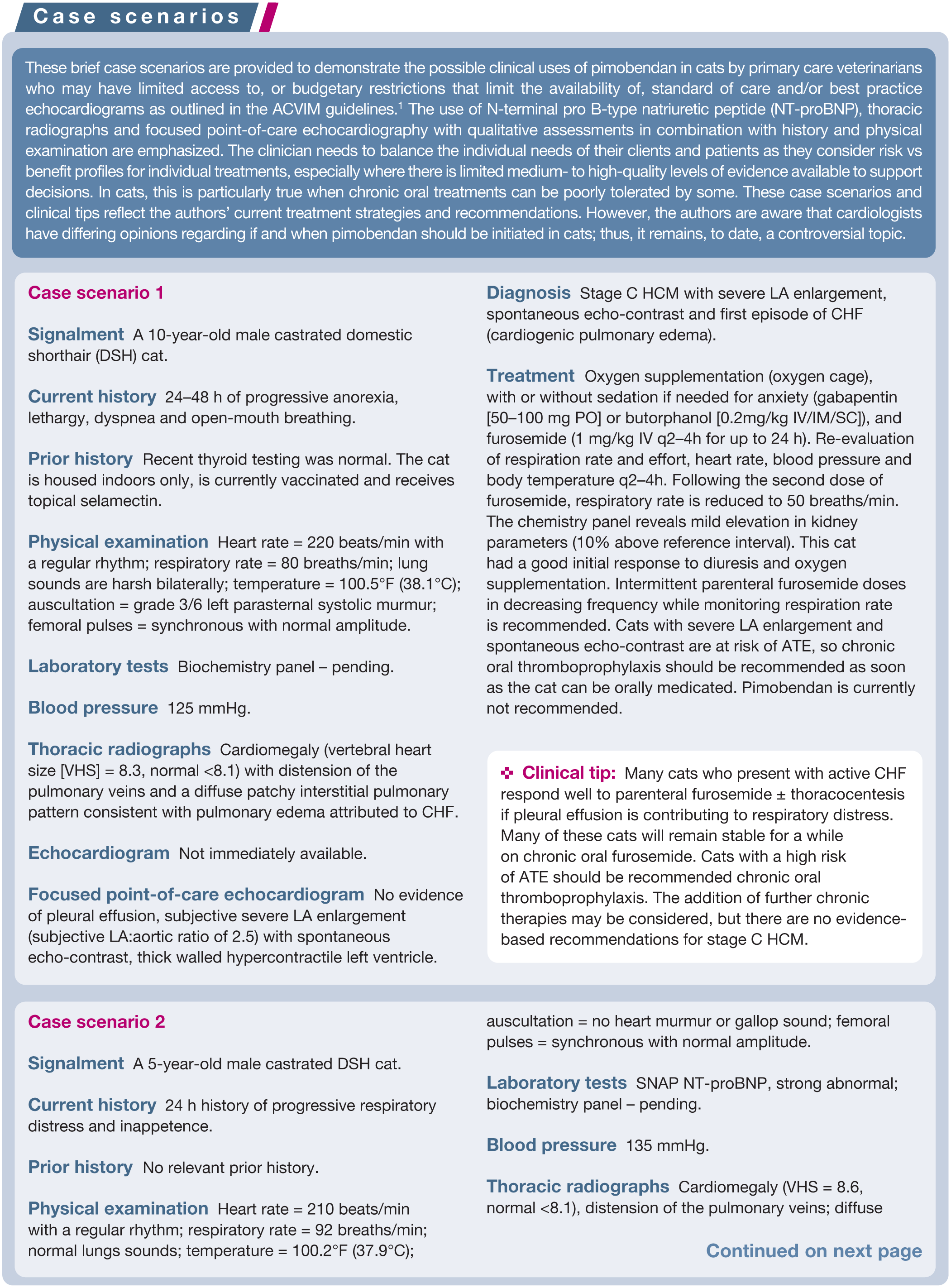
Abstract
Practical relevance:
Cardiomyopathies are a common condition and the leading cause of congestive heart failure (CHF) in cats; however, to date there have been limited therapeutic options available. Commonly used therapeutics include diuretics for CHF and antithrombotics 2 to reduce the risk of arterial thromboembolism (ATE), which do not directly affect myocardial function. This review summarizes the evidence for the use of pimobendan, a phosphodiesterase III inhibitor and calcium channel sensitizer, in feline cardiomyopathies. Potential benefits of pimobendan in cats include improved left ventricular systolic and diastolic function, reduced platelet aggregation, reduction in left atrial size, and improved left atrial and auricular systolic function.
