Abstract
Objectives
The aim of the study was to compare the neutrophil:lymphocyte ratio (NLR) and platelet:neutrophil ratio (PNR) in cats with or without hypertrophic cardiomyopathy (HCM) and cardiogenic arterial thromboembolism (CATE) and to evaluate their prognostic significance in cats with HCM.
Methods
Healthy cats and cats with HCM and CATE were prospectively enrolled between 2018 and 2020. Complete blood count and echocardiogram were performed. NLR and PNR were compared between cats in healthy, HCM and CATE groups. Outcome data, including cause of death, time to death and progression of HCM, were collected via review of medical record and owner interviews. Associations between time to cardiac-related mortality, NLR and PNR were analyzed using Kaplan–Meier curves and the Cox proportional hazards regression model.
Results
A total of 110 cats were enrolled, including 42 healthy cats, 57 cats with HCM (37 stage B1, 9 stage B2, 11 stage C) and 11 cats with CATE. Cats with CATE had the highest NLR (5.8, range 3.1–12.7) compared with HCM (P = 0.03) and healthy cats (P ⩽0.0001), while NLR was higher in cats with HCM (3.3, range 1.8–5.1) compared with healthy controls (P = 0.04). PNR was lower in cats with CATE (16.6, range 13.2–27.9) compared with healthy cats (P = 0.0027). Although PNR did not differ between cats in the healthy and HCM groups (P >0.9), HCM-affected cats with PNR below 40 had a significantly lower median survival time (1093–1185 days) compared with those with PNR above 40 (P = 0.03). Cats with PNR below 40 at the time of HCM diagnosis also had a significant risk (hazard ratio 9.8; P = 0.03) of cardiac-related mortality.
Conclusions and relevance
PNR is an accessible and cost-effective hematological biomarker that outperforms NLR and echocardiographic findings in cats with early subclinical HCM. Alterations in NLR and PNR in cats with HCM and CATE suggest a potential role of systemic inflammation in feline HCM.
Keywords
Introduction
Hypertrophic cardiomyopathy (HCM) is the most common feline cardiomyopathy, affecting up to 15% of cats in the general population.1 –4 HCM is defined as a diffuse or regional increase in left ventricular (LV) wall thickness with a non-dilated LV chamber. 2 Over time, cats with HCM may develop life-threatening sequelae, such as sudden death, congestive heart failure (CHF) and cardiogenic arterial thromboembolism (CATE). 2 Of the complications, CATE, which occurs because of embolism of an intracardiac thrombus, remains a major contributor to morbidity and mortality in cats, with a devastating survival rate of under 40% of all treated cats. 5
There remains limited information on hematological markers associated with prognosis in cats with HCM and CATE. Current methods of identifying risk factors of cardiac-related complications rely largely on echocardiographic findings, such as presence of spontaneous echocardiographic contrast (SEC), left atrial (LA) enlargement and LA dysfunction. This identification is complicated by the fact that many cats at risk of CATE are apparently healthy and do not have clinical signs or auscultatory abnormalities; hence, they are unlikely to be selected for echocardiographic screening. 6 The neutrophil:lymphocyte ratio (NLR) is a readily available and cost-effective marker of systemic inflammation that has shown promise as a prognostic marker in a range of conditions in human medicine, including sepsis, neoplasia, cardiac disease and infections.7 –12 An elevated NLR reflects physiological stress and a proinflammatory response, as increased inflammatory cytokines and cortisol promote lymphocyte apoptosis and redistribution from lymphoid tissues, while simultaneously stimulating neutrophil production and release from the bone marrow. 13 Previous research by Fries et al 14 demonstrated that an NLR below 5 in cats with advanced HCM was independently associated with cardiac-related mortality. Despite these findings, the prognostic value of NLR in cats with early-stage HCM and CATE remains unclear and warrants further investigation.
The platelet:neutrophil ratio (PNR), distinct from the platelet:lymphocyte ratio, is an emerging marker of thrombotic risk that reflects the interactions between platelets and neutrophils, which are central to the complex relationship between inflammation and thrombosis. A decreased PNR may indicate formation of platelet-neutrophil aggregates or increased platelet consumption due to systemic inflammation. 15 In human medicine, the clinical utility of PNR appears to vary by disease context. For instance, one study reported a direct association between a lowered PNR and poorer outcomes in patients with acute ischemic stroke, while another study investigating epithelial ovarian cancer found that elevated PNRs correlated with a lower survival rate in patients.16,17 Despite its prognostic and diagnostic potential, PNRs have received limited attention in veterinary medicine. Given that veterinarians have limited reliable tools to identify HCM-affected cats at risk of CATE and other complications associated with HCM, further investigations of accessible and cost-effective biomarkers such as NLR and PNR may enhance early risk stratification and potentially improve clinical outcomes.
