Abstract
Objectives
The aim of the present study was to evaluate the sedative and echocardiographic effects of dexmedetomidine (DEX) administered via intranasal (IN) and intramuscular (IM) routes in cats.
Methods
This randomised, blinded crossover study involved eight healthy adult cats. Cats were randomly allocated to receive DEX 10 μg/kg via either the IN or IM routes. Sedation, mechanical nociception and muscle relaxation were subjectively assessed and physiological variables recorded at baseline and at 5 min intervals for up to 40 mins after drug delivery. Echocardiography was performed 15 mins after delivery.
Results
In both treatment groups, sedation assessment scores significantly increased compared with baseline values (P <0.05). At 25–35 mins after delivery, only the IN group exhibited a significant decrease in mechanical nociception scores compared with baseline (P = 0.041, P = 0.042, P = 0.026). DEX delivery via both routes resulted in significant reductions in pulse rate (P <0.05). In the IM group, mean arterial blood pressure measurements 35–40 mins after delivery were significantly lower than baseline (P = 0.012, P = 0.012). Fractional shortening significantly decreased in both the IN and IM groups compared with baseline (P = 0.016 and P = 0.049, respectively). Both routes caused reductions in cardiac systolic function, with no significant difference between the two routes. Vomiting occurred in half of the IN group (4/8) and in all cats of the IM group (8/8), with a significantly lower incidence in the IN group (P = 0.046).
Conclusions and relevance
IN delivery of-DEX provided comparable sedation, increased tolerance to mechanical nociception and muscle relaxation effects while causing fewer adverse effects than IM-DEX. Both routes similarly reduced cardiac contractile function. Thus, IN-DEX at a dose of 10 μg/kg is a viable alternative to IM-DEX for sedation in healthy cats.
Introduction
Dexmedetomidine (DEX) is widely used in human and veterinary medicine because of its sedative, analgesic and anxiolytic properties. 1 As a highly selective and potent alpha-2 adrenoceptor agonist, DEX induces sleep-like sedation by suppressing the noradrenergic neuronal firing of the locus ceruleus in the brainstem 2 and activating the endogenous sleep-promoting pathway. 3 Several studies have shown that intramuscular (IM) administration of DEX (IM-DEX) and intravenous (IV) administration of DEX can produce suitable levels of sedation and analgesia but cause cardiovascular depression in dogs and cats.4 –7 As such, careful consideration of these potential cardiovascular effects is warranted when employing DEX for sedation.
The intranasal (IN) administration route has emerged as a viable alternative for delivering some injectable drugs. 8 In addition to rapid absorption into the bloodstream via nasal cavity capillaries, this delivery method facilitates the bypassing of the blood–brain barrier through the trigeminal and olfactory pathways, enabling direct drug access to the brain in humans. 9
IN delivery of DEX (IN-DEX) has been used in paediatric patients for decades, providing reliable sedation in a simple and well-tolerated manner. 10 Sedation is crucial in veterinary medicine, particularly for managing cats exhibiting fear or agitation. 11 Previous studies12,13 have investigated the sedative, cardiovascular and pharmacokinetic effects of IN-DEX in dogs, indicating that IN administration could serve as a feasible alternative to IM administration of DEX, with effective sedation and less bradycardia in healthy dogs. A recent study has suggested the sedation scores were comparable when using IN-DEX and IM-DEX at a dose of 5 μg/kg in dogs. 14
From a search of the literature, it was discovered that there are no published studies evaluating the sedative and echocardiographic effects of IN-DEX in cats. The primary objective of our study was to objectively compare changes in physiological and echocardiographic parameters, as well as the incidence of adverse effects between IN-DEX and IM-DEX at 10 μg/kg. Secondary objectives included a subjective evaluation of sedative effects, tolerance to mechanical nociception and muscle relaxation.