Drug details:
Pimobendan is indicated for the treatment of CHF in conditions associated with systolic dysfunction (including dilated cardiomyopathy, restrictive cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy and some cases with non-specific phenotypes) and has been used in the treatment of CHF secondary to hypertrophic cardiomyopathy. A dosage of pimobendan of 0.25 mg/kg PO q12h is generally well tolerated, and an approved oral solution is now available in the UK, EU, USA, Japan and Australia, which may facilitate administration in some cats; it should be noted, however, that pimobendan is not licensed for use in cats and marketing authorizations may vary between countries. Adverse events are uncommon with pimobendan but include gastrointestinal upset (anorexia, vomiting), transient changes in heart rate and possible worsening arrhythmias at higher doses. The use of pimobendan is contraindicated in cats with fixed obstructions of the left or right ventricular outflow tract (ie, congenital heart disease – pulmonic or aortic valvular stenosis).
Audience:
This article reviews the use of pimobendan in cats for primary care veterinarians and includes practical case examples that reflect the typical use and recommendations for usage of pimobendan by the authors.
Introduction
Cardiomyopathies are common in cats, with hypertrophic cardiomyopathy (HCM) being the most common phenotype (see ‘HCM in cats’ box).16–19 Despite limited evidence, conventional treatments for cats with cardiomyopathy and CHF include diuretics, renin–angiotensin– aldosterone system inhibitors (ie, angiotensin-converting enzyme inhibitors, spironolactone) and antithrombotic therapy (ie, clopidogrel, factor Xa inhibitors) for thromboembolism prophy-laxis.1,20–27 Pimobendan (Vetmedin; Boehringer Ingelheim) has also been increasingly used across the globe in recent years,1,28 primarily in cats with clinical cardiomyopathies (ACVIM stages C–D),29,30 with the LV systolic dysfunction of some cats with HCM, as described in the ‘HCM in cats’ box, providing a potential rationale for the use of this drug in these patients.4–6 Pimobendan is a phosphodiesterase III inhibitor and calcium channel sensitizer that prolongs survival times in dogs with CHF secondary to myxomatous mitral valve disease and dilated cardiomyopathy (DCM), and delays the onset of CHF in dogs with ACVIM stage B2 myxomatous mitral valve disease and subclinical DCM.31–33 However, while the inotropic, lusitropic and systemic vasodilatory effects of pimobendan have been documented in dogs, similar published data in cats are so far limited.31,34–37

Studies reporting the use of pimobendan in cats with CHF secondary to a variety of cardiomyopathies show that this drug is well tolerated, although efficacy data are controversial and limited.30,27,38–40 One retrospective study, which included treatment of cats with documented mild LVOTO, reported that the median survival time in the cats with stage C HCM increased with pimobendan treatment compared with control cats (626 days vs 103 days, respectively). 41 However, a 6-month prospective, multicenter, double-blind randomized placebo-controlled non-pivotal exploratory field study in cats with CHF secondary to HCM with or without LVOTO failed to demonstrate a difference between placebo and pimobendan in the proportion of cats completing the planned 6-month followup period. This study did confirm that pimobendan was well tolerated, with no difference in adverse events reported between placebo and pimobendan. 40
