Abstract
Objectives
The primary aim of this prospective observational study was to document clinical progression, survival, remission and relapse in New Zealand cats with feline infectious peritonitis (FIP) that were treated with compounded oral remdesivir with or without initial parenteral remdesivir therapy. The secondary aims were to determine the optimal monitoring protocols and report the adverse effects of treatment and complications associated with FIP.
Methods
A total of 29 client-owned cats with a clinical diagnosis of FIP were prospectively recruited. Cats were administered oral remdesivir (30 mg/kg q24h), rounded up to the nearest capsule size as the sole treatment, or after initial parenteral remdesivir (15–30 mg/kg q24h). Rechecks were recommended at weeks 1, 2, 4, 8 and 12 during treatment, and at 2 and 12 weeks after treatment. A full physical examination (including neurological and ophthalmic examination) and point-of-care ultrasound were performed at each recheck. A complete blood count and biochemistry panel were performed at weeks 4, 8, 12 and 24. Molnupiravir was offered as a rescue therapy for cats that relapsed.
Results
In total, 25 (86%) cats entered remission and survived beyond 6 months (range 6–27). A total of 22 (75%) cats achieved remission within 84 days while receiving oral remdesivir. Three cats received subsequent molnupiravir rescue therapy to achieve remission. Five cats (20%) experienced relapse: four with non-effusive disease and one with effusive disease. Notably, 4/8 (50%) non-effusive cases relapsed, compared with only 1/20 (5%) with effusive disease.
Conclusions and relevance
This study demonstrates that oral remdesivir at a dose rate of 30 mg/kg q24h is an effective treatment for effusive FIP. The survival rate in non-effusive cats was significantly lower; therefore, an increased dose rate or frequency of administration should be considered in these cats. Oral remdesivir is a viable antiviral option where GS-441524 is unavailable.
Keywords
Introduction
Feline infectious peritonitis (FIP) is a disease that affects domestic cats caused by virulent feline coronavirus (FCoV). 1 In a minority of exposed cats, FIP may develop, causing pyogranulomatous vasculitis and serositis.2 –4
The clinical presentation of FIP can be broadly divided into an effusive form, which is characterised by the presence of high-protein effusion within body cavities, and the non-effusive form, characterised by pyogranulomatous mass lesions. Either forms may demonstrate concurrent ocular or neurological signs and likely represent a disease continuum.1,5
Without treatment, almost all cats that develop FIP will die of the disease. 6 The successful treatment of FIP using the antiviral drug GS-441524 was first published in 2018. 7 Since that publication, treatment of FIP with GS-441524 and its prodrug remdesivir (RDV) has demonstrated high survival rates (81–100%) and favourable safety profiles.8 –14
GS-441524, the prominent intermediate metabolite of RDV, is only available in a small proportion of countries and provides an alternative to daily RDV injections. At the time of publication, there is no legal source of GS-441525 in New Zealand and veterinarians rely on compounded RDV to treat FIP.
RDV is a nucleotide analogue that inhibits viral RNA synthesis. 15 In New Zealand, RDV is available as a compounded medication.
Clinical experience has demonstrated that RDV is painful on subcutaneous (SC) injection and cost-prohibitive for many cat owners. 9 This has prompted New Zealand veterinarians to prescribe compounded oral RDV as an alternative to parenteral RDV in many cases.
However, the bioavailability of oral RDV in cats with FIP has not been determined, whereas the bioavailability of oral RDV in dogs and people has been demonstrated to be low.16,17 A recent study demonstrated that the metabolism of RDV is different in cats compared with dogs and humans, with conversion to GS-441524 occurring in whole blood (not requiring phase 1 hepatic metabolism). 18 Bioavailability data collected by Cook et al 19 in three healthy cats treated with oral RDV revealed that GS-441524 plasma concentrations greater than the established corresponding EC50 were achieved and sustained for 24 h with dosages of 25 mg/kg PO q24h. This suggests that oral RDV may have higher bioavailability in cats than other species. It is important to note that the EC50 determined from in vitro cell cultures may not necessarily reflect the drug’s distribution within tissues (particularly immune-privileged sites such as the central nervous system and eye). Moreover, GS-441524 undergoes intracellular conversion to a pharmacologically active triphosphate, and the relationship between plasma drug concentrations and the active triphosphate concentrations within tissue macrophages (the primary target cell) remains unknown.
