Abstract
Objectives
This study aims to characterise the presence and nature of ocular findings in cats diagnosed with feline infectious peritonitis (FIP) and to evaluate the effects of antiviral treatment with GS-441524 on ocular manifestations.
Methods
Study participants were prospectively recruited over a 1-year period by the FIP study team at the Vetsuisse Faculty, University of Zurich. A total of 118 cats diagnosed with FIP underwent a comprehensive standardised semi-quantitative ophthalmic examination. Cats with ophthalmic abnormalities at the baseline examination received a follow-up examination on days 7 and 42, after commencement of the oral antiviral therapy, GS-441524. Changes throughout the treatment period were recorded and reviewed.
Results
At the baseline examination, 48/118 (40.1%) cats had ocular abnormalities, with the most common associated findings being anterior uveitis, keratic precipitates and chorioretinitis. Throughout the course of treatment with GS-441524, significant improvements were observed in all ocular segments, with rapid improvement noted during the first 7 days of treatment in most cases. Residual chorioretinal lesions and vitreal cells persisted in a minority of cases.
Conclusions and relevance
This study suggests that ocular abnormalities in cats with FIP are more common than previously reported. Potentially vision-threatening posterior segment changes occurred frequently, and treatment with oral GS-441524 led to rapid improvement in most ocular lesions, particularly uveitis and chorioretinitis. These findings support the inclusion of ophthalmic examination as part of the clinical evaluation of cats with suspected or diagnosed FIP.
Introduction
Feline coronavirus (FCoV) is a ubiquitous RNA virus affecting felids worldwide. In a small percentage of infected cats, spontaneous mutation enables FCoV to effectively target monocytes and macrophages,1,2 leading to severe disease known as feline infectious peritonitis (FIP). FIP manifestations range from abdominal and/or thoracic effusions to granulomatous inflammation of parenchymatous organs, including the kidneys, mesenteric lymph nodes, bowel wall, liver, central nervous system and eyes.1,3 –8 Although previously fatal, FIP-diseased cats have shown an excellent response to antiviral therapies in recent years, particularly to the nucleoside analogue remdesivir (prodrug) and its active form GS-441524.4,9 –15 With the recognition of effective and tolerated treatments, there is a paradigm shift from preventing mortality to mitigating morbidity in diseased individuals. 7
Ocular manifestations of FIP arise from infiltration of infected monocytes and macrophages into ocular tissues. Disruption of the blood–ocular barrier allows FCoV-laden macrophages to access the usually immune privileged eye, contributing to fibrinous uveitis and granulomatous inflammation throughout ocular tissues. 16 In the anterior chamber, inflammatory cells may deposit on the corneal endothelium as keratic precipitates. The inflammatory response also results in pyogranulomatous vasculitis, manifesting clinically as anterior uveitis and chorioretinitis.4,15,16 Along with pain, uveitis can cause adhesions between the iris and the lens (posterior synechiae), cataract formation and pre-iridal fibrovascular membranes, with potential development of secondary glaucoma. In the posterior segment, exudates may accumulate in the subretinal space, leading to retinal detachments. Chorioretinitis and retinal detachments cause vision impairment, and if significant and bilateral, can lead to complete and possibly permanent blindness.17,18
Several studies have reported clinical ocular findings in cats with FIP.3,6,19 –23 Many of these were retrospective in nature or involved relatively small cohorts, reflecting the challenges inherent in investigating this disease. Histopathological studies have identified ocular involvement in a higher proportion of affected cats, suggesting that some ocular manifestations may not be evident on routine clinical examination.16,21 More recently, a limited number of studies have reported recovery in cats with ocular manifestations of FIP during antiviral therapy, although detailed descriptions of specific ocular changes and follow-up duration have varied between studies.15,19,23,24
To date, no prospective studies using a standardised, semi-quantitative ophthalmic examination have reported on the prevalence or characterisation of ocular findings associated with FIP. In addition, there is a knowledge gap regarding the progression of ocular manifestations in cats receiving antiviral therapy. The primary aim of this study was to document, characterise and report the presence and nature of ocular findings in cats diagnosed with FIP. The secondary aim was to describe the clinical changes in ocular manifestations throughout the course of antiviral treatment.
Materials and methods
Study design
Study population
This study was approved by the governmental veterinary office (TVB number ZH124/2022; 34964). Client-owned cats with all clinical manifestations of FIP were prospectively enrolled as part of an ongoing study evaluating a 42-day oral antiviral treatment with GS-441524, its long-term adverse effects and prognostic factors for disease reoccurrence. Cats were included in this study if they met the following inclusion criteria: a confirmed diagnosis of FIP (see ‘Diagnosis of FIP’) and were available for the baseline examination and at least one follow-up examination if ophthalmic findings were identified at baseline. Cats were excluded if they were in a moribund state at presentation, if they were subsequently determined to not have FIP based on diagnostic testing, if the owners were unable to administer oral tablets for 42 days or if the owners could not attend follow-up examinations. Cats that died or were euthanased after completion of the baseline ophthalmic examination were to be retained in the study and included in the final analysis.
