Abstract
Objectives
This study aimed to assess the prevalence of hypocobalaminaemia (B12 <400 pg/ml) and hypercobalaminaemia (B12 >1000 pg/ml), describe the clinicopathological abnormalities and the diagnostic imaging findings in a referral population of cats in the UK, and identify the underlying disease processes associated with both conditions.
Methods
A retrospective study of cats that had their serum cobalamin concentration assessed between December 2016 and December 2023 at a single referral hospital; 216 cats were included. Patient outcome was established from the clinical records.
Results
A total of 76 (35%) cats had hypocobalaminaemia and 67 (31%) cats had hypercobalaminaemia. The most common diagnoses were chronic enteropathy (CE) in 39/76 (51%) hypocobalaminaemic cats and 39/67 (58%) hypercobalaminaemic cats (P = 0.001), and high-grade lymphoma in 14/76 (18%) hypocobalaminaemic cats and 11/67 (16%) hypercobalaminaemic cats (P = 0.438). The most common clinical signs were chronic vomiting in 36/76 (47%) hypocobalaminaemic cats and 24/67 (36%) hypercobalaminaemic cats (P = 0.005), hyporexia in 40/76 (53%) hypocobalaminaemic cats and 21/67 (31%) hypercobalaminaemic cats (P <0.001), and chronic diarrhoea in 12/76 (16%) hypocobalaminaemic cats and 21/67 (31%) hypercobalaminaemic cats (P = 0.001). The most common abnormalities identified on abdominal ultrasound were lymphadenomegaly and thickened intestines in 49/76 (64%) hypocobalaminaemic cats and 28/67 (42%) hypercobalaminaemic cats (P = 0.0025). Median survival time was 274 days in the hypocobalaminaemic group and 711 days in the hypercobalaminaemic group (P = 0.001). The hypocobalaminaemic cats exhibited significantly reduced survival time compared with hypercobalaminaemic cats (odds ratio 2.4 vs 0.4, respectively) (P <0.001).
Conclusions and relevance
This study suggests that cobalamin has limited diagnostic utility in differentiating between underlying disease processes; chronic diarrhoea and CE are more common in hypercobalaminaemic cats in contrast with the previous literature. Hypocobalaminaemia is associated with reduced survival in this cohort of cats; therefore, early cobalamin supplementation is recommended.
Keywords
Introduction
Cobalamin (vitamin B12) is a water-soluble vitamin that plays an essential role in many metabolic intracellular reactions and is widely used in small animal practice as a marker of gastrointestinal disease. Hypocobalaminaemia has been reported in a variety of diseases in veterinary patients, including many diseases of the gastrointestinal tract, liver and pancreas, including exocrine pancreatic insufficiency, chronic severe ileal disease, small intestinal dysbiosis and inherited conditions.1 –3 In dogs, congenital cobalamin malabsorption secondary to genetic disorders has been identified in several breeds such as the Border Collie and Giant Schnauzer.1,2 In humans, a rare inherited selective B12 malabsorption exists, also called Imerslund–Gräsbeck syndrome. 4 Congenital disorders of cobalamin absorption appear to be very rare in cats, with only one report of a suspected case published. 5
Hypocobalaminaemia in cats, similarly to dogs, has been associated with exocrine pancreatic insufficiency, intestinal disease such as CE and lymphoma, biliary disease or a combination of these diseases.6 –8 There have only been a small number of studies in cats that have evaluated the prevalence of hypocobalaminaemia with gastrointestinal disease, which generated different results.6 –8 The prevalence of hypocobalaminaemia in feline patients appears to be similar to the canine population,9 –12 and was reported to occur in up to 43% of cats with CE8,13 and up to 78% of cats with intestinal lymphoma.8,13,14
In dogs, hypocobalaminaemia has been identified as a negative prognostic factor in exocrine pancreatic insufficiency,15,16 CE 9 and lymphoma. 17 There are currently no published studies that have evaluated the detection of hypocobalaminaemia as a risk factor for a negative outcome in cats.
Few studies have investigated the clinical relevance of hypercobalaminaemia in companion animals. 2 In dogs, it was reported to be associated mainly with gastrointestinal and hepatic disease but was also associated with endocrine and neurological conditions. 18 Similarly, studies in human medicine have associated hypercobalaminaemia with solid neoplasms, haematological malignancies, and hepatic and renal diseases. 19 In cats, it was previously reported to be associated with hepatic disease and solid neoplasm; 20 however, a recent study failed to identify those associations. 21 It is currently unknown whether the presence of hypercobalaminaemia is clinically significant in cats.