Our primary objective was to assess and compare several hematological parameters, including the NLR and PNR among clinically healthy cats, HCM-affected cats and those presenting with CATE. Our secondary objective was to compare the NLR and PNR between clinical and subclinical HCM-affected cats. Finally, we investigated the prognostic significance of NLR and PNR by examining their association with cardiac-related mortality in HCM-affected cats. We hypothesized that cats with HCM and CATE would have an increased NLR and decreased PNR. These changes in NLR and PNR at the time of HCM or CATE diagnosis would be significantly associated with cardiac-related death.
Materials and methods
Animals
The study was approved by the Institutional Animal Care and Use Committee at the University of California, Davis (protocols 21037).
All cats underwent physical examination and echocardiogram, and whole blood was obtained for biochemistry and complete blood count (CBC). As part of a large-scale study evaluating hematological changes in cats with or without HCM, client-, staff- or student-owned cats were prospectively enrolled for HCM screening between October 2018 and August 2020. Cats were considered healthy if echocardiograms, physical examination, biochemical profiles and CBCs were within normal limits at the time of blood collection. Cats with clinical HCM and CATE were enrolled at the William R Pritchard Veterinary Medical Teaching Hospital at the University of California, Davis. Diagnosis of CHF included consistent physical examination and imaging findings. Consistent imaging findings included echocardiographic evidence of LA or biatrial enlargement, as well as radiographic evidence of pleural effusion and pulmonary edema and/or point-of-care ultrasound findings consistent with severe LA enlargement and pulmonary edema, pleural effusion or pericardial effusion. CATE was diagnosed according to published criteria. 18 Briefly, cats must have had at least four of the following physical examination findings: (1) sudden onset of vocalization; (2) paralysis or paresis of one or more limbs; (3) absent motor function with absent skin sensation of one or more limbs; (4) absent femoral and/or dorsal pedal pulses; (5) pale or cyanotic food pads/nailbeds of one or more limbs; (6) firmness of the cranial tibial or gastrocnemius muscles; and (7) low rectal temperature (<37.6°C or <99.7°F). Medical treatments of cats in the HCM and CATE groups were left to the discretion of the primary clinicians. Additional diagnostic tests, including total thyroxine (T4) (VC2; Zoetis), and blood pressure measurement using Doppler sphygmomanometry were performed in cats in the HCM and CATE groups according to the clinicians’ discretion. All cats with CATE had full echocardiographic examinations. Exclusion criteria included the following: (1) previous treatment with an antiplatelet drug and/or anticoagulant; (2) uncooperative behaviors during echocardiogram and blood collection; (3) hyperthyroidism (total T4 ≥4.8 µg/dl); and (4) systemic hypertension (systolic blood pressure ≥180 mmHg). Cats with systemic diseases requiring therapies were also excluded. Based on echocardiographic findings and clinical signs, cats were grouped into healthy, HCM and CATE groups.
Blood collection and complete blood count
All blood samples were collected from the medial saphenous vein or jugular vein using a 21 or 23 G butterfly needle within 2–3 h of the echocardiogram being performed. Blood was drawn from the jugular vein or directly aspirated from an indwelling intravenous catheter in cats with CATE. After transferral to 3.2% sodium citrate tubes, CBC was analyzed using an automated hematology analyzer (HM5; Zoetis). The platelet histograms generated by the analyzer were inspected for clumps, and all platelet counts were confirmed by manual blood smear evaluation by the corresponding author. In blood samples with multiple areas of clumps on manual blood smears with consistent histograms, a CBC and smear analysis were repeated after blood was rewarmed at 37°C for 15 mins and mixed thoroughly. Blood samples were recollected if the aforementioned method failed to resolve platelet clumping with persistent low platelet concentration (<100 ×109/l). Platelet counts were corrected by a factor of 1.1 to account for the dilutional effect of sodium citrate. 19 In addition, platelet counts were considered an underestimate if histograms and manual blood smears confirmed the presence of platelet clumping.
Echocardiogram
Transthoracic echocardiographic evaluations were performed by board-certified veterinary cardiologists or trainees under direct supervision of board-certified veterinary cardiologists, as previously described. 18 Diagnosis of HCM was based on identification of regional or global LV wall thickness ≥ 6 mm using two-dimensional or M-mode echocardiography in the absence of systemic hypertension and hyperthyroidism. The inclusion of moderator band insertion sites was avoided during measurement of LV wall thickness. All echocardiographic assessments were performed as described. 20 Echocardiographic measurements were evaluated using commercially available software (Syngo Dynamics Workspace 10.0.01; Siemens Medical Solutions). 21 The presence or absence of any observed intracardiac thrombi along with their location was recorded. SEC, defined as a dynamic and organized swirling pattern visualized within any of the cardiac chambers, at standard gain setting for the remainder of the examination was recorded as present or absent.