Diagnosis of FIP
A diagnosis of FIP followed the current recommendations established by the European Advisory Board on Cat Diseases, 7 with diagnostics and interpretation undertaken by the FIP study team at the Vetsuisse Faculty, University of Zurich, under the supervision of board-certified American College of Veterinary Internal Medicine/European College of Veterinary Internal Medicine diplomates. An FIP diagnosis was confirmed through identification of high viral loads in effusion, tissues (sampled by ultrasound-guided fine-needle aspiration), cerebrospinal fluid or aqueous humour by semi-quantitative RT-PCR (RT–qPCR) or direct detection of the FCoV antigen in macrophages via immunocytochemistry in a cat with compatible anamnestic, clinical and clinicopathological findings.7,8,25
Data collection
Patient signalment, including breed, age, sex and neuter status, was collected for all patients enrolled in this study. On the first day of enrolment, before administration of GS-441524, a comprehensive assessment involving detailed medical history, thorough physical examination, thoracic and abdominal ultrasound, haematology, serum biochemistry, measurement of acute phase proteins including serum amyloid A (SAA) and alpha-1-acid glycoprotein (AAG) along with urinalysis was performed. Cats were tested for retrovirus infections (rapid testing feline immunodeficiency virus antibodies and feline leukaemia virus [FeLV] antigen, followed by Western Blot and FeLV provirus PCR, respectively, in case of positive results). History of prior or concurrent ocular complaints was recorded if reported. Finally, if cats were to meet the inclusion criteria (see ‘Study population’), an ophthalmic examination was performed.
Standardised ophthalmic diagnostic evaluation
Ophthalmic examination
A complete ophthalmic examination was performed on all enrolled cats, with a ‘baseline examination’ defined as taking place during the first 3 days after study enrolment and initiation of antiviral therapy. Examinations were conducted by board-certified European College of Veterinary Ophthalmologists (ECVO)/American College of Veterinary Ophthalmologists (ACVO) diplomates, by ECVO-trained residents and by ophthalmology specialty interns from the Ophthalmology Section at the Vetsuisse Faculty, University of Zurich, under the supervision of ECVO/ACVO diplomates. Ophthalmic examinations included evaluation of menace response and reflexes (palpebral, dazzle, indirect and direct pupillary light reflex), slit-lamp biomicroscopy (Kowa SL–17 and Kowa SL–19; Kowa), rebound tonometry (TonoVet tonometer; iCare) and fluorescein staining. Indirect ophthalmoscopy (Heine Omega 500 and 600 binocular ophthalmoscopes; Heine Optotechnik) was performed 20 mins after instillation of tropicamide (Tropicamide 0.5%; Théa Pharma) using 28D, 20D (BIO Lens; Volk Optical) and 2.2D (Pan Retinal; Volk Optical) condensing lenses. Cats with no ophthalmic abnormalities identified during the baseline examination were not examined again during the study time frame. If abnormalities were identified, with the exception of minor fluorescein stippling or conjunctivitis, a complete ophthalmic examination was repeated on days 7 and 42, after commencement of antiviral therapy. Additional rechecks were performed between days 7 and 42 as needed for patients with more severe clinical signs and therapy was adjusted accordingly. As with baseline examination assessments, rechecks on days 7 and 42 were permitted within 3 days of these time points to encourage compliance.
Quantification of disease severity
For each scheduled ophthalmic examination, a standardised examination protocol was used (supplementary material A). Severity of ocular disease was quantified using a modified McDonald-Shadduck Scoring System,26 –28 enabling semi-quantitative assessment of clinical findings based on well-defined criteria (supplementary material B).
Photographic documentation
Macrophotography was used to document anterior segment changes if present (Z6 mirrorless camera and AFsS VR Micro Nikkor 105 mm lens; Nikon). Similarly, if fundic abnormalities were identified, fundic photography was used to document changes (Aurora Retinal Module; Optomed).
Treatment and standardised therapy
Systemic medication
All enrolled patients underwent treatment with GS-441524 (GS-441524; BOVA UK), which was administered orally at a dose of 15 mg/kg (rounded to the nearest quarter of a 50 mg tablet) once daily for 42 days. Patients with laboratory or clinical signs suggestive of secondary infections were treated with one or more systemic antimicrobials. As some referred patients were already receiving systemic antimicrobial therapy, a standardised antimicrobial regimen was not applied. Cats with neurological involvement were administered anti-epileptic medication when indicated, such as levetiracetam (Levetiracetam; UCB-Pharma). Systemic prednisolone (Prednisolon; Graeub) was administered to patients exhibiting clinically significant secondary immune-mediated inflammatory responses or neurological signs. When indicated, additional therapy, such as anti-emetics, analgesics, appetite stimulants and intravenous fluid therapy, was administered. Dosages and combinations of systemic medications (excluding GS-441524) were determined based on patient presentation and clinical acumen.
Topical medication
Patients received topical steroid treatment 1% w/v prednisolone acetate (Pred Forte; Allergan) if anterior uveitis was present in the absence of corneal disease. The decision to treat with topical steroids, as well as duration and frequency of administration, was based on individual assessment and clinician discretion. Topical lubrication, including Carbopol 980 NF (Ocrygel; Dômes Pharma), 1.2% Hyaluronate (AnHypro; An-vision) and/or 5% Dexpanthenol (Bepanthen; Bayer), was initiated if corneal erosions were present.