Materials and methods
Animals
Eight purpose-bred cats, comprising three males and five females (one castrated male, two sexually intact males and five sexually intact females), with a mean (± SD) weight of 3.30 ± 0.46 kg and age range of 1–6 years, were included in our study. Cats were determined to be healthy (American Society of Anesthesiology physical status classification score I/V) based on physical examination, complete blood count, serum biochemistry analysis and echocardiographic assessment. The study was approved by the Laboratory Animal Welfare and Ethics Committee of Nanjing Agricultural University (reference number 20210702102). Cats were fasted for 12 h before the experiment, with water available, and were brought to a quiet room at least 15 mins before the experiment.
Study design
A prospective, randomised, blinded crossover study design with a 2-week washout period was used. Cats were randomly assigned to receive DEX (Dexdomitor; Zoetis) at 10 μg/kg via either the IN or IM routes. Randomisation was achieved using the web-based tool ‘research randomizer’ (https://www.randomizer.org/). The treatment drug was diluted with saline to a final volume of 0.4 ml by an investigator uninvolved in any aspect of data collection or drug administration, knowing only the drug’s concentration and treatment allocation. The investigator who diluted the drug differed from the investigators administering the drug. For IN administration, a nasal atomisation device (Wuxi NEST Biotech) was used to deliver 0.2 ml of the prepared drug solution into each nostril by an investigator who was blinded to treatment allocation. After drug delivery, each cat’s head was tilted backwards 45° for 1 min. For IM injection, the treatment drug was injected into the semimembranosus muscle using a 25 G (25.4 mm) needle by the same investigator as the IN group, who was blinded to treatment allocation.
The variables in this paragraph were recorded by three investigators blinded to treatment. In our study, a previous published feline multiparametric scale was employed to evaluate sedation, mechanical nociception and muscle relaxation (Table 1). 4 The sedation score (SS), with a range of 0–13, was assessed subjectively, scoring the cat’s posture, gait stability, resistance to lateral recumbency, resistance to hindlimb extension (without pinching), resistance to stretching the stifle, and reaction to sound (hand clap) and tactile (air blown in the ear) stimuli. 4 A higher SS indicates deeper sedation. The mechanical nociceptive score (MNS), with a range of 0–6, was evaluated subjectively by observing the cat’s nociceptive response to digital pad clamp and tail clamp using haemostatic forceps (Shanghai Medical Equipment Co) applied to the first interlocking tooth. Reactions such as vocalisation, limb movement, tail twitching, muscle fasciculation or other escape behaviours indicated a response to nociceptive stimuli, with lower MNS values reflecting increased tolerance to the noxious stimulus. The muscle relaxation score (MRS), in the range of 0–4, was assessed based on hindlimb muscle tone and resistance to mouth opening, where a higher MRS indicated greater muscle relaxation.
Sedation, mechanical nociceptive and muscle relaxation assessment 4

Physiological variables
Physiological variables were recorded before (TB) and at various times (T) after drug delivery: at 5, 10, 15, 20, 25, 30, 35 and 40 mins. In addition, any observed adverse effects during sedation were documented. The following physiological variables were recorded: pulse rate (PR), respiratory rate (RR), mean arterial blood pressure (BP) and rectal temperature (RT). PR and BP measurements were obtained using a Doppler ultrasonic device (Model 811B; Parks Medical Electronics). The cuff was positioned at the base of the tail, with the cuff diameter chosen to be approximately 40% of the tail’s circumference. RT was measured using a certified digital thermometer (Model MT1961; Hualue Electronics Co). RR was determined by observing thoracic excursions over 1 min.
Echocardiography
A pilot study was performed to determine the timing for echocardiography (Echo). In this preliminary study, the same eight cats were divided equally into two groups – IN and IM treatments – following the same procedures as in the full study. Based on the onset of sedation observed in this pilot study, echocardiograms were scheduled to be performed before and at T15 after drug delivery. All Echo examinations were performed on the cats in lateral recumbency by an experienced echocardiographer, blinded to treatment, using an ultrasound machine (Mylab Sigma VET; Esaote) in the same room where DEX was administered and all variables were recorded.