Pimobendan’s potential combination of increased inotropic and systemic vasodilatory effects has prompted concerns that it could cause, or worsen, dynamic LVOTO or mid-LV obstruction in cats with HCM by increasing the severity of obstruction while simultaneously lowering periperhal pressure. This could, in turn, lead to theoretical increases in myocardial oxygen demand, tachycardia, arrhythmias or peripheral hypotension. Research evaluating the safety and efficacy of pimobendan in cats with dynamic LVOTO is minimal and conflicting. One study reported worsened dynamic LVOTO in a cat with complex congenital heart disease including mitral valve dysplasia receiving pimobendan, 42 while a more recent study reported that dynamic LVOTO did not worsen in cats with HCM receiving a single dose of pimobendan. 39 In two studies, cats with HCM and dynamic LVOTO were either excluded from the study at the onset 30 or excluded from statistical analysis (n = 3) due to worsening dynamic LVOTO despite a lack of clinical decline. 40 However, two relatively large retrospective studies included cats with HCM and LVOTO and reported no adverse outcomes.29,30 The balance of this evidence suggests that, in some cases, pimobendan may cause a mild increase in LV outflow tract velocities, but it does not appear to cause clinical concern or reportable adverse events. However, to the authors’ knowledge, there are no studies to date that have evaluated the effects of pimobendan in cats with moderate to severe baseline LVOTO. 28
What is known about pimobendan in cats?
Studies describing the use of pimobendan in cats are summarized in Table 1. Most pharmacokinetic and pharmacodynamic data are derived from normal healthy animals and, as such, continue to be a potential limitation of their application in diseased animals. To improve clarity, the following sections are separated into healthy normal cats and cats with cardiomyopathy.
Studies describing pimobendan use in cats

ACEi = angiotensin-converting enzyme inhibitor; ARVC = arrhythmogenic right ventricular cardiomyopathy; CHF = congestive heart failure;
CM = cardiomyopathy; DCM = dilated cardiomyopathy; HCM = hypertrophic cardiomyopathy; HR = heart rate; LA = left atrial; LV = left ventricular; LVOTO = left ventricular outflow tract obstruction; MST = median survival time; NSCM = non-specific cardiomyopathy; PDA = patent ductus arteriosus; TMT = transient myocradial thickening
Healthy normal cats
Pharmacokinetic data evaluating pimobendan administration in healthy cats reveals a high volume of distribution and a rapid absorption after oral administration.43,44 Maximal plasma concentration is achieved 40 mins and 20 mins after oral doses of 0.625 mg and 1.25 mg, respectively. 44 Cats appear to achieve higher plasma concentrations of pimobendan than dogs at similar mg/kg doses.43,44,56 After administration, pimobendan is rapidly converted to the biologically active metabolite O-desmethyl-pimobendan (ODMP). Maximal plasma concentrations of ODMP are achieved after 1 h. 44 In contrast to dogs, cats convert less pimobendan to ODMP, which may be due to species differences in cytochrome P-450 isoenzyme activities. 44 The elimination half-life for pimobendan in cats is between 0.7 and 0.8 h. 44
The pharmacodynamic effects of pimoben-dan have been explored in various studies. Administration of single oral doses of 0.625 mg or 1.25 mg of pimobendan to healthy cats changed several echocardiographic parameters associated with systolic function; however, the magnitude of the changes was small and thus may not be clinically rele-vant. 44 A single oral dose study in healthy cats showed no significant effect on echocardiographic parameters of LV systolic function, but demonstrated improved LA function 46 in addition to positive effects on multiple echocardiographic parameters of right ventricular and right atrial function. 44 Another study of five healthy cats comparing doses of 0.125, 0.25 and 0.5 mg/kg of pimobendan q12h for 14 days found improved LV systolic function in the 0.25 and 0.5 mg/kg groups, with significant correlation between cardiac function and plasma concentrations of pimobendan and ODMP, highlighting the effectiveness of a higher dose of pimobendan; however, again, the magnitude of the changes were small and thus may not be clinically relevant. 51 Short-term administration of pimobendan (0.25 and 0.5 mg/kg PO q12h) increased peak longitudinal strain rate in healthy cats, despite a lack of change in fractional shortening or peak systolic velocity of the lateral mitral annulus. 50 These data suggest that advanced echocardiographic measurements may be required to fully assess the effects of oral pimobendan on systolic function in cats. Intravenous pimo-bendan (0.15 and 0.3 mg/kg), on the other hand, increased LV fractional shortening, peak systolic velocity of the lateral mitral annulus and echocardiographically calculated cardiac output in healthy sedated cats. 54
