Abstract
Objectives
The aim of the study was to evaluate the efficacy and safety of capromorelin to manage weight loss in cats with unintended weight loss, as occurs in chronic kidney disease (CKD), in a randomized, masked, placebo-controlled, multicenter clinical field study.
Methods
A total of 176 client-owned cats with existing CKD and unintended weight loss of 5% or more were enrolled. Cats were randomized 2:1 to receive capromorelin or a vehicle placebo orally once daily for 55 days. Changes in body weight and safety parameters were monitored throughout the study.
Results
Body weight increased progressively with time in the capromorelin group and decreased in the placebo group. For the effectiveness population data sample (n = 112), mean change in body weight from day 0 to day 55 was +5.18% (95% confidence interval [CI] 3.45–6.91) with capromorelin and –1.65% (95% CI –3.82 to 0.55) with placebo. The treatment effect (capromorelin minus placebo) from day 0 to day 55 was +6.81% (95% CI 4.21–9.42) with P <0.0001, representing +0.25 kg (95% CI 0.15–0.35) body weight. Hypersalivation was observed only in the capromorelin group (P <0.0001). For all other adverse events (AEs), there was no significant difference between the treatment groups: in the capromorelin group 96/118 (81.4%) cats and in the placebo group 41/58 (70.7%) cats had at least one reported AE (P = 0.3650).
Conclusions and relevance
Capromorelin was safe and effective, and provides a valuable new option to maintain or increase body weight in cats with CKD.
Introduction
Unintended weight loss in cats is observed in chronic conditions leading to cachexia and sarcopenia. Both are associated with higher morbidity and mortality rates due to depletion of lean body mass (LBM), although sarcopenia refers to loss of LBM attributed to aging, even in the absence of disease. 1 Weight loss in cats is frequently associated with chronic kidney disease (CKD),2 –4 and can begin up to 3 years before the disease is diagnosed and worsens after diagnosis. 5 Several factors contribute to this weight loss, including reduced food consumption, metabolic changes related to aging, and/or secondary to physiological changes due to CKD itself or comorbid conditions. 6
Accurate CKD diagnosis and a tailored treatment plan positively influence a cat’s prognosis and may extend life expectancy. Managing sequelae including dehydration, hyperphosphatemia, hypokalemia, hypertension, proteinuria and anemia are mainstays. Nutritional support has been an important factor in managing the disease as well as promoting weight gain in these patients. However, concomitant inappetence complicates attempts to increase caloric intake or utilize therapeutic renal diets. Shifting metabolism to promote weight gain, as well as stimulation of food intake, could promote optimal nutrition, restore weight and muscle mass, and potentially increase life expectancy in cats.
Capromorelin is a selective ghrelin receptor agonist (GRA), mimicking the effects of the endogenous hormone ghrelin. Ghrelin and GRAs bind to receptors in the hypothalamus to stimulate appetite and in the pituitary gland to stimulate secretion of growth hormone (GH); hence, the receptor is also referred to as the growth hormone secretagogue receptor 1a. 7 Increased GH stimulates insulin-like growth factor 1 (IGF-1) release from the liver, which has a negative feedback action on GH secretion. 8 The ghrelin/GH/IGF-1 axis is closely connected with growth via anabolic metabolism and many conditions are associated with abnormalities in this pathway. 9 Notably, ghrelin was shown to be dysregulated in cats with CKD. 10
Recent research has indicated a more nuanced role of ghrelin acting as a type of nutrient sensor wherein stimulation of nutrient intake (‘hunger’) is only one effect. 11 In addition to the orexigenic effects (stimulating food-seeking behaviors, desire to eat and satisfaction with eating), ghrelin also promotes weight gain with increases in both adipose and LBM.9,12 Independent of ghrelin’s stimulation of GH release, it appears to target muscle cells directly, resulting in improved muscle strength and survival. 13 Elderly humans treated with capromorelin for 12 months 14 or ibutamoren (MK-677) for 24 months 15 demonstrated sustained gains in body weight and LBM. GRAs have also been reported to increase body weight and LBM in cancer patients treated for cancer-associated anorexia and cachexia syndrome.16 –18
Capromorelin is the only GRA authorized for use in animals for the management of weight loss in chronically diseased cats (Elura/Eluracat; Elanco Animal Health) and for appetite stimulation in dogs (Entyce; Elanco Animal Health). During safety evaluations of capromorelin in healthy laboratory cats receiving capromorelin for up to 6 months, increased food consumption and body weight gain were observed.19,20 This clinical field study evaluated capromorelin’s effectiveness in reversing unintended weight loss and promoting weight gain in client-owned cats with CKD, a condition commonly seen in feline practice.