Cats were gently restrained in right and then left lateral recumbency for the Echo examination. The Echo technique followed established methods,5,15 and all measurements were made by averaging three consecutive cardiac cycles. Specifically, the interventricular septal thickness, the left ventricular (LV) internal dimensions (LVIDs) and the LV posterior wall thickness were measured during both end systole and end diastole from the right parasternal long-axis views using M-mode. After obtaining the LVIDs, the LV factional shortening (FS) and the ejection fraction were calculated using the Teicholz method.
Two-dimensional echocardiography was used to measure the aortic diameter (Ao), left atrial diameter (LA) and LA:Ao ratio from the right parasternal short-axis views. Pulsed-wave Doppler measurements included peak pulmonic flow velocity (PV Vmax) and acceleration time across the pulmonic valve from the right parasternal short-axis pulmonary artery view. Left apical four- and five-chamber Doppler assessments included transmitral flow velocities (peak E, peak A), E:A ratio, lateral mitral annular velocities (Ea lat, Aa lat) and their respective ratios (E:Ea lat, Ea:Aa lat). Additional parameters measured were isovolumetric relaxation time and deceleration time of early diastolic transmitral flow. Valvular regurgitation was assessed using colour flow Doppler.
Statistical analysis
Data analyses and visualisation were performed using commercial software (SPSS version 26.0; IBM and GraphPad Prism version 9.2.0; GraphPad Software, respectively). Data normality was assessed using the Shapiro–Wilk test. Differences between the IN and IM routes of drug administration at specific time points were evaluated using the Wilcoxon signed-rank test for matched pairs. Repeated measure variables (SS, MNS, MRS, physiological and echocardiographic variables) were analysed for differences from baseline among different time points using the Wilcoxon signed-rank test with Bonferroni’s adjustment for multiple comparisons. The incidence of vomiting was assessed using Fisher’s exact test with Bonferroni’s adjustment. Values of P <0.05 were considered significant. Data are expressed as median (interquartile range).
Results
Sedation
SS increased significantly in the IN group from T15 to T40 when compared with baseline (P <0.05) (Figure 1a). The IM group showed a similar pattern with significant elevations in SS, compared with TB, from T5 to T40 (P <0.05). Between the IM and IN groups, no significant differences in SS were detected at any of the corresponding time points (P >0.05).

(a) The sedation score (higher values representing increased sedation), (b) the mechanical nociceptive score (lower values representing increased tolerance to the noxious stimulus) and (c) muscle relaxation score (higher values representing increased relaxation) in eight cats after intranasal (IN) or intramuscular (IM) administration of dexmedetomidine (DEX) at 10 μg/kg before and during the 40 mins after DEX administration. Time points are represented as TB (before drug delivery), T5, T10, T15, T20, T25, T30, T35 and T40 (5–40 mins after drug delivery). Data are expressed as box and whisker plots: box range is between the first and third quartiles of the data, whiskers range is between minimum and maximum values, and median is displayed as the horizontal line within the box. Exact P values are provided in the main text. There were no significant differences between groups for SS, MNS or MRS at any corresponding time point
The MNS decreased significantly in the IN group from T25 to T35, with P values of 0.041, 0.042 and 0.026, respectively (Figure 1b), but did not change in the IM group at any time points. There were no significant differences in MNS between the two groups at any of the corresponding time points (P >0.05).
The MRS in the IN group increased significantly at T30 and T35 (P = 0.041, P = 0.041) (Figure 1c). The IM group showed no significant changes in MRS at the examined time points. Between the IM and IN groups, no significant differences in MRS were detected at any time (P >0.05).
Physiological variables
PR in the IN group significantly decreased at T25, T35 and T40 compared with TB (P = 0.041, P = 0.035, P = 0.049) (Table 2). By contrast, PR in the IM group decreased significantly beginning at T5 and remaining significantly lower than TB throughout the assessment period (P <0.05). BP in the IN group did not show significant changes after drug delivery compared with TB, whereas the IM group decreased significantly in BP compared with TB for T35 to T40 (P = 0.012, P = 0.012). Notably, BP in the IN group at TB and T5 was significantly lower than in the IM group at the same time points (P = 0.036, P = 0.043, respectively) with no differences found between groups from T10 to T40 (P >0.05).
Pulse rate (PR), blood pressure (BP), respiratory rate (RR) and rectal temperature (RT) in cats after intranasal (IN) or intramuscular (IM) administration of dexmedetomidine (DEX) at 10 μg/kg before DEX administration (TB) and during the 40 mins after DEX administration

Values are median (interquartile range). The data highlighted in bold are statistically significant
Significantly different between groups (P <0.05)
Significantly different from TB (P <0.05)
RR in the IM and IN groups did not differ significantly from TB at any time (P >0.05) (Table 2). At T40, RR in the IN group was significantly higher than that in the IM group (P = 0.039). RT in the IN group did not show any significant differences between T5 and T40 when compared with TB (P >0.05), whereas RT at T20 in the IM group was significantly higher than TB (P = 0.021). In addition, at T20, T30, T35 and T40, RT in the IN group was significantly lower than that of the IM group (P = 0.027, P = 0.042, P = 0.044, P = 0.049).
Echocardiography
After drug delivery, a significant increase in the LVIDs (P = 0.025 for both groups) and a decrease in FS (P = 0.016, P = 0.049) were observed in both the IN and IM groups, compared with their baseline measurements (Table 3). A significant decline in peak A was shown in the IM group compared with baseline (P = 0.023).
Echocardiographic variables in eight cats at baseline (BL) and 15 mins after administration of dexmedetomidine (DEX) via intranasal (IN) or intramuscular (IM) routes