Results
Study cohort
Study population demographics
The study cohort comprised 147 cats provisionally enrolled over a 1-year period (July 2023 to June 2024), as recorded at the time of manuscript preparation. A total of 29 cats were subsequently excluded from the study: 21 because of lack of owner compliance or comorbidities that rendered them unsuitable for oral therapy and necessitated an alternative (intravenous) treatment; five for insufficient evidence of FIP after further diagnostic evaluation, as laboratory results were pending or equivocal at the time of recruitment; and three that died or were euthanased before a baseline ophthalmic examination could be performed. An additional five cats died or were euthanased after completion of the baseline examination; however, their baseline examination findings were included in the analysis (see ‘Baseline examination’). The final study cohort therefore consisted of 118 cats (75 males, 43 females; age range 3–165 months; median age 12.5 months) (Table 1).
Description of the study cohort including breed, age, sex and neuter status distribution (n = 118)

Data are n (%)
Siberian, Persian, Ragdoll, Siamese, Somali, Savannah (with each breed accounting for less than five cats in total)
BLH = British Longhair; BSH = British Shorthair; DSH = domestic shorthair
Ophthalmic examination time points and patient numbers
A total of 118 cats underwent baseline examinations, with 70 cats showing no ocular findings. In total, 118 left eyes and 117 right eyes were examined, as one cat had previously undergone unilateral enucleation. A total of 48 cats demonstrated ophthalmic lesions related to FIP, of which 42 received follow-up ophthalmic examinations; 37 were examined on day 7 and 39 on day 42 (Figure 1). Six cats with ocular findings at the baseline examination did not receive a follow-up examination, either per study protocol for only having inactive chorioretinal lesions that were interpreted as being unrelated to FIP (n = 3) or since lost to follow-up (n = 3, two with anterior uveitis, one with active chorioretinitis). The mean (±SD) time between the baseline examination and day 7 examination was 5.6 ± 1.0 days, and the mean time between the baseline examination and day 42 examination was 40.2 ± 1.2 days. Figure 1 illustrates patient numbers and study time points.

Flow diagram illustrating patient enrolment, ophthalmic examination time points and patient numbers. Of the 147 cats provisionally enrolled, 29 were excluded, resulting in a final cohort of 118 cats that underwent baseline ophthalmic examination. Ophthalmic abnormalities were identified in 48 cats at baseline. Follow-up examinations were performed in 37 cats on day 7 and in 39 cats on day 42. FIP = feline infectious peritonitis
Baseline examination
Overall results and affected population
Abnormalities were most frequently observed in the posterior segment, followed by the cornea and anterior chamber. Summarised findings by ocular segment are presented in Table 2, with detailed findings in supplementary material C.
Distribution of unilateral and bilateral ocular abnormalities in cats (n = 118)
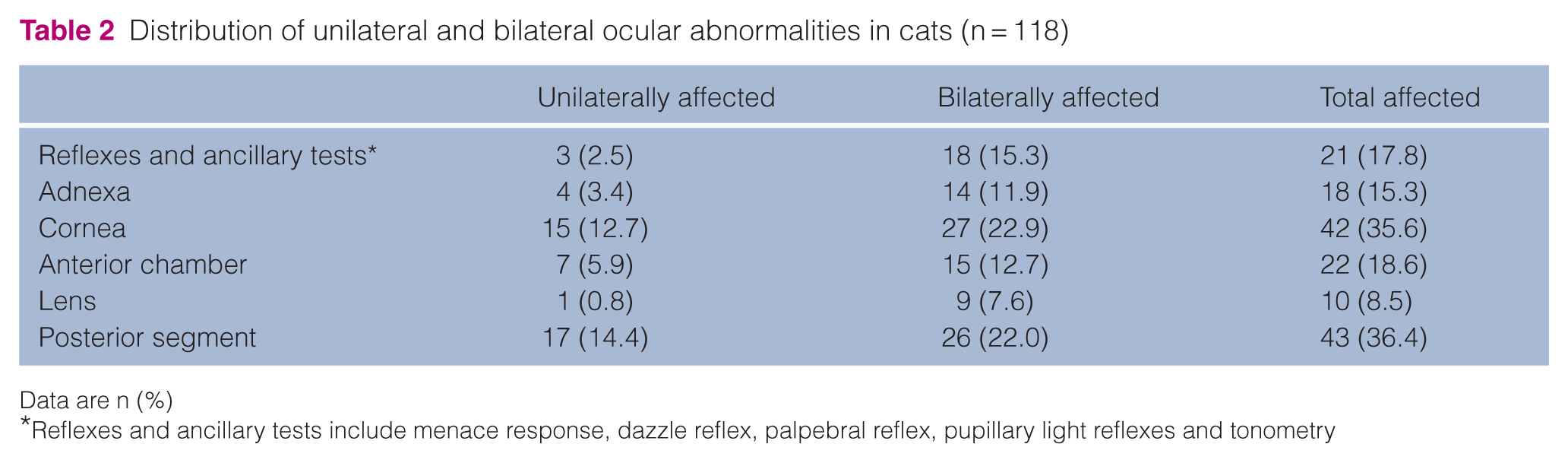
Data are n (%)
Reflexes and ancillary tests include menace response, dazzle reflex, palpebral reflex, pupillary light reflexes and tonometry
Anterior chamber
Patients exhibited a wide array of clinical findings (Figure 2a–d), including anterior chamber flare and/or cells, keratic precipitates, fibrin (Figure 2b), hyphaema, iris swelling and pre-iridal fibrovascular membranes (Figure 2a). Anterior chamber changes were noted in 22/118 (18.6%) cats, with anterior uveitis diagnosed in 20. The severity of clinical signs varied, as exampled in the fine nasoventral keratic precipitates of Figure 2c to more dense and/or haemorrhagic ‘mutton-fat’ precipitates seen in Figure 2d. A broader overview of the variability of clinical presentations can be seen in supplementary material C.