The primary aim of this prospective observational study was to document clinical progression, survival, remission and relapse in New Zealand cats with FIP, treated with compounded oral RDV with or without parenteral RDV therapy. The secondary aims were to determine the optimal monitoring protocols, report the adverse effects of treatment and complications associated with FIP.
Materials and methods
Client-owned cats with a clinical diagnosis of FIP were prospectively recruited. All cats had clinical and clinicopathological signs consistent with FIP and achieved a presumptive diagnosis based on positive FCoV RT-PCR as per European Advisory Board on Cat Diseases (ABCD) guidelines as determined by a board-certified specialist. 1 At the time of writing, FCoV immunohistochemistry (IHC) is not available within New Zealand.
Cats were excluded if treated with antiviral medication before enrolment.
Oral and injectable RDV was sourced from a compounding pharmacy (Optimus Healthcare). All cats received oral RDV, either as the sole treatment or after initial parenteral RDV. Treatment protocols were non-randomised: owners chose the treatment protocol after consultation with an internal medicine specialist. Parenteral RDV was administered at a dosage of 15–20 mg/kg q24h in some cats before transitioning to oral treatment. Table 1 summarises the parenteral treatment protocols. Five cats received a single intravenous (IV) injection of RDV 30 mg/kg as part of a separate pharmacokinetic study. Parenteral RDV was administered as a slow IV bolus over 20 mins in the hospital and SC after discharge. All cats received RDV (30 mg/kg PO q24h) rounded up to the nearest 30 mg capsule. Owners were encouraged to administer capsules whole without food. All cats were treated for a minimum of 84 days.
Parenteral dose rates of remdesivir

At the time of enrolment and at all subsequent rechecks, cats had a full physical, neurological and ophthalmic examination performed by an internal medicine specialist. Point-of-care ultrasound was performed at each appointment to assess effusions. All cats had 2.6 ml of blood collected with a 21 G needle and 3 ml syringe into a 1.3 ml EDTA and 1.3 ml non-additive tube for a complete blood count and biochemistry panel before starting RDV.
Rechecks were recommended at weeks 1, 2, 4, 8 and 12 during treatment and at 2 and 12 weeks after finishing treatment.
The dose of RDV administered was revised with weight gain to ensure cats received a minimum dose rate of 30 mg/kg. The dose was also increased by 30 mg if specific clinical or clinicopathological milestones (Table 2) were unmet.
Treatment milestones

Remission is defined as being free of clinical and clinicopathological signs associated with FIP 3 months after treatment cessation. Owners of cats that did not enter remission or relapsed had the option to either increase the dose of RDV and extend treatment duration or change to molnupiravir (MPV). Relapse was defined as recurrence of initial clinical/clinicopathological signs or development of new clinical/clinicopathological signs known to be associated with FIP.
Long-term follow-up was performed via appointment, telephone call or email.
This study was approved by the Massey University Animal Ethics Committee (AEC 22/67).
Analysis of compounded RDV capsules
The quantity of RDV contained within capsules that were dispensed as 60, 90, 120 mg were measured using liquid chromatography equipped with fluorescence detection (excitation: 250 nm/emission: 475 nm) (Nexera XR LC system; Shimadzu Corporation) modified from our previously established method. 20 Accordingly, we utilised an XBridge C18 (Waters), 3.5 µm, 4.6 × 150 mm, and an isocratic mobile phase comprising a 20 mM ammonium acetate buffer (pH 4.5 adjusted with acetic acid) and 58.5% of acetonitrile to separate and elute RDV. The retention time for RDV was 2.9 mins and was quantified using an external calibration curve prepared with RDV standard (GS-5734, CAS 1809249-37-3) in the mobile phase. The calibration curve demonstrated an accuracy and precision of over 99%. The measured RDV content, expressed as mean ± SD (n = 3), in the 60, 90 and 120 mg capsules was 58.16 ± 4.07, 89.87 ± 1.37 and 123.58 ± 0.83, respectively, corresponding to 97–103% of claimed amounts (see Table 1 in the supplementary material).
Statistical analysis
All statistical analyses were conducted in R Studio (v2024.04.0+735) using R version 4.4.0. Figures were produced using ggplot2.
