Abstract
Six African grey parrots (Psittacus e erithacus) were diagnosed with cardiomyopathy and congestive heart failure based on gross and microscopic findings. Ages ranged from 15 days to 8 years, and 5 of 6 parrots were either neonates or juveniles at the time of diagnosis. Two neonates and 2 juveniles came from the same breeding aviary; the 2 juveniles were born to the same breeding pair. The 2 other parrots were kept as pets. Clinical signs included distention of the coelomic cavity (4 of 6), rales (3 of 6), weakness (4 of 6), bradyarrhythmia (1 of 6), growth retardation (1 of 6), crop stasis (1 of 6), and regurgitation (1 of 6). Three parrots were euthanized and 3 died. Gross findings included cardiomegaly due to biventricular, right-, or left-sided cardiomyopathy (6 of 6); coelomic effusion (6 of 6); whitish or yellow foci in the liver (6 of 6); atrophy of the liver (particularly, the left lobe; 5 of 6); reddened or grey lungs (5 of 6); subcutaneous edema (2 of 6); hydropericardium (1 of 6); and bilateral thyroid gland enlargement (1 of 6). Relevant microscopic findings included passive hepatic congestion (6 of 6) and pulmonary congestion (2 of 6), lymphocytic thyroiditis (2 of 6), and diffuse thyroid follicular hyperplasia (2 of 6). Microscopically, the heart was unremarkable (2 of 6) or had mild lymphocytic myocarditis (2 of 6), mild multifocal cytoplasmic vacuolation of cardiomyocytes (2 of 6), mild lymphocytic myocardial (Purkinje cell) ganglioneuritis (1 of 6), and mild multifocal interstitial fibrosis and nuclear hypertrophy of cardiomyocytes (1 of 6). One parrot had concurrent proventricular dilatation disease (systemic ganglioneuritis). The cause of cardiomyopathy in these parrots was not determined.
Keywords
Cardiac disease is infrequently documented in birds, with the exception of some forms of cardiomyopathy in domestic turkeys and chickens and infectious and parasitic myocarditis in a variety of species. Numerous causes of cardiac disease have been identified in birds, including viruses (eg, avian leukosis subgroup J, 23 West Nile virus, 25,29 bunyavirus 22 ), bacteria, 20 parasites (eg, toxoplasmosis, 26 sarcocystosis, 10,27 schizont infection 12 ), valvular degeneration/insufficiency, 2,15 intoxications (eg, salt, 14 selenium, 11 avocado, 3 ionophore antibiotics, 1 furazolidone 18 ), pulmonary hypertension (eg, in rapidly growing chicks, 5 high altitude disease, 4 pulmonary disease 15 ), and nutritional diseases (eg, vitamin E deficiency, 13 phosphorus deficiency 9 ). Endocrine disease as a cause of cardiomyopathy is well documented in mammals—particularly, humans and domestic carnivores; however, to the best of our knowledge, cardiac disease in birds with endocrinopathies has not been reported.
Except for infectious and parasitic disease, most of this information involves domestic avian species, and cardiomyopathy with congestive heart failure appears to be rarely reported in nondomestic birds. In a retrospective study of cardiac disease in 26 parrots of various species, 15 had congestive heart failure, 2 of which were African grey parrots (Psittacus e erithacus); 15 however, details of cardiac gross pathology are limited. Avian neonatal/juvenile cardiomyopathy is also rarely documented in nondomestic birds; in the aforementioned study, 15 the age of affected parrots was not provided. Here we document the clinical and pathological findings of cardiomyopathy with congestive heart failure in 6 African grey parrots, including 2 neonates and 3 juveniles; of these, 2 juvenile parrots had hatched from the same female in different clutches and had prominent thyroid lesions.
Table 1 details the signalment, clinical findings, and outcome for the 6 parrots. Ages ranged from 15 days to 8.8 years, and 5 of 6 parrots were either neonates or juveniles at the time of diagnosis. Two neonates and 2 juveniles (Nos. 1–4) originated from a single breeding aviary with 350 breeding pairs; the other 2 parrots (Nos. 5 and 6) were kept as pets. Two parrots (Nos. 1 and 2) were born in 2004 and 2005 to the same breeding pair. Overall, this breeding pair produced numerous infertile eggs, particularly in 2001 and from late 2005 to date (January 2009), periods in which no fertile egg/clutches were produced. From late 2002 to roughly mid-2005, this pair had 18 chicks and 1 fertile egg with embryonic death. Of the 18 chicks, 2 had cardiomyopathy (Nos. 1 and 2), and 3 from 2 different clutches in 2003 were cardiomyopathy suspects based on similar clinical findings and limited pathology reports (eg, cardiomegaly, coelomic effusion). The remaining 13 chicks were alive as of the writing of this article (January 2009) and with no clinical evidence of cardiomyopathy. Clinical signs included marked distention of the coelomic cavity (4 of 6), rales (3 of 6), weakness (4 of 6), ruffled plumage (1 of 6), growth retardation (1 of 6), crop stasis (1 of 6), regurgitation (1 of 6), and paralytic ileus and hematochezia with marked anemia (packed cell volume, 9% rising to 28% after three blood transfusions; 1 of 6). Radiographically, parrot No. 6 had mild cardiomegaly 24 and coelomic effusion; by electrocardiography, bradyarrhythmia with low voltage waves was detected. Duration of disease ranged from 2 days to 2 months. It was not recorded exactly in 4 parrots (Nos. 1–4) but was less than 5 days in Nos. 3 and 4 and about 1 week in Nos. 1 and 2, according to the breeder; however, Nos. 1–3 were euthanized, so the duration of disease might have been longer.