Outcome analysis
To evaluate the prognostic significance of NLR and PNR, outcome data were collected after thorough review of the electronic medical system (William R Pritchard Veterinary Medical Teaching Hospital, University of California, Davis). If medical records were insufficient or if outcomes could not be determined by the database, owners were contacted by telephone and/or email. Their responses were collected and recorded using a standardized online questionnaire (see File 1 in the supplementary material). Information regarding mortality, including date, location and cause of death, was collected. The causes of death were further categorized as cardiac-related or non-cardiac-related. Cardiac-related mortality, based on modified published criteria, was defined as naturally occurring deaths or euthanasia due to decompensation of existing HCM, such as worsening of CHF, acute onset of CATE or sudden death. 22 Decompensated CHF was defined by the presence of pleural effusion or pulmonary edema that became increasingly refractory to heart failure medications. Owners’ perceived progression of CHF was characterized as acute onset of respiratory distress, such as tachypnea and open-mouth breathing. Cardiac-related death secondary to CATE was defined as death or euthanasia after newly diagnosed or recurrence of CATE, as stated above. Sudden death was considered cardiac-related if the patient was found unconscious without any apparent cause and if the owners perceived the patient to be apparently well in the preceding 24 h. Cats in the CATE group were not included in the initial outcome analysis if they were euthanized within 6 h of diagnosis and hospitalization. Survival time was calculated based on the days between the date of initial CBC and echocardiogram and the date of outcome data collection. For the purposes of this study, only HCM-affected cats with previously confirmed CHF were considered in stage C, as cats with CATE were analyzed as a separate group.
Statistical analysis
Normality of continuous data was tested using visual inspection of histograms and the Shapiro–Wilk test, which determined all data to be non-parametric. Data were, therefore, presented as median and interquartile range (IQR). Comparison of continuous data between the three groups was analyzed using Kruskal–Wallis testing with post-hoc analysis being Dunn’s multiple comparison testing. Continuous data between two groups was compared using the Mann–Whitney U-test. P <0.05 was considered statistically significant. Categorical data were analyzed using Fisher’s exact test. Survival data were graphically assessed using Kaplan–Meier survival curves and compared using a two-sided log-rank test. Specifically, cutoffs of NLR and PNR were generated based on quartiles (25th, 25th–75th and 75th percentiles rounded to the nearest whole number) in the HCM group. All the aforementioned statistical analyses were performed using Prism v.10 (Graphpad Software). Survival analysis was conducted using the Cox proportional hazards model to first examine the associations between NLR, PNR and known risk factors, such as left atrium to aortic root (LA:Ao) ratio, left auricular flow velocity, stage of cardiac disease, presence of SEC and the time to cardiac-related death using commercially available software (SPSS Statistics version 29.0.1.0; IBM). Due to the non-linear relationships between NLR and PNR and probability of survival, a semiparametric approach was taken, with the assumption that NLR above 4 and PNR below 40 and the log hazard rate have a linear relationship. Data of the Cox proportional hazards model were presented as hazard ratios (HRs) with 95% confidence intervals (CIs).
Results
Study population characteristics
Paired CBC and echocardiograms from 110 cats evaluated between 2018 and 2020 met the inclusion criteria (Figure 1). Of the 110 cats, 42 were healthy, 57 were diagnosed with HCM and 11 were diagnosed with CATE. The signalment of all the cats is summarized in Table 1. Of the 57 cats in the HCM group, 37 (65%) were in stage B1, nine (15.8%) were in stage B2 and 11 (19.3%) were in stage C at the time of CBC and echocardiogram. Of the nine cats in stage B2, one (11.1%) had evidence of SEC and one had SEC and an intracardiac thrombus in the left auricle. Five cats in stage C had SEC; of them, two were found to have an LA thrombus. None of the cats were in stage D. Since 28 cats were lost to follow-up, outcome data were available for further analysis in 82 cats (Figure 1). Eight cats (72.7%) in the CATE group were excluded from the outcome analysis because of euthanasia within 48 h of initial echocardiogram and CBC. The echocardiographic findings are summarized in Table 2.

Flow chart illustrating the number of cats included for analysis of neutrophil:lymphocyte and platelet:neutrophil ratios and the number of cats available for outcome analysis. CATE = cardiogenic arterial thromboembolism; CBC = complete blood count; HCM = hypertrophic cardiomyopathy
Baseline demographic parameters and complete blood count

Data are median (interquartile range)
Difference between groups (P <0.05)
Healthy vs CATE (P <0.05)
HCM vs CATE (P <0.05)
Healthy vs HCM (P <0.05)
CATE = cardiogenic arterial thromboembolism; FI = female intact; FS = female spayed; HCM = hypertrophic cardiomyopathy; MC = male castrated; MI = male intact; RI = reference interval; U = unsexed
Comparison of selected echocardiographic data

Data are median (interquartile range)
P <0.0001 between groups
2D = two-dimensional; CATE = cardiogenic arterial thromboembolism; HCM = hypertrophic cardiomyopathy; IVSd = interventricular septum at end diastole; LA:Ao = left atrial:aortic root ratio; LV = left ventricular; LVPWd = left ventricular posterior wall thickness at end diastole; N/A = not applicable; SEC = spontaneous echocardiographic contrast
Hematology findings
Pertinent hematological findings are presented in Table 1. Overall, platelet count was significantly different between the three groups (P = 0.03) (Table 1). Peripheral blood smear evaluation showed that platelet clumping was present in all samples and, therefore, platelet count was considered an underestimation in all cats. Cats with HCM had a significantly higher platelet count compared with healthy cats (P = 0.02). No significant difference in platelet count was found between HCM-affected cats with or without CATE (P = 0.05). The neutrophil count was significantly different between the three groups (P <0.0001). Cats in the CATE group had the highest neutrophil count compared with the HCM (P = 0.02) and healthy groups (P <0.0001), while no significant difference was found when comparing between healthy and HCM-affected cats (P = 0.05).
