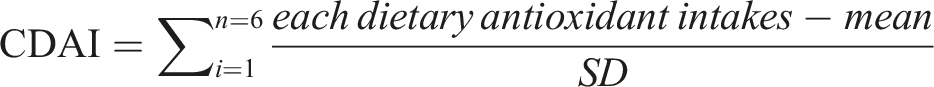Abstract
Keywords
Introduction
Gout is the most common inflammatory arthritis caused by prolonged hyperuricemia, leading to the formation of sodium urate crystals in the joints, tendons, and periarticular tissues. Gout has severely affected patients’ multiple aspects of daily life, including mobility, sleep quality, work productivity, and psychological status (Díaz-Torné et al., 2023; Garcia-Guillen et al., 2022; Stewart et al., 2020). In the USA, Europe, and Australia, the prevalence of gout in adults is about 3.9, 2, and 1.6%, respectively (Chen-Xu et al., 2019; González-Chica et al., 2018; Sigurdardottir et al., 2022); 7.4 million incident cases every year and 1.3 million Years Lived with Disability (YDL) globally caused by gout in 2017 (Kuo et al., 2015; Robinson et al., 2015; Ryu et al., 2014; Safiri et al., 2021). Established risk factors for gout are obesity, alcoholic drinking, smoking, hypertension, type 2 diabetes, and chronic kidney disease (CKD; Borghi et al., 2020; Nieradko-Iwanicka, 2022; Rohm et al., 2022; Song et al., 2022). Due to its high incidence and the significant negative impact of gout flare on patients’ psychological and physical issues, gout has become a major public health problem.
Oxidative stress is regarded as an imbalance between pro-oxidative and anti-oxidative factors, with excessive reactive oxygen species (Zamudio-Cuevas et al., 2015). Numerous researches have revealed a strong relationship between oxidative stress and gout (Albu et al., 2020; Sánchez-Lozada et al., 2008). The formation of Monosodium Urate (MSU) and hyperuricemia result in inflammation, endothelial oxidative damage, and oxidative stress by stimulating Nicotinamide Adenine Dinucleotide Phosphate (NADPH) oxidases, reducing endothelial levels of antioxidant nitric oxide (NO), increasing reactive oxygen species (ROS) and activating pro-inflammatory biomarkers (Borghi et al., 2015; Gherghina et al., 2022). Consequently, how to effectively reduce or prevent oxidative stress has attracted attention from clinical physicians and nurses.
The daily dietary intake of antioxidants may inhibit oxidative stress to ameliorate gout and lower serum uric acid concentration by increasing antioxidant defense and reducing oxidative stress and inflammation (Ryu et al., 2014). Although antioxidants such as vitamin E have been evidenced to benefit the Deoxycorticosterone (DOCA)-salt-treated rat models by decreasing blood pressure, lowering urinary uric acid, and increasing urinary excretion of uric acid, their relationship with gout patients remains unclear (Seifi et al., 2011). Some randomized clinical trials attempted to establish the effects of the intake of antioxidants on developing gout in patients (Juraschek et al., 2022; Stamp et al., 2013). However, the conclusions of these studies were conflicting and just considered one single food or specific dietary antioxidants, and the effect of the total antioxidant capacity of the diet on gout cannot be reflected.
To date, the CDAI seems to be a scientific and nutritional evaluation method to assess an individual diet’s overall antioxidant characteristics (Maugeri et al., 2019). The CDAI, evaluated with zinc, selenium, carotenoids, and vitamins A, C, and E, have shown low CDAI was correlated with an increased risk of various types of diseases and associated with inflammatory biomarkers including IL-1β and TNFα, which were also established inflammatory biomarkers of gout (Luu et al., 2015). Previous studies found a negative association between CDAI and depression in US adults, increased intake of dietary antioxidants may protect from depressive symptoms and improve the prognosis of stroke patients (Fattori et al., 2020; Xu et al., 2023; Zhao et al., 2022). However, the influence of a combination of dietary antioxidants (such as CDAI) in gout has not been conducted. Therefore, we analyzed the NHANES database to explore the link between gout in the US population aged 20 and older and CDAI.