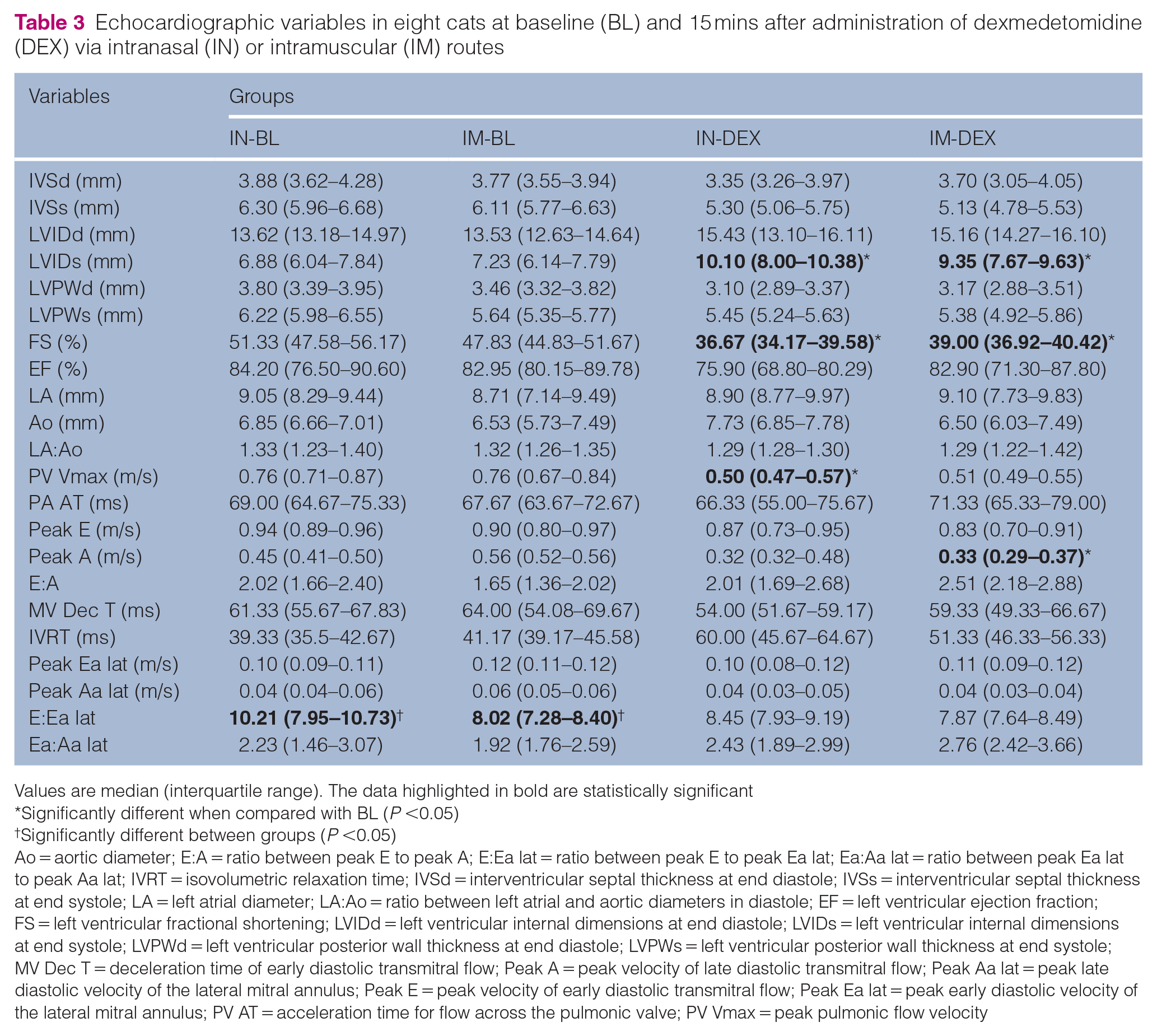
Values are median (interquartile range). The data highlighted in bold are statistically significant
Significantly different when compared with BL (P <0.05)
Significantly different between groups (P <0.05)
Ao = aortic diameter; E:A = ratio between peak E to peak A; E:Ea lat = ratio between peak E to peak Ea lat; Ea:Aa lat = ratio between peak Ea lat to peak Aa lat; IVRT = isovolumetric relaxation time; IVSd = interventricular septal thickness at end diastole; IVSs = interventricular septal thickness at end systole; LA = left atrial diameter; LA:Ao = ratio between left atrial and aortic diameters in diastole; EF = left ventricular ejection fraction; FS = left ventricular fractional shortening; LVIDd = left ventricular internal dimensions at end diastole; LVIDs = left ventricular internal dimensions at end systole; LVPWd = left ventricular posterior wall thickness at end diastole; LVPWs = left ventricular posterior wall thickness at end systole; MV Dec T = deceleration time of early diastolic transmitral flow; Peak A = peak velocity of late diastolic transmitral flow; Peak Aa lat = peak late diastolic velocity of the lateral mitral annulus; Peak E = peak velocity of early diastolic transmitral flow; Peak Ea lat = peak early diastolic velocity of the lateral mitral annulus; PV AT = acceleration time for flow across the pulmonic valve; PV Vmax = peak pulmonic flow velocity
In addition, PV Vmax was significantly reduced in the IN group compared with baseline (P = 0.016). No statistical differences were found between the IN and IM groups or between treatment groups and baselines for the remaining echocardiographic measurements (P >0.05). No valvular regurgitation (mitral, tricuspid, aortic, pulmonic) was detected via colour Doppler evaluations in either group.
Adverse effects
Vomiting occurred in half (4/8) of the cats in the IN group and all (8/8) of the cats in the IM group. The incidence of vomiting (IV) was significantly lower in the IN group, with a median of 0.5 (range 0–1), compared with the IM group, which had a median of 1 (range 1–1) (P = 0.046).