The aims of this study were to assess the prevalence of hypocobalaminaemia, defined as a serum cobalamin concentration below 400 pg/ml, and hypercobalaminaemia, defined as a serum cobalamin concentration over 1000 pg/ml, to describe associated clinicopathological abnormalities and diagnostic imaging findings in a referral population of cats in the UK, and to identify underlying disease processes associated with both conditions. A secondary objective was to evaluate whether the detection of hypocobalaminaemia is a negative prognostic factor in this cohort of cats.
Materials and methods
This study was approved by the RCVS Ethics Review Panel (2023-112) and the owners provided written informed consent for their pets’ participation.
This retrospective study was conducted at a single private referral institution between December 2016 and December 2023.
The clinical pathology database was retrospectively searched for feline serum cobalamin results during the above-mentioned study period. Serum cobalamin concentration was measured using a chemiluminescent enzyme immunoassay (Immulite 2000 Vitamin B12 system; Siemens).
Cats were included if they had a serum cobalamin concentration measured, serum biochemistry performed and abdominal imaging (ultrasound or CT) conducted by a board-certified diagnostic imaging specialist or a supervised resident.
Exclusion criteria included prior cobalamin supplementation within 12 weeks before measurement, incomplete clinical information or unavailable final diagnosis. Clinical variables retrieved from the achieved clinical notes included age, sex, breed, body weight, body condition score (BCS), presenting clinical signs, duration of clinical signs, diagnosis, abnormal haematology and serum biochemistry findings, serum cobalamin concentration and date of measurement, abnormal abdominal imaging findings, abdominal imaging modality used, and time and reason of death. Additional information that was collected, if available, included modality of biopsy collection, histopathology results and ancillary tests performed, including immunohistochemistry (IHC), clonality test using PCR for antigen receptor rearrangements (PARR) or flow cytometry, type of cobalamin supplementation, duration of supplementation and serum cobalamin after supplementation.
The prevalence of hypercobalaminaemia, which was defined as a serum cobalamin concentration above 1000 pg/ml, and hypocobalaminemia, defined as a serum cobalamin concentration below 400 pg/ml, was estimated. The cutoff of 400 pg/ml was chosen according to the recommendations of the Gastrointestinal Laboratory at Texas A and M University. 22 Normocobalaminaemia was defined as a serum cobalamin concentration in the range of 400–1000 pg/ml.
CE was defined, according to the 2023 American College of Veterinary Internal Medicine (ACVIM) consensus statement, 23 as signs of gastrointestinal disease for a duration of at least 3 weeks, where extragastrointestinal, metabolic and infectious causes have been ruled out. Cats with CE were further diagnosed with either lymphoplasmacytic enteritis (LPE), eosinophilic enteritis (EE) or low-grade intestinal T-cell lymphoma (LGITL) based on histopathology. LPE was defined as per the 2023 ACVIM consensus statement 23 as inflammatory lesions in the gastrointestinal tract that are dominated by lymphocytic infiltration in the lamina propria, while the term LGITL was used for lesions characterised by a monomorphic infiltration of the lamina propria or epithelium or both of cats with small, mature, neoplastic (clonal) T lymphocytes. The term EE was used for inflammatory lesions with significant eosinophilic infiltrate. The term chronic inflammatory enteropathy (CIE) was used to refer to cats with histopathologically confirmed LPE or EE. When a definitive histopathological diagnosis was not possible, a presumed diagnosis of CE was made.
High-grade lymphoma was diagnosed on cytology based on the presence of a monomorphic population of large, atypical lymphocytes exhibiting cytological features of malignancy.
Pancreatitis was presumptively diagnosed based on a combination of compatible clinical signs (such as vomiting, lethargy, anorexia and abdominal pain), ultrasonographic findings suggestive of pancreatitis (enlarged, hypo- or hyperechoic pancreas, hyperechoic mesentery and focal peritoneal effusion) and increased serum pancreatic lipase activity (1,2-o-dilauryl-rac-glycero-3-glutaric acid-(6’-methylresorufin) ester [DGGR] lipase >26 IU/l) and/or feline pancreatic lipase immunoreactivity (fPLI >4.5 μg/l).