NLR was significantly different between the three groups (P <0.0001) (Figure 2a). As expected, cats with CATE had the highest NLR compared with cats in the HCM (5.8 [IQR 3.1–12.7] vs 3.3 [IQR 1.8–5.1]; P = 0.03) and healthy groups (2.1 [IQR 1.1–3.1]; P <0.0001). Cats with HCM also had a higher NLR compared with healthy cats (P = 0.04).

Scatter plots comparing neutrophil:lymphocyte ratio (NLR) and platelet:neutrophil ratio (PNR) in healthy cats without hypertrophic cardiomyopathy (HCM), cats with HCM and cats with cardiogenic arterial thromboembolism (CATE). NLR and PNR were calculated at the time of HCM and CATE diagnoses. (a) NLRs in cats with HCM or those with CATE were significantly higher compared with healthy cats. Cats with CATE also had higher NLRs compared with cats with HCM. (b) PNR was not significantly different between cats with or without HCM; however, cats with CATE had a significantly lower PNR. Bar represents the median and error bars represent the interquartile range. **P <0.005
PNR was different between the three groups (P = 0.0027). Although no significant difference between healthy and HCM-affected cats was noted (41.4 [IQR 26.0–54.3] vs 43.0 [IQR 18.7–66.5]; P >0.9), PNR was significantly lower in cats with CATE (16.6 [IQR 13.2–27.9]) when compared with healthy cats (P = 0.002) and cats with HCM (P = 0.006) (Figure 2b).
Subgroup analysis
As a result of the limited number of cats diagnosed with stage B2 HCM, cats with stages B1 and B2 HCM were analyzed as a single subclinical group and compared with those with clinical signs (CHF and CATE combined). When comparing NLR between HCM-affected cats, we found that those with clinical disease (CHF and/or CATE) had a significantly higher NLR compared with subclinically affected cats (5.8 [IQR 2.7–8.8] vs 3.4 [IQR 1.8–5.5]; P = 0.02). Among stage C cats, NLR did not differ between those with SEC or intracardiac thrombosis and those with CHF alone (3.9 [IQR 2.0–8.4] vs 5.8 [IQR 3.1–12.7]; P = 0.4).
Similarly, subclinical HCM-affected cats in stage B1 and B2 had a higher PNR (41.9 [IQR 20.0–69.0]) compared with cats with clinical disease (20.5 [IQR 14.7–43.7]; P = 0.02). No statistical difference in PNR was found between stage C cats with CHF and those CHF-affected cats with SEC or intracardiac thrombosis (37.1 [IQR 16.4–48.5] vs 16.6 [IQR 13.2–37.8]; P = 0.2).
Clinical outcome
Of the 26 healthy cats, 23 (88%) were alive at the time of data collection; none of the cats had any reported cardiac disease. One cat (4%) was euthanized because of severe hyperthyroidism with possible carcinoma, one cat (4%) was euthanized owing to large cell lymphoma and one cat (4%) was euthanized because of being found comatose from presumed diabetic crisis. Of the 45 cats in the HCM group, 18 (40%) were dead at the time of data collection. Of these 18 cats, 15 (83.3%) died of cardiac-related causes and three (16.7%) died from non-cardiac related causes, which included declining quality of life (n = 1), difficulty ambulating and progressive neurological decline (n = 1), progressive urinary tract infection and renal disease (n = 1), nasopharyngeal obstruction (n = 1), neoplasia (n = 1) and severe hyperthyroidism (n = 1). Of the 15 cats that died of cardiac-related causes, six (40%) died or were euthanized because of progressive CHF, four (26.7%) died of sudden death and five (33.3%) were euthanized as a result of acute onset of CATE. Of the 11 cats in the CATE group, eight were euthanized within 6 h of hospitalization; therefore, they were not included in the outcome analysis. The median survival time (MST) of the three remaining cats in the CATE group was 219 days; none of the cats were alive at the time of writing. Of these three cats, one (33%) lived for 682 days and was euthanized owing to recurrence of CATE, one (33%) lived for 219 days and was euthanized because of anorexia and worsening lethargy, and one (33%) lived an additional 38 days and died of sudden death. The last cat was hospitalized twice for two separate CATE events within 6 months before progression to CHF.