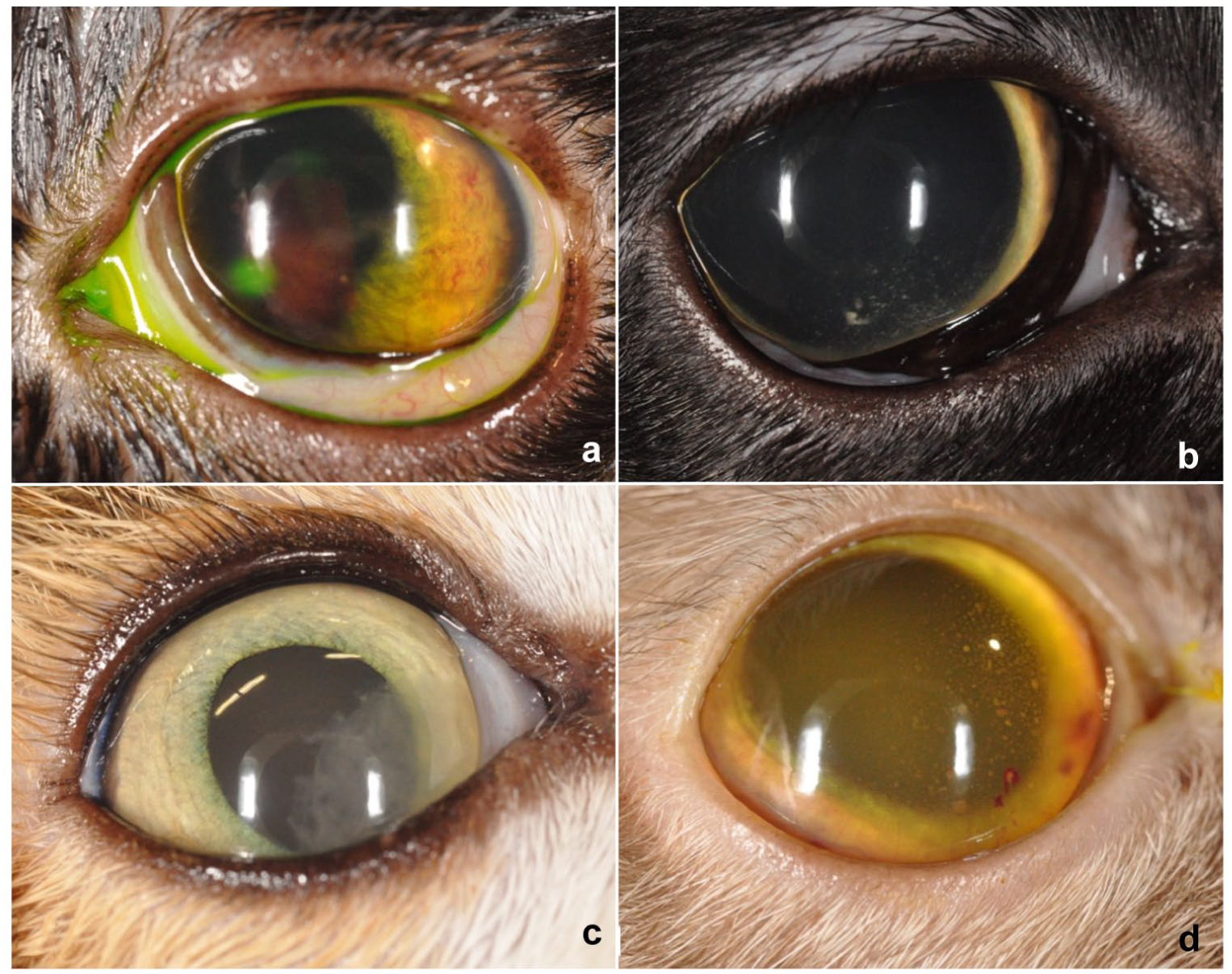
Anterior segment images of study cats diagnosed with feline infectious peritonitis. (a) A 5-month-old male entire British Longhair with fibrinous anterior uveitis and pre-iridal fibrovascular membranes. (b) A 14-month-old female spayed domestic shorthair with nasoventral keratic precipitates. (c) A 3-year-old male castrated domestic shorthair with fibrinous anterior uveitis. (d) A 9-month-old male entire Siamese with corneal neovascularisation and ‘mutton-fat’ and haemorrhagic keratic precipitates accompanied by hyphaema
Posterior segment
Posterior segment changes were noted in 43/118 (36.4%) cats, with most cats having one or more bilateral findings (Table 2 and Figure 3a–f). The most frequently identified finding was chorioretinitis, reported in 30/118 (25.4%) cats (supplementary material C), with examples in Figure 3a,b. Active chorioretinitis was observed in 16/118 (13.6%) cats, while inactive chorioretinitis was noted in 14/118 (11.9%) cats. Severe anterior chamber and/or lenticular changes precluded examination of the posterior segment in three cats. Optic nerve atrophy and cupping were not identified in any study patient.