Animal Data and Clinical Findings in 6 African Grey Parrots With Cardiomyopathy
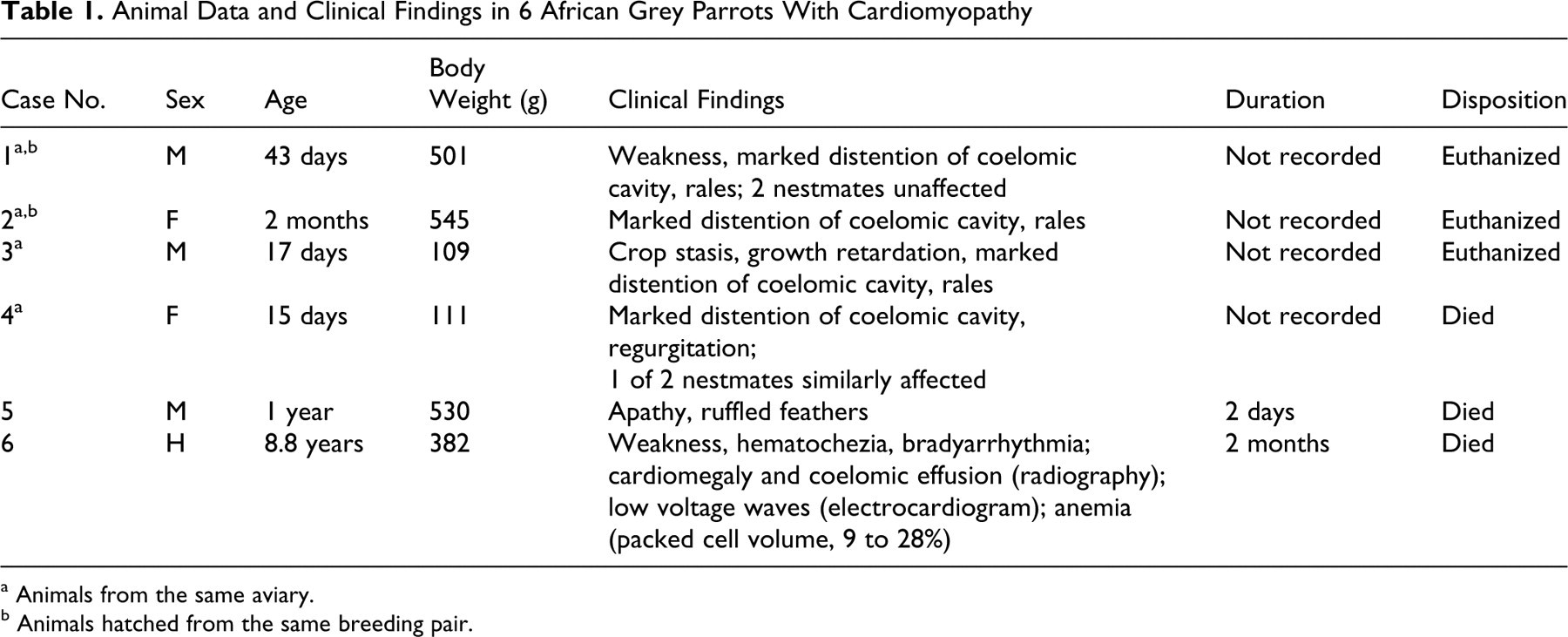
a Animals from the same aviary.
b Animals hatched from the same breeding pair.
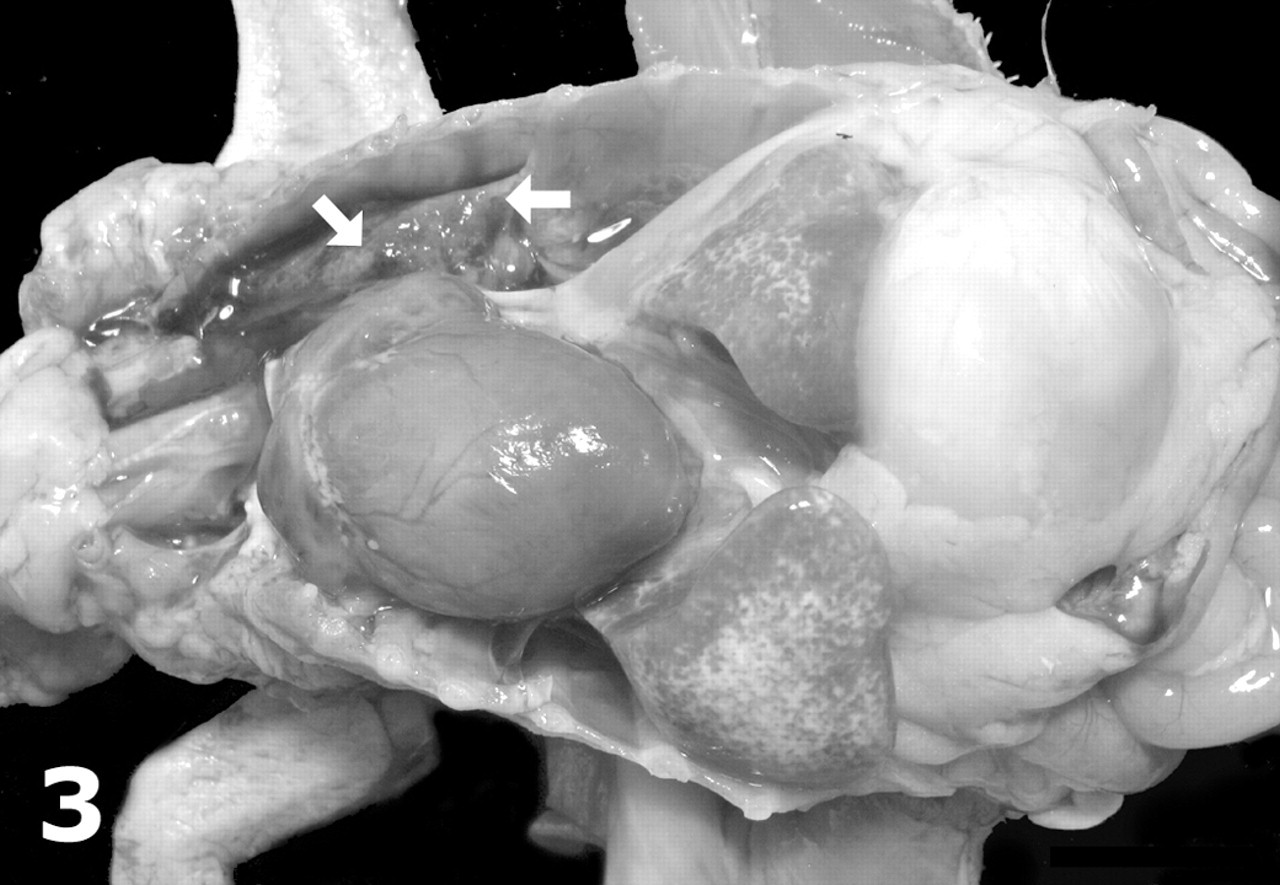
Table 2 details the gross findings in these parrots. All parrots had varying degrees of dilatation of the coelomic cavity due to accumulation of yellow translucent fluid (Figs. 1, 2) with a few fibrin strands; in two cases (Nos. 2 and 5), this coelomic effusion partly clotted after opening the coelomic cavity. In one parrot (No. 5), the serosal surfaces of the liver, spleen, and air sacs were thickened and moderately opaque, and there were adhesions between intestinal loops. Mild ventral subcutaneous edema was observed in 2 parrots (Nos. 4 and 5), and parrot No. 5 had mild hydropericardium. All parrots had enlarged hearts with rounded vertices and variable cardiac chamber dilatation with ventricular thinning (Figs. 2–5, Table 2); ventricular wall thickening was apparent in only 2 parrots (Nos. 3 and 5). Cardiomegaly was most severe in the chicks (Nos. 1–4): Parrot Nos. 1–3 had biventricular enlargement, and No. 4 presented with right-sided enlargement. Parrot No. 5 had left-sided hypertrophic cardiomyopathy; milder cardiomegaly in this parrot was attributed to mild ventricular dilatation. Details on the gross findings in parrot No. 6 were not recorded, but based on gross photographs provided by the referring veterinarian, cardiomegaly was mild; the pericardium was opaque and thickened; and there were adhesions between the parietal and visceral pericardium.
Gross Findings in 6 African Grey Parrots With Cardiomyopathy a
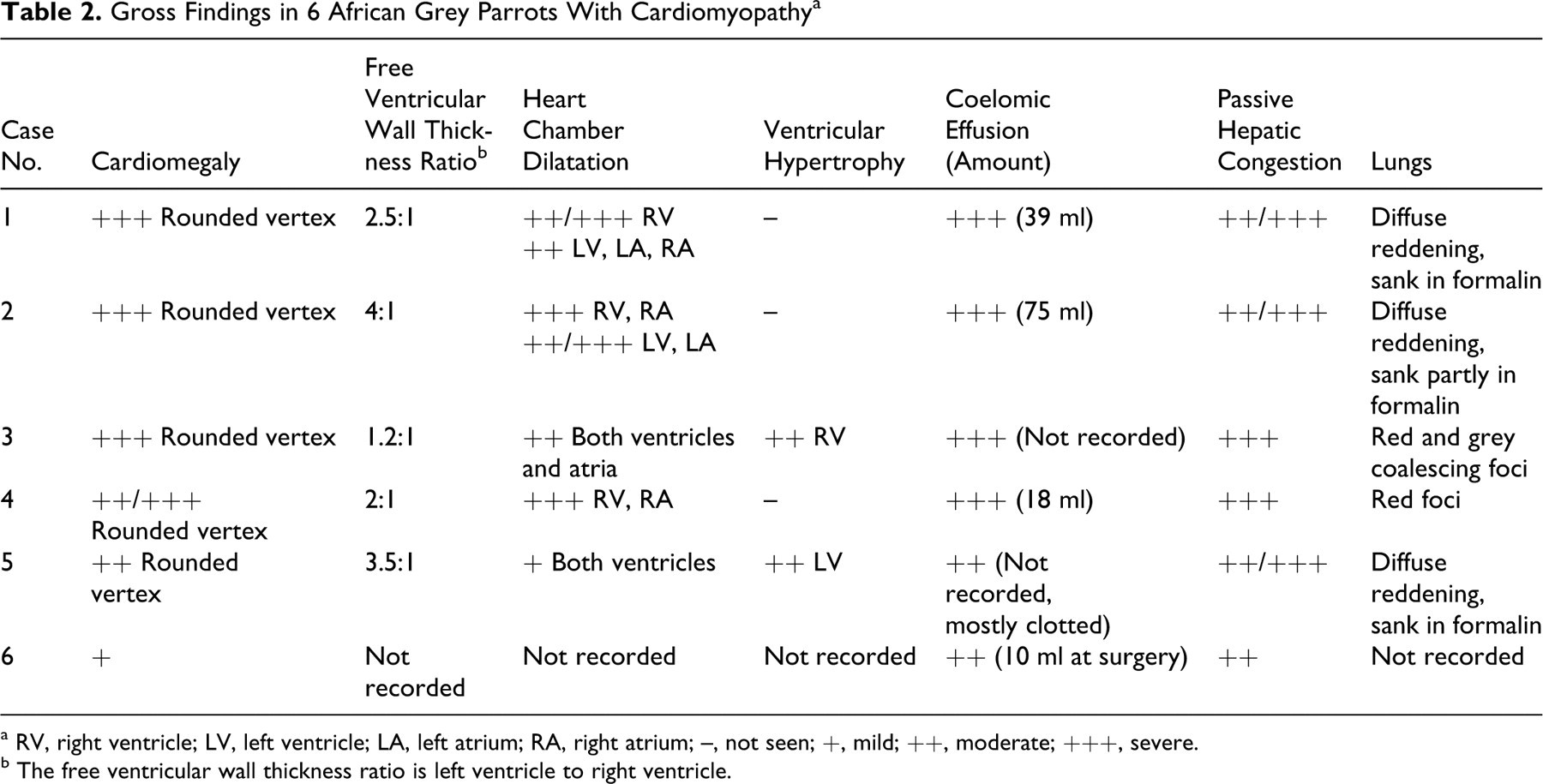
a RV, right ventricle; LV, left ventricle; LA, left atrium; RA, right atrium; –, not seen; +, mild; ++, moderate; +++, severe.
b The free ventricular wall thickness ratio is left ventricle to right ventricle.
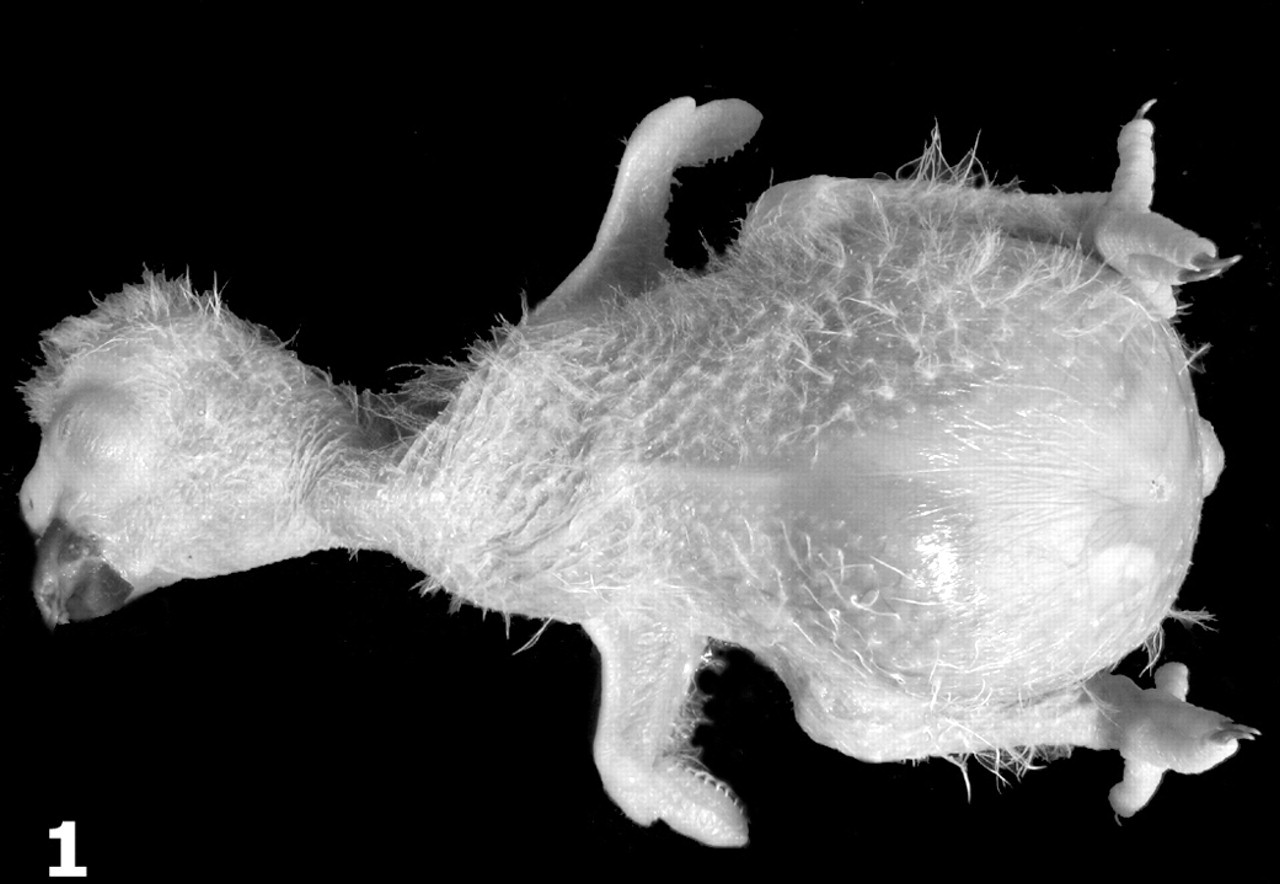
African grey parrot, case No. 3. Note marked distention of the coelomic cavity.
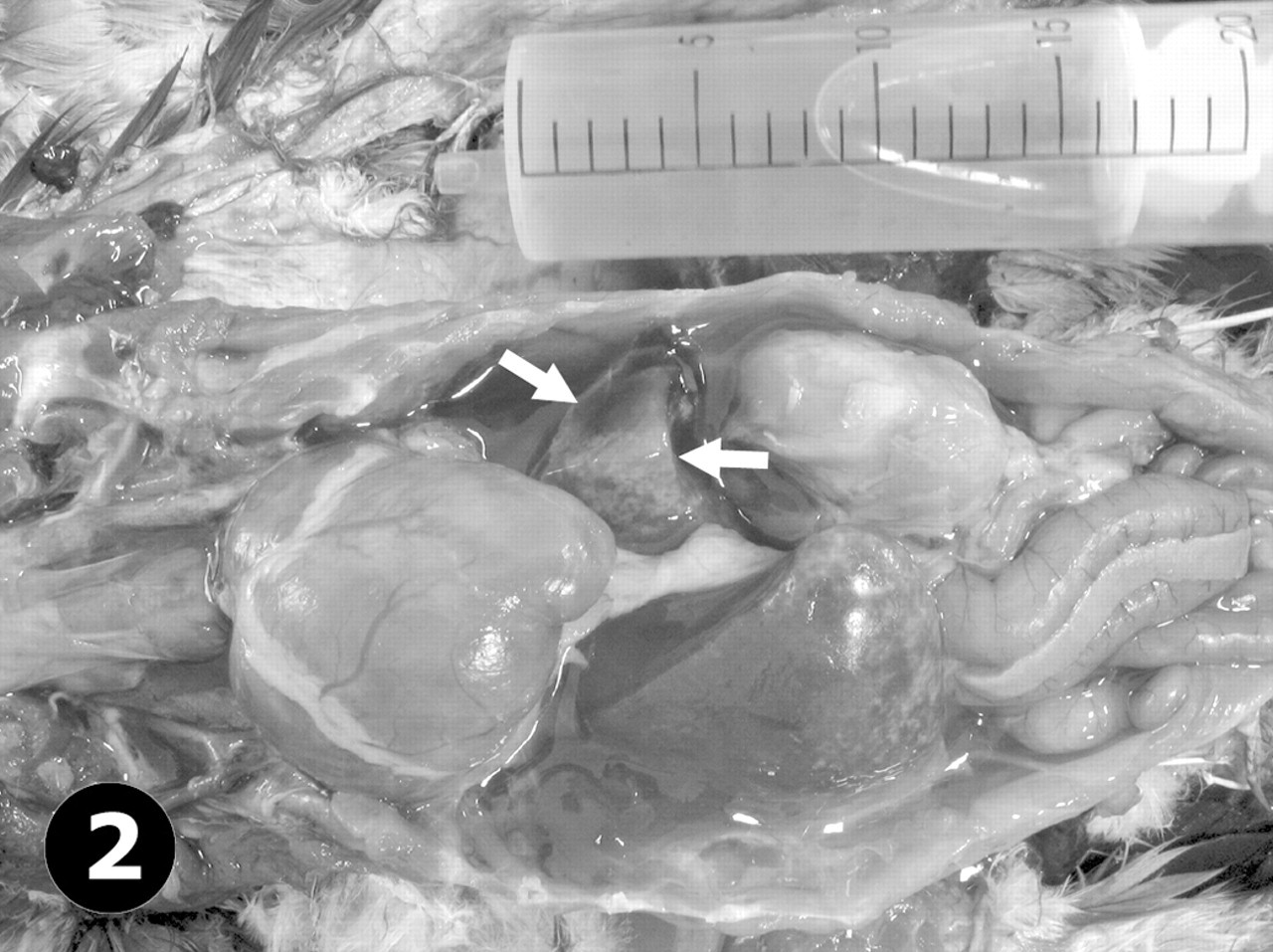
African grey parrot, case No. 2. Prominent coelomic effusion (syringe) and cardiomegaly are observed. Note severe dilatation of the right ventricle and atrium and a rounded vertex. The liver—particularly, the left lobe (arrows)—is atrophied and contains numerous pale foci.
African grey parrot, case No. 3. Note marked cardiomegaly with a rounded heart, as well as numerous pale foci on the surface of the liver, which is mildly atrophic.
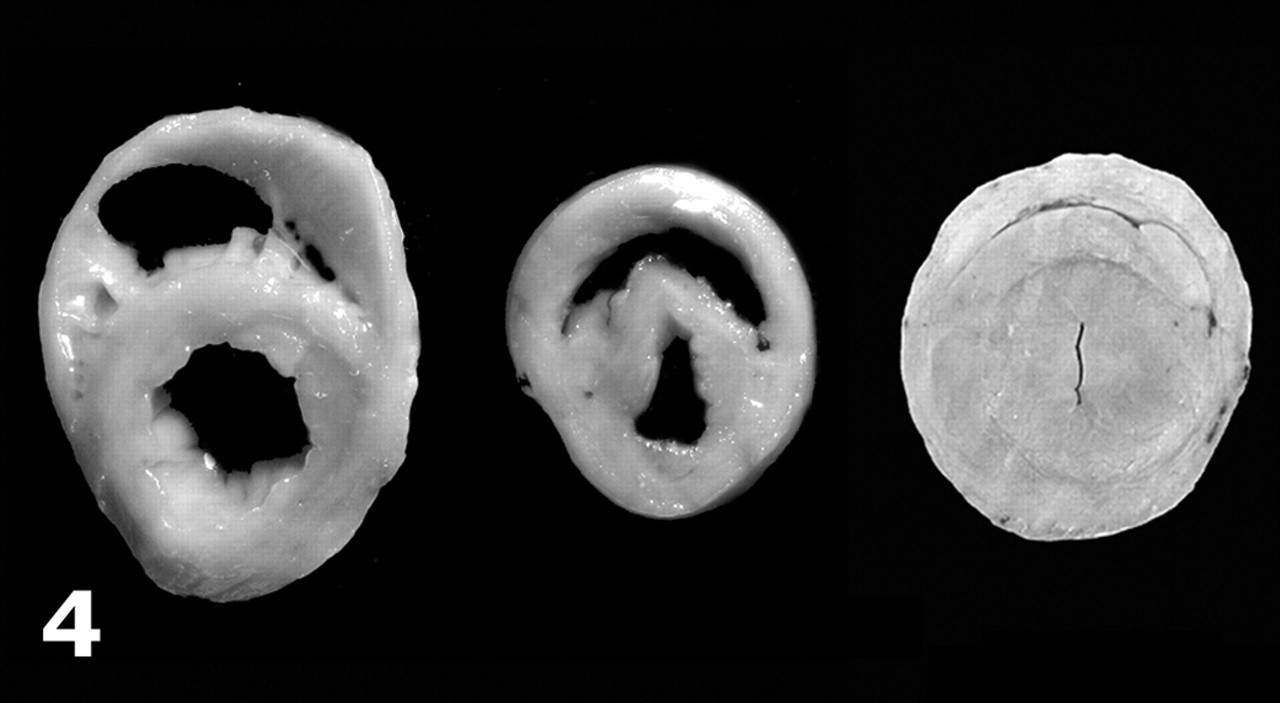
African grey parrots, case Nos. 2 (left) and 3 (center) and control parrot (right); formalin-fixed heart. Transverse sections of the heart in which biventricular dilation is prominent (case No. 2) or mild (case No. 3). Hypertrophy of the right ventricle is prominent in case No. 3. Compare with a normal heart from a 530-g adult African grey parrot (right).
In 5 parrots, the consistency of the liver was increased, and there was hepatic atrophy, which was most severe in the left hepatic lobe of Nos. 2 and 5. In all 6 parrots, abundant white or yellow coalescing foci were found in the liver, most prominently in subcapsular areas (Figs. 2 and 3). Multifocal to coalescing or diffuse red or grey discoloration of the lungs (Fig. 3) was observed in 5 parrots; for 3 parrots, the lungs sank in formalin. Moderate enlargement of both thyroid glands was noted in parrot No. 1 (the left thyroid was 7 × 5 × 5 mm; the right, 5 mm in diameter); the glands were pale and had an irregular external surface (Fig. 5). Four parrots (Nos. 1–4) were in good body condition, and parrot Nos. 5 and 6 had severe atrophy of adipose tissue; parrot No. 5 also had moderate to severe muscular atrophy. Moderate luminal hemorrhage in the intestine was noted in parrot No. 6.
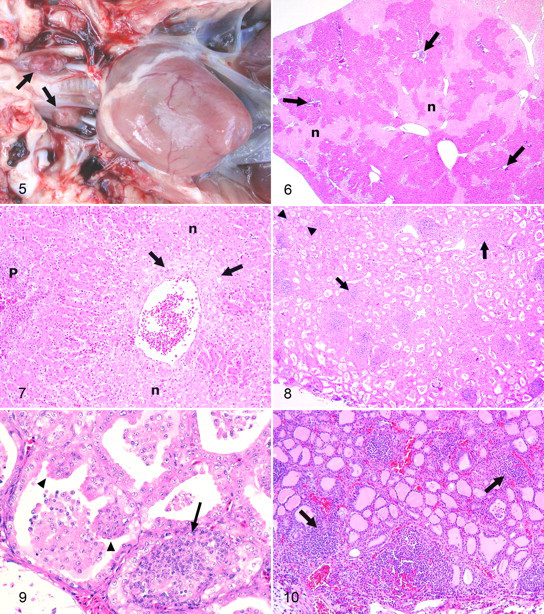
Microscopically, the heart was unremarkable (2 of 6) or had mild multifocal lymphocytic or lymphoplasmacytic interstitial myocarditis (Nos. 2 and 6), mild multifocal cytoplasmic vacuolation of cardiomyocytes (Nos. 1 and 5), mild lymphocytic myocardial (Purkinje cell) ganglioneuritis (No. 6), and mild multifocal interstitial fibrosis and nuclear hypertrophy of cardiomyocytes (No. 5). In all parrots, hepatic lesions consisted of chronic passive congestion characterized by varying degrees of dilatation and fibrosis of centrilobular veins with centrilobular coagulative hepatocellular necrosis and some mild infiltration of necrotic tissue by heterophils (Figs. 6, 7). These lesions were more often intense in the subcapsular areas of the parenchyma. In some cases, fibrosis extended from the centrilobular veins to the Disse space of adjacent hepatocyte cords. Two parrots (Nos. 3 and 5) had microscopic evidence of passive pulmonary congestion, which consisted of extravasation of erythrocytes within the lumen of air capillaries, phagocytosis by macrophages of erythrocytes, hemosiderosis of luminal and interstitial macrophages, interstitial fibrosis, and/or hypertrophy of the respiratory epithelium.
Two parrots (Nos. 1 and 2) had moderate to severe bilateral multifocal, often nodular, interstitial infiltration of lymphocytes in the thyroid glands (Figs. 8–10); fewer heterophils were observed in the infiltrates of No. 2. Inflammation caused varying degrees of collapse and compression atrophy of adjacent follicles (Figs. 8–10). In addition, follicles in the thyroid glands of No. 1 contained scarce or no colloid and were lined by cubical or cylindrical epithelium that frequently formed papillary projections into the follicular lumen, which was variably collapsed and/or contained desquamated follicular cells (Figs. 8, 9). Similar multifocal follicular lesions were observed in parrot No. 5 but were mild and lacked papillary projections of the follicular epithelium.
Parrot No. 6 had concurrent proventricular dilatation disease (PDD; systemic ganglioneuritis). Other lesions consisted of acquired atelectasis and acute luminal hemorrhage (parrot Nos. 1, 3, 4, and 6) and/or proteinaceous edema (parrot Nos. 2 and 6) in the lungs, as well as acute interstitial pneumonia (parrot No. 6).
Six parrots, including 4 neonates/juveniles, were diagnosed with cardiomyopathy and congestive heart failure based on pathologic findings (cardiac lesions, coelomic effusion, and passive congestion in the liver and lungs). When present, microscopic myocardial lesions were mild and nonspecific and did not contribute to an etiological diagnosis of the cardiac disease. Myocarditis was apparent in only 2 parrots and was mild; thus, infectious or parasitic disease seems an unlikely cause of cardiomyopathy in these animals. Of these, 1 parrot had concurrent PDD, the likely cause of myocarditis; 28 visceral inflammation not involving nervous ganglia can be observed in a variety of tissues in psittacines with PDD. Microscopic myocardial lesions suggestive of specific causes of cardiac disease (eg, vitamin E deficiency, ionophore antibiotic toxicity, parasitism, or bacterial infection) were not present in any parrot, and cardiovascular malformations were not observed. Nutritional studies and toxicological analyses were not carried out to evaluate other possible nutritional and toxic cardiomyopathies.
Endocrine and genetic disease was considered a possible cause of cardiomyopathy in 2 chicks (Nos. 1 and 2) hatched from the same female, based on the familial occurrence of prominent lymphocytic thyroiditis in these parrots and the diffuse colloid goiter in 1 of them (No. 1). In humans with Grave disease (autoimmune thyroiditis), in which autoantibodies stimulate the thyroid-stimulating hormone receptor in the thyroid follicular epithelium and can migrate transplacentally, similar thyroidal lesions and cardiomyopathy can occur in the fetus and neonate. 16 There is also an avian model of genetic autoimmune lymphocytic thyroiditis in the obese strain of chickens, as characterized by thyroglobulin autoantibodies with development of thyroiditis during the neonatal period; 6 however, cardiomyopathy has not been reported in these birds. Inherited cardiomyopathy in turkeys also develops during the neonatal/juvenile period, 7,17 but it is not associated with thyroiditis.
Of the other 16 chicks produced by the same breeding pair, 3 from two clutches in 2003 had similar clinical signs and were cardiomyopathy suspects, but detailed pathology records were not available and were not included in this study. Again, during the preparation of this article, the remaining 13 chicks were alive, with no clinical evidence of cardiomyopathy. Of 14 additional grey parrots submitted for necropsy from the same breeder (4 chicks [including case Nos. 3 and 4] and 10 adults), only 1 chick had thyroiditis, which was lymphocytic and effaced most of the thyroid glands, with only a few hyperplastic follicles visible, and which was also associated with mild left-sided hypertrophic cardiomyopathy. This chick hatched to a different breeding pair and was not included in this study, because cardiomyopathy was mild and not accompanied by congestive heart failure. The possibility of an inherited or congenital component in the etiology of thyroiditis and thyroid follicular hyperplasia in these chicks requires further evaluation of affected breeding females. Clinical evaluation with hematological and endocrinological testing of these breeding females and any affected and unaffected chicks from future clutches and comparison with unaffected, age-matched parrots, as well as gross and histopathologic exams of any dead embryo, may be helpful in further evaluating this possibility but a recommendation has been made not to use the breeding pair of cases Nos. 1 and 2 for breeding.
Diffuse thyroid follicular hyperplasia is common in captive birds and often attributed to dietary iodine deficiency or excess and/or the presence of goitrogenous compounds in the diet or drinking water. 8,19 In humans with Grave disease, goiter can be a component of autoimmune thyroid disease. 21 No other cases of thyroid hyperplasia have been diagnosed in parrots from this center; thus, nutritional or toxic causes seem unlikely.
Interestingly, parrot No. 4 had 1 of 2 nestmates similarly affected, according to clinical and gross findings, and one subsequent clutch had cardiomyopathy suspects; however, detailed pathology records are not available and were not included in this report. Parrot No. 4 had thyroid glands histologically within normal limits.
Hypoxia occurring in rapidly growing chicks can induce primary pulmonary hypertension and right-sided cardiomyopathy with ascites in poultry; 5 this might have contributed to the development of cardiomyopathy in parrot Nos. 1–4, which originated from a single parrot breeding center.
In conclusion, cardiomyopathy with mostly right-sided congestive heart failure was diagnosed in 6 African grey parrots based on gross and histopathologic findings; the causes of cardiac disease were not identified in any parrot. However, 5 of these parrots were either neonates or juveniles, 4 of them from a single parrot breeding center; hypoxia-induced pulmonary hypertension in rapid growth in chicks may have favored the development of cardiac disease in the younger chicks. Additionally, 2 of these chicks, hatchlings from a same breeding pair, had advanced thyroidal lesions, which suggests the possibility of genetic endocrine disease in the pathogenesis of cardiomyopathy, but further testing is needed in this regard. The remaining parrot had advanced PDD with mild myocarditis and myocardial (Purkinje cell) ganglioneuritis, which might have contributed to cardiomyopathy in this case.
Footnotes
Acknowledgements
We thank ConZOOlting Wildlife Management for support in conducting this study, as well as an anonymous parrot breeder for submission of 4 cases and for providing detailed histories of affected chicks and breeding pairs.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