Fisher’s exact test was used to compare the proportion of cats that relapsed in the non-effusive group with the proportion that relapsed in the effusive group. A P value <0.05 was considered significant, implying a difference in proportions.
Results
Study cohort
A total of 29 cats with a clinical diagnosis of FIP were included. Of the cats, 22 (76%) were aged ⩽1 year at diagnosis. Tables 3 and 4 show the breeds and neuter status, respectively.
Breeds of cats (n = 29)

Neuter status of cats (n = 29)

At the time of diagnosis, 21 cats had effusive FIP and eight had non-effusive FIP. Of the non-effusive cats, five had neurological signs, of which three also had anterior uveitis. One cat had anterior uveitis, one had abdominal lymphadenomegaly and one had an ileocolic mass. Neurological signs, where present, included seizures, ataxia, paresis and altered mentation (see Table 2 in the supplementary material for more case details).
Of the cats with effusion, 16 (76%) had abdominal effusion, four (19%) had pleural effusion and one had pleural and pericardial effusions.
Tables 5 and 6 show the presenting clinical and clinicopathological signs, respectively.
Clinical signs observed at the time of diagnosis in 29 cats with feline infectious peritonitis

Clinicopathological abnormalities observed at time of diagnosis in 29 cats with feline infectious peritonitis

Table 7 summarises the diagnostic tests used to confirm FIP. In total, 28 cats had a positive feline enteric coronavirus antigen RT-PCR, whereas one cat had a positive FCoV IHC on an omental biopsy.
Cats with effusive and non-effusive feline infectious peritonitis (FIP) that tested positive to each confirmatory test

CSF = cerebrospinal fluid; IHC = immunohistochemistry
Antiviral treatment regimens
A total of 20 cats received parenteral RDV before transitioning to oral RDV. Of these cats, seven received one dose, six received two doses, six received 14 doses and one cat received 28 doses parenterally. The mean dose rate of parenteral RDV was 37 mg/kg.
Four cats received MPV 15 mg/kg PO q12h as rescue therapy.
Remission and survival
Figure 1 displays the outcomes of individual cats. In total, 25 (86%) cats entered remission and survived beyond 6 months (range 6–27), whereas 22 (75%) cats achieved remission within 84 days while receiving oral RDV. Three cats received MPV rescue therapy to achieve remission.
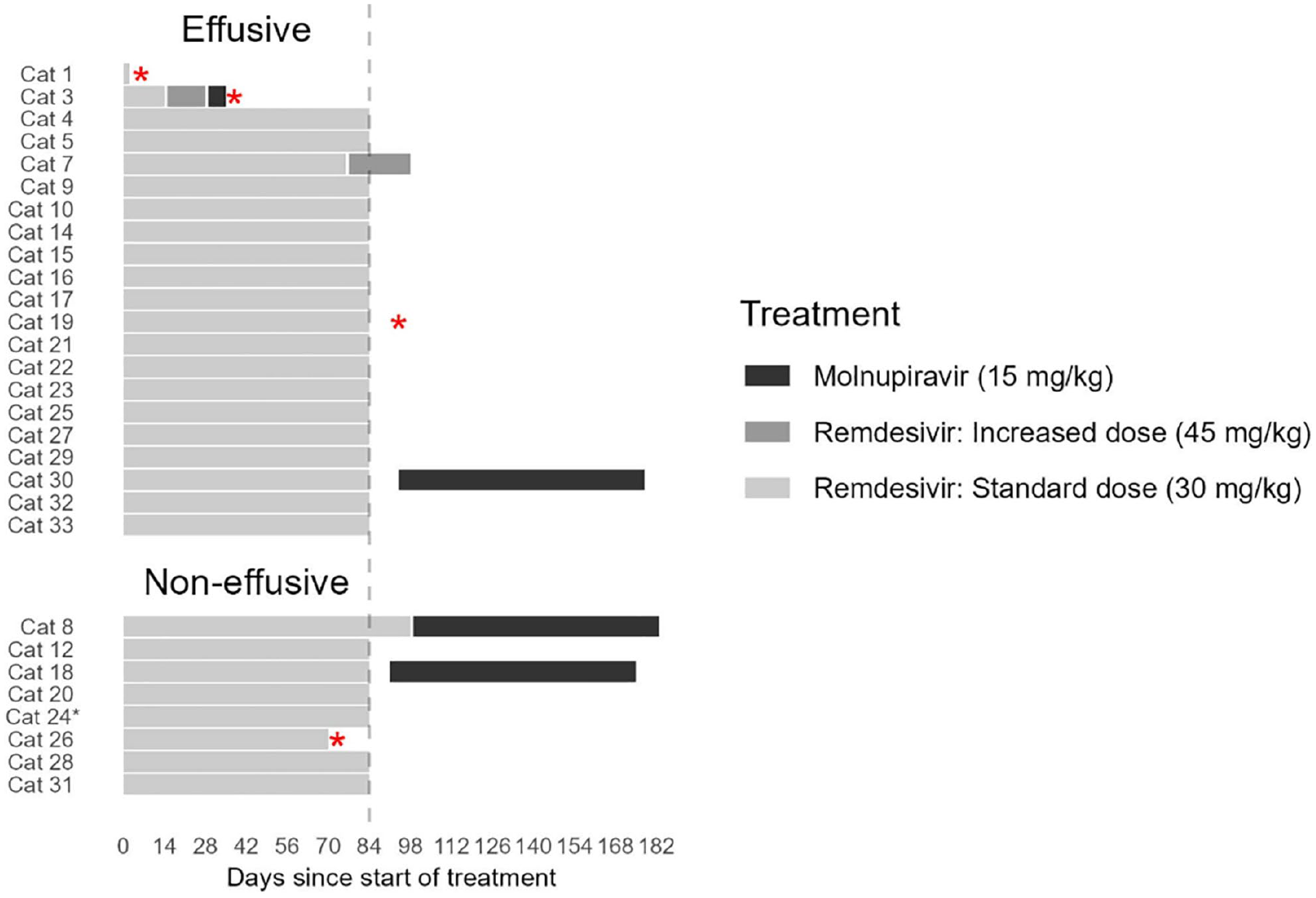
Duration of treatment and treatment protocol received in 29 cats with feline infectious peritonitis primarily treated with remdesivir (RDV) and rescue therapy with molnupiravir (MPV). Red asterisks indicate deaths. *Cat 24 relapsed 509 days after starting the primary treatment and was treated with a second course of RDV for 84 days. It relapsed a second time on day 628 and was treated with MPV. The cat was lost to follow-up after starting MPV
