in all children less than 8 years and an intraocular lens implanted where indicated. A record of any other medication/devices used for mydriasis was kept. All these patients were operated under general anesthesia and pulse rate and blood pressure was monitored immediately before and after injection to assess any systemic side effects.
Figure 1 shows a representative photo depicting how pupillary diameter was measured. Pupil size was measured on table before the start of surgery (T1), after injection of PC (T2), after viscoelastic injection (T3), just before IOL implantation (T4), and at the end of surgery (T5), from screen captures from recorded videos using image J software by an independent observer who was masked to patient data. Snapshots at the required time points were exported to the Image J software. The scale of measurement was set by the pre-measured corneal diameter and the pupillary diameter was auto-calculated (Figure 1).
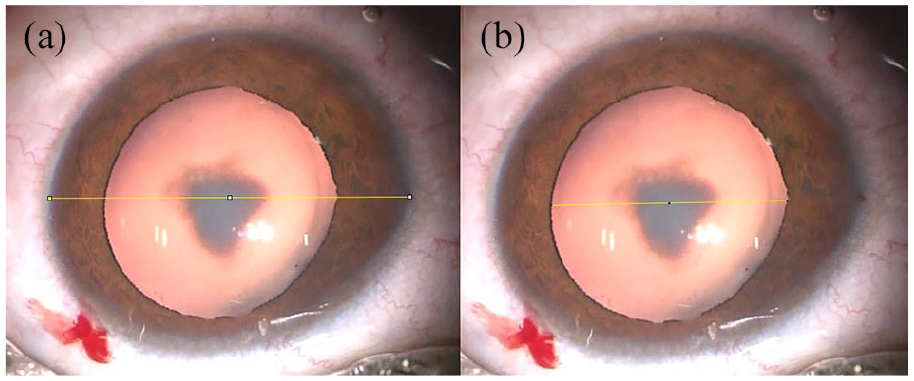
Representative photo depicting how pupillary diameter was measured: (a) horizontal corneal diameter entered manually set as a scale for measurement of the pupillary size as shown in (b).
Postoperatively, all children received antibiotics and steroid as a routine care. Children were examined on the first post-operative day, third post-operative day and then after 2 weeks. Intraocular pressure was measured on the third post-operative day by a handheld rebound tonometer (ICare; Tiolat, Helsinki, Finland).
Statistical analysis was done using SPSS software version 21. Since it was a pilot study, we did not fix the required sample size. All those who consented within the study period were included. Descriptive statistics, including mean and standard deviations was calculated. The paired t-test was used to compare the maximum pupillary dilation size from baseline. Correlation analysis was done using Pearson correlation coefficient.
The primary outcome measure was the completion of capsulorhexis and the concluding step in surgery (defined as either intraocular lens implantation or aphakia) without the need for using additional mydriatics or devices. Secondary outcomes included time for obtaining adequate mydriasis, total time of surgery, need for mechanical pupillary dilatation/additional mydriatic top-up (intracameral adrenaline), unexpected inflammation/corneal edema on the first post-operative day when examined on slit lamp, or any other adverse events. Observation for possible local and systemic adverse events was also kept.
Results
At the end of the study period, we had recruited 16 eyes of 13 patients. The mean age of the patients was 4.1 ± 3.9 years (range 1 month–10 years). There were eight males and five females with equal distribution of right and left eyes. There were four patients with microcornea/microphthalmos (six eyes). The baseline characteristics of the patients are detailed in Table 1.
Baseline characteristics of the patients.