When comparing the NLR between survivors and non-survivors due to cardiac-related deaths, NLR in non-survivors was significantly higher than in survivors (6.0 [IQR 3.0–9.2 vs 3.1 [IQR 1.1–4.2]; P = 0.003) (Figure 3a). The PNR in survivors (48.4 [IQR 22–82.8]) was also significantly higher compared with non-survivors that died of cardiac-related deaths (17.8 [IQR 13.9–36.3; P = 0.003) (Figure 3b). To determine the prognostic significance of NLR, the distribution and the probability of survival among HCM cats were categorized based on NLR cutoffs in the HCM group (<2, 2–4, >4) (Figure 4). 14 Kaplan–Meier analysis showed a difference in survival between the three NLR cutoffs (P <0.003). Cats with an NLR above 4 had an MST of 959 days, while more than 50% of cats with an NLR of 4 or below were alive at the time of data analysis. Survival in cats with an NLR above 4 and those with an NLR in the range of 2–4 was also significantly different (P = 0.04).
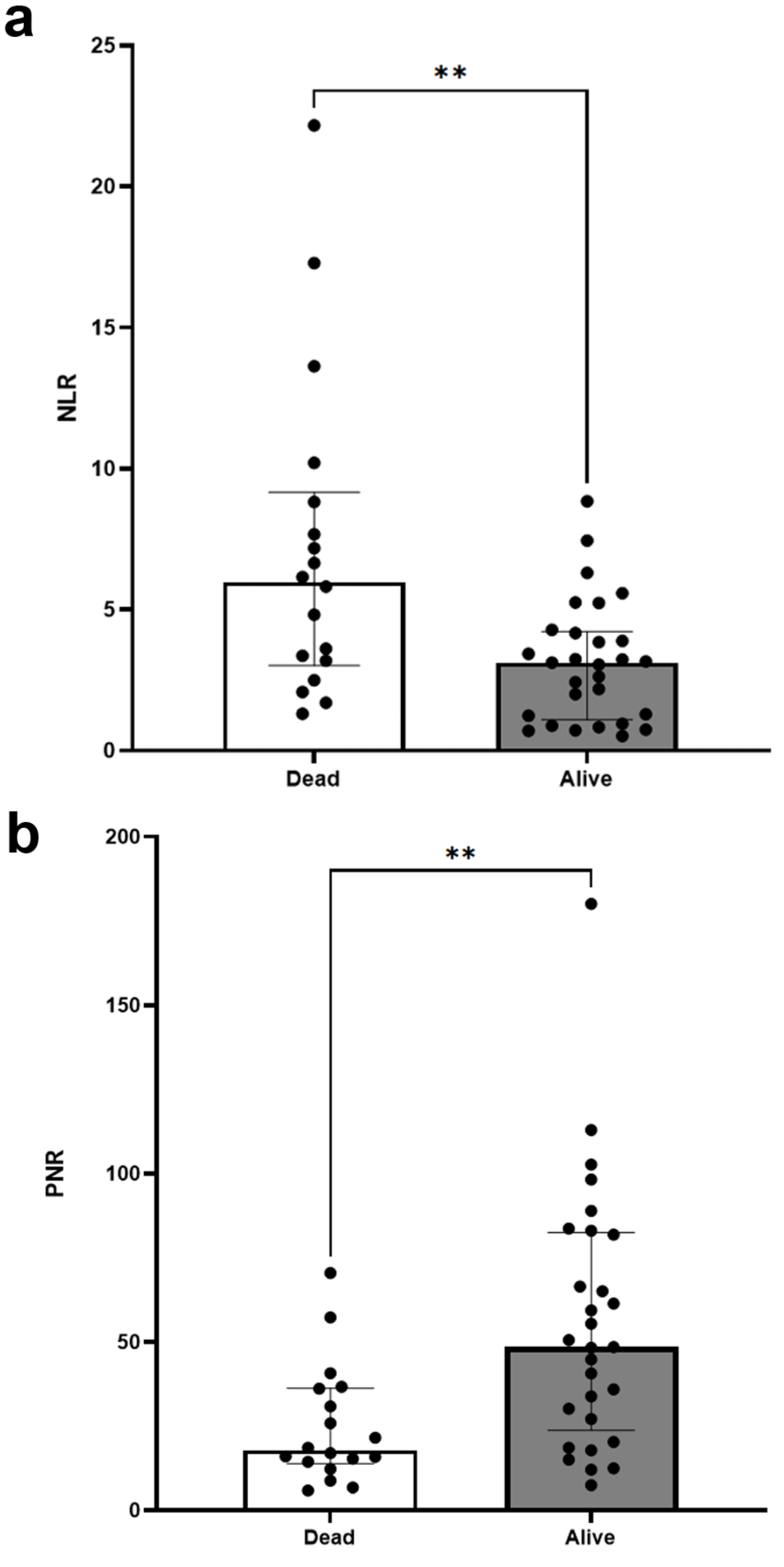
Scatter plots comparing neutrophil:lymphocyte ratio (NLR) and platelet:neutrophil ratio (PNR) between hypertrophic cardiomyopathy (HCM)-affected cats that succumbed to cardiac-related death and HCM-affected cats that were alive by the time of data collection. (a) Dead cats had a significantly higher NLR compared with alive cats. (b) PNR was significantly lower in dead cats compared with alive cats. Bar represents the median and error bars represent the interquartile range. **P <0.005

(a) Distribution of survivors and non-survivors grouped according to neutrophil:lymphocyte ratio (NLR) quartiles (<2, 2–4, >4). The Kaplan–Meier curve demonstrates a significant difference (P <0.003) in survival probability from cardiac-related mortality in cats with hypertrophic cardiomyopathy according to NLRs. Cats with NLR >4 had the shortest median survival time (MST) of 959 days. (b) Distribution of survivors and non-survivors grouped accorded to platelet:neutrophil ratio (PNR) quartiles (<20, 20–40, >40). Kaplan–Meier statistics showed a significant difference in overall survival (P = 0.003). The number of non-survivors when PNR was <40 was significantly higher with an MST of 1092.5 days (PNR 20–40) and 1184.5 (PNR <20). Nearly all HCM cats with PNR ≥ 42 survived, while cats with PNR < 42 had an MST of 1184.5 days
