Abstract
Purpose
Surgical removal of a vascularized pupillary membrane may be challenging with the risk of intraoperative bleeding and postoperative recurrence. We present a case of a 4-week-old who presented with anterior persistent fetal vasculature (PFV) and dense vascularized pupillary membrane in which the use of intracameral and intravitreal bevacizumab may have contributed to successful treatment.
Observation
A 4-week-old-month-old otherwise healthy girl was referred to Boston Children's Hospital for evaluation of cataract. Ocular examination revealed right microcornea and vascularized pupillary membrane. The left eye exam was unremarkable. Only three weeks after surgical excision of the pupillary membrane and cataract extraction, recurrence of a vascular pupillary membrane was noted. Repeat membranectomy with pupilloplasty and use of intracameral bevacizumab was performed. The pupillary opening was further opened 5 months later, after repeat (intravitreal) bevacizumab, and the pupil has remained open and stable with >6 months’ follow-up.
Conclusion and importance
This case suggests a role for bevacizumab in the management of PFV, however, a cause-and-effect relationship cannot be proven. Further prospective comparative studies are needed to confirm our findings.
Keywords
Introduction
Persistent fetal vasculature (PFV), formerly known as persistent hyperplastic primary vitreous, is a congenital disorder that results from the failure of regression of the hyaloid vascular system. Although it is mostly unilateral, subtle peripheral retinal vascular abnormalities have been reported in the contralateral eyes of unilateral cases. Clinically it is classified into anterior, posterior, or combined PFV. Presentations are variable including shallow anterior chamber, vascularized pupillary membrane, cataract, elongated ciliary process, Mittendorf dot, vascular stalk, or retinal detachment. 1 Several ocular anomalies may be associated with PFV including increased corneal thickness, microcornea, and chorioretinal coloboma.2,3 Pupillary membranes in case of anterior PFV may be densely vascularized which may result in intraoperative hyphema and vitreous hemorrhage when the lens is removed. Additionally, rapid postoperative recurrence of dense vascularized membrane occluding the pupil may occur.
In our patient, intracameral bevacizumab (Avastin®, Genentech, Inc., South San Francisco, CA) was used to help delay the reformation of the pupillary membrane, and to control bleeding so that pupilloplasty could be completed. While this experience suggests a role for bevacizumab in the management of PFV cases with dense vascularized pupillary membrane, a cause-and-effect relationship cannot be proven. Larger prospective comparative studies are needed to confirm our findings.
Case report
A 4-week-old healthy term-born girl was referred for a right eye cataract evaluation, first noted at the age of 2 weeks. Family history was unremarkable for childhood eye disorders. Pertinent abnormal examination findings OD included mild microcornea (9 mm horizontal corneal diameter OD vs 10 mm OS), an extensive vascularized anterior pupillary membrane extending across the iris to Schwalbe's line, covering the lens (Figure A). Intraocular pressures measured by iCare rebound tonometry (iCare Finland, Oy) were normal (6 mmHg OD, 7 mmHg OS), and dilated fundus examination showed a grossly normal retina in the visible fundus area around the pupillary membrane. At surgery, the membrane was found to be adherent to the lens capsule, so removal of the membrane centrally as well as cataract extraction with posterior capsulotomy and anterior vitrectomy, with a peripheral iridectomy were performed (Figure 1B). Three weeks later, the patient presented with a recurrent vascular pupillary membrane (Figure 1C). After discussion and with parental consent, repeat membranectomy with pupilloplasty and use of intracameral bevacizumab (.03 mL of .25 mg/0.02 mL concentration) was performed. During the procedure, the central membrane was opened (Figure 1D), but pupilloplasty could not be completed due to bleeding from the iris membrane, despite cautery. This resulted in a small vitreous hemorrhage that resolved spontaneously in 2 weeks. The pupillary opening constricted slightly so 5 months later we performed an examination under anesthesia, noting the persistent vascularization of the iris (Figure 1E) for which we performed an intravitreal bevacizumab injection (.02 mL of .5 mg/0.02 mL concentration) in advance of a second pupilloplasty that was planned one week later. This pupilloplasty was completed as planned without significant bleeding, and the pupil has remained open and stable with 9 months’ follow-up (Figure 1F). We did not notice any intra- or postoperative adverse events related to the bevacizumab injection, this patient had good health and normal development. Aphakia was managed with a contact lens (initially +29 diopters) and amblyopia was managed by patching OS (up to 50% of waking hours). At the final follow-up, 9 months after the last procedure, the patient showed good fixation OD and OS with good grating visual acuity scores of 20/89 OU (Teller cards).