Cholangitis was defined by the presence of ultrasonographic findings suggestive of hepatobiliary disease, including gallbladder or common bile duct wall thickening, hyperechoic gallbladder content, cholelithiasis, biliary duct dilation, altered hepatic echotexture or hepatomegaly. Diagnosis was supported by concurrent hyperbilirubinemia when pre-hepatic causes were excluded. Further confirmation required the presence of bactibilia and/or inflammatory cells on bile cytology with a positive bile culture, or histopathological evidence of neutrophilic or lymphocytic infiltration, with or without bacterial infection.
Hepatic lipidosis was defined by the presence of an enlarged, hyperechoic liver on ultrasound, cytological or histopathological evidence of a predominantly vacuolar hepatopathy, and a cholestatic pattern on serum biochemistry characterised by a disproportionate increase in alkaline phosphatase (ALKP) relative to gamma-glutamyl transferase (GGT) and alanine aminotransferase (ALT), along with concurrent hyperbilirubinemia after exclusion of pre-hepatic causes. Triaditis was defined as concurrent pancreatitis, cholangitis and CE.
Patient outcomes were determined from medical records or, if unavailable, by contacting the primary care practices.
Data were analysed using Minitab Statistical Software Version 21.1.0. Continuous variables were presented as median (range) and categorical variables as number and percentage per category.
Comparisons between hypocobalaminaemic, normocobalaminaemic and hypercobalaminaemic cats were performed using one-way ANOVA (or the Kruskal–Wallis test if assumptions were unmet) for scale data, Kruskal–Wallis tests for ordinal data and χ2 tests for categorical data. Survival was compared using log rank tests and Kaplan–Meier plots.
Sample size calculations were based on a power of 0.80 and a significance level of 0.05. For the one-way ANOVA, Cohen’s (1992) medium effect size estimate indicated a requirement of 52 animals per group. The Kruskal–Wallis test required a 15% increase in sample size (60 per group). For χ2 tests, an R package estimated a minimum of 72 animals per group, assuming a 3 × 2 contingency table with one group showing a 40% incidence and the other two groups 20%. P values <0.05 were considered statistically significant.
Results
During the study period, a total of 266 cats that had serum cobalamin concentration assessed were identified in the clinical pathology database. Of these cats, 86 were in the hypercobalaminaemic group, 91 were in the hypocobalaminaemic group and 89 were in the normocobaminaemic group. After excluding 50 cats because of incomplete clinical records, 216 cats were included in the study and classified according to their cobalamin status. A total of 76 (35%) cats were hypocobalaminaemic, 67 (31%) were hypercobalaminaemic and the remaining 73 (34%) cats had normal cobalamin.
Signalment details are summarised in Table 1. The study population included 130 domestic shorthairs, 14 domestic longhairs and 72 purebred cats. The most common breeds were British Shorthairs (12/216) and Maine Coons (9/216) (Table 2). There were 105 (49%) female and 111 (51%) male cats, of which 211 (98%) were neutered. The median age of the population was 9 years (range 6 months to 26 years), median BCS was 4 (range 2–8) and median body weight was 3.8 kg (range 1.4–11.2). Sex, reproductive status and body weight did not differ significantly between groups (P = 0.988, P = 0.280 and P = 0.055, respectively) (Tables 1 and 2); however, age and BCS were statistically significant (P <0.001 and P = 0.016, respectively) (Figures 1 and 2). Hypercobalaminaemic cats were significantly younger (median age 7 years; range 10 months to 18 years) than hypo- and normocobalaminaemic cats (median age 11 years; range 6 months to 26 years and median age 9 years; range 10 months to 19 years, respectively) (Figure 3).