Four cats were euthanased during the study period: one was euthanased 3 days after starting treatment because of acute deterioration; one failed to respond to treatment and developed congestive heart failure at week 5; one was euthanased after relapse; and one presented with gastrointestinal blood loss 1 week after cessation of treatment. Case details are provided in Table 3 in the supplementary material.
Long-term follow-up information was available for 21/25 surviving cats. All 21 cats were alive 6–27 months after completing treatment.
Relapse
Five cats relapsed during the study period, with six relapse events in total. Four cats had non-effusive disease (n = 8) and one had effusive disease (n = 21). Fisher’s exact test supports that the true proportion of relapse in non-effusive cats is higher than in effusive cats (P = 0.013)
Two cats relapsed during the primary course of oral RDV after initial improvement and meeting the milestones in Table 2. One cat entered remission after treatment with MPV, whereas the second cat was euthanased because of severe neurological disease.
Four relapse events occurred after cessation of treatment. Two cats achieved remission after receiving 84 days of MPV. One cat relapsed twice, with one relapse occurring after entering remission and the second relapse 5 weeks after finishing treatment. This cat was lost to follow-up after its second relapse.
Of the five cats that relapsed, four presented with clinical signs different from their initial signs. See Table 4 in the supplementary material for details on the cats that relapsed.
RDV dose escalation
The dose rate of RDV was increased during treatment seven times across five cats. Table 8 shows the justification for the dose escalation.
Reasons for remdesivir dose increases during treatment

Cat 7 re-presented at week 9 for acute onset vomiting and lethargy. An abdominal ultrasound revealed marked fluid distension of the stomach and duodenum, consistent with a mechanical intestinal obstruction. An exploratory laparotomy revealed that a fibrous adhesion arising from the mesentery had caused focal narrowing of the duodenum and dilation of duodenum orad to the adhesion. FCoV RT-PCR was performed on this tissue and was negative. The oral RDV dose rate was increased at this time, with the cat receiving 14 weeks of RDV in total. This cat entered remission without further complications.
Clinicopathological findings during treatment
Figure 2 shows resolution of clinicopathological parameters at weeks 0 to 12.