The effect of pimobendan on heart rate and cardiac rhythm in healthy cats has also been investigated in numerous small studies (4–11 cats). The results are variable and challenging to interpret, however, as any documented changes are small in magnitude and of unknown clinical significance in a species where the presence and burden of arrythmias is not well characterized in either health or the presence of cardiomyopathy. A study analyzing heart rates obtained by echocardiogram after a single oral dose of pimobendan demonstrated a mild increase in heart rate, 46 while another study reported a decrease in heart rate. 44 The reported heart rate changes in both studies would not be considered clinically relevant.44,46 Twenty-four hour Holter electrocardiogram monitoring in healthy cats before and after short-term oral administration of twice daily placebo or 0.125, 0.25 or 0.5 mg/kg of pimobendan did not change the total number of heart beats or number of ventricular premature complexes, or change the minimum, mean or maximum heart rate in a 24 h period, suggesting that oral pimobendan administration may not affect heart rate in normal cats; however, this study was also small. 50 A higher number of idioventricular/ idiojunctional beats and escape complexes were observed in cats administered 0.5 mg/kg q12h pimobendan. 50 While these arrhythmias did not appear to be malignant in these healthy cats, the results suggested that a degree of caution should be exercised when using higher doses of pimobendan in cats with heart disease pending more thorough exploration of these potential effects. Holter data following intravenous administration of pimobendan (0.15 mg/kg q12h for 3 days) in five healthy cats revealed a significant increase in the total number of heart beats in 24 h and in mean heart rate compared with placebo. 55 Two of the five cats in this study had increased numbers of arrhythmias posttreatment (including a paroxysm of ventricular tachycardia in one cat), while the other three cats showed a decreased number of arrhythmias post-treatment. 55 To date, limited conclusions can be drawn with regard to expected heart rate and rhythm parameters associated with pimobendan use in cats, particularly in cats with heart disease.
In addition to cardiovascular effects, phosphodiesterase inhibitors also have the potential to reduce platelet aggregation. Several phosphodiesterase III inhibitors used in humans have demonstrable antiplatelet activ-ity.57–62 In healthy cats administered two clinically relevant doses of pimobendan (0.625 and 1.25 mg/cat q12h) for 7 days, pimoben-dan did not alter a limited assessment of platelet function. 53
Cats with HCM
Varying pharmacodynamic effects of pimo-bendan have been demonstrated in cats with HCM. In a study of purpose-bred research colony cats with HCM and normal LA size to mild LA enlargement (stage B1), no changes in LV systolic function or exacerbation of dynamic LVOTOs were observed after a single 1.25 mg oral dose of pimobendan, but increased LA fractional shortening was observed. 39 A single-dose study in client-owned cats with HCM and normal LA size to mild LA enlargement (stage B1) replicated the increase in LA fractional shortening 90 mins after a 1.25 mg oral dose of pimobendan, documented a mild increase in LV systolic function with increased peak systolic velocity of the mitral annulus, and increased left and right ventricular outflow tract velocities. 48 A study focusing on echocardiographic parameters associated with LA size and function in client-owned cats with stage B or C stable HCM after short-term administration of pimobendan (0.25 mg/kg q12h) demonstrated modest improvements in LA systolic function. 47
In summary, to date the pharmacodynamic effects of pimobendan in cats with cardiomyopathy characterized by normal to mild LA enlargement include improved LA function, a possible small increase in LV systolic function and a mild increase in outflow tract velocities.