Box plot representing the age distribution between the three groups (hypocobalaminaemia, normocobalaminaemia and hypercobalaminaemia)
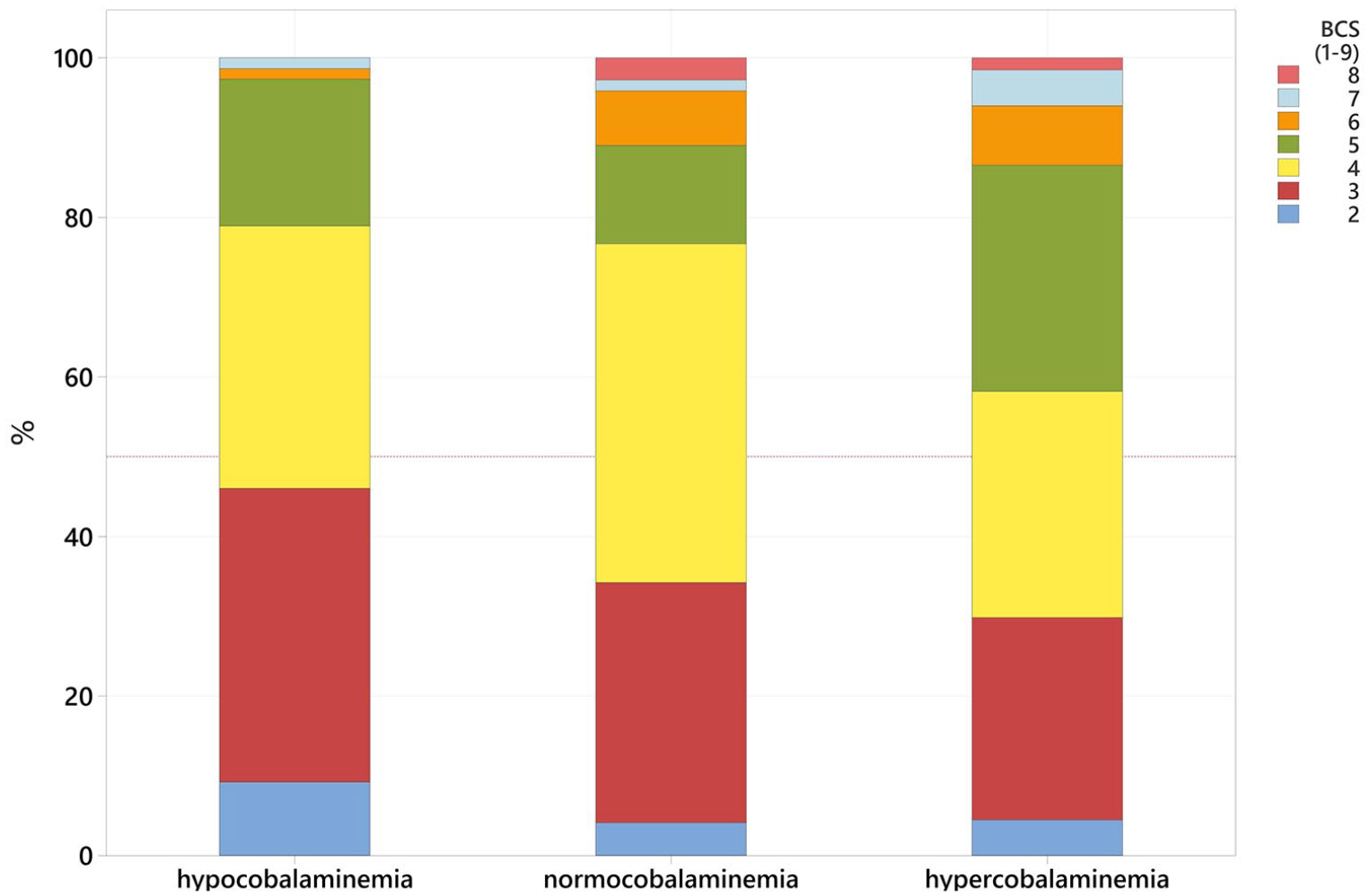
Bar chart representing the body condition score distribution between the three groups (hypocobalaminaemia, normocobalaminaemia and hypercobalaminaemia)

Bar chart representing the percentage (%) of cats that display the three main clinical signs identified: chronic vomiting (v+), hyporexia and chronic diarrhoea (d+)
Signalment data for the three groups of cats (hypercobalaminaemia, hypocobalaminaemia and normocobalaminaemia)

Data are n (%) or median (range)
BCS = body condition score; BW = body weight
Breed distribution between the three groups of cats (hypercobalaminaemia, hypocobalaminaemia and normocobalaminaemia)

Data are n (%)
Presenting clinical signs are reported in Table 3. The most common clinical signs that achieved statistical significance were chronic vomiting in 24/67 (36%) hypercobalaminaemic cats and 36/76 (47%) hypocobalaminaemic cats (P = 0.005), hyporexia in 21/67 (31%) hypercobalaminaemic cats and 40/76 (53%) hypocobalaminaemic cats (P <0.001), lethargy in 16/67 (24%) hypercobalaminaemic cats and 32/76 (42%) hypocobalaminaemic cats (P = 0.043), and chronic diarrhoea in 21/67 (31%) hypercobalaminaemic cats and 12/76 (16%) hypocobalaminaemic cats (P = 0.001) (Figure 3).