Materials and methods
Animal enrollment
This was a multicenter, placebo-controlled, randomized and masked clinical field study conducted in client-owned cats under owner consent at 20 general veterinary practices in the USA. The study protocol was concurred by the Food and Drug Administration Center for Veterinary Medicine and the sponsor’s Institutional Animal Care and Use Committee before study initiation (AT002-FCL-16-004). The study was conducted according to Good Clinical Practices (VICH GL9) and this manuscript followed CONSORT guidelines. 21
There were no restrictions on age, breed, sex or baseline weight.
To be eligible, cats needed to have been diagnosed with CKD at least 30 days prior, had a documented unintended weight loss of at least 5% over the past 3 years, and other medical conditions and treatment regimens were stable. To avoid interference with the study objective, cats with confirmed congestive heart failure, cancer, diabetes insipidus or diabetes mellitus were excluded. Any stage of CKD per International Renal Interest Society (IRIS) Guidelines 2023 was acceptable. 22
Randomization, masking and study schedule
Before the study, randomization tables unique for each site were generated using SAS Proc Plan (SAS version 9.3). Upon inclusion, cats were randomly assigned to 1/3 treatment codes, with a block length of three, where two codes corresponded to capromorelin and the remaining code corresponded to placebo. Only the treatment dispenser, owner and study monitors knew the assigned treatment code. The investigator, examining veterinarian, study support staff, safety monitor, statistician and sponsor representative remained masked to treatment information throughout the study.
Screening of cats could take place up to 7 days before enrollment and administration of the first dose (day 0). Before treatment, physical examination, body weight measurement, body condition scoring (BCS),23,24 muscle condition scoring (MCS)24,25 and clinical pathology tests (hematology, serum chemistry, total serum thyroxine measurement, urinalysis and fecal analysis) were performed. Owners listed the foods their cat ate and were instructed to maintain this diet, making changes only after consultation with the investigator. Physical examination, body weight and clinical pathology testing (hematology, serum chemistry and urinalysis) took place during visits on day 15 ± 2, day 27 ± 3 and day 55 ± 4 (Figure 1). Unscheduled visits occurred as needed. All clinical pathology samples collected during the study were submitted to a central laboratory (IDEXX Laboratories).

Schedule of study events. AEs = adverse events
Study medication
Capromorelin oral solution (Elura; Elanco Animal Health) was administered at a dose of 2 mg/kg (0.1 ml/kg) for 55 days. The placebo group received a vehicle control (identical oral solution without the active ingredient) at the same volume (0.1 ml/kg) and duration. The dose was calculated based on the body weight on day 0 and was not adjusted during the study to not draw attention to changes in weight. The owner administered the first dose in the clinic under guidance of the treatment dispenser, and the remaining doses were given at home once daily by the owner using an oral dosing syringe. Owners were advised to offer a meal at least 30 mins after dosing if their cats were fed meals. A missed dose was defined as follows: dose was not administered or not documented; partial dose was administered and the cat was not re-dosed; or the cat vomited within 15 mins of dosing and was not re-dosed.
Although treatments interfering with the study’s objectives were prohibited and would lead to non-enrollment or withdrawal, necessary treatments for illness or injury were always allowed.
Effectiveness and safety assessments
Effectiveness was assessed by body weight measurements in kilograms (kg) or pounds, recorded to at least one-tenth of a kg or ounce. Safety was monitored through clinical pathology tests, physical examinations and adverse events (AEs). AEs were identified based on owner observations and veterinarian evaluations. AEs included any unfavorable, unintended observations after the use of study medication, whether considered to be treatment related or not.
Analysis populations
The intention to treat population included all randomized cats that received at least one dose of medication and is referred to as the safety population (SP). The per-protocol population included all cats that were not withdrawn or excluded and is further referred to as the effectiveness population (EP). EP analyses required each site to have two evaluable cases in each treatment group. Based on a power analysis using results from a pilot study (data on file) and regulatory expectations for enrollment, we aimed for 150 cases but enrolled 176 cases to ensure sufficient cases for effectiveness analyses. The sample size of 100 capromorelin-treated cats and 50 placebo-treated cats of evaluable cases provides more than 99% power (alpha = 0.05, two-sided) to detect a difference in the treatment success between groups. Figure 2 depicts case flow for the SP and EP.
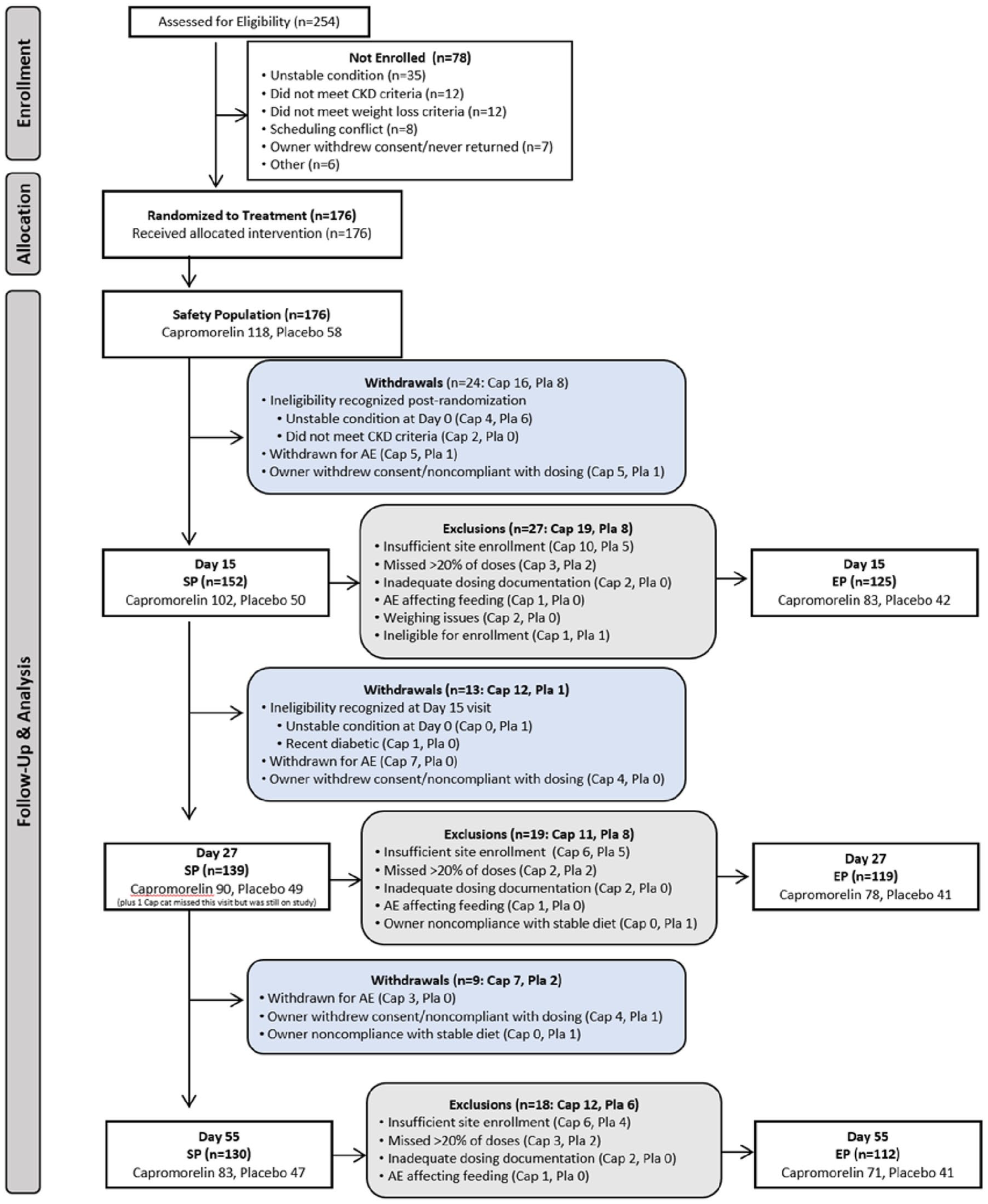
Case flow for the safety population (SP) and effectiveness population (EP). AE = adverse event; Cap = capromorelin; CKD = chronic kidney disease; Pla = placebo
Statistical analyses
Statistical analyses were conducted using SAS (SAS Institute) with the individual patient as the experimental unit and alpha = 0.05.