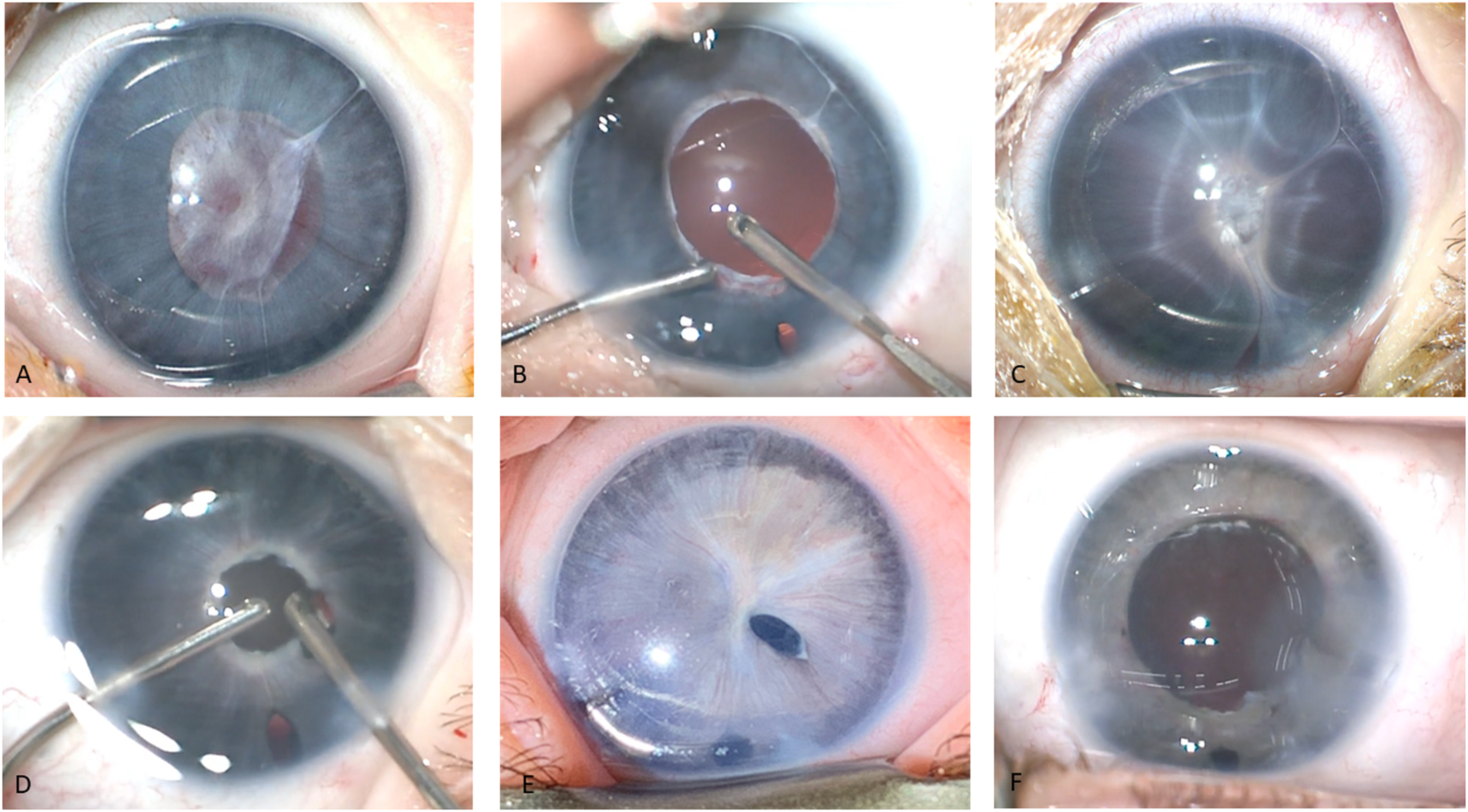
All surgical images are the surgeon's view. A) Extensive iris-pupillary membrane with vascular component; B) Intra-operative image after removal of the central membrane, cataract, and peripheral iridectomy; C) Recurrence of vascular pupillary membrane 3 weeks after surgery; D) Intra-operative image after removal of the central membrane, cautery to the edge of the pupil, and bleeding after initiation of pupilloplasty; E) Persistent vasculature at an examination under anesthesia for intravitreal bevacizumab injection; F) Anterior segment image of final pupillary status.