Presenting clinical signs for the three groups of cats (hypercobalaminaemia, hypocobalaminaemia and normocobalaminaemia) and their respective P values

Data are n (%). The χ2 test was used to compare the three groups. P values in bold are statistically significant
N/A = not applicable
In the hypercobalaminaemic group, the most common diagnoses were CE (of which 12% were diagnosed with CIE, 45% presumed CE and 1% LGITL) in 39/67 (58%) cats, high-grade lymphoma in 11/67 (16%) cats and chronic kidney disease in 10/67 (15%) cats. In the hypocobalaminaemic group, the most common diagnoses were CE (of which 8% were diagnosed with CIE, 33% presumed CE and 11% LGITL) in 39/76 (51%) cats, high-grade lymphoma in 14/76 (18%) cats and triaditis in 7/76 (9%) cats. The diagnosis of CE reached statistical significance (P = 0.001), particularly for presumed CE (P = 0.026) and LGITL (P = 0.002) (Table 4, Figure 4). Table 4 shows the proportion of cats in each disease category among the groups.
Categories of disease for the three groups of cats (hypercobalaminaemia, hypocobalaminaemia and normocobalaminaemia) and their respective P values

Data are n (%). χ2 test was used to compare the three groups. P values in bold are statistically significant
N/A = not applicable
Endoscopic intestinal and gastric biopsies were performed in 12/216 (6%) cases and confirmed the diagnosis of LPE in 9/12 (75%) cases, EE in 1/12 (8%) cases and LGITL in 2/12 (17%) cases.
Exploratory laparotomy was performed in 26/216 (12%) cases. Of these, 14 (54%) underwent full-thickness intestinal biopsies, confirming a diagnosis of LPE in 6/14 (43%) cases, EE in 2/14 (14%) cases, LGITL in 5/14 (36%) cases and visceral mast cell tumour in 1 (7%) case. The remaining 12/26 (46%) cats were diagnosed with pyogranulomatous lymphadenitis (6/12, 50%), chronic hepatitis (2/12, 17%), hepatic lymphoma (1/12, 8%), squamous cell carcinoma (1/12, 8%), metastatic carcinoma (1/12, 8%) and chronic