Discussion
The IM route is the only labelled route of administration for DEX in cats. However, alternative extralabelled extravascular routes, such as oral transmucosal, sublingual and IN administration, have gained popularity in recent years.8,16,17 The main benefits of IN administration include its non-invasiveness and ease of use. DEX is rapidly absorbed into the bloodstream through the nasal cavity capillaries, bypassing the blood–brain barrier via the trigeminal and olfactory pathways and allowing direct access to the brain in humans. 9 In addition, IN administration avoids hepatic first-pass metabolism, resulting in improved bioavailability compared with oral administration. 18
There are few studies assessing the sedative and echocardiographic effects of DEX administered via IN or IM routes in healthy cats. The results of the present study show that IN-DEX produces comparable sedation to IM-DEX, while providing measurable antinociceptive and muscle relaxation effects, along with a lower incidence of vomiting compared with IM-DEX. Both administration routes induced changes in physiological indices, but significant changes in RT and BP were observed only in the IM group. In addition, both IN-DEX and IM-DEX reduced the contractile function while having minimal impact on diastolic function in healthy cats.
There was no difference in the SS between IN and IM groups. The SS consistently remained significantly higher than TB starting from T15 in both groups, which aligns with findings from a previous pharmacokinetic study of IM-DEX in cats. 19 In a recent study, the sedation scores for cats in the IN groups were significantly lower than those observed in the IM group during the 20 mins after DEX administration at 20 μg/kg. 20 The variation in results from our study can be attributed to the use of higher doses of DEX and a shorter observation period. In a prior study in dogs, there were no differences in the onset of sedation between IN-DEX and IM-DEX at a dose of 5 μg/kg. 14 However, in our study, the IM-DEX group exhibited a significant sedative effect with an earlier onset at T5 compared with baseline, whereas sedation in the IN-DEX group was delayed, beginning at T15. Variations in the onset of sedation with IN-DEX across species may be attributed to anatomical and physiological differences in the vascularisation and innervation of the olfactory mucosa and bulbs, which could influence drug diffusion from the nasal cavity to the brain. 12
Using the same evaluation criteria for MNS and MRS, previous research in cats demonstrated that IM-DEX at 20 μg/kg significantly elevated tolerance to mechanical nociception and improved muscle relaxation compared with baseline. 4 Our study extends these observations by showing that IN administration of DEX at half the dose (10 μg/kg) significantly reduced responses to mechanical nociceptive stimuli and achieved comparable muscle relaxation relative to baseline. In contrast, IM administration at the same dose failed to produce significant effects, emphasising the comparable efficacy of the IN route at lower doses. However, it should be emphasised that these results are specific to mechanical nociceptive responses; a comprehensive assessment of analgesic efficacy would require evaluation across multiple nociceptive modalities.
DEX, an alpha-2 adrenergic receptor agonist, reduces heart rate via both central and peripheral mechanisms. Centrally, it inhibits norepinephrine release, lowering sympathetic tone and circulating catecholamine levels. Peripherally, initial vasoconstriction and the subsequent increase in BP may activate the baroreflex, enhancing vagal (parasympathetic) tone and further contributing to bradycardia.21,22 In our study, both the IN and IM groups showed significant declines in PR, with median reductions of 18.8% and 28.4%, respectively, from baseline to T40. A previous study in healthy dogs reported 67.2% median PR reduction after IN-DEX at 10 μg/kg from TB to T60. 5 In addition, both IN-DEX and IM-DEX initially increased, then decreased, BP relative to baseline, consistent with the known biphasic effect of alpha-2 agonists: initial vasoconstriction followed by vasodilation via post-synaptic alpha-2 receptor activation on vascular endothelial cells.22,23 However, a significant BP reduction was observed only in the IM group at T30, T35 and T40, whereas no significant changes in BP were observed in the IN group. This aligns with previous findings in dogs, where IN-DEX mitigated BP decreases under anaesthesia. 24 These findings highlight the importance of considering different administration routes and species-specific responses when evaluating the impact of DEX on BP. Regarding RT, a previous study in cats reported a significant decrease after IM-DEX at 5 μg/kg at T20 compared with baseline. 25 In contrast, our study found an initial significant increase in RT in the IM group, followed by a decline that remained within normal limits. IN administration had no significant effect on RT. These findings highlight the need for further investigation into how different routes and doses of DEX influence thermoregulation in cats.