Aim of the Study
The aim of this study was to explore the relationship between CDAI and gout to provide data support for preventing gout through dietary intervention. We hypothesized that (1) the CDAI was related to gout; (2) the higher CDAI was related to a lower prevalence of gout.
Methods
Data Sources
We selected 6 consecutive survey cycles of the National Health and Nutrition Examination Survey (NHANES; 2007–2008, 2009–2010, 2011–2012, 2013–2014, 2015–2016, 2017–2018), and the overall sample of adults (age ≥20) with no missing data for any variables is 26,117. The 26,117 NHANES participants represented the 177 million noninstitutionalized adult population of the United States. The inclusion criteria were as follows: those who had complete data about age, gout, demographics, BMI, smoke status, alcohol status, hypertension, diabetes, CDAI, and CKD. We excluded those with missing data. Participants with missing data were discarded by listwise deletion. Participants’ selection, exclusion criteria, and additional details are shown in Figure 1. Flowchart of the study participants.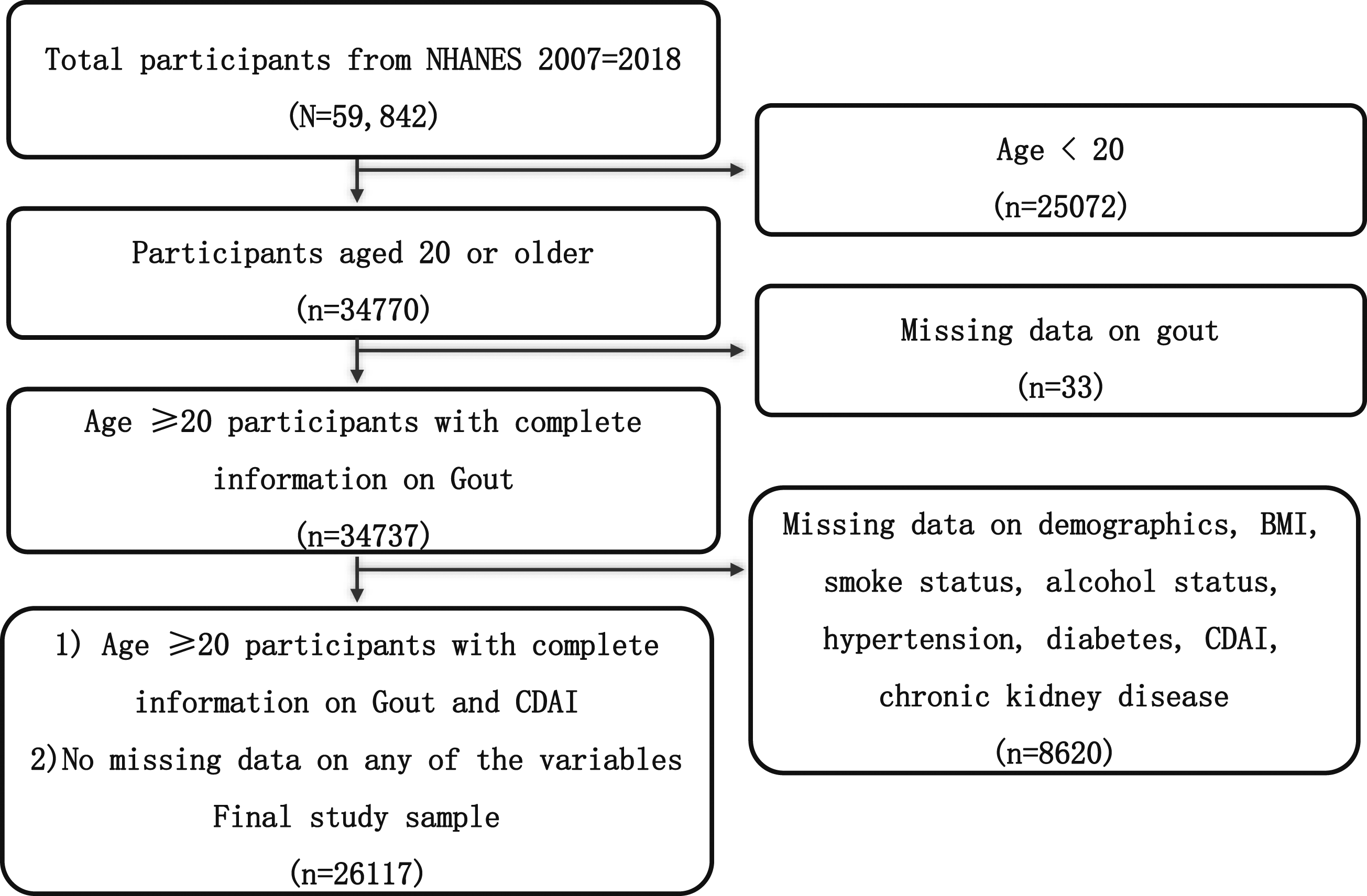
Composite Dietary Antioxidant Index Calculation
Participants from NHANES are eligible for the 24-h dietary recall. The calculation of CDAI was previously validated in another cross-sectional study (Maugeri et al., 2019), and the CDAI included 6 antioxidants (zinc, selenium, carotenoids, and vitamins A, C, and E). To assess the synergistic effects of CDAI on gout, the CDAI was calculated for all participants using the method recommended by Maugeri et al. (2019). Each of the 6 dietary antioxidants was standardized by subtracting the sex-specific mean and dividing by the sex-specific standard deviation; and then calculated the CDAI by summing the standardized 6 dietary antioxidant intakes.
Gout
All participants were asked, “Has a doctor or other health professional ever told you that you had gout?” and the response options were “Yes” or “No.” Those participants whose response was “Yes” were defined as having gout and those who answered “No” were defined as non-gout (Chen-Xu et al., 2019).
Covariates
The following covariates were included in this study: sex (male, female), age (20–39, 40–59, 60–79, ≥80), race (black, Mexican, white, other races), marital status (married, never married, other including divorced), poverty income ratio (PIR level (<1.3, ≥1.3), education level (junior high school or below, high school, college or above), BMI (<18.5, 18.5–24.99, 25–29.99, ≥30), smoking status (never, former, now), alcohol status (never, former, mild, moderate, heavy), hypertension (yes, no), diabetes (diabetes mellitus, impaired fasting glycaemia, impaired glucose tolerance, no) and chronic kidney disease (yes, no).
Composite dietary antioxidant index were classified according to quartiles, with quartile 1 (Q1) and quartile 4 (Q4). To account for the complex survey design and to obtain results that would be generalizable to the total population living in the US, we used the survey weight from the mobile examination center (MEC12 YR = 1/6*wtmec2yr; Johnson et al., 2013). Categorical variables were presented as weighted percentages. We compared categorical variables among different CDAI quartile groups using the weighted chi-square test. The weighted univariate and binary logistic regression analysis were used to test the association between variables and gout. The weighted multivariable logistic regression analysis was used to test the association of CDAI and gout in 4 different models. In the crude model, covariates were not adjusted. Model 1 adjusted for age, gender, and race. Model 2 adjusted for age, gender, race, marital status, PIR level, and education level. Model 3 adjusted for age, gender, race, marital status, PIR level, education level, smoke status, alcohol status, hypertension, diabetes, and CKD. We further performed stratification and interaction analyses by diabetes, marital status, alcohol status, hypertension, PIR level, education level, BMI, smoke status, age, sex, race, and CKD. All analyses were performed using R Studio (4.2.2, http://www.R-project.org), and a p-value <.05 was statistically significant.
Results
Baseline Characteristics of Participants
Characteristics of Participants in 4 CDAI Quartile Groups [n (%)].