cystitis (1/12, 8%). CIE was histopathologically confirmed in 18/99 (18%) cats, of which 15 were diagnosed as LPE and three as EE. Two cats were diagnosed with LGITL via cytology of the mesenteric lymph node in combination with either PARR or flow cytometry. Of the remaining seven cats that underwent gastrointestinal histopathology, one cat underwent PARR, two cats underwent IHC and one cat had both ancillary tests.
The most common clinicopathological abnormalities are summarised in Table 5. Hypocobalaminaemic cats most commonly exhibited mild non-regenerative anaemia (P = 0.002), moderate leucocytosis (P = 0.005), mild-to-moderate neutrophilia (P <0.001), mild-to-moderate monocytosis (P = 0.023) and mild-to-moderate hypoalbuminaemia (P = 0.002). Hypercobalaminaemic cats were more likely to have mild-to-moderate hypercholesterolaemia (P = 0.024 and moderate-to-marked ALKP elevation (P = 0.037) Figure 5).

Bar chart representing the percentage of cats with the diagnosis of LGITL, LPE, EE and presumed CE, among the three groups: hypocobalaminaemia, normocobalaminaemia and hypercobalaminaemia. Most cats diagnosed with LGITL have hypocobalaminaemia, while most cats with presumed CE have hypercobalaminaemia. CE = chronic enteropathy; EE = eosinophilic enteritis; LGITL = low-grade intestinal T-cell lymphoma; LPE = lymphoplasmacytic enteritis
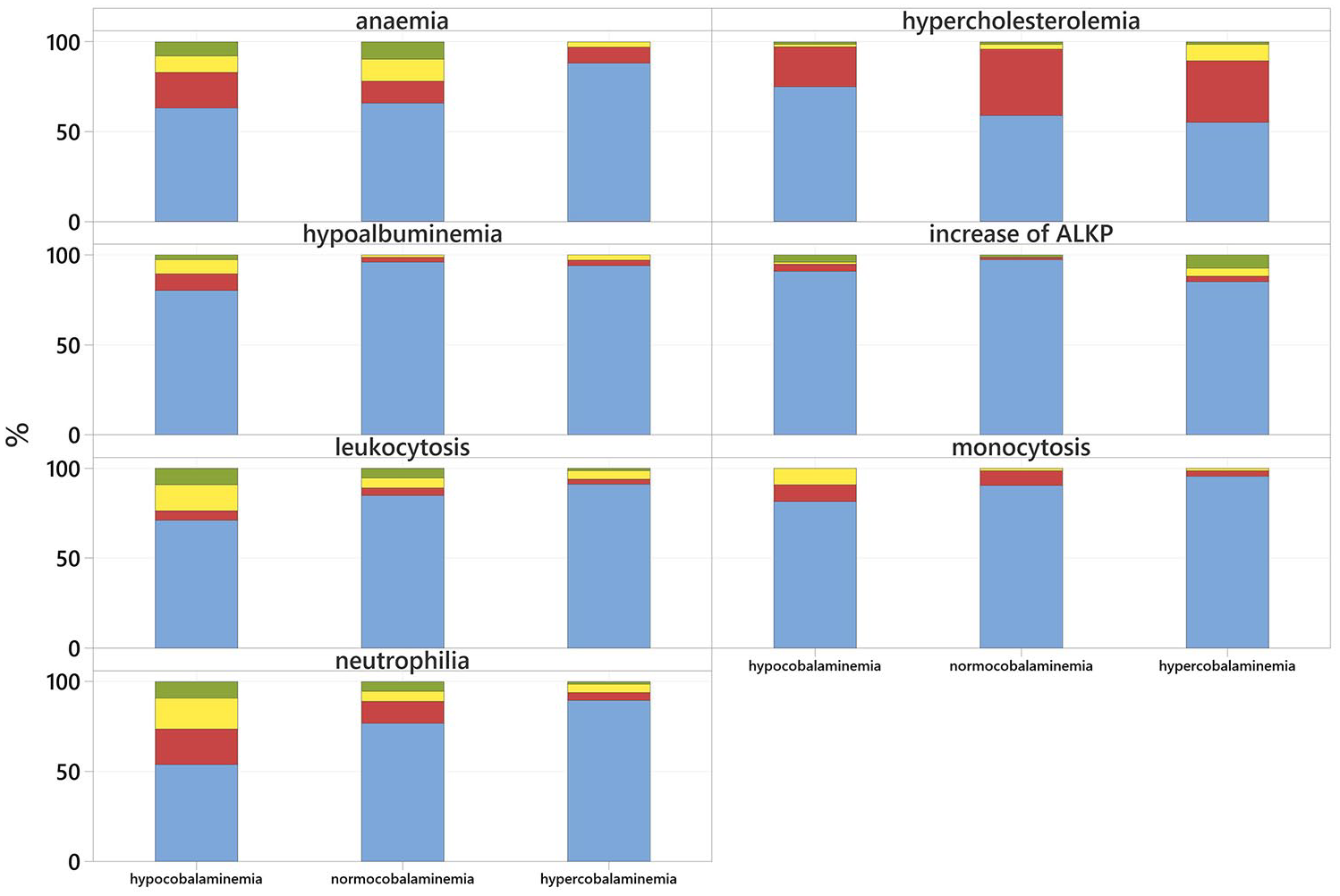
Stacked bar representing the proportion of cats in the hypocobalaminaemic, hypercobalaminaemic and normocobalaminaemic group with anaemia, leucocytosis, neutrophilia, monocytosis, hypoalbuminaemia, increased alkaline phosphatase and hypercholesterolaemia. Cats with normal values are shown in blue, those with mild values in red, those with moderate values in yellow and those with severely abnormal values in green
Most common clinicopathological abnormalities for the three groups of cats (hypercobalaminaemia, hypocobalaminaemia and normocobalaminaemia) and their respective P values