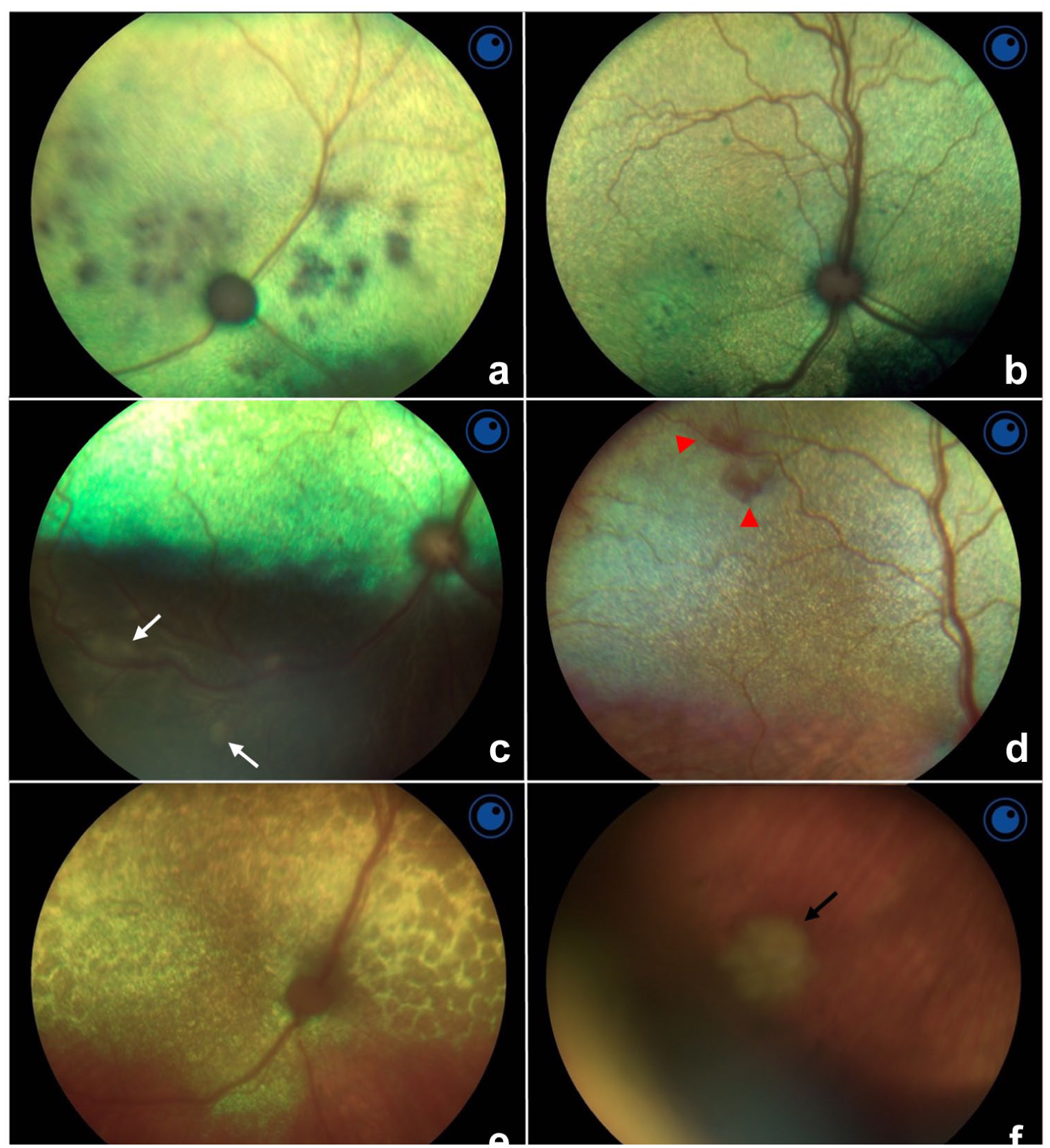
Fundic images of study cats diagnosed with feline infectious peritonitis. (a) A 5-month-old male entire British Longhair with multifocal chorioretinitis. (b) A 14-month-old female spayed domestic shorthair with mild multifocal chorioretinitis. (c) A 2-year-old domestic shorthair with perivascular infiltrates (also called ‘perivascular cuffing’), indicated by white arrows. (d) A 7-month-old male castrated Siamese with small focal bullous retinal detachments with cellular infiltrates, indicated by the red arrowheads. (e) A 9-month-old male entire Siamese with diffuse retinal oedema and cellular infiltrates. (f) A granulomatous lesion adjacent to the temporoventral ora serrata of the cat described in (e), indicated by the black arrow
Days 7 and 42 follow-up
The severity and frequency of ocular abnormalities observed decreased throughout the observation period, during which patients were treated with GS-441524. Anterior uveitis occurrence decreased from 34.7% (33/95 eyes) to 19.2% (14/73 eyes) within the first 7 days, and a further reduction to 6.5% (5/77 eyes) was observed at the ocular examination on day 42. Similarly, cases of active chorioretinitis reduced from 31.6% (30/95) to 16.4% (12/73) in the first week, with only one cat having active chorioretinitis on day 42. More perivascular infiltrates were observed at day 7 (8/73 eyes) compared with baseline examination (7/95 eyes), although this had decreased by day 42 (2/77 eyes). Although the overall frequency of retinal detachments did not change significantly from the baseline examination to day 7, changes in retinal appearance suggested partial improvement in retinal attachment (Figure 4). Retinal detachments were observed in only one cat on day 42 (Table 3).
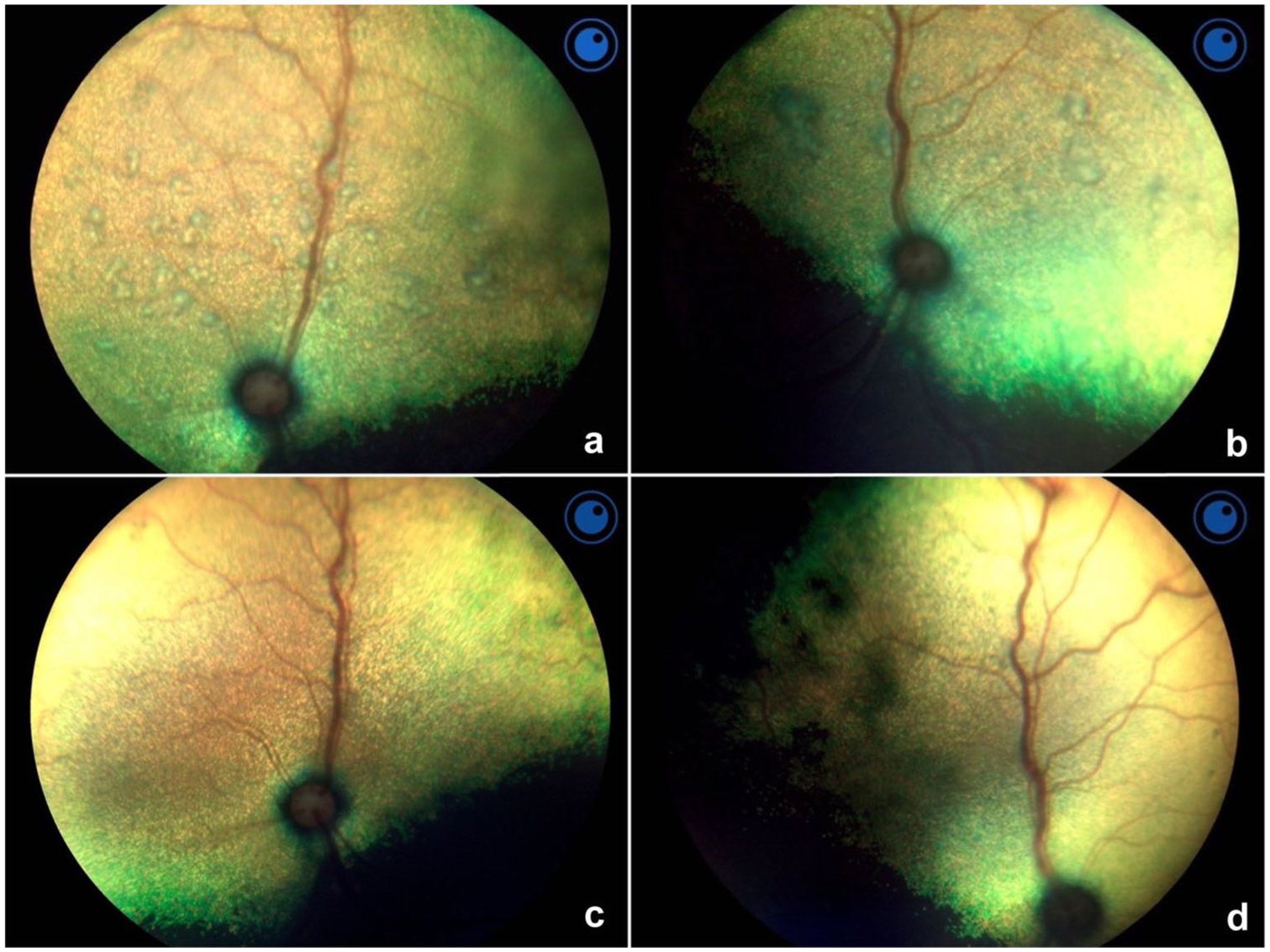
Fundic images of a two-year-old female spayed domestic shorthair cat. (a,b) Fundic images of the right and left eyes, respectively, at baseline examination showing multifocal bullous retinal detachments. (c, d) Fundic images of the right and left eyes, respectively, 5 days after commencing treatment with GS-441524, showing post-inflammatory hyperpigmented lesions
Characterisation of commonly observed ocular findings at baseline examination, day 7 and day 42 in cats affected with feline infectious peritonitis

Data are n or n (%)
Systemic steroid treatment
Oral steroid treatment was administered to 15/118 (12.7%) cats during the follow-up period. Of these, only one cat received oral corticosteroids because of marked ophthalmic manifestations, which included bilateral retinal detachments and severe panuveitis. In this case, mild anterior uveitis was noted on day 7, with subtle flare persisting bilaterally on day 42. Retinal reattachment was also noted on the day 42 examination. In total, 10 cats with significant ocular findings warranting ophthalmic follow-up received oral steroids for non-ocular reasons, the most common indication being FIP-related neurological involvement. In addition, one cat each was treated with oral steroids for secondary immune-mediated haemolytic anaemia, suspected inflammatory bowel disease, chronic rhinitis or severe pleuritis. Anterior uveitis was noted in at least one eye of four of these cats at baseline examination and had reduced in all cases by day 7, with complete resolution of uveitis by day 42. Signs of posterior uveitis were noted at baseline examination in at least one eye of two cats, with persistence at day 7 and resolution in one cat at the day 42 examination. Three cats had evidence of active chorioretinitis at baseline examination, with signs of active chorioretinitis persisting in at least one eye on day 7; however, no active lesions were observed on day 42.
Discussion
This prospective observational study investigates ocular findings in cats with FIP and reports changes observed during treatment with oral GS-441524. Almost three-quarters of the reported cats in this patient population were aged less than 2 years, similar to previous studies,6,29 –34 even though cats aged over 5 years made up almost 20% of the study population. Consistent with the published literature,13,14,20,29 –31,34 male cats were more frequently diagnosed with FIP than female cats; domestic shorthair cats were the most commonly affected breed.32 –34 Of the 118 cats, 48 (40.1%) had ophthalmic abnormalities, higher than in previous reports. A recently published study reported the prevalence of ocular findings in 4.4% of cats with FIP in Thailand, similar to the 8.6% (9/105) found in Wuhan, China.6,20 A retrospective study by Tršar et al 22 in Slovenia reported ocular findings in 28.5% of cases; however, posterior segment changes were not reported. This was similar to the 29% reported by Kipar and Meli, 4 although the original data source is unclear. Pederson 3 found varying rates between effusive (36.2%) and non-effusive (5.5%) forms of the disease, although significant crossover between the two forms challenge this simplified categorisation. 7 In the UK, Green et al 19 first reported ocular findings in 18.8% (6/32) of cats with FIP; however, a more recent observational study by Andrews et al 23 reported FIP-associated ocular findings in 33% of cats. Higher detection in this publication may reflect ophthalmic examinations by board-certified or residency-trained clinicians and the prospective study design.
Regarding specific ocular findings, 18.6% (22/118) of cats had anterior segment changes at their baseline examination, with most being bilateral. Anterior uveitis was most frequent, affecting 16.9% (20/118) of cats, lower than the 24.6% (15/61) observed in the UK, 23 although higher than the 12% (4/42 cats) reported in Sydney, Australia. 30 Specifically, pyogranulomatous uveitis in cases of FIP is characterised by the production of fibrin, infiltration of macrophages and other inflammatory cells, likely explaining keratic precipitates in over 10% (12/118) of cats in this study. Despite the pyogranulomatous nature of FIP-associated ocular manifestations, observation of uveitis in the reported cases cannot definitively be attributed to FIP alone as not all causes of uveitis were excluded.
Posterior segment changes were observed in 36.4% (43/118) of cats, with chorioretinitis affecting over one-quarter of the study population. Of chorioretinitis cases, over one-third were considered inactive at baseline examination. Although chorioretinitis may be unrelated to FIP, another proposed explanation is the tendency towards a protracted or recrudescent manifestation of the disease, historically associated with ocular involvement.3,35,36 Small or focal chorioretinal lesions were also difficult to distinguish as active or inactive and were graded according to observer discretion. Consistent with previous studies,16,17,37,38 chorioretinitis was observed frequently and granulomatous infiltrates were seen in the posterior segment. This is also consistent with histological studies, as morphological features of granulomatous vasculitis in venules of the iris, choroid, retina, optic nerve and sclera are recognised in cats with FIP.36,39 Although the observation of chorioretinitis was not specifically reported by Andrews et al, 23 26% (16/61) of cats had posterior uveitis, similar to the 25% (29/118) of cats with posterior segment changes in our study when inactive chorioretinitis was excluded.
Adnexal and corneal changes were common in this population, with conjunctival inflammation, corneal opacity and ulceration or erosion most frequently observed. Corneal oedema has been previously documented in cats with FIP,6,20,40 while conjunctivitis was reported in only one study. 22 Histological investigations have identified inflammatory infiltrates containing viral antigen-laden macrophages in the conjunctiva and nictitating membrane, suggesting that FIP can involve adnexal tissues. However, the virus appears to have lower affinity for these structures than other ocular tissues, as viral antigens were identified in the nictitating membrane of only 8% of infected cats. 41 Affinity for corneal tissue may be higher, as histological evidence of keratitis was found in 16.7% of cats in another study, 36 similar to the rate of corneal opacification observed in our population (14.4%).