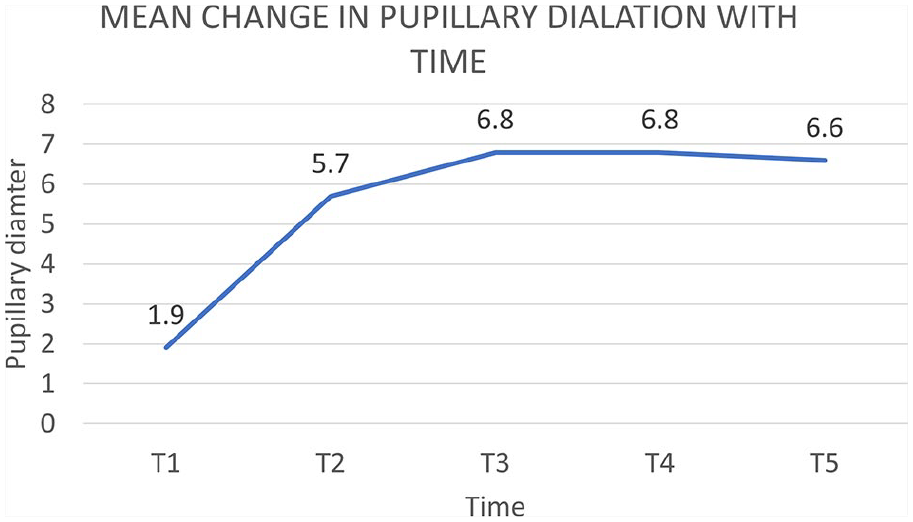
All surgeries were done by a single surgeon (JS) and recorded. The pupillary dynamics are described in Table 2. The mean pupillary diameter changed increased from 1.92 to 5.68 mm after injection of one unit of PC (p < 0.0001). There was a fair positive correlation between baseline pupillary diameter and axial length (R = 0.58) as well as corneal diameter (R = 0.54) and also between baseline pupil diameter and maximum pupillary dilation achieved (R = 0.66). There was a strong positive correlation of the pupillary dilation with axial length (R = 0.86) as well as horizontal corneal diameter (R = 0.81) (Table 2). Mean pupillary dilation achieved after excluding patients who had microcornea, was 6.6 ± 1.3 mm. Patients with microcornea had less pupillary dilation, but in all cases, surgery could be completed without the need for mydriatic supplementation. Twelve patients (75%) had a pupillary diameter >6 mm at all time points during surgery.
Mean pupillary dilation achieved with intracameral mydriatic at different time points of surgery.
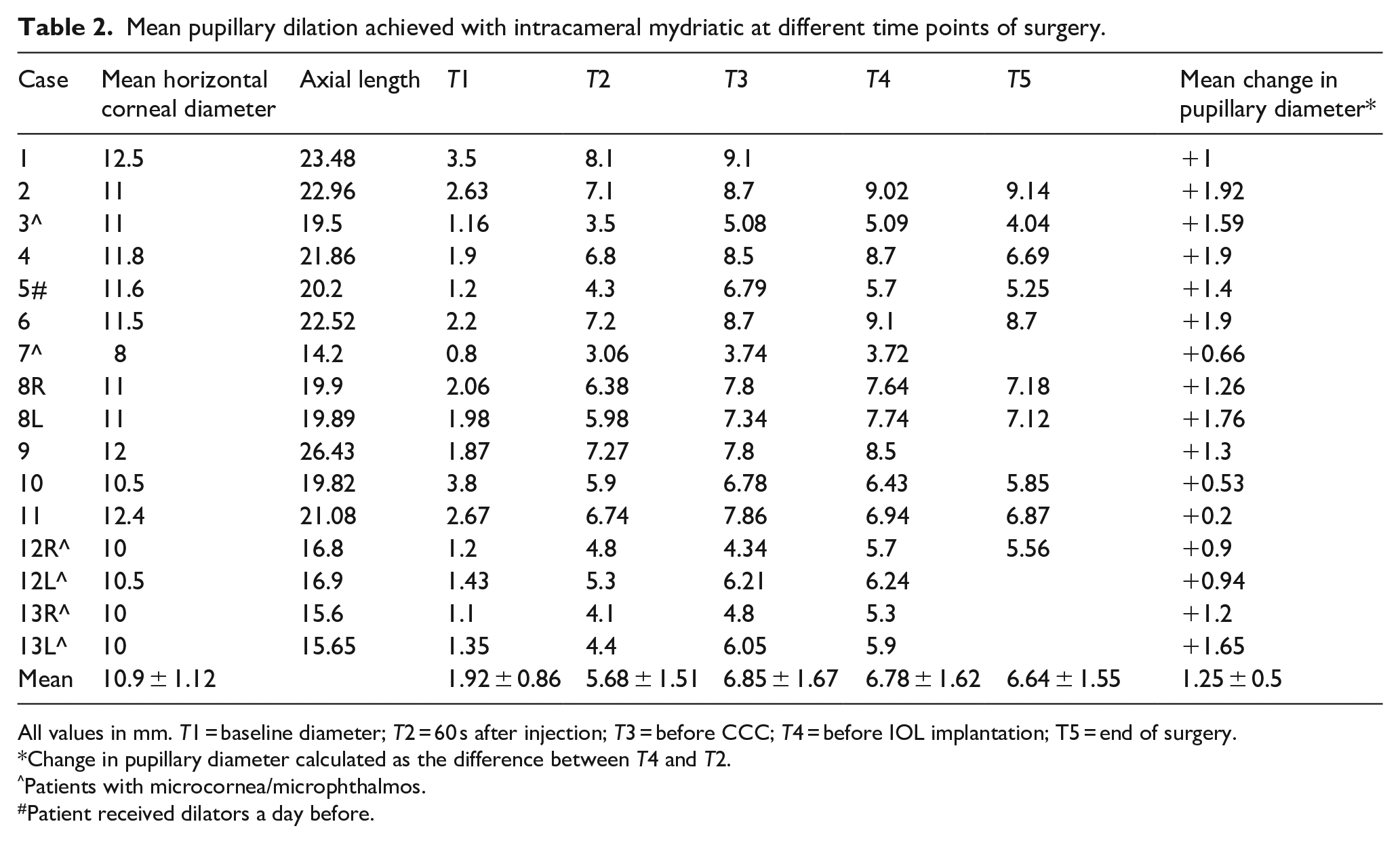
All values in mm. T1 = baseline diameter; T2 = 60 s after injection; T3 = before CCC; T4 = before IOL implantation; T5 = end of surgery.
Change in pupillary diameter calculated as the difference between T4 and T2.
Patients with microcornea/microphthalmos.
Patient received dilators a day before.
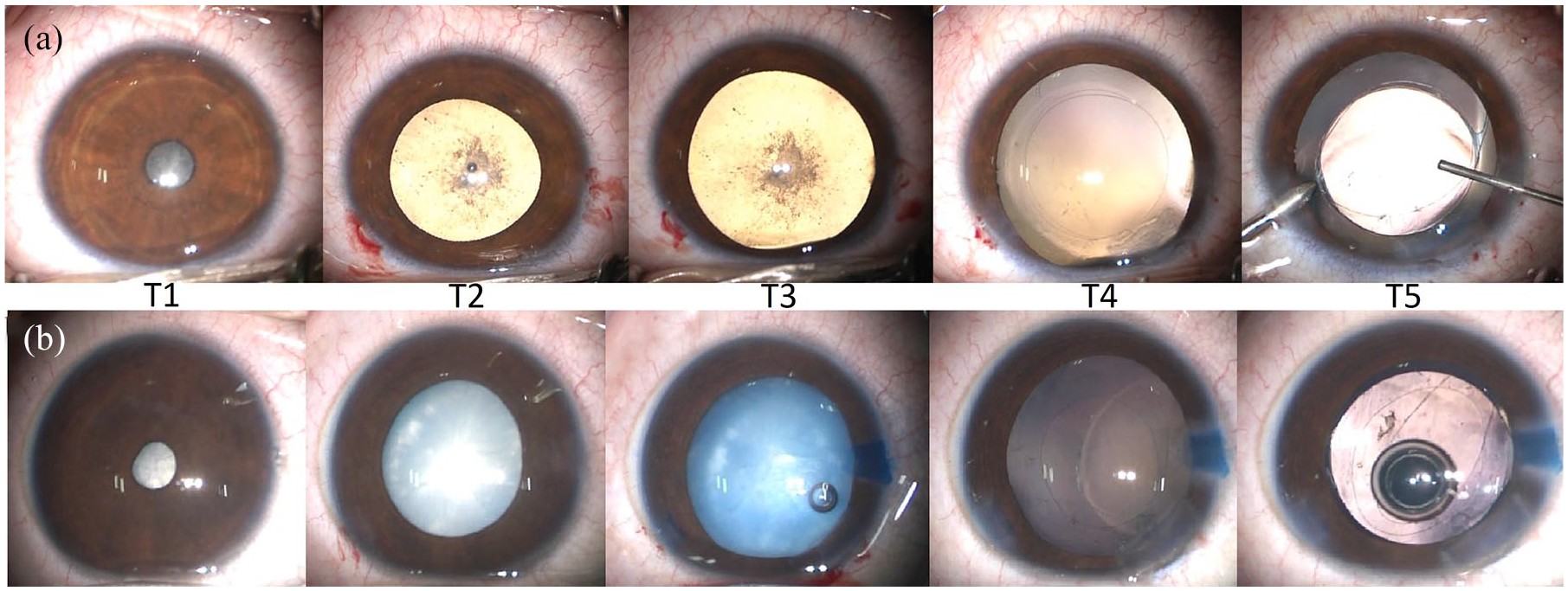
Figure 2 shows the pupillary dilation achieved in a representative case. There was no case of intraoperative miosis, iris bleeding, iris damage, or floppy iris noted. The pupil dilatation remained quite stable throughout the surgery. Mean variability in pupillary diameter was 1.45 ± 0.47 mm throughout the surgery. The average change in pupillary size from injection of drug till end of surgery was positive in all cases (mean change in pupillary diameter from injection till the end of surgery was +1.25 mm). There was a consistent increase in pupillary dilation throughout the surgery (Figure 3). Two eyes required augmentation(Case 3 and 7) because the pupillary dilation achieved by single dose was less than 4 mm. Eleven cases had a significant increase in pupillary diameter (⩾1 mm). Two eyes required augmentation. The mean change in pupillary diameter in these patients was only 0.6 ± 0.1 mm after injecting double the amount. No additional mydriatic or device was need during the entire duration of surgery. There were no cases of significant change in pulse/blood pressure noted during surgery in any patient. Postoperatively there was no case of unexpected post-operative corneal haze or inflammation or retained cortex. Intraocular pressure was not elevated in any patient.
Line graph showing mean change in pupillary diameter over time in case 5 (a) and case 9 (b).
Serial photographs of two representative cases depicting pupillary dilation with intracameral mydriatic-anesthetic combination. T1 = baseline pupillary diameter; T2 = 60 s after injection; T3 = before capsulorhexis; T4 = before IOL implantation; T5 = after viscoelastic removal.
Discussion
The ease of performing cataract surgery is mainly dependent on the size of the pupil. Conventionally in children, the pupil is dilated with a standard regime of topical drops (diluted for infants), given preoperatively starting a few hours before surgery.13,14 There are potential disadvantages of topical mydriatics. First, they need to be instilled repeatedly prior to surgery to ensure adequate intraoperative mydriasis, which is time-consuming. In addition, nearly half of the drug goes into systemic circulation that can cause medication errors, cardiovascular, and pulmonary side effects, besides ocular surface toxicity with preservatives. Systemic side effects of topical mydriatics are even more pronounced in children.1–5 Thirdly, the onset of action takes time, and their effect wanes off during surgery.6–8
The use of intracameral injection obviates the need for topical drops and their associated drawbacks. Several studies and trials have established the efficacy and safety of a fixed dose intracameral combination of phenylephrine (0.31%), tropicamide (0.02%), and lidocaine (1%) in phacoemulsification surgery in adults.7–12 The formulations have been proven to be effective and are a safe alternative to standard eye drops for initiating and maintaining intraoperative mydriasis and analgesia. Not only are they efficacious, but also reportedly comfortable to an adult patient. 12 However, there is no study on their use in children.
A child’s eye behaves very differently from that of an adult. Poorly dilating pupil is one of the major preoperative hurdles in pediatric cataract surgery.14–17 Maintaining adequate and consistent mydriasis during the entire surgery can be a challenge, thus adversely affecting the entire surgical procedure. Repeated touching of the iris in a small pupil can increase surgical time and leads to fibrinous complications, which in turn can lead to visual axis obscuration. These complications are more worrisome than adults because of the risk of amblyopia.
The advantages of the intracameral drug combination, were extrapolated to children in this pilot study and our results have shown that this mode of pupillary dilatation was effective, fast, and targeted. The average pupillary dilation achieved (6.2 mm) was comparable to that reported (from 6.5 to 7.5 mm) in adults. This dilation was stable and increased as the surgery continued, in contrast, to decrease in pupillary diameter with topicals as the surgery progresses.6–8 Moreover, the surgery could be comfortably completed in all cases without the need for additional drugs. The average time taken for dilation was 60 s which was a much faster onset than topical mydriatics (takes a minimum of 20 min due to slow absorption through the cornea). This was more than that reported in adults. 7 The pupil dynamics varied about 1.5 mm during the surgery with no additional benefit being obtained with a second injection. This stability of the pupil could also be attributed to the lidocaine component of the formulation. 18
None of the studies in adults have reported any serious adverse effects with the formulation.9–12 In India, the only combination available is a 1 ml ampule of preservative-free Phenocaine Plus (Entod Pharma, Gujarat, India). In children, we observed that 0.025 ml of the solution was enough in many cases, and increasing the amount for injection did not have a significant effect on further increasing the dilatation. The concentration of the constituents in the commercially available solution is exceptionally low compared with topical mydriatics, which ensures greater safety and lower side effects. There is presently only one study that has analyzed the efficacy of intracameral phenylephrine and ketorolac combination in pediatric patients. 19 The study concluded the combination to be safe and effective to maintain mydriasis in pediatric cataract surgery. We also did not observe any change in blood pressure or pulse rate during surgery when intracameral combination was used. The use of intracameral mydriatics in patients with glaucoma or patients with synechia was not studied.
It has been shown in adults that the combination works well in all types of iris pigmentation. 12 Indian eyes are heavily pigmented, so hypothetically, patients with lightly pigmented iris may benefit even more. Our series, which had children, and all were darkly pigmented has proven this targeted delivery to be efficacious. The endothelial cell loss observed in adults is comparable to routine phacoemulsification surgery. 20 In children, the endothelium is more robust as compared to adults. With a dose much less than that used in adults, endothelial integrity may be believed to be well maintained, which can be studied by imaging in the future.
In our setting, where the children are staggered with issues of when they are taken up for surgery, intracameral mydriatics are an ideal cost-effective way to obtain mydriasis as and when required. We found this very useful in COVID-19 times when staggering of surgical cases is recommended and awaiting reports leads to delay in the start of surgery. Many times the mothers feed wailing children, which again leads to a delay in general anesthesia. It is very normal for children to cry when the drops are instilled, and so pupillary dilatation may not be achieved as the topical drops are continuously washed out. Corneal factors which may lead to decreased absorption are bypassed by intracameral injections. The concerns with preservatives in topical drops are done away with intracameral use. In cases where a surgeon aims at bilateral simultaneous cataract surgeries and/or when one eye surgery might take variable time, surgery in the second eye can be performed comfortably by intracameral mydriasis. The use of epinephrine in irrigating solutions is supported by many pediatric ophthalmologists 21 but is anticipated to have systemic side effects. 22 Gonioscopy examination with an undilated pupil can be performed just prior to surgery, followed by intracameral mydriasis and then completion of surgery. It can be safely used in those with an allergy to Atropine, and Cyclopentolate. Adequate pupillary dilation reduces intraoperative complications due to iris touch and improves instrument maneuverability.
The study has limitations as the number of participants are less (being a pilot study) and there is no comparison arm. With the proven suitability of this combination for pediatric cataract surgery from our pilot project, we are in the process of recruiting patients for a randomized study comparing topical versus intracameral mydriatics in pediatric cataract surgery, where we have included pain for evaluation as well. The results of this study reveal prompt and stable mydriasis with intracameral injection in children undergoing cataract surgery. Coupled with so many advantages in children with a minuscule dose, we recommend this as an effective and safe alternative to topical drops.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