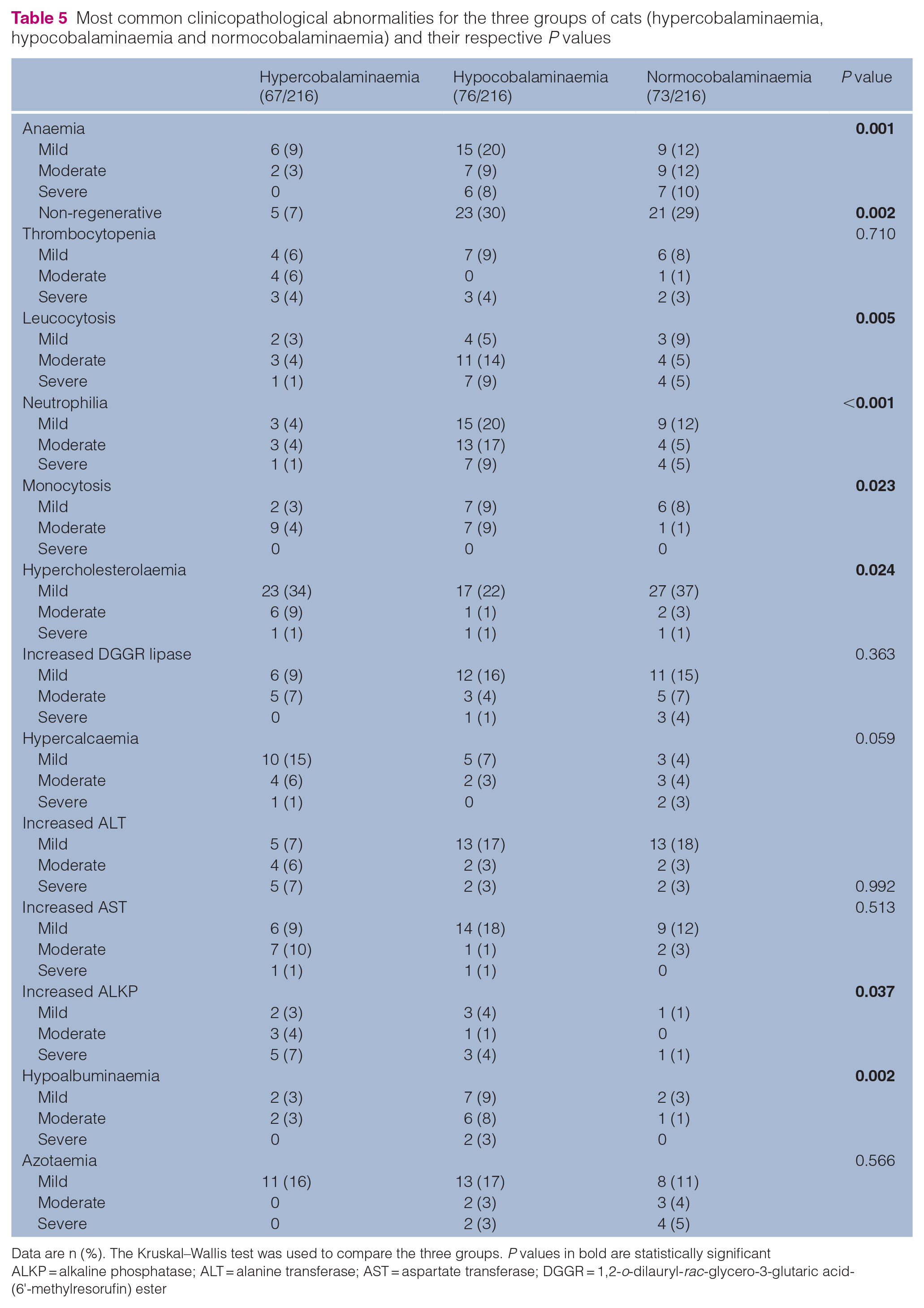
Data are n (%). The Kruskal–Wallis test was used to compare the three groups. P values in bold are statistically significant
ALKP = alkaline phosphatase; ALT = alanine transferase; AST = aspartate transferase; DGGR = 1,2-o-dilauryl-rac-glycero-3-glutaric acid-(6'-methylresorufin) ester
A total of eight cats underwent CT, while 211 cats had abdominal ultrasound, with three cats undergoing both imaging modalities. The imaging findings are shown in Table 6. The most common abnormalities identified that achieved significance were lymphadenomegaly in 49/76 (64%) hypocobalaminaemic cats and 28/67 (42%) hypercobalaminaemic cats (P = 0.0025), thickened intestines in 49/76 (64%) hypocobalaminaemic cats and 28/67 (42%) hypercobalaminaemic cats (P = 0.025), ascites in 38/76 (50%) hypocobalaminaemic cats and 17/67 (25%) hypercobalaminaemic cats (P = 0.008), and gallbladder sludge in 26/76 (34%) hypocobalaminaemic cats and 13/67 (19%) hypercobalaminaemic cats (P = 0.024).
Most common diagnostic imaging findings for the three groups of cats (hypercobalaminaemia, hypocobalaminaemia and normocobalaminaemia) and their respective P values

Data are n (%). The χ2 test was used to compare the three groups. P values in bold are statistically significant
In the hypocobalaminaemic group, 42/76 (55%) cats received injectable hydroxocobalamin supplementation for a median of 6 weeks (range 1–12), while 25/76 (33%) cats received oral cobalamin supplementation for a median of 12 weeks (range 2–24). In addition, 7/76 (9%) cats received a combination of both types of supplementation. A total of 15 (20%) cats did not receive any supplementation. Among the cats that had cobalamin reassessed after supplementation (24/67, 36%), 14/24 (58%) had a cobalamin concentration exceeding 1000 pg/ml, while 9/24 (38%) were within the normocobalaminaemic range and 1/24 (4%) remained in the hypocobalaminaemic range.