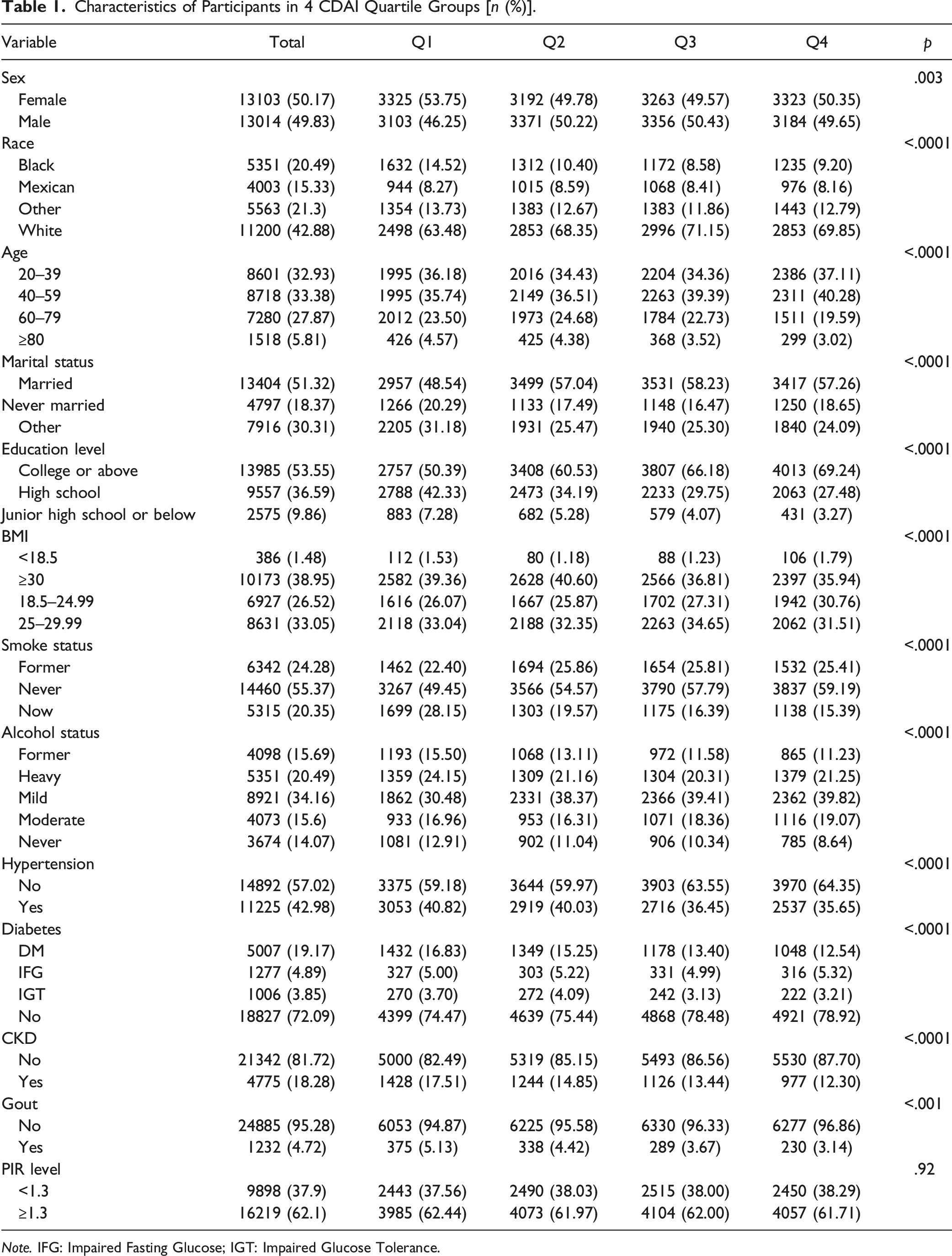
Note. IFG: Impaired Fasting Glucose; IGT: Impaired Glucose Tolerance.
Weighted Univariate and Multivariable Analysis of Gout
Weighted Univariate and Multivariate Logistic Analysis of Gout.
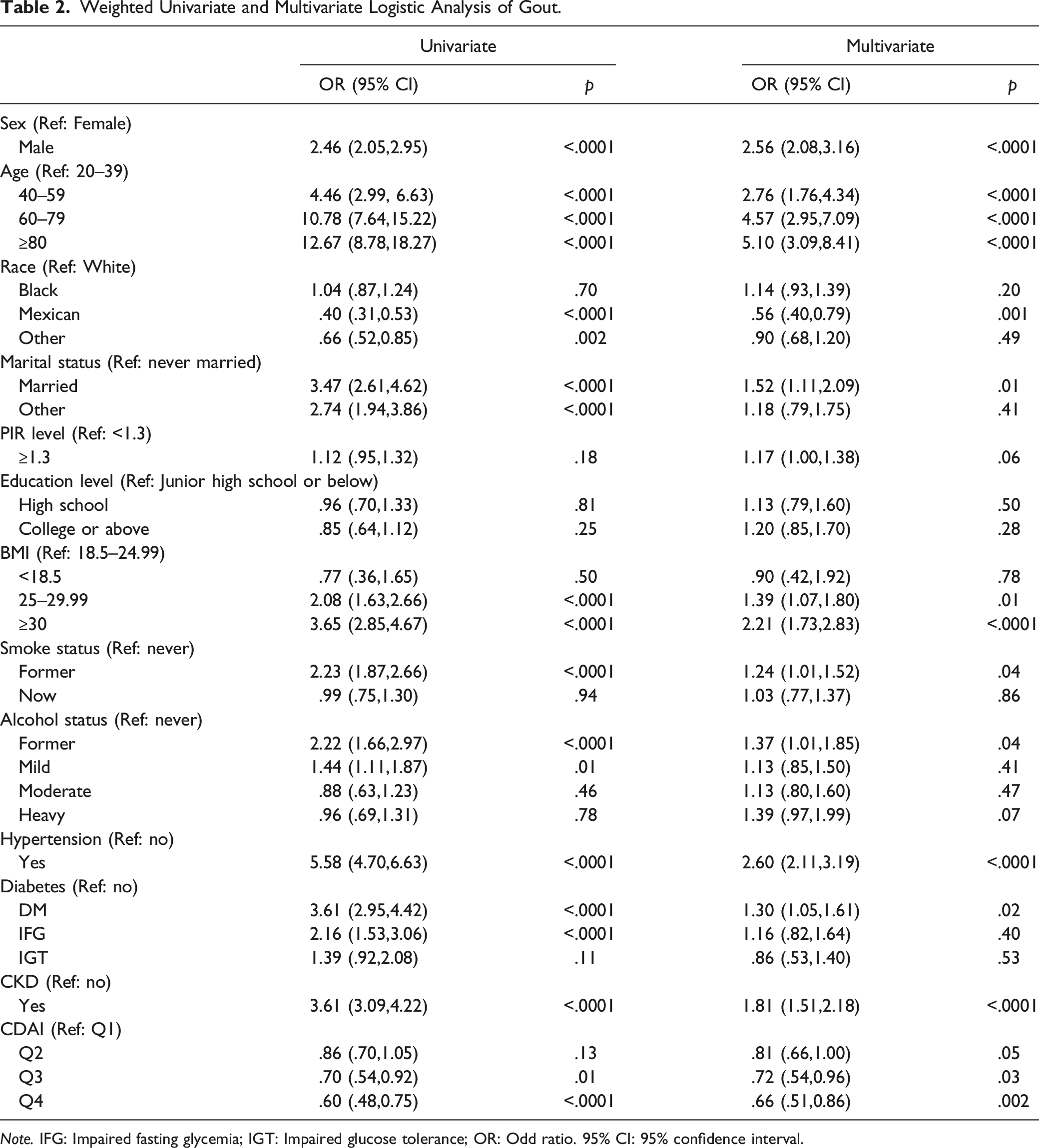
Note. IFG: Impaired fasting glycemia; IGT: Impaired glucose tolerance; OR: Odd ratio. 95% CI: 95% confidence interval.