Efficacy analyses
Body weight was the only efficacy endpoint assessed. A model with treatment as a fixed effect, and site and site*treatment as random effects, was used. There was no formal imputation for missing data. Two-tailed P values and 95% confidence intervals (CIs) are reported for change in body weight. Assumptions of normality of residuals were investigated for the least square mean (LSM) percent change from day 0 to day 15, 27 or 55. If the normality assumption was rejected at the 1% level by the Shapiro–Wilk test, 26 then an analysis using rank transformation was performed. The main effectiveness variable was the LSM percent change in body weight from day 0 to day 15, day 27 and day 55 in the EP. The SP sample underwent the same analyses for supportive effectiveness data.
An additional analysis was conducted on the EP on day 55. A cat was classified as a treatment success if its body weight on day 55 was equal to or greater than its day 0 weight or a treatment failure if it was lower.
Safety analyses
Safety was assessed through the occurrence of AEs and physical examination, hematology, serum chemistry and urinalysis variables. As the main safety variable, AEs were coded, tabulated in frequency tables and evaluated by treatment group using descriptive statistics. Clinical pathology data were tested for normality and analyzed using a variance model with treatment and day as fixed effects. The LSM and standard errors were calculated along with 95% CI about the means. The differences (and standard errors) between capromorelin and placebo were also estimated along with the 95% CI.
Results
Demographics
A total of 254 cats were screened, of which 176 were enrolled. Although all 176 cats were diagnosed with CKD on day 0, laboratory results from samples collected on day 0 were subsequently found to be inconsistent with CKD in two cases assigned to the capromorelin group.
The SP data set consisted of 176 cats (capromorelin n = 118, placebo n = 58). Enrolled cats had a mean age of 15.1 years (range 4.4–22.1) with a mean body weight of 4.1 kg (range 1.8–6.8) at enrollment, summarized in Table 1. Comorbidities were common and included, in descending incidence: dental disease; moderate or severe muscle loss; heart murmur; history of vomiting and gastrointestinal disease; hyperthyroidism; and hypertension (Table 2).
Demographic characteristics of cats enrolled in the study

Data are n (%) or mean ± SD (range). Body condition score (BCS) was in the range of 1–9; 1 = emaciated, 8 = obese and 9 = grossly obese were not utilized. Muscle condition score (MCS) was in the range of 0–3. Body weight differences between the groups were compared by t-test, and chronic kidney disease (CKD) stage, BCS and MCS frequencies by χ2 analysis
Statistically significant difference (P ⩽0.05)
IRIS = International Renal Interest Society
Summary of comorbid conditions on day 0

Data are n (%). Comorbidity frequencies of the two groups were compared using χ2 analysis
Statistically significant difference (P ⩽0.05)
Before study completion, 46/176 cases were withdrawn: 35 (29.7%) from the capromorelin group and 11 (19.0%) from the placebo group; of these, 15 capromorelin-treated cat and one placebo-treated cat were withdrawn because of AEs.
After completion of the study, cats were excluded from the EP mainly for lack of two evaluable cases in both groups per site, missing more than 20% of doses or inadequate documentation of dosing (Figure 2).
Effectiveness of capromorelin
Capromorelin was effective over the entire 55-day treatment period with body weight increasing in the capromorelin group and decreasing in the placebo group.
In the EP, the LSM percent difference in body weight from day 0 at day 15 was 3.26% (capromorelin n = 83/+3.26%, placebo n = 42/±0.00%; P = 0.0002), at day 27 4.68% (capromorelin n = 78/+3.80%, placebo n = 41/–0.88%; P <0.0001) and at day 55 6.81% (capromorelin n = 71/+5.18%, placebo n = 41/–1.64%; P <0.0001) (Figure 3).

Percentage change in body weight from day 0 (effectiveness population). Least square mean (LSM) percentage change in body weight from day 0; whiskers represent the range and asterisks indicate statistically significant differences. P values were derived from analysis of variance modeling of percentage change in body weight (day 15) or rank-transformed percentage change in body weight (days 27 and 55)
The results in the SP were similar, with LSM percent difference in body weight at day 15 of 3.14% (capromorelin n = 102/+3.18%, placebo n = 50/+0.04%; P <0.0001), at day 27 4.26% (capromorelin n = 90/+3.58%, placebo n = 49/–0.67%; P <0.0001) and at day 55 5.84% (capromorelin n = 83/+4.16%, placebo n = 47/–1.67%; P <0.0001) (Figure 4). The actual body weights are summarized in Table 3.

Percentage change in body weight from day 0 (safety population). Least square mean (LSM) percentage change in body weight from day 0; whiskers represent the range and asterisks indicate statistically significant differences. P values were derived from analysis of variance modeling of percentage change in body weight (day 15) or rank-transformed percentage change in body weight (days 27 and 55)
Descriptive statistics of body weight (in kg) of the effectiveness population (EP) and safety population (SP) from baseline to days 15, 27 and 55

The success analysis of treatment with capromorelin on day 55 showed success rates of 83.1% and 41.5% for the capromorelin and placebo groups (P = 0.001), respectively.
Safety of capromorelin
Capromorelin was well tolerated. All 176 enrolled cats were dosed at least once and included in the SP. At least one AE was reported in 96/118 (81.4%) cats in the capromorelin group and 41/58 (70.7%) cats in the placebo group. Except for hypersalivation, AEs were not significantly different between the groups. The overall difference in AEs was not significantly different, whether hypersalivation was included or not, at P = 0.0563 and P = 0.3650, respectively (Table 4). Hypersalivation was observed in 25 (21.2%) capromorelin-treated cats but not in placebo-treated cats. Of these cats, eight were reported to have salivated on more than 50% of study days and completed the study with an average increase of body weight of 6.1%.
Adverse events reported in more than 5% of cases in either group

Data are n (%). Frequency of adverse events in the two groups was compared using χ2 analysis
Statistically significant difference (P ⩽0.05)
CKD = chronic kidney disease; SDMA = symmetric dimethylarginine
Results for serum chemistry, hematology and urinalysis were overall similar between groups regarding both results by study day and changes from day 0. At baseline, a higher proportion of cats in the capromorelin group were above the reference intervals for alkaline phosphatase, blood urea nitrogen, cholesterol, creatinine, glucose, symmetric dimethylarginine, monocytes, neutrophils, white blood cells and urine specific gravity. The LSM of blood calcium in the capromorelin group was higher than the placebo group with statistical significance on days 15, 27 and 55. The LSM of hemoglobin was statistically significantly lower in the capromorelin-treated cats on days 15 and 55. Complete results for clinical pathology data are presented in the supplementary material.
Discussion
In this study, capromorelin was well tolerated and effective in promoting weight gain in cats with unintended weight loss. This study demonstrates that the GRA capromorelin can be useful in managing unintended weight loss in cats with CKD and other comorbidities. In the EP, the capromorelin-treated cats already exhibited a mean weight gain after 2 weeks; over the 55-day treatment period they reached an increase of 5.2% while the placebo control group had a mean loss of 1.6%. There was a mean weight change of 0.25 kg between the groups, with a gain of 0.18 kg in the capromorelin group and a loss of 0.07 kg in the placebo group. This difference is even more clinically remarkable considering the baseline mean weight in the capromorelin group was 0.4 kg lower than that in the control group (3.9 vs 4.3 kg).
AEs were common in both groups, as expected in the test population of cats with CKD and weight loss plus additional comorbidities. Many of the AEs were attributable to pre-existing diseases rather than the test articles. Assessment of AEs is complicated by the fact that, despite randomized treatment allocation, the cats assigned to capromorelin had an overall poorer health status at baseline, as indicated by a statistically significant higher number of pre-existing conditions other than CKD and statistically significant lower body weights than the placebo group. This imbalance was considered in the statistical effectiveness analyses (body weight) but not for the safety assessments. It is unclear if the higher number of AE-related withdrawals in the capromorelin group is due to progression of disease or the treatment itself.
The most common AE attributable to capromorelin was hypersalivation, which was very commonly reported in the capromorelin group (21.2%) but not at all in the placebo group. It had no apparent negative effect on effectiveness, as a 6% average weight increase was recorded in the eight cats that salivated after more than 50% of doses. Hypersalivation resolved within a few minutes after dosing. For about one-third of the affected cats, it occurred after each administration. Two cats were withdrawn because of hypersalivation. Increased salivation has also been noticed in healthy dogs after administration of capromorelin. 27 Although the bitterness of the active ingredient plays a role, ghrelin receptors have been found in salivary glands in humans and suggested to be associated with the function of appetite regulation. 28 Thus, the pharmacology of capromorelin could also be a factor for hypersalivation.
Vomiting, inappetence, anemia and lethargy were reported very commonly (>10%) in both the capromorelin and placebo groups, but with a higher frequency in the capromorelin group. It is not clear if any of these signs reflect genuine actions of capromorelin vs the underlying CKD or other comorbid conditions. Although increases in the stage of CKD were reported as AEs, it could not be determined if this was related to treatment or progression of the disease. The higher number of cats showing progression in CKD in the capromorelin group (6.8%) compared with the placebo group (3.3%) is consistent with the previously mentioned AEs and could be a result of their poorer health status from the onset. Vomiting was also reported frequently (twice per week) in cats with CKD treated with placebo in an independent field study. 29 It is unlikely that the reported inappetence could be due to capromorelin, which has been clearly shown to increase food consumption (and body weight) in studies in cats. 20 The high frequency of anemia in both groups can be explained by the fact that normocytic, normochromic and non-regenerative anemia is one sequela of CKD due to reduced renal production of erythropoietin. 30 The clinical relevance of the higher frequency of non-specific lethargy with capromorelin administration is unclear, but may be related to transient reductions in heart rate and blood pressure, as reported in healthy laboratory cats. 19
Low body weight and/or poor body condition have been associated with a shorter survival time in cats with CKD, 5 cancer 31 and heart failure, 32 as well as in a general population of cats visiting a veterinary clinic, with or without illness. 33 In cats with chronic conditions, several mechanisms contribute to weight loss, including inflammation, malabsorption, increased energy requirements and decreased appetite. The orexigenic and anabolic activity of capromorelin, as demonstrated in healthy cats, 19 is likely not limited to cats with CKD, as the diverse comorbidities in this study suggest. Capromorelin offers an opportunity as an adjunct therapy to manage weight loss in other chronic conditions, such as hyperthyroidism, congestive heart failure, cancer and arthritis, in which unintended weight loss is a negative prognostic indicator for cats. Stimulating food consumption and metabolic changes to increase weight could improve the quality of life and health span of cats with chronic conditions, including CKD.
This study should be interpreted in the context of its limitations. BCS and MCS were not measured throughout the study, only at enrollment to characterize the cats’ health status. Hence, conclusions on the impact of capromorelin treatment on these parameters cannot be drawn. Transient reductions in blood pressure and heart rate have been reported after capromorelin administration. 19 Blood pressure was not measured as part of the study, so we were unable to assess the incidence of hypotension after capromorelin administration in this study population. Although heart rate was measured at each visit and no AE was reported for change in heart rate, the timing relative to treatment was not recorded and thus any acute effect on heart rate in this study is unknown.
Conclusions
Capromorelin is expected to be safe and efficacious for long-term management of unintended weight loss in cats with CKD and various comorbidities.
Supplemental Material
Supplemental Material
Safety – least square means and 95% confidence interval for clinical pathology data
Footnotes
Acknowledgements
The authors thank Shilpa Rani for supporting the writing of manuscript, Margie Bell and George Milliken for additional statistical analyses, and the investigators for managing the clinical cases.
Author note
JAW’s affiliation at the time of publication is Animerra. A short abstract summarizing this study was presented at the ANZCVS Annual Meeting, 24–26 July 2025.
Supplementary material
The following file is available as supplementary material:
Safety – least square means and 95% confidence interval for clinical pathology data.
Conflict of interest
JAW is a former employee and EH a current employee of Elanco Animal Health and both are stockholders. AMM was a consultant for Elanco Animal Health.
Funding
The study was funded by Elanco Animal Health.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