Practical considerations for pimobendan use in cats
Dose
No pimobendan formulations have been approved for use in cats. Given the pharmacodynamic data available to date, 1.25 mg PO q12h has become the most clinically utilized dosage of pimobendan in cats, with minimal to no adverse effects attributed to this dosing scheme. For an average-sized cat this corresponds to a weight-based dose of approximately 0.25 mg/kg. Adjustments may be considered based on an individual cat’s weight and according to clinical response.
Pimobendan is available as approved tablets and capsules in various strengths. 63 The availability of approved oral solutions could facilitate easier and more accurate dosing, therefore increasing its potential use in cats for both acute and chronic treatment.
Until further investigation into cardiac rhythm and rate changes are investigated in cats with heart disease treated with higher doses of pimobendan, dose escalation above 0.25 mg/kg q12h should be pursued with caution and close monitoring. In a clinical setting, pimobendan is often given in conjunction with other medications including diuretics, antiplatelet drugs and dobutamine without apparent interactions; however, this has not specifically been investigated to date.29,30,38,41,42,64
Indications, contraindications and adverse effects
Pimobendan has been evaluated as a therapy for managing CHF in cats; relevant conditions and recommendations are summarized in Tables 2 and 3. Ventricular systolic dysfunction can be a feature of many types of cardiomyopathy, including HCM, a cardiomyopathy that traditionally features diastolic dys-function.10,11 Feline cardiomyopathies typically associated with ventricular systolic dysfunction include dilated cardiomyopathy, restrictive cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy and some cases with nonspecific phenotypes. Systolic dysfunction also occurs secondarily to myocardial infarction or as an end-stage presentation of any cardiomyopathy. Systolic dysfunction is characterized on echocardiographic examination by focal areas of reduced wall motion or global reduction in ventricular systolic function.
Recommendations for pimobendan use in cats with cardiomyopathy diagnosed by echocardiography

Note: Strong evidence-based therapeutic options are limited for cats with cardiomyopathy. Most cats with stage C cardiomyopathy can be stabilized with furosemide. The clinical use of most other chronic medications are controversial in cats with stages B2, C and D cardiomyopathy. The above reflects the authors’ current treatment strategies and recommendations
Consultation with a cardiologist would be beneficial
ARVC = arrhythmogenic right ventricular cardiomyopathy; CHF = congestive heart failure; LA = left atrial
Recommendations for pimobendan use in cats with heart disease NOT confirmed and characterized by echocardiography

Note: For some diagnoses, peer-reviewed clinical data are limited or absent; some recommendations are therefore based on anecdotal reports and the authors’ experience
Consultation with a cardiologist could be beneficial
CHF = congestive heart failure; LA = left atrial
Pimobendan use has been reported in cats with conditions associated with systolic dys-function,30,38,42 and has also been used in the treatment of CHF secondary to HCM with and without dynamic LVOTO, where global routine measures of ventricular systolic function are normal.4,29,40 Dynamic LVOTO in cats with HCM can be intermittent and heart rate dependent, and in cats where pimobendan is associated with the new onset or exacerbation of dynamic LVOTO or mid-LV obstruction, the magnitude and therefore clinical significance of any worsening of the obstruction varies.40,47 Clinical decompensation has generally not been identified when pimobendan is used in cats with dynamic LVOTO, but can occur in rare cases.30,40–42 ,48 The development of systemic hypotension, weakness, collapse, sinus tachycardia or worsening arrhythmias, or the presence of a new or louder murmur suggesting exacerbation of an obstruction, should prompt re-evaluation of the patient, and pimobendan should be discontinued if considered likely to be the causative factor. Routine monitoring, including auscultation, electrocardiography, indirect systemic blood pressure and echocardiography to assess dynamic LVOTO, can detect complications early and guide treatment adjustments.