The distribution of PNR among survivors and non-survivors is shown in Figure 4b. PNR cutoffs of below 20, 20–40 and above 40 were established based on the data distribution among cats in the HCM group. The number of non-survivors with a PNR above 40 at the time of HCM diagnosis was significantly higher compared with those with a PNR below 40 (P = 0.0007). In addition, Kaplan–Meier statistics showed a significant difference in overall survival between the three groups (P = 0.003). The MST of cats with a PNR below 20 was 1184.5 days, and those with a PNR in the range of 20–40 had an MST of 1092.5 days. Survival between PNR above 40 and PNR of 20–40 was not significantly different (P = 0.8). Only 1/18 (5.6%) cats with a PNR above 40 died of cardiac-related causes at the time of data collection (Figure 4b).
Cox proportional hazards models were utilized to evaluate the relationship between survival time in the cats in the HCM group and known risk factors on echocardiogram and CBC (Table 3). We found that of all the echocardiographic variables in the model, only the presence of LA enlargement (LA:Ao >1.6) was significantly associated with a 10-fold increased risk of cardiac-related mortality. When the model was created based on hematological variables only, only a PNR below 40 at the time of HCM diagnosis was significantly associated with mortality. When combining both echocardiographic and hematological variables, LA enlargement (LA:Ao >1.6) was associated with a three-fold increase in cardiac-related mortality, while cats with a PNR below 40 at the time of HCM or CATE diagnosis were approximately 10 times more likely to experience cardiac-related mortality (HR = 9.8, 95% CI 1.2–79.9; P = 0.03). Table 3 summarizes the findings of the Cox proportional hazard models. Details of the Cox proportional hazards regression model are in File 2 of the supplementary material.
Summary of Cox proportional hazards model for cardiac-related mortality in hypertrophic cardiomyopathy-affected cats
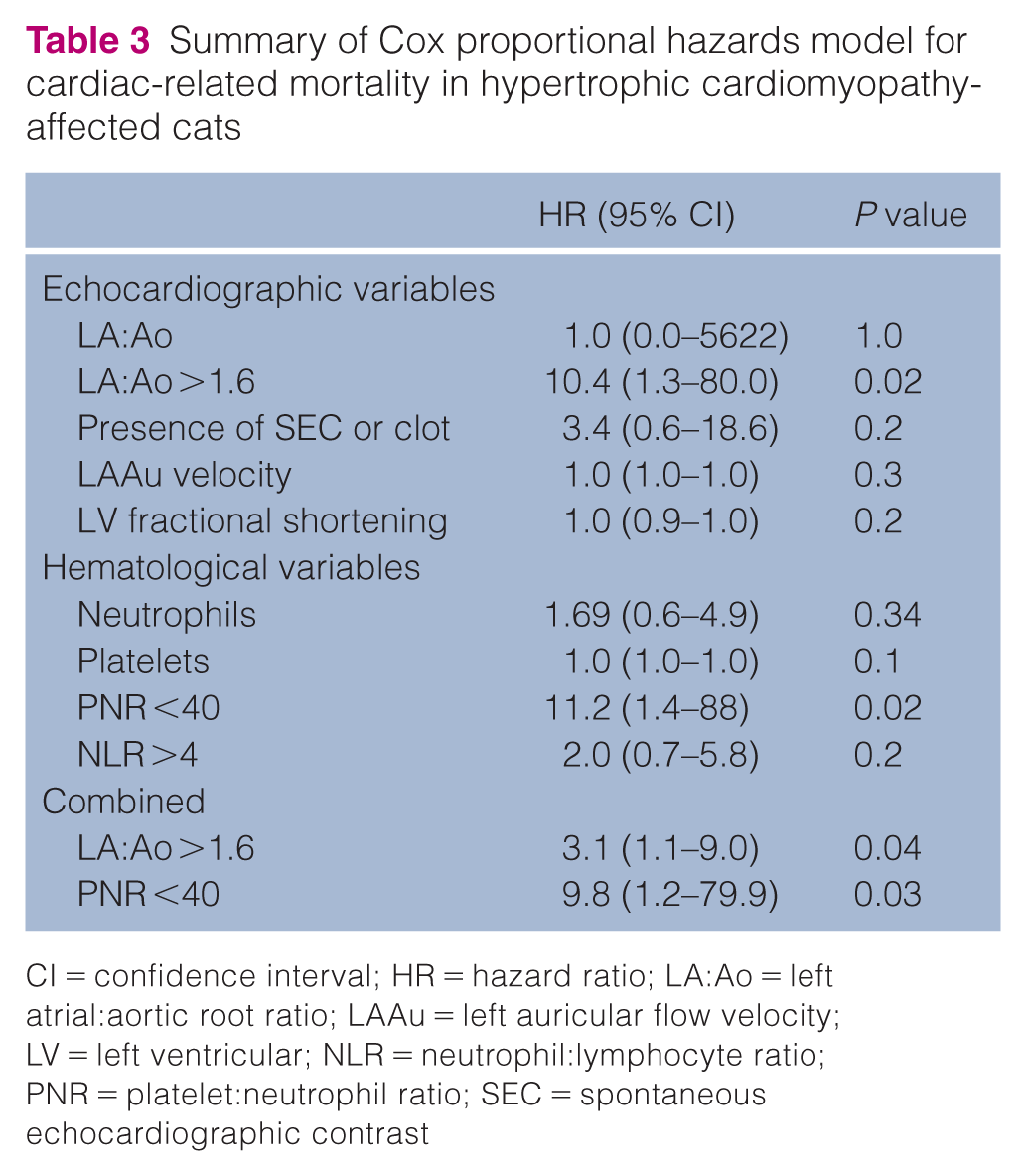
CI = confidence interval; HR = hazard ratio; LA:Ao = left atrial:aortic root ratio; LAAu = left auricular flow velocity; LV = left ventricular; NLR = neutrophil:lymphocyte ratio; PNR = platelet:neutrophil ratio; SEC = spontaneous echocardiographic contrast
Discussion
In this study, we found that cats with HCM had an increased NLR compared with healthy cats. The PNR was significantly lower in cats with CATE. After excluding non-survivors within the first 6 h of CATE diagnosis, a decreased PNR of below 40 at the time of HCM diagnosis was significantly associated with cardiac-related mortality.
The NLR was previously evaluated in cats with HCM. Similar to our findings, a retrospective study by Fries et al 14 found an increased NLR in cats with HCM compared with healthy controls. However, a notable difference is that the NLR failed to have any prognostic significance in our cohort. This discrepancy may be secondary to the marked difference in survival times, which is likely attributed to the early diagnosis of HCM, since most of our HCM-affected cats were diagnosed with stage B1 HCM. For instance, HCM cats with NLRs above 5 had an MST of 275 days in the previous study, while our cohort with NLRs above 4 had an MST of 959 days. This is further supported by the absence of significant LA enlargement and dysfunction in the HCM group relative to healthy controls. These findings also indicate that LA function and size may be less useful at predicting outcomes in cats with subclinical HCM until their disease advances to stages B2 and beyond. Another explanation is that the prospective nature of our study eliminated the selection bias that could otherwise occur in a retrospective cohort study since CBCs are only usually performed if clinically indicated. In the authors’ experience, a CBC is not part of a routine work-up for cats with HCM unless the patient is critically ill or exhibiting atypical clinical signs.
Despite this, an overall increase in NLR among cats with HCM was found when compared with healthy controls. Elevation in NLR indicates the presence of systemic inflammation, which could be caused by an imbalance of innate and adaptive immunity, previously noted in human HCM patients. 23 However, the link between inflammation and myocardial hypertrophy is not well understood. An in vitro study in cardiomyocytes extracted from humans with end-stage HCM showed profound levels of oxidative stress markers and proinflammatory cytokines such as tissue necrosis factor-alpha and interleukin 6, which were associated with diastolic dysfunction and cardiac fibrosis. 24 Reactive oxygen species have also been demonstrated to have detrimental effects on excitation–contraction coupling and diastolic function. 24 A multiomics study in cats with HCM also supported the notion that cats with HCM may be in a chronic inflammatory state secondary to upregulation of proteins associated with complement activation and inflammatory responses. 25 It is worth noting that leukocyte counts, including neutrophils, lymphocytes and monocytes, were not significantly different between healthy cats and cats with HCM. However, increased NLR despite leukocyte counts within the reference interval is associated with myocardial damage, ventricular tachycardia and syncope in humans with coronary syndrome and HCM. 26 Elevation in NLR should prompt clinicians to monitor their feline patients more closely for disease progression and occurrence of cardiovascular-related complications.