Mortality was significantly higher in the hypocobalaminaemic group (55/76, 72%) compared with the hypercobalaminaemic (22/67, 33%) and normocobalaminaemic groups (38/73, 52%) (P <0.001). Death was primarily due to disease progression, with one unrelated case of aortic thromboembolism. Most cats (109/115, 95%) were euthanased; of the remaining cats, 2/115 (2%) suffered cardiopulmonary arrest and 4/115 (3%) died suddenly at home.
Survival curve analysis (Figure 6) revealed a median survival time of 274 days in the hypocobalaminaemic group, 389 days in the normocobalaminaemic group and 711 days in the hypercobalaminaemic group (P = 0.001).

Kaplan–Meier curve depicting survival time for 216 cats at the time of cobalamin measurement
Binary logistic regression indicated increased risk of mortality in hypocobalaminaemic cats (odds ratio [OR] 2.4, 95% confidence interval [CI] 1.22–4.76) and a decreased risk in hypercobalaminaemic cats (OR 0.45, 95% CI 0.22–0.89) when both were compared with normocobalaminaemic cats (P <0.001). Fitted probability of death were 72% for the hypocobalaminaemic cats, 52% for the normocobalaminaemic cats and 33% for the hypercobalaminaemic cats.
After age adjustment to counteract for the age difference between groups, the hypocobalaminaemic cats remained at an increased risk of mortality (OR 2, 95% CI 0.98–4.08) and the hypercobalaminaemic cats remained with a decreased risk (OR 0.5, 95% CI 0.25–1.04) (P <0.001). Fitted probability of death was also very similar: 69% for the hypocobalaminaemic cats, 53% for the normocobalaminaemic cats and 37% for the hypercobalaminaemic cats.
Stratifying the hypocobalaminaemic group revealed a significantly increased mortality risk in cats with severe hypocobalaminaemia (<200 pg/ml) (OR 5.06, 95% CI 1.89–13.54), while cats with moderate hypocobalaminaemia (200–400 pg/ml) showed a lesser risk (OR 1.35, 95% CI 0.60–3.00) (P = 0.001).
Revised survival curve analysis (Figure 7) demonstrated a median survival time of 120 days for the severe hypocobalaminaemia subgroup and 364 days for the moderate subgroup (P = 0.001).

Kaplan–Meier curve depicting survival time for 216 cats at the time of cobalamin measurement
Discussion
This study suggests that more than half of cats diagnosed with CE could either present with hypercobalaminaemia or hypocobalaminaemia. The latter is presumed to result from reduced absorption of intrinsic factor–cobalamin complexes in the distal small intestine, sometimes in combination with small intestinal dysbiosis. 2 Conversely, the underlying mechanism of increased serum cobalamin in gastrointestinal disease remains unclear, although direct diffusion through a disrupted mucosal barrier or dysbiosis are possible explanations. 18 When multiple comorbidities are present, several factors may contribute to increased serum cobalamin, such as release from hepatocytes in liver disease, reduced renal clearance with kidney disease or increased dietary intake.2,18,19
The prevalence of hypocobalaminaemia in this study population was 35%, aligning with previous literature.7,8,13 The most common diagnoses, aside from CE in 51% of cats, were high-grade lymphoma and triaditis in 18% and 9% of cases, respectively, with only the former reaching statistical significance. Notably, although only a small subset was diagnosed with LGITL, 8/9 (89%) cats had a low cobalamin concentration, a statistically significant finding. Thus, we confirmed what other authors previously reported:8,13,14 that LGITL appears to be associated with increased prevalence of hypocobalaminaemia.
The prevalence of hypercobalaminaemia in this population was 31%, with the most common diagnoses being CE in 58% of cases, followed by high-grade lymphoma and chronic kidney disease in 16% and 15% of cases, respectively. To the author’s knowledge, only three studies have explored the significance of hypercobalaminaemia in cats.2,20,21 Similarly to one of these studies, 2 our study found a high proportion of cats diagnosed with CE.
Histopathology was performed in only 25/99 (25%) cats, of which 18 were diagnosed with LPE or EE and seven with LGITL. The majority of cats had a presumed diagnosis of CE (72/99, 73%), largely because of financial constraints, