Abstract
Practical relevance:
Cardiovascular and kidney diseases are highly prevalent and frequently coexistent in cats, particularly those of advanced age. Several pathways of interaction between the heart and kidneys likely contribute to progressive functional decline of these systems during disease of one or both. Identification of heart or kidney disease in a cat should prompt the clinician to consider how the disease and its treatment might impact the function of the other organ and how coexisting primary disease of the other might impact case management and patient prognosis.
Clinical challenges:
A common goal in the management of congestive heart failure, acute kidney injury or chronic kidney disease is to maintain optimal body fluid balance; however, many therapies for disease of one system are at odds with the best interests of the other. For example, treatment of kidney disease often includes parenteral fluid therapy with the goal of maintaining mean arterial pressure and adequate renal blood flow, while diuretics are almost always necessary to reduce capillary hydrostatic pressure in cats with heart failure. Overly aggressive volume expansion or reduction during the management of kidney or heart failure, respectively, can lead to injury to or excessive stress on the other organ system.
Evidence base:
Recommendations are based on available peer-reviewed literature and expert consensus guidelines when available.
Audience:
This article is aimed at all veterinary practitioners who work with cats.
Introduction
Heart disease is common in domestic cats. The most frequently diagnosed of these diseases – idiopathic primary hypertrophic cardiomyopathy (HCM) – affects 15–16% of apparently healthy cats1,2 and results in cardiovascular death in 28% of cases. 3 In one large study of cats presented to primary care practices in the UK, cardiac disease was the attributed cause of death in 4.6% of those aged 5 years or older. 4
Kidney disease is also one of the most common causes of morbidity and mortality in cats, particularly those of advanced age. The reported prevalence of chronic kidney disease (CKD) in cats depends on the population studied and diagnostic criteria used, ranging from 2–50% in cats of all ages5,6 and up to 80% in cats aged 15 years or more. 5 In the large UK study referenced above, ‘renal disorder’ was the most common cause of death in cats aged 5 years or older, cited in 13.6%. 4 In addition, cats with kidney disease are frequently diagnosed with systemic arterial hypertension, which has important pathophysiologic consequences for both the kidneys and the heart.
Given the relatively high prevalence of heart and kidney diseases in cats, the intimate relationship between these systems in health and disease, and the fact that disease (or treatment) of one can cause direct or indirect injury or dysfunction of the other, it is unsurprising that clinicians commonly encounter cats with cardiovascular and renal comorbidities. Indeed, in a recent study, 51 (44%) of 116 cats treated for congestive heart failure (CHF) were azotemic prior to initiation of parenteral diuretics. 7 Likewise, ‘cardiovascular events’ were recorded in 104 (62%) of 168 cats admitted at a different institution for urinary tract obstruction. 8
In this article, we review the relationships between diseases of the heart and kidneys and present some considerations for the management of cats affected by both.
The cardiorenal axis and cardiovascular–renal disorders
The functions of the heart, blood vessels and kidneys are highly interrelated. Consideration of an integrated ‘cardiorenal axis’ is a useful lens through which to view these complex relationships. As defined by the veterinary Cardiovascular–Renal Axis Disorders Consensus Group, cardiovascular–renal disorders of dogs and cats are ‘disease, toxin or drug-induced structural and/or functional damage to the kidney and/or cardiovascular system, leading to disruption of the normal interaction between these systems, to the ongoing detriment of one or both’. 9 This approach encourages clinicians to consider connections between the cardiovascular system and kidneys during disease of either or both, and their interactions with other disease conditions. Further, it emphasizes the pathways, often bidirectional, by which disease progression in either or both might be mediated.
Although little is known regarding whether and how frequently primary disease of the kidneys or heart can initiate disease in the other organ, several pathways of interaction between the heart and kidneys likely contribute to progressive functional decline during disease of one or both (Figure 1).

Proposed hemodynamic, neurohormonal and inflammatory pathways linking the heart and kidneys during injury or dysfunction of either. GFR = glomerular filtration rate; RAAS = renin–angiotensin–aldosterone system; RBF = renal blood flow; SNS = sympathetic nervous system. Created in BioRender. Lourenço, B. (2025) https://BioRender.com/z0jijvq
How might heart disease and failure, or the treatment of these, impact kidney function?
Cats with severe heart disease can experience reductions in cardiac output and are at risk of developing systemic arterial hypotension, CHF and arterial thromboembolic complications.
RAAS and SNS activation
While initially compensatory and supportive of glomerular filtration rate (GFR), activation of the renin–angiotensin–aldosterone system (RAAS) and sympathetic nervous system (SNS) leads to chronic myocardial and kidney stress. 10 Heart failure can be viewed as an inflammatory condition,11–13 with the production of reactive oxygen species and pro-inflammatory mediators leading to endothelial dysfunction and tissue fibrosis. These effects can impair renal blood flow, oxygen delivery and GFR; damage corticomedullary renal tubular cells; and promote renal interstitial inflammation and fibrosis, both part of a final common pathway of disease progression in CKD. 14 In human patients with severe heart disease, the major pathophysiologic role of renal venous congestion and increased intra-abdominal pressure in kidney injury is also well recognized;15,16 whether this is a factor in cats is currently unstudied. Finally, the pattern (eg, whether sustained, low-level insults or more severe, episodic injuries are to blame) and time course (eg, at which point in the course of cardiac disease or its treatment) of the above effects on the kidney are unclear. 17
Arterial thromboembolism leading to renal infarction
Cats with heart disease frequently experience arterial thromboembolism (ATE). 3 While this most commonly manifests as distal aortic thromboembolism (ie, ‘saddle thrombus’), the kidneys can also be a direct target. 18 The prevalence of renal artery thromboembolism in cats with heart disease is undetermined. However, cats with renal infarction (Figure 2) appear more likely to have echocardiographic evidence of heart disease than those without.19,20
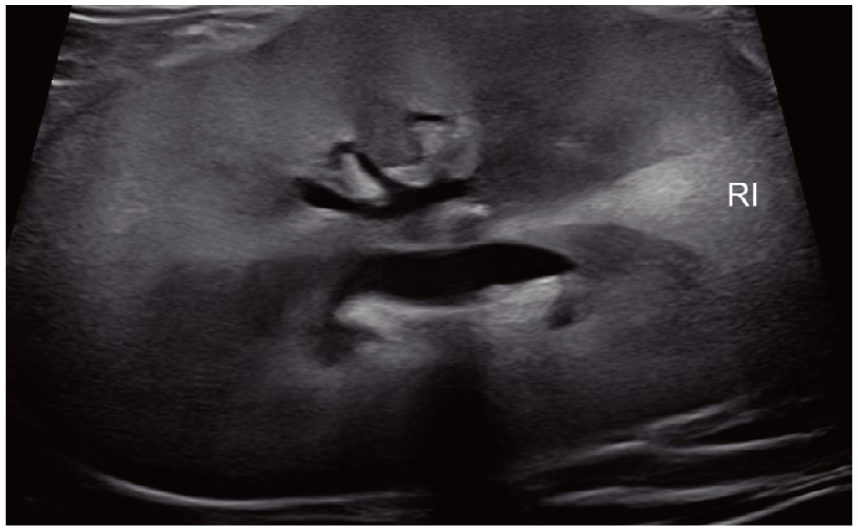
Two-dimensional transabdominal ultrasound image taken in a sagittal plane through the right kidney of a cat with chronic kidney disease. There are undulant cortical margins, wedge-shaped hyperechoic renal cortical infarcts (RI) and a diffuse decrease in corticomedullary distinction
Pharmacologic agents used for cardiovascular disease and heart failure
Drugs commonly used to treat heart disease and failure (eg, loop diuretics, angiotensinconverting enzyme inhibitors [ACEis]) or systemic arterial hypertension (eg, angiotensin receptor blockers [ARBs]) can affect kidney perfusion, renal autoregulation and glomerular hemodynamics, particularly when administered at high dosages alone or in combination, or when given in the setting of decreased circulating blood volume.21–23 While they represent a cornerstone of therapy for both acute and chronic life-threatening CHF in cats,24,25 loop diuretics can impair renal autoregulation and reduce renal blood flow under certain conditions by interfering with tubuloglomerular feedback.22,26
How common is kidney injury in cats with primary heart disease?
Azotemia is common in cats with apparently asymptomatic cardiomyopathy 27 and in those with acute CHF even before CHF has been treated.7,12,27,28 Serum creatinine concentrations >2.1 mg/dl (186 µmol/l) or >1.6 mg/dl 140 µmol/l) were documented prior to furosemide therapy in 11 (24%) of 45 cats 28 and 51 (44%) of 116 cats 7 hospitalized for acute CHF, respectively. In the larger of these studies, only 21 (18%) of 116 cats had known CKD at intake, suggesting that kidney injury due to primary heart disease might have explained pretreatment azotemia in some of the cases, although interpretation is complicated by the cats’ advanced age (median [range], 10 years [0.8–19.3]) and challenges in diagnosing early-stage CKD. In the same study, 75% of cats experienced a serum creatinine concentration >1.6 mg/dl (140 µmol/l) at some point during hospitalization, and the frequency of serum creatinine concentration >1.6 mg/dl remained high (ie, 45–69% of surveilled cases) post-discharge. 7 Increases ⩾0.3 mg/dl in serum creatinine concentration, consistent with the study’s definition of kidney injury, were experienced by 49% of cats during hospitalization, 7 in keeping with previous studies.28,29 While, in general, acute increases in serum creatinine have been linked to worse outcomes in cats, 30 kidney injury (as defined above) coincident with acute CHF treatment was not associated with poorer long-term prognosis in the study performed by Rogg and colleagues. 7
How might kidney disease and its treatment impact cardiovascular function?
The kidney regulates extracellular fluid volume and composition (including electrolyte concentrations) and systemic arterial blood pressure (BP). Due to renal functional derangements or drugs and therapies used in their treatment, cats with kidney disease can experience excesses or deficits of blood volume and pressure that affect cardiovascular function. Aside from the established effects of systemic arterial hypertension on the heart, whether these other derangements can induce heart disease or failure de novo is unclear; however, for cats with underlying heart disease, they are likely relevant.
Hypo- and hypervolemia
Uremic cats can experience hypovolemia due to obligatory polyuria if not met by sufficient intake, or fluid losses associated with vomiting or diarrhea, leading to decreases in cardiac output. Conversely, kidney disease can be associated with hypervolemia due to aggressive parenteral fluid administration, abnormal renal sodium and water handling, activation of the RAAS and chronic anemia, all of which can exacerbate heart disease and failure by increasing the workload of the heart.
Cats with kidney disease frequently require parenteral fluid therapy to address excessive renal and (when uremic) gastrointestinal fluid losses. While supplemental fluids can restore euvolemia and euhydration, their use – particularly of sodium-rich, isotonic crystalloids – can promote excessive extracellular fluid volume if not carefully optimized to patient needs. 31 This can be particularly problematic when administration is for long periods or at high rates to cats with underlying heart disease. Similarly, blood transfusion for severe anemia can cause transfusion-associated circulatory overload. 32
Systemic arterial hypertension
Systemic arterial hypertension is noted in 40–61% of cats with CKD33,34 and 28% of those with acute kidney injury (AKI). 35 It is the most well established pathophysiologic connection between kidney and heart diseases. The relationship between kidney disease and systemic arterial hypertension, 36 and the importance of the heart, vasculature and kidneys as targets of hypertensive end-organ damage, are detailed elsewhere.37,38 In small cross-sectional studies, 48–85% of systemically hypertensive cats have echocardiographic evidence of hypertensive cardiomyopathy (Figure 3).39–42 However, the clinical relevance of these changes is unclear. Although a cardiovascular cause of death or complication at the time of euthanasia was assigned for 4 (21%) of 19 hypertensive cats who died in one study, 43 the authors of a separate report found no significant difference in mean survival time between hypertensive cats with or without echocardiographic abnormali-ties. 39 It is uncommon for a cat to develop CHF or arterial thromboembolism secondarily to systemic arterial hypertension alone. 44 However, its consequences are relevant in patients with pre-existing heart disease or those undergoing additional hemodynamic stressors (eg, blood transfusion, intravenous fluid therapy), in whom CHF might be more easily precipitated.
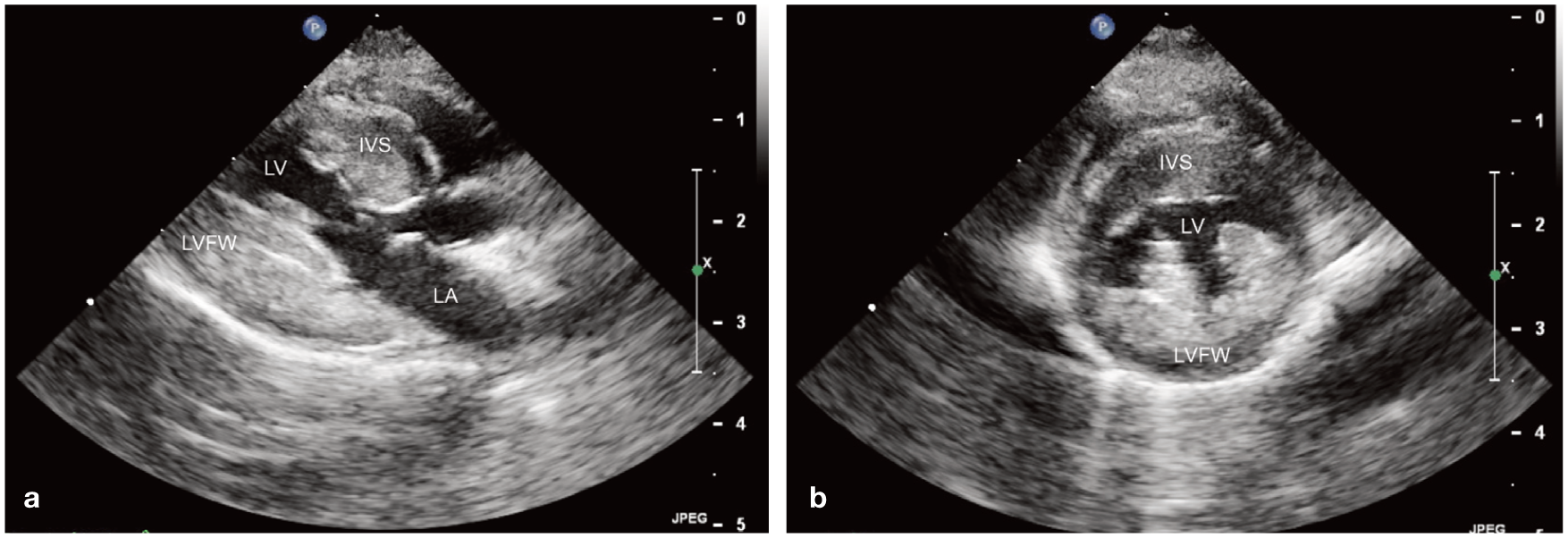
Two-dimensional (a) long- and (b) short-axis transthoracic echocardiographic images taken from a 10-year-old spayed female cat with chronic kidney disease and untreated systemic arterial hypertension (average indirect systolic blood pressure, 200 mmHg). Each image was taken at end-diastole from a right parasternal view. There is moderate-to-severe concentric hypertrophy of the left ventricular wall. IVS = interventricular septum; LA = left atrium; LV = left ventricle; LVFW = left ventricular free wall
Electrolyte derangements
Kidney disorders are frequently accompanied by electrolyte derangements. These include hypo- and hyperkalemia, hypo-and hypercalcemia, and hyperphosphatemia, which are common in cats with AKI45–48 or CKD45,49–51 and can affect cardiovascular function and structure.
Potassium abnormalities can alter cardiac excitability and lead to brady- or tachyarrhythmias (eg, sinus bradycardia, transient atrial standstill, sinoventricular rhythm, ventricular tachyarrhythmias, asystole)52,53 or abnormalities of cardiac conduction (Figure 4). In one retrospective study of 96 azotemic cats with acute ureteral or urethral obstruction, hyperkalemia was reported in 45 (46%) and serum potassium concentration was significantly associated with the occurrence of cardiovascular events – 34% of which were arrhythmias – based on multivariate analysis. 8 Conversely, hypokalemia is more common with polyuria and is therefore most likely in cats with CKD who are not fed a potassium-supplemented therapeutic renal diet, 49 especially those with mild or moderate decreases in GFR (ie, International Renal Interest Society [IRIS] CKD stages 2 or 3) or those with diuresis following relief of urethral obstruction. 54

Lead II surface electrocardiograms (paper speed, 50 mm/s; sensitivity, 20 mm/mV) taken (a) before and (b) after treatment of severe hyperkalemia (serum potassium concentration, 8.18 mmol/l [reference interval (RI), 3.41–4.76]) in a 7-year-old castrated male cat with aortic and renal arterial thromboembolism. In both recordings, the rhythm is sinus. After treatment, there is normalization of P wave amplitude (0.05–0.2 mV), PR interval (0.10–0.07 s [RI, 0.05–0.09]), QRS duration (0.07–0.03 s [RI <0.04 s]) and T wave morphology and amplitude (RI ± 0.3 mV)
Calcium is critical for normal cardiomyocyte contraction, cardiac automaticity and nodal conduction, and excitation–contraction coupling.55,56 In humans, low blood calcium concentrations can cause systemic arterial hypotension due to clinically relevant compromises in myocardial and vascular smooth muscle contract-ility.57,58 Conversely, sustained hypercalcemia, which can occur in CKD– mineral and bone disorder (CKD– MBD), can promote vascular mineralization and is associated with increased cardiovascular morbidity and mortality in people.59–62 Although vascular mineralization is recognized in cats with CKD,63,64 the impact of calcium disorders on cardiovascular outcomes remains understudied.
Finally, decreased renal excretion of phosphate stimulates the production of fibroblast growth factor 23 (FGF-23), which is central to the pathophysiology of CKD–MBD. 65 Increased circulating phosphate and FGF-23 concentrations are important risk factors for CKD progression and mortality in cats,66–68 and strongly predict worse cardiovascular outcomes in humans, likely due in part to the dystrophic mineralization they promote.69–71
CKD-associated anemia
The anemia of CKD increases in prevalence and severity with disease stage. 50 In addition to exacerbating renal hypoxia and oxidative stress and accelerating CKD progression in cats, 72 chronic anemia triggers detrimental adaptations (eg, RAAS and SNS activation) that expand extracellular fluid volume, increase cardiac workload, and promote left ventricular and left atrial hypertrophy. 73 These effects can be clinically relevant (Figure 5): in one report of eight severely anemic (ie, hematocrit ⩽18%) cats without underlying heart disease or systemic arterial hypertension, left heart enlargement and CHF were noted in 100% and 71%, respectively. 74 While these data are almost certainly influenced by selection bias – included cats had to have undergone echocardiography – they illustrate the potentially serious cardiovascular consequences of anemia.
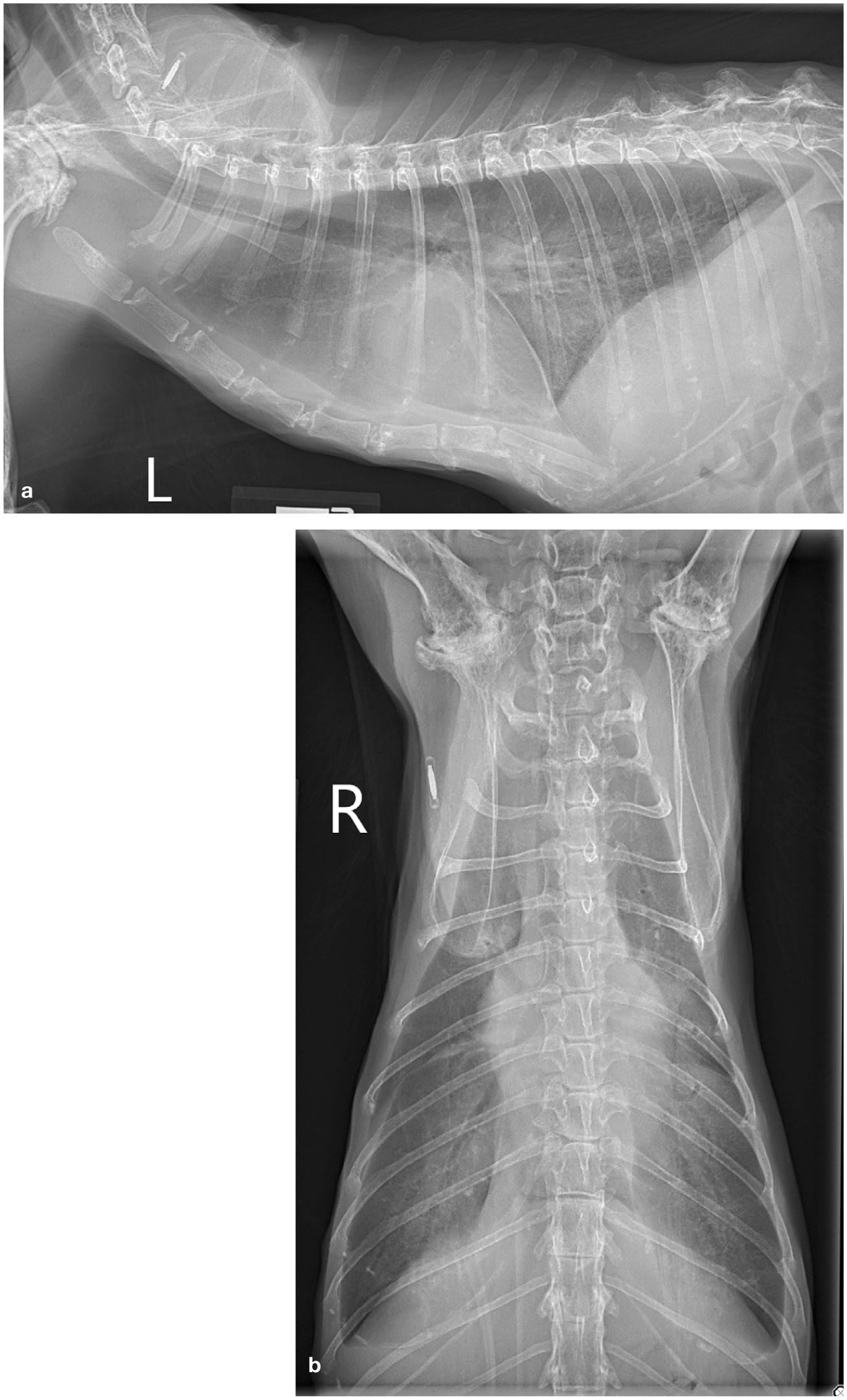
(a) Left lateral and (b) ventrodorsal thoracic radiographs taken from a 10-year-old spayed female cat with chronic kidney disease (International Renal Interest Society stage 3, normotensive) and chronic non-regenerative anemia (packed cell volume, 25%), receiving once daily treatment with subcutaneous fluids. There is mild-to-moderate cardiomegaly, a mild amount of pleural effusion and a multifocal interstitial coalescing-to-patchy alveolar pulmonary pattern, consistent with congestive heart failure
Erythropoiesis-stimulating agents (eg, dar-bepoetin) are used in cats with declining quality of life due to CKD-associated ane-mia, 75 potentially benefitting the kidneys and heart in addition to improving clinical signs. However, systemic arterial hypertension is a well-known potential adverse effect of this drug class,76,77 so careful monitoring of BP is recommended during therapy. Molidustat, a hypoxia-inducible factor prolyl hydroxylase inhibitor that stimulates endogenous production of erythropoietin, and for which currently available data do not support an effect on BP in cats, 78 might represent a potentially viable alternative for this indication. At the time of writing, molidustat has conditional approval in the USA for the treatment of non-regenerative anemia in cats with CKD. However, drugs of the same class are associated with the development of systemic arterial hypertension in humans, 79 suggesting that BP monitoring is advisable in treated cats.
RAAS and SNS activation
As in heart disease, excessive activation of the RAAS and SNS occurs in kidney disease independent of anemia (Figure 1). As mentioned above, the chronic hemodynamic (eg, increases in blood volume and pressure) and non-hemodynamic (eg, pro-inflammatory, pro-fibrotic, pro-oxidative stress) effects can harm the kidneys and cardiovascular system. 80 While evidence for activation of the circulating RAAS in cats with kidney disease is inconsistent, activation of the local, independently regulated, intrarenal RAAS is likely.81–85
Reduced renal clearance of cardiovascular pharmacologic agents
Abnormal kidney function can reduce the renal clearance of various pharmacologic agents. This is potentially clinically relevant for cardiovascular drugs whose parent compound or active metabolites (eg, atenolol, enalapril and its active metabolite, enalapri-lat) are primarily eliminated by the kidneys (Table 1), as this would raise concerns for drug accumulation and increased risk of dosedependent adverse effects.86,91 Therefore, dose reductions or the selection of other drugs in the same class with no or less renal elimination might be necessary in cats with kidney function impairment. For example, the clearances of benazepril and benazeprilat, which largely undergo biliary elimination, are unaffected by reduced kidney function in cats. 92
Mode of endogenous elimination for drugs commonly used in the treatment of cardiovascular disease of cats

Drugs for which greater than half of the dose is eliminated by the kidneys in cats (if data for this species are available) or in at least one other species (if data for cats are not available)
Pharmacokinetic data generated in cats are available. In all other instances, information was extrapolated from other species
Suboptimization of heart failure protocols
Finally, there is a general lack of evidence to guide heart failure therapies in cats with advanced kidney disease, which might impact cardiovascular function and outcomes. An easily imagined effect is the underprescription and suboptimization of heart failure protocols – an idea that is supported by experience in human medicine. 100 For example, in one retrospective study of 20,902 elderly patients in heart failure, prescription of ACEis, beta-adrenergic blockers or both, was associated with similar reductions in 1-year mortality risk in patients with and without azotemic CKD (ie, serum creatinine >2.0 mg/dl) compared with patients treated with neither drug. 101 However, in that study and anoth-er, 102 these drugs were less often prescribed to patients with azotemic CKD compared with those without.

Recommendations for diagnostic testing in patients with cardiovascular or kidney disease
Identification of heart or kidney disease in a cat should prompt the clinician to consider how the disease and its treatment might impact the function of the other organ and how coexisting primary disease of the other might impact case management and patient prognosis. This might include complete disease staging and screening for other diseases and conditions (eg, hyperthyroidism, systemic arterial hypertension) that might explain or exacerbate disease in one or both systems.
Assessment of kidney function in patients with heart disease
The IRIS classification systems for AKI and CKD 75 are useful for the characterization and grading (AKI; Table 2) or staging (CKD; Table 3) of kidney function in any patient, including those with heart disease.
International Renal Interest Society (IRIS) grading and subgrading criteria for acute kidney injury (AKI) 109

Lower limits differ from published values (ie, 141 µmol/l and 1.7 mg/dl); changed here after personal communication with an IRIS board member Cr = creatinine; RRT = renal replacement therapy; SDMA = symmetric dimethylarginine; UOP = urine output
International Renal Interest Society (IRIS) staging and substaging of chronic kidney disease (CKD) in cats 109
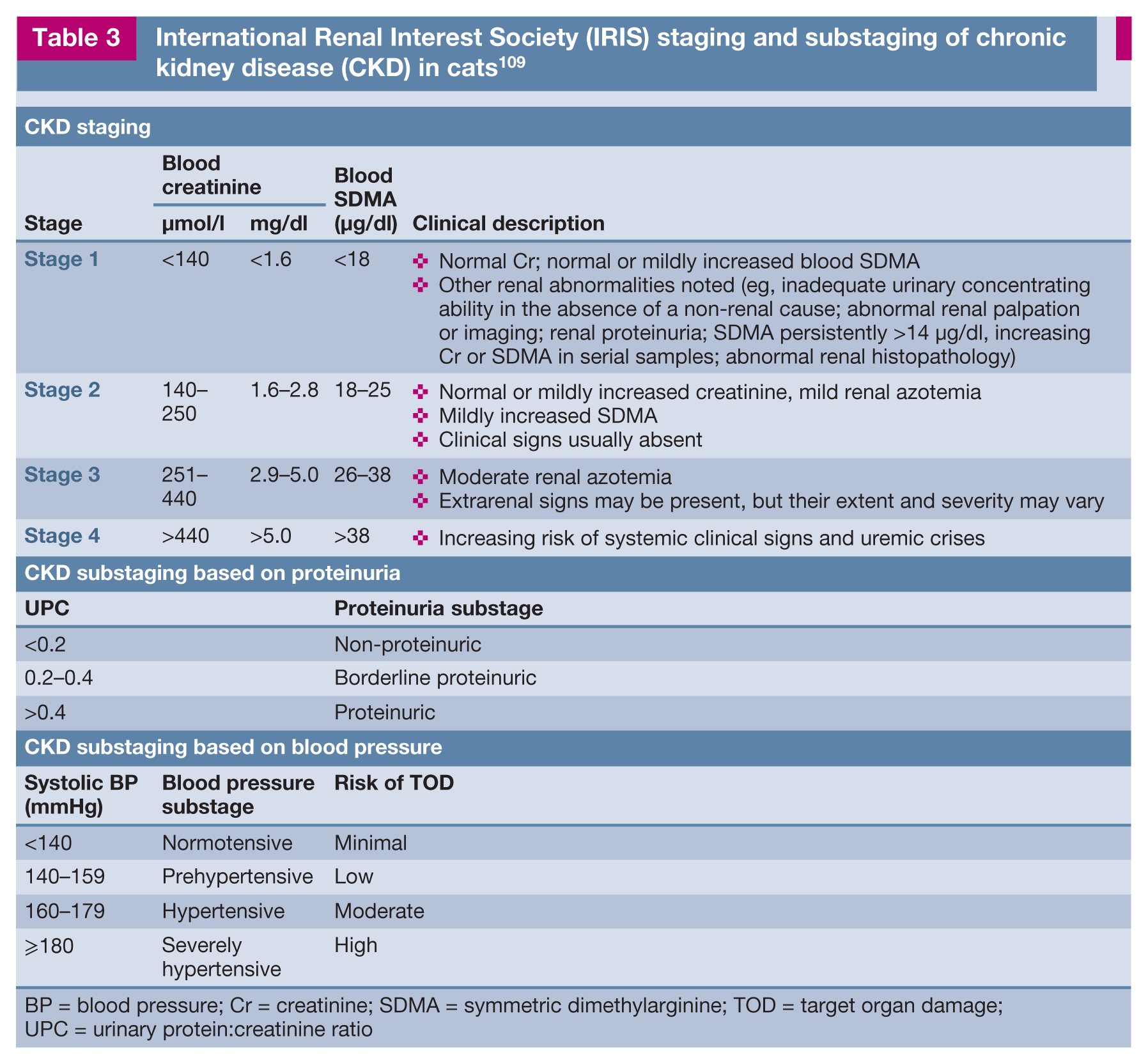
BP = blood pressure; Cr = creatinine; SDMA = symmetric dimethylarginine; TOD = target organ damage; UPC = urinary protein:creatinine ratio
Evaluation of routine blood and urinary biomarkers – including markers of GFR (ie, serum creatinine, symmetric dimethylarginine [SDMA] and urea nitrogen concentrations), serum electrolyte and albumin concentrations, urine specific gravity, urinary protein:creatinine ratio, and presence of renal glucosuria or cylindruria – allows the clinician to screen for kidney injury with reasonable sensitivity. However, functional markers like serum creatinine and SDMA concentrations are poorly sensitive and specific for mild injury characterized by little (or current) impact on glomerular filtration. Furthermore, the influences of heart function and medications for cardiovascular disease on renal blood flow and GFR can substantially impact the utility of these markers to diagnose intrinsic kidney disease in cats with concurrent heart disease (Table 4). Repeated sampling and documentation of progressive increases in serum creatinine or SDMA concentrations within the normal reference interval are likely to increase the sensitivity of these markers for the detection of decreased kidney function.
Potential impacts of kidney or heart dysfunction on tests used to monitor or diagnose disease of the other organ

BUN = blood urea nitrogen; CKD = chronic kidney disease; CHF = congestive heart failure; GFR = glomerular filtration rate; HCM = hypertrophic cardiomyopathy; NT-proBNP = N-terminal prohormone of B-type natriuretic peptide; SDMA = symmetric dimethylarginine
For cats with heart disease whose treatment includes a diuretic, it can be challenging to differentiate among volume-responsive (previously, ‘prerenal’) azotemia, azotemia due to acute kidney injury and chronic cardiovascular disease leading to progressive CKD (ie, type 2 cardiovascular–renal syndrome). Importantly, because of the urine-diluting effects of diuretics, urine specific gravity measurements cannot be used to help determine whether azotemia is most likely due to extrarenal causes. Ultrasonographic changes to the kidneys (see below), senior or geriatric patient age, and the presence of non-regenerative anemia increase concern for intrinsic renal azotemia. There is growing interest in the use of urinary biomarkers of kidney injury (eg, urinary cystatin B, clusterin, neutrophil gelatinase-associated lipocalin) to detect early or active/ongoing damage.111–114 Of these, only a test for urinary cystatin B is commercially available at the time of writing. The effects of extrarenal conditions, including heart and vascular diseases, on these markers deserve future study.
In cats with heart disease, radiographic or ultrasonographic evaluation of kidney structure can be useful to diagnose concurrent kidney disease. Imaging should be considered for cats whose kidney health status is questioned but unclear based on clinicopathologic findings. Small, irregularly shaped kidneys, greater than normal echogenicity of the renal cortex and medulla, decreased cortico-medullary distinction, loss of normal internal architecture or a combination of these supports a diagnosis of CKD.115,116 An intrinsic acute nephropathy is suggested by enlarged kidneys and increased cortical echogenicity with preserved corticomedullary distinction, whereas marked pyelectasia or hydronephrosis, hydroureter or ureteral calculi can signal obstructive nephropathy.115,116
Assessment of cardiac structure and function in cats with kidney disease
Imaging of the cardiopulmonary structures is necessary for accurate and comprehensive assessment of heart structure and function, and is recommended for cats with kidney disease who also have historical or clinical findings suggestive of heart disease or failure. The American College of Veterinary Internal Medicine feline cardiomyopathy staging system can be used to characterize the clinical impact of cardiomyopathy in cats (Table 5). 25
American College of Veterinary Internal Medicine staging of feline cardiomyopathy 25

ATE =arterial thromboembolism; CHF = congestive heart failure
In an ideal world, and assuming no practical or financial limitations, all cats with known kidney disease would undergo assessment of cardiac structure and function using complementary information from echocardiography and thoracic radiography. This is because in this species, physical examination findings (eg, presence of a heart murmur or gallop) and thoracic radiography alone have poor sensitivity for the identification of subclinical heart disease and heart enlargement, respectively.1,117,118 Instead, the clinical gold standard for assessment of cardiac structure and function is echocardiography, which allows for the diagnosis, specific phenotyping and staging of heart disease. The findings of thoracic radiography complement echocardiographic data by determining whether signs of CHF are present.
Echocardiography is not feasible in all cases or for all owners, however. The most practical reason to screen for heart disease in cats with kidney disease is to identify individuals who, compared with cats without heart disease, are at meaningfully increased risk for complications (eg, CHF, ATE, cardiac arrhythmias) when challenged with certain interventions or stressors. This largely equates to cats with moderate or severe heart disease/injury and enlargement. 119
The accuracy of widely available circulating cardiac biomarker (eg, N-terminal prohormone B-type natriuretic peptide [NT-proBNP], cardiac troponin I) concentrations to establish the presence of subclinical heart disease or injury in cats with kidney disease is limited by several factors. Among the most relevant is that both biomarkers undergo renal clearance, which decreases their specificity for cardiac injury in cats with abnormal kidney function (Table 4).120,121 In addition, little is known about their performance compared with echocardiography in cats with AKI or CKD. Therefore, the use of cardiac biomarker concentrations in isolation to rule in heart disease or injury in this situation is not advised. However, NT-proBNP concentration might be a useful component of screening protocols in certain cases, as discussed below. 122
In the authors’ practice, echocardiography is most strongly recommended for cats with kidney disease who have one or more of the following, particularly when treatments (ie, parenteral fluids, blood transfusion) that increase the workload of the heart are planned: (1) historical or clinical findings suggestive of current or previous heart disease or failure; (2) thoracic radiographic findings suggestive of cardiomegaly, cardiogenic edema or effusions, or both; or (3) physical examination findings that, when present, imply increased risk of cardiomyopathy (eg, loud [⩾grade IV/VI] heart murmur, gallop sound or arrhythmia).25,119 The information provided can allow for the formulation of a rational treatment plan that minimizes the risk of excess hemodynamic stressors on a diseased heart. This approach is supported by the findings of one study in which 61 (51%) of 120 cats with CHF experienced antecedent events (eg, intravenous fluid administration, anesthesia, surgery, corticosteroid administration) that might have precipitated the episode. 123
In cats for whom echocardiography is not feasible, the combined results of serum NT-proBNP concentrations and thoracic radiographs can help to identify cases that are unlikely to have moderate or severe heart disease or failure. Importantly, this approach will misclassify some cats with mild forms of disease (false negative) or normal hearts (false positive), although ‘clinically relevant’ disease is less likely in older cats with normal findings in both tests. 124 However, this approach has not been rigorously tested in cats with kidney disease.
Assessment of systemic arterial BP in cats with heart or kidney disease
Given the link between BP abnormalities and heart and kidney disease, the authors, as well as a consensus panel of veterinary cardiologists and nephrologists, 9 recommend routine monitoring of systemic arterial BP in cats with diseased kidneys, with the goal of identifying and correcting pathologic systemic arterial hypertension or hypotension. In the authors’ practice, routine BP monitoring is also performed in cats with moderate or severe heart disease who have risk factors for systemic hypertension, clinical signs of CHF or are receiving medications known to impact systemic BP.
Considerations for the management of patients with concurrent cardiovascular and renal diseases
A common goal in the management of CHF, AKI or CKD is to achieve and maintain optimal body fluid balance. However, many therapies for disease of one system are fundamentally at odds with the best interest of the other. For example, the treatment of kidney disease often includes parenteral fluid therapy with the goal of maintaining mean arterial pressure and adequate renal blood flow, while diuretics are almost always necessary to reduce capillary hydrostatic pressure in cats with CHF. Overly aggressive volume expansion or reduction during the management of kidney or heart failure, respectively, can lead to injury to or excessive stress on the other system. Two commonly encountered clinical scenarios are described on the following pages to illustrate the balance required to manage cats with concurrent underlying disease of both systems. In general, the approach should be to treat the system whose decompensation is responsible for the patient’s clinical signs in a manner that minimizes risk to the other system and the cardiovascular–renal axis.


Key Points
✜ Heart and kidney diseases frequently coexist in the same cat.
There are several pathways of interaction between the heart and kidneys during health and disease, and it is helpful to think of these systems as belonging to an integrated ‘cardiovascular–renal axis’.
✜ One of the most well-described links between heart and kidney diseases is systemic arterial hypertension, which is frequently associated with CKD in cats, and can lead to progressive damage to both. Given the link between BP abnormalities and heart and kidney diseases, routine monitoring of systemic arterial BP and the use of antihypertensive therapy (eg, amlodipine, telmisartan) in cats with diseased kidneys is recommended.
✜ The general approach to the treatment of cats with both conditions should be to treat the system whose decompensation is responsible for the patient’s clinical signs in a manner that minimizes risk to the other system and the cardiovascular–renal axis.
Footnotes
Conflict of interest
Amanda Coleman declares no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Bianca Lourenço has received research funding, speaker honoraria, and consulting fees from Ceva Animal Health and Boehringer Ingelheim Vetmedica, and research funding from Elanco Animal Health.
Funding
The authors of this commissioned Clinical Spotlight review received an honorarium; as for all JFMS articles, this Clinical Spotlight article went through peer review.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