Corneal epithelial or stromal compromise was also frequent, with fluorescein uptake noted in over one-third of cats. Several extrinsic factors may have contributed to these corneal changes. Cats with respiratory disease often required oxygen supplementation via oxygen cages, which have been shown to increase the risk of development of ulceration in hospitalised patients after anaesthesia. 42 Neurological impairment may have reduced blink frequency, further predisposing to corneal erosion. In addition, some cats received opioid analgesics or mild sedation for ancillary tests. Although the effects of sedatives on feline tear production are poorly described,40,43,44 canine studies suggest they reduce tear production and blink frequency, promoting corneal desiccation and epithelial defects.45 –48 Hospitalisation itself has been shown to disrupt normal tear production in cats 49 and may contribute to recrudescence of ocular signs in those co-infected with feline herpesvirus type-1.50,51 Thus, it is of the authors’ opinion that the development of conjunctival and corneal changes in the present study are multifactorial in nature and should be interpreted with caution with regard to causative association with FIP.
The secondary aim of the study was to observe the progression of ocular findings during treatment with GS-441524. Cases of anterior and posterior uveitis, keratic precipitates, active chorioretinitis and retinal detachments decreased throughout the 42-day observation period, while cases of inactive chorioretinitis increased overall. Perivascular infiltrates and retinal detachments were noted more frequently on day 7 compared with baseline, but reduced to baseline values or below by day 42. This may be due to delayed development or obscured fundic visualisation due to corneal changes or uveitis at the baseline examination. Other than these exceptions, there appears to be a rapid improvement of inflammation within the first week of treatment, with evidence of persistent inflammation in very few cats on day 42, possibly due to varying drug properties in differing states of ocular disease and the tendency for vitreal cells to persist even after the active inflammation has settled down. Overall, significant improvement in vision-threatening ocular disease was observed throughout therapy with GS-441524. Timing variability in rechecks, with mean intervals of 5.6 ± 1 and 40.2 ± 1.2 days should be noted, as there was an overall shorter-than-intended recheck interval.
Epidemiological factors associated with FIP may influence regional differences in clinical presentation, limiting the generalisability of this study’s findings. 30 Although difficult to implement, a key limitation of this study is the absence of a control group. To represent the study cohort, control cases would need to be of similar ages and breeds originating from comparable geographic locations to be representative. Regarding ocular examinations, the involvement of multiple examiners with various experience may have led to inferred inter-observer variability; however, this was limited in nature because of the use of a standardised ophthalmic examination protocol. Furthermore, 15/118 (12.7%) cats received topical and systemic medications (notably corticosteroids) in addition to GS-441524, making it difficult to attribute improvements solely to antiviral therapy. Of the 15 cats in the study cohort receiving oral steroids, 11 were re-examined at days 7 and 42 because of FIP-related ophthalmic findings, even though steroids were prescribed for non-ophthalmic reasons in most cases. In the patients who received corticosteroids, it cannot be determined whether the improvement in ocular disease was attributable to systemic antiviral therapy alone.
The data presented reflects preliminary findings from the first 118 cats in an ongoing larger scale prospective study. This early descriptive publication aims to provide clinicians with valuable insights into ocular findings in cats affected by FIP. Although statistical analysis is not yet included, the focus here is on observational data. Future studies will aim to compare cases with and without adjunctive systemic or topical steroid therapy, as well as post-treatment observations. In addition, associations between clinical markers such as systemic signs and laboratory parameters and ophthalmic findings will be explored to assess their potential prognostic value for the resolution of ocular disease (Spiri et al, manuscript in preparation). Such analyses will be valuable given recent reports of FIP recurrence after cessation of antiviral therapy,52,53 as well as possible adverse drug reactions.
Conclusions
The present results suggest a higher occurrence of ocular findings in cats with FIP than previously reported. The most common ocular findings observed – anterior uveitis, presence of keratic precipitates and chorioretinitis – were consistent with previous studies. Corneal pathology was frequent in examined cats, although its development is likely multifactorial. Posterior segment changes were observed in 36.4% of cats, thus clinicians managing cases of FIP are encouraged to perform thorough ocular examinations, including posterior segment evaluation using mydriatics, at the time of FIP diagnosis.
Supplemental Material
Supplementary material A
Ophthalmic examination protocol.
Supplemental Material
Supplementary material B
Modified McDonald-Shadduck Scoring System.
Supplemental Material
Supplementary material C
Results of ocular findings in each segment at baseline examination.
Footnotes
Acknowledgements
The authors would like to thank Alessandro Contadini, Jeannine Dumelin Dobler, Arno Hirtler and Niklas Holz, Angelie Shukla, Andrea Isler and Simone Salathe for their involvement in ophthalmic examinations. Gratitude is also extended to Celia de Witt Curtius, Julia Stachowski and Jennifer Wenk from the University of Zürich for their involvement in participant recruitment and data collection. The laboratory work was performed using the logistics of the Centre for Clinical Studies at the Vetsuisse Faculty of the University of Zürich.
Author note
Data are available upon reasonable request from the authors.
Supplementary material
The following files are available as supplementary material:
Supplementary material A: Ophthalmic examination protocol.
Supplementary material B: Modified McDonald-Shadduck Scoring System.
Supplementary material C: Results of ocular findings in each segment at baseline examination.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was partly funded by the Schweizerische Vereinigung für Kleintiermedizin (Swiss Association of Small Animal Medicine), the Stiftung für Kleintiere (Foundation for Small Animals) of the University of Zürich Vetsuisse Faculty, the University of Zürich Global Strategy and Partnerships Funding and the Stiftung für wissenschaftliche Forschung (Foundation for Scientific Research) of the University of Zürich.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