Clinicopathological parameters during treatment at weeks 0, 4, 8 and 12. A:G ratio = albumin:globulin ratio; ALT = alanine transaminase; PCV = packed cell volume
Adverse events
Of the eight cats that received SC RDV, four (50%) had adverse reactions, including crusting, superficial ulceration, subcutaneous swelling and pain at the injection site. Two cats experienced severe reactions. One cat showed aggression to their owner when injections were being given, which was perceived to be due to a stinging sensation. This cat was also reported to be lethargic for 2 h after the injection. The second cat developed a skin ulcer at the injection site measuring 1 cm × 2 cm. No adverse reactions to IV or oral RDV were reported.
Post-mortem examinations
A post-mortem examination was performed on cat 26 after euthanasia because of severe relapse of neurological signs. Histology revealed mild to moderate multifocal lymphoplasmacytic vasculitis and spongiosis within sections of brainstem, cerebellum and cerebrum. All other tissues examined were within normal histological limits. Post-mortem examinations were declined by the owners of the other three cats that were euthanased.
Discussion
To the authors’ knowledge, this is the largest study to report the successful treatment of cats with effusive and non-effusive FIP using oral RDV. It is also the first publication on the treatment of FIP in New Zealand cats.
The survival rate in this study (86%) is comparable to that published by Cosaro et al, 21 who reported a survival rate of 79% in nine cats with effusive FIP treated with oral RDV. Survival rates of cats treated with oral GS-441524, parenteral RDV and MPV are also similar to this study, and are in the range of 78–96%.8,9,22
The relapse rate in this study was 17%, which is similar to those reported in studies treating cats with GS-441525 and RDV.9,13,21 In this study, 4/5 (80%) cats that relapsed had neurological and ocular disease at initial diagnosis. Of the cats with ocular/neurological signs, 5/6 (83%) had poor outcomes, with either relapse or a lack of a sufficient response to treatment. In contrast, of the cats that relapsed in the study by Coggins et al, 9 83% had effusive disease. The non-effusive population in this study had predominantly ocular/neurological signs, whereas only one of the non-effusive cats in the study by Coggins et al 9 had ocular disease and none had neurological disease. The two non-effusive cats without neurological/ocular signs in this study had uncomplicated remissions. This suggests that the presence of ocular or neurological disease may impact treatment success more than the presence or absence of effusion.
A greater proportion of cats in the non-effusive group (50%) relapsed compared with the effusive group (5%). The blood–brain barrier and blood–ocular barrier are suspected to contribute to incomplete response to treatment or relapse because of reduced concentrations of active metabolites within the cerebrospinal fluid and aqueous humour. 7 Murphy et al 7 reported that the combined levels of GS-441524 in aqueous humour and cerebrospinal fluid was one-eighth of blood plasma after SC administration in healthy cats. Consequently, higher doses of RDV and GS-441524 have been recommended in cats with ocular or neurological forms of FIP.10,22,23 The dose of RDV selected for this study was based on the oral dose required to achieve plasma concentrations above the EC50 (half maximal effective concentration) determined by Cook et al. 19 While the effusive cases in this study achieved similar survival rates to other treatment protocols, the non-effusive cases had significantly poorer outcomes. This supports that the dose rate used was appropriate for cats with effusive disease, but a higher dose rate or increased dosing frequency should be considered for non-effusive disease.
Of the five cats that relapsed, four (80%) developed clinical signs that were different from those shown on initial presentation. Similarly, Taylor et al 8 reported that 76% of cats that relapsed did so with clinical signs different from their initial presentation. Furthermore, the first sign of relapse in one of the cats was the development of oculonasal discharge and sneezing, which is not a classic presentation of FIP. One explanation for this is reinfection with FCoV with different tissue tropisms or the emergence of an antiviral-resistant FCoV with different tissue tropisms. This emphasises the importance of being vigilant for new clinical signs during or after treatment.
Four cats were treated with MPV after relapsing, with three subsequently entering remission. The potential causes of relapse responsive to MPV include acquired antiviral resistance to RDV, inadequate dosing, differences in drug tissue penetration or re-infection with resistant viral strain during treatment.
A post-mortem examination was performed on the cat that was euthanased. Histopathological abnormalities associated with FIP were only present in the brainstem and cerebrum. There were no lesions in the rest of the body consistent with FIP, suggesting that the virus was either successfully eradicated from the rest of the body or only developed a tropism for the central nervous system.