DEX administration caused a significant decrease in FS and an increase in LVIDs compared with baseline in both groups. Decreases in peak A in both groups indicate that either administration route could diminish left atrial systolic function in healthy cats at 10 μg/kg, especially the IM injection route, where the changes were more pronounced. These align with previous reports which demonstrated that DEX administration causes an increase in the LVID and a decrease in systolic function in dogs and cats.5,13,26 –28
No significant differences were observed in diastolic parameters after administering 10 μg/kg of DEX, which aligns with previous studies5,13,26,27 showing that DEX, whether administered IM or IN, does not impair LV diastolic function. However, it is interesting that the E:Ea lat ratio was reduced with a slight reduction in E wave and LA:Ao ratio in both IN and IM groups. Although not clinically significant, these changes suggest that DEX administration might cause minor reductions in LV filling pressures and improve diastolic function in healthy cats. Further research is needed to assess DEX’s potential impacts on diastolic parameters when used in cats as sedatives at different doses or routes.
No significant changes in pulmonary, mitral or aortic regurgitation were detected with 10 μg/kg of DEX. This is in contrast with a recent study in cats, where higher doses (40 μg/kg IM) caused valvular regurgitation due to increased preload and end-diastolic volume. 27 It also differs from prior reports of regurgitation with IM-DEX and IV-DEX in dogs.5,26 As a result, we suspect that using DEX in dogs or high doses of DEX may be more likely to result in valvular regurgitation. However, further investigation is warranted to confirm if valvular regurgitation is associated with species or the dose of DEX.
When assessing adverse effects in cats, the incidence of vomiting of the IN group (50% of cats) was significantly lower than that in the IM group (100% of cats). A previous study reported that 7 μg/kg of IM-DEX induced vomiting in 58% of cats, 29 while in a retrospective study, vomiting occurred in 100% of cats receiving IM-DEX and 67% receiving IV-DEX, with a median dose of 7 μg/kg (range 0.96–10.0). 30 These findings indicate that the IN route may be a preferable option for reducing the risk of emesis associated with DEX administration in cats.
The present study has some limitations. The sample size was limited by the number of available experimental animals and may provide only a rough estimate of true differences, with considerable uncertainty. In cases where no significant differences were observed, a type II error (false negative) may have occurred, potentially overlooking important differences between treatments. In addition, an untreated control group was not included, as the clinical and pharmacological effects of DEX in cats have been well described in previous studies.4,27,31 The scoring systems for SS, MNS and MRS involved a degree of subjectivity. Echocardiography may influence sedation scores as a result of the stimulation and restraint required. Although no visible drug residue remained in the atomisation device, the use of an atomiser may still result in some medication loss. IN administration may also be limited by the risk of immediate expulsion via sneezing or swallowing, which could lead to first-pass metabolism. 13 Finally, no concurrent pharmacokinetic studies of IN-DEX in cats was conducted. Future pharmacokinetic studies are essential to better understand the relationship between the pharmacokinetic profile and clinical effects of IN-DEX in cats.
Conclusions
Administration of both IN-DEX and IM-DEX in healthy cats significantly reduced pulse rate and cardiac systolic function, with no difference between the two routes. IN-DEX produced comparable sedation with fewer adverse effects, making it a practical alternative for cats that are sensitive to the stress associated with needle aversion.
Footnotes
Author note
The original data are available upon request.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Meng Li’s startup fund of Nanjing Agricultural University (804127).
Ethical approval
The work described in this manuscript involved the use of experimental animals and the study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