The PNR reflects the balance between platelets and neutrophils and the intricate connection between thrombosis and inflammation. We found that cats with CATE had an overall decreased PNR, which is likely caused by decreased circulating platelets due to consumption as a result of thrombosis, and rising neutrophils caused by systemic inflammation and thromboinflammation. 27 To demonstrate the prognostic significance of PNR, we excluded cats with CATE that were euthanized within 48 h of admission. Not only did we find a difference in PNR between survivors and non-survivors, we also found that PNR was independently associated with a near 10-fold increased risk of cardiac-related mortality. This observation is in agreement with a previous study in humans with ischemic stroke, which showed that a decreased PNR after thrombolysis therapy was associated with poor treatment outcomes such as delayed cognitive recovery and stroke recurrence. 28 Increased platelet activation, which has previously been shown in multiple feline HCM studies, could play a key role in decreasing PNR in cats with HCM and CATE.29 –31 Platelet activation, which is crucial to the formation of platelet-neutrophil aggregates, not only causes platelet consumption but also activates neutrophils and systemic inflammation. 32 Increased neutrophil activation leads to the formation of neutrophil extracellular traps (NETs), which are cell-free DNA decorated with granular proteins and histones.32,33 NETs have also been demonstrated to stimulate thrombosis through multiple mechanisms.34 –36 In cats, circulating NETs in the form of cell-free DNA and citrullinated histones are increased proportionally to HCM severity and risk factors of CATE, such as LA:Ao and left auricular flow velocity, suggesting that decreased PNR may occur before the formation of active NETs. 18 Further studies are needed to investigate if early detection of decreased PNR could serve as a biomarker of platelet activation, NET formation and systemic hypercoagulability in cats with subclinical HCM.
Given the variability in CBC results arising from differences in analyzer types and anticoagulants used by practitioners, reporting rounded values of NLR and PNR cutoffs acknowledges this inherent imprecision and reflects clinically meaningful thresholds more realistically. Such rounding also improves clarity, facilitating the development of practical guidelines and enhancing ease of application for general practitioners. Although rounding may theoretically introduce a rounding error, our analyses and modeling indicate that this has no impact on Kaplan–Meier statistics or the Cox proportional hazards model.
This study has several limitations. First, a lack of standardization of HCM treatments, thromboprophylaxis and patient re-evaluations are potential confounding variables even though the study took place in a single tertiary institution. Second, as we relied on owners’ observations and review of the medical records for outcome assessments, determining the exact causes of death was limited without necropsies. Third, as all blood samples were taken only at the time of HCM diagnosis, this prevented us from evaluating the changes of NLR and PNR in association with the disease progression and its variable outcomes. Fourth, given that our sample size of CATE-affected cats was significantly reduced after excluding those euthanized within 48 h of diagnosis, it is possible that selection bias was introduced and cats that died because of the disease process were excluded. Lastly, the blood samples utilized in this study were anticoagulated in citrated tubes given that the feline subjects were also enrolled in a large-scale platelet function study. Citrate is a commonly used anticoagulant when studying platelet physiology because of its minimal effect on pH and calcium-chelating property preventing excessive platelet clumping that is commonly encountered when analyzing feline blood. 37 However, because EDTA is the most commonly utilized anticoagulant agent in the clinical setting, the presence of platelet clumping that occurs commonly in feline blood samples may limit the diagnostic utility of PNR in the clinical setting using traditional EDTA samples.
Despite these limitations, our findings suggest that abnormalities of NLR and PNR can be detected early in subclinically HCM-affected cats, and PNR may have better prognostic significance in predicting outcomes in cats with HCM. A future prospective observational cohort study involving serial CBC measurements would allow us to characterize hematological changes with disease status. As a result of the limited sample size, future research to support these findings to create more standardized management guidelines is needed.
Conclusions
In this prospective study, we found that NLR and PNR were significantly different between healthy controls, HCM-affected cats and cats with CATE. Changes in NLR and PNR are suggestive of long-term outcome. Cats with a PNR below 40 at the time of HCM diagnosis were approximately 10 times more likely to experience cardiac-related mortality compared with those with a PNR above 40, suggesting that increased platelet-neutrophil interactions and inflammation may lead to a shorter survival time. This hematological change was more predictive of outcome than LA enlargement. This is important to provide a more accurate prognosis for owners and optimize treatment and monitoring strategies. Future prospective studies to monitor serial hematological ratios with disease progression are needed to further validate these findings.
Supplemental Material
File 1
Hypertrophic cardiomyopathy retrospective study client feedback form.
Supplemental Material
File 2
Details of Cox proportional hazards models.
Footnotes
Acknowledgements
The authors would like to thank Nghi Nguyen, Avalene Tan, Yu Ueda, Cheyenne Chen, Joshua Stern and the University of California, Davis Veterinary Cardiology and Emergency Services for their assistance with patient screening and enrollment.
Author note
Data are available upon request.
Supplementary material
The following files are available as supplementary material:
File 1: Hypertrophic cardiomyopathy retrospective study client feedback form.
File 2: Details of Cox proportional hazards models.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the Morris Animal Foundation (D18FE-014, D20FE-601, D20FE-805) and the Center for Companion Animal Health (2019-30-F).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