Transient myocardial thickening of the left ventricle can mimic HCM and can also result in CHF. Transient myocardial thickening is often associated with an antecedent event and elevated cardiac troponin I, and is characterized by eventual clinical improvement with resolution of CHF and reverse remodeling of the heart to normal within months. 65 Pimobendan has been used in the management of CHF secondary to transient myocardial thickening caused by Toxoplasma gondii. 52
Pimobendan has also been used to manage CHF due to a variety of congenital heart conditions, predominantly in diseases characterized by left-to-right shunting (patent ductus arteriosus, septal defects) or in the presence of marked atrial enlargement.66–68 Pimobendan use is not recommended in cats with fixed obstructions of the left or right ventricular outflow tract (ie, aortic or pulmonic stenosis or fixed midventricular obstructions). In general, consultation with a cardiologist is recommended when considering pimobendan use in cases of congenital heart disease.




Reported adverse effects with pimobendan administration are uncommon and are predominantly characterized by gastrointestinal upset (ptyalism, anorexia, vomiting, diarrhea), transient changes in heart rate and possible worsening arrhythmias at higher doses.30,38,39,41,43,44
Conclusions
Pimobendan appears to be well tolerated in cats, with minimal adverse effects at a dosage of 1.25 mg PO q12h. Although the current level of evidence for pimobendan use in cats with cardiomyopathy is low (eg, retrospective case series, studies in other species and expert opinion), the reported short-term pharmacodynamic effects (eg, improved LA function; mild, possibly clinically insignificant increase in LV systolic function; absence of clinical concerns related to mild LVOTO) support a favorable potential clinical utility in cats with ACVIM stages C and D cardiomyopathies in the absence of clinically significant LVOTO or other heart disease associated with a fixed obstruction. The development of evidence-based recommendations for pimobendan use in feline cardiomyopathy with and without LVOTO will require adequately powered, randomized controlled studies with pragmatic endpoints.
Key Points
✜ Cardiomyopathies are a common condition of cats for which evidence-based therapeutic options are limited.
✜ Most cats with stage C cardiomyopathy can be stabilized with furosemide. The addition of additional chronic medications are controversial in cats with stage C cardiomyopathy. In cats with stage C cardiomyopathy and no evidence of LVOTO, pimobendan can be considered; however, the risk vs benefit profile of polypharmacy strategies in cats should be considered when initiating new medications, and owners should be informed when recommendations are not evidence based.
✜ In cats with stage C cardiomyopathy and no confirmation of LA size or risk factors for ATE, the empirical use of chronic thromboprophylaxis is controversial. In stage B2 or C cardiomyopathies characterized by the presence of high-risk factors for ATE, empirical use of thromboprophylaxis can be considered.
✜ The safety of pimobendan in cats with severe LVOTO has not been established.
✜ A new approved pimobendan oral solution is available in some countries (UK, EU, USA, Japan and Australia) and could facilitate accurate and easier dosing of cats.
✜ While further research is needed to establish strong evidence-based recommendations for pimobendan use in cats, available data suggest that oral pimobendan in cats is generally well tolerated with minimal adverse effects.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. All authors have received funding from Boehringer Ingelheim Animal Health within the past 5 years for some or all of the following activities: research, travel, speaking fees, consultancy fees and preparation of educational materials.
Funding
The authors of this commissioned Clinical Spotlight review received an honorarium; as for all JFMS articles, this Clinical Spotlight article went through peer review. All authors have received funding from Boehringer Ingelheim Animal Health within the past 5 years for some or all of the following activities: research, travel, speaking fees, consultancy fees and preparation of educational materials.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. For any animals or people identifiable within this publication, additional informed consent for publication was obtained.