Discussion
Vascular endothelial growth factor (VEGF) plays a vital role in the pathogenesis of several pediatric vascular disorders such as retinopathy of prematurity (ROP) and familial exudative vitreoretinopathy. Although the exact pathogenesis of PFV is unknown, VEGF and placental growth factor have been reported to play a role in hyaloid vasculature regression and the development of PFV. Gajjar and colleagues 4 proposed that an imbalance in the levels between antiangiogenic factors such as arresten and matrix metalloproteinases may play a role in the development of PFV.
Bevacizumab is a recombinant humanized monoclonal antibody against VEGF. Intravitreal anti-VEGF injections are widely used in the pediatric population with ROP or Coats disease; however, their role in PFV is unclear. By inhibiting VEGF, bevacizumab is proposed to reduce leakage from the abnormal PFV vessels and induce their regression. Prakhunhungsit and Berrocal 5 have suggested injecting anti-VEGF intraoperatively after surgical management of PFV to reduce the risk of bleeding and postoperative inflammation;however, no studies are currently available to support this theory. Off-label intracameral anti-VEGF injection in the pediatric population has been tried by Nallasamy and Colby 6 after surgical removal of a dense retroprosthetic membrane that developed over Boston type I Keratoprothesis (Massachusetts Eye and Ear Infirmary, Boston, MA) that was implanted after surgical removal of limbal dermoid. However, the authors did not comment on its efficacy. In adults, several reports have demonstrated regression of active fibrovascular membranes over the iris after intracameral bevacizumab.7–9 Raghuram and colleagues 9 described a 31-year-old female patient who presented with a blind painful eye. Examination disclosed anterior chamber inflammation and iris neovascularization with fibrovascular membrane. They reported a reduction in the anterior chamber inflammation and regression of the iris neovascularization after intracameral bevacizumab (1 mg) injection and the effect was sustained for up to 6 months. Gristani and colleagues 8 reported reduced leakage of iris neovascularization after intracameral injection of bevacizumab in adult patients with neovascular glaucoma. Another study showed regression of rubeosis and decreased leakage in 16 eyes with neovascular glaucoma injected with 1.25 mg of bevacizumab intracamerally. 7 In an animal study, Shin and colleagues 10 showed that intracameral bevacizumab was safe on corneal endothelium without any significant changes in the morphology or function of endothelial cells after injection.
In our patient, the rapid recurrence of a vascular pupillary membrane after the first surgery motivated us to utilize intracameral bevacizumab at the end of the second procedure, but we were unable to complete the pupilloplasty due to intra-operative bleeding. While there was no central recurrence of a pupillary membrane in the area of pupilloplasty, there was still incomplete regression of the iris vessels so we performed a second intravitreal bevacizumab injection (now unicameral eye) one week prior to the successful pupilloplasty. The uneventful postoperative course after this procedure suggests that the adjunctive use of bevacizumab was effective. In hindsight, such cases of anterior PFV with a significant persistent vascular network may benefit from an injection prior to initial surgery. Since the effect of bevacizumab is temporary, unless complete regression of the abnormal vasculature is noted, some patients may need reinjection. On the other hand, most cases of PFV cataracts are mild and do not exhibit persistence of the abnormal vasculature, and the use of an anti-VEGF agent is not warranted. We recognize that intraocular use of bevacizumab is off-label, and any intracameral medication should be confirmed to be preservative-free to avoid corneal toxicity.
This report demonstrates the use of intracameral and/or intravitreal bevacizumab as adjunctive therapy to reduce the risk of perioperative bleeding and recurrence for surgical treatment of a vascularized pupillary membrane or cataract in the setting of PFV. Pediatric cataract surgeons should be aware of the potential benefit of bevacizumab and consider the application of this therapy in select cases.
Literature search
We performed a literature search in the electronic databases of PubMed CENTRAL, Google Scholar, and Ovid MEDLINE using the following one or more of the following words: “bevacizumab”, “Avastin”, “VEGF”, “anti-VEGF”, “intracameral”, “persistent fetal vasculature”, “PFV”, and “pupillary membrane”.
Footnotes
Meeting presentation
Annual meeting of the American Association of Pediatric Ophthalmology and Strabismus (AAPOS) 2022, Scottsdale, Arizona.
Disclosure
The authors claim no relevant financial disclosures.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Boston Children's Hospital Ophthalmology Foundation, Inc.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
The study adhered to the tents of the declaration of Helsinki version 2013
Patient consent
Consent was not needed as no identifying information was published.