None of the cats received dose increases based solely on haematologic milestones. Haematologic parameters were normal by week 4 in the majority of cats and in all cats by week 8. These findings are similar to those published by Coggins et al. 9 Monthly monitoring of haematological parameters beyond resolution of abnormalities may not be required in cats that are clinically well.
While all the cats that entered remission met the milestones, the milestones were not always predictive of treatment success in the cats that relapsed or failed to respond to treatment. One cat with effusive disease in this study failed to clinically respond at all to RDV, despite dose escalations (maximum dose of 44 mg/kg q24h), and was euthanased 4 weeks after initiating treatment. This cat had met all haematological and biochemical milestones despite persistent clinical signs (worsening ascites, hyporexia and vomiting). Potential reasons for failure to respond to RDV include intrinsic resistance to RDV, inadequate bioavailability of oral RDV and an incorrect diagnosis of FIP.
Of the five cats that had dose increases, only one had an uncomplicated remission.
Cat 7 is the first case of fibrous intestinal adhesions in a cat with FIP to be reported. Before re-presenting at week 9 with an intestinal obstruction, this cat had met the milestones in Table 2. We theorised that the intestinal adhesions leading to obstruction were the result of fibrin-rich abdominal exudate caused by FCoV. No gross lesions indicative of ongoing FIP infection were observed during surgery, and RT-PCR testing of the excised tissue was negative for FCoV. This patient’s RDV course was extended by 2 weeks during postoperative recovery and the patient has remained in remission for 18 months to date. This may represent an underdiagnosed complication of FIP and should be considered in cats with FIP that present with new gastrointestinal signs during or after treatment.
There were no adverse reactions associated with oral or IV RDV. Adverse reactions to SC injections were reported in 4/8 (50%) cats that received SC injections. All four cats received a minimum of 2 weeks of RDV SC injections. Discomfort at the injection site and local skin reactions associated with subcutaneous RDV have been reported in previous studies.8 –10 Two-thirds of the cats in this study that received parenteral RDV (n = 18) received IV injections only, and none of these had adverse reactions. In cases where parenteral treatment is elected, IV administration negates the risk of local skin reactions.
The limitations of this paper include the lack of randomisation of cats into treatment groups, the small sample size and the predominance of effusive cases. Treatment could not be randomised, primarily because of the marked difference in cost between parenteral and oral RDV. Of the cats in this study, 72% had effusive disease and only 18% had non-effusive disease. Of the cats with non-effusive disease, eight had neurological/ocular disease and only two had other presentations.
Oral RDV was administered by the owners. Although compliance was not reported to be an issue by any owners, it is a potential limitation.
Another limitation of this study is that only one post-mortem examination was performed; therefore, the definitive cause of death in the other cats is unknown.
Conclusions
Oral RDV with or without parental RDV resulted in a survival rate of 86%, which is comparable to studies treating FIP with RDV, GS-441524 and MPV.
As a result of the significantly higher proportion of relapse in cats with ocular/neurological disease, higher dose rates or increased dosing frequency should be considered in these cats. Randomised, prospective studies with larger numbers of ocular/neurological cases are required to further elucidate this finding. In contrast, oral RDV at dose rates of 30–40 mg/kg was an effective treatment for cats with effusive FIP.
As no changes were made to the treatment regime based on haematological findings, these may be less clinically relevant for assessing treatment response than clinical signs, serum bilirubin and globulins.
Supplemental Material
Supplemental Material
Table 1: Amount of RDV (mg) in the 60 mg, 90 mg and 120 mg compounded RDV from Optimus Healthcare, New Zealand.
Table 2: Details of cats included in this study.
Table 3: Details of the cats that were euthanased during or after finishing treatment.
Table 4: Details of the cats that relapsed during or after finishing treatment.
Footnotes
Supplementary material
The following files are available as supplementary material:
Table 1: Amount of RDV (mg) in the 60 mg, 90 mg and 120 mg compounded RDV from Optimus Healthcare, New Zealand.
Table 2: Details of cats included in this study.
Table 3: Details of the cats that were euthanased during or after finishing treatment.
Table 4: Details of the cats that relapsed during or after finishing treatment.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by Healthy Pets New Zealand.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