The weighted multivariable analysis showed that participants who were Mexican (OR = .56, 95% CI: .40, .79, as compared to white) and had CDAI Q4 (OR = .66, 95% CI: .51,0.86, as compared to Q1) were associated with lower prevalence of gout. However, those who aged 40–59, 60–79, ≥80 (40–59: OR = 2.76, 95% CI: 1.76, 4.34; 60–79: OR = 4.57, 95% CI: 2.95, 7.09; ≥80: OR = 5.10, 95% CI: 3.09, 8.41, as compared to aged 20–39), were male (OR = 2.56, 95% CI: 2.08, 3.16, as compared to female), had married (OR = 1.52, 95% CI: 1.11, 2.09, as compared to never married), had BMI 25–29.99, ≥30 (OR = 1.39, 95% CI: 1.07, 1.80; OR = 2.21, 95% CI: 1.73, 2.83, as compared to BMI = 18.5–24.99), were former smokers (OR = 1.24, 95% CI: 1.01, 1.52, as compared to never smokers), were former drinkers (OR = 1.37, 95% CI: 1.01, 1.85, as compared to never drinkers), had hypertension (OR = 2.60, 95% CI: 2.11, 3.19, as compared to no hypertension), had DM (OR = 1.30, 95% CI: 1.05, 1.61, as compared to no DM), had CKD (OR = 1.81, 95% CI: 1.51, 2.18, as compared to no CKD) presented an increased prevalence of gout.
The increased risk of gout due to other marital status, BMI 25–29.99, mild drinkers, and IFG observed on univariable analysis was not presented on multivariable analysis. The decreased risk of gout due to other races was not presented on univariable analysis was not presented on multivariable analysis.
Weighted Logistic Regression Analysis of CDAI and Gout
Logistic Regression Analysis of CDAI and Gout.
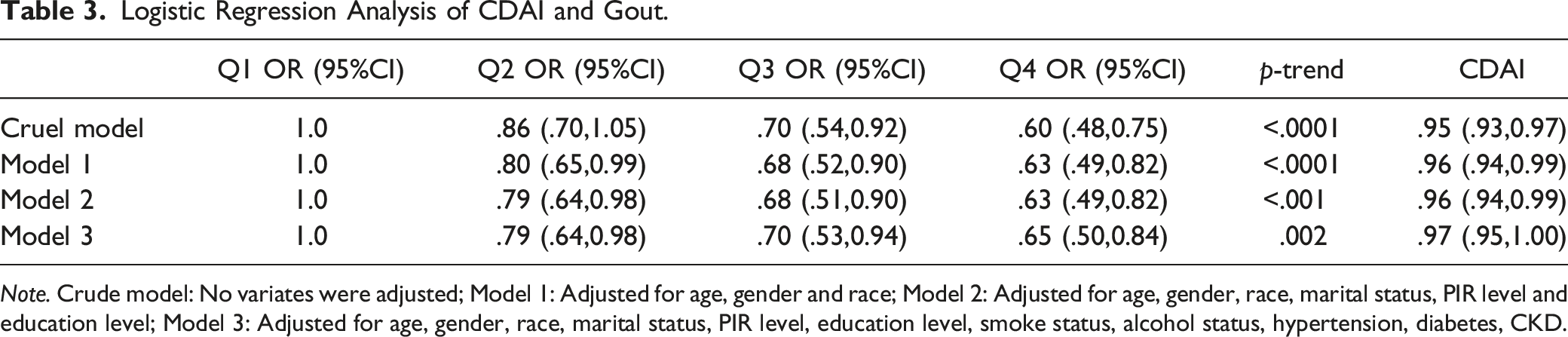
Note. Crude model: No variates were adjusted; Model 1: Adjusted for age, gender and race; Model 2: Adjusted for age, gender, race, marital status, PIR level and education level; Model 3: Adjusted for age, gender, race, marital status, PIR level, education level, smoke status, alcohol status, hypertension, diabetes, CKD.
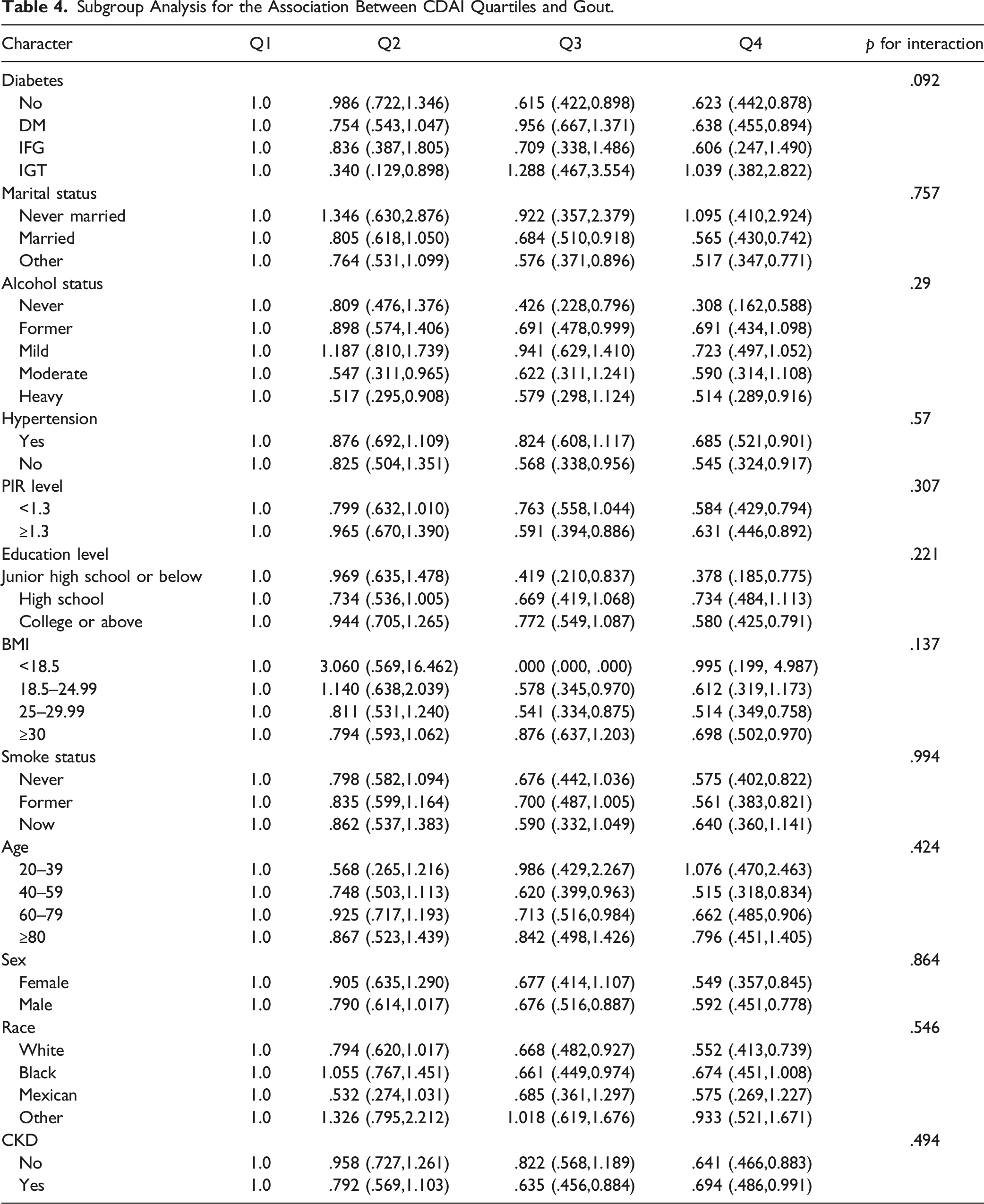
Subgroup Analysis for the Association Between CDAI Quartiles and Gout
Subgroup Analysis for the Association Between CDAI Quartiles and Gout.
Discussion
In our cross-sectional study with 26,117 participants enrolled, we suggest that participants with higher CDAI show a decreased likelihood of gout. Subgroup analysis and interaction tests show that this relationship is consistent in a diverse demographic setting. To our best knowledge, this is the first study to explore the relationship between CDAI and gout in the general population.
Although existing research on the association between gout and CDAI is still relatively rare, the use of specific dietary antioxidants to intervene in the management of gout has been a hot topic of research today. A study found that an anti-inflammatory-antioxidant rich diet reduced intestinal inflammation and improved clinical symptoms in progressive forms of multiple sclerosis (Moravejolahkami et al., 2022). A large, prospective research showed higher vitamin C intake, an essential antioxidant, was beneficial in lowering the risk of incidence of gout (Choi et al., 2009). Results from the NHANES survey 2007–2014 also suggested that dietary serum 25 hydroxyvitamin D was inversely associated with gout in US adults. Our results also confirmed a negative association between CDAI and gout, in line with previous studies.
The mechanism of the negative association between CDAI and gout still needs to be further explored, and there may be possible explanation. Oxidative stress and inflammation may be critical in the association between CDAI and gout. A previous cross-sectional study has found patients with gout were higher in oxidative stress and inflammation-related markers, including hi3-nitrotyrosine, tumor necrosis factor, C-reactive protein, interleukin (IL)-1β, and interleukin-6 (Alduraibi et al., 2023). Oxidative stress can cause excessive levels of reactive oxygen species and endothelial dysfunction; meanwhile, inflammation also causes the release of neutrophils and the increase in reactive oxygen species by activating NADPH oxidase, involving the occurrence and progression of certain diseases, such as gout, renal disease, heart failure, stroke, and atherosclerosis (Liu et al., 2021; So & Martinon, 2017). Therefore, a better understanding of the underlying mechanism may lead to the development of new treatments and dietary interventions for gout.
CDAI is another new method used to evaluate oxidative stress and inflammation status of certain diseases. Previous studies showed that dietary total antioxidant capacity (DTAC) is negatively associated with oxidative stress of hyperuricemia, rheumatoid arthritis, hypertension, and CKD (Choi et al., 2009; Cyuńczyk et al., 2022; Moradi et al., 2022; Yang et al., 2022). These results are consistent with our findings that DTAC is associated with oxidative stress. Patients with gout have been reported to have higher oxidative stress, higher levels of inflammation, and a lower intakes of dietary antioxidants (Krishnan, 2010; Rai et al., 2017; So & Martinon, 2017). Therefore, dietary antioxidants are important for patients with gout. Numerous dietary antioxidants can protect against oxidative stress and reduce the level of uric acid serum by reducing lipid peroxidation, lowering insulin resistance, and preventing inflammation (Choi et al., 2009; Ryu et al., 2014; Yokose et al., 2021). Carotenoids, zinc, and selenium were important antioxidants in decreasing the levels of oxidative stress. Seaweed, which is high in antioxidant content of carotenoids, helps to reduce serum uric and increase the excretion of uric by inhibiting xanthine oxidase and preventing kidney reabsorption (Zhang et al., 2022). Zinc, which is an important hydrophilic antioxidant in human plasma, can eliminate oxygen radicals to retard the oxidative process (Powell, 2000; Xie et al., 2015). As an essential trace element for the human body, selenium exerts its biological functions through selenoproteins. It exerts antioxidant functions by inhibiting the NF-κB-cascade and anti-inflammatory effects, thereby reducing the production of inflammatory mediators (Hariharan & Dharmaraj, 2020).
Limitations
Our research has some limitations. First, the diagnosis of gout was based on the self-report from subjects. Therefore, gout prevalence in this cross-sectional study may be inflated, similar to prevalence estimates for other diseases from the NHANES. Additionally, this design is a cross-sectional study, and we could not obtain a causal association and avoid bias from confounding factors.
Conclusion
Composite dietary antioxidant index was inversely associated with gout in US adults, and dietary antioxidant intervention might be a promising method in the therapy of gout, and greater emphasis should be placed on zinc, selenium, carotenoids, vitamins A, C, and E.
Footnotes
Acknowledgments
We acknowledged participants were involved in the NHANES, and the NHANES research team was collecting and sharing the data.
Author Contributions
Wanqin Hu contributed to acquisition, analysis, and interpretation drafted manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Ziwei Ye contributed to conception critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Zeya Shi contributed to acquisition critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy Tao Li contributed to design critically revised manuscript gave final approval agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The data given this article are publicly available datasets were analyzed in this study. This data can be found here: ![]() .
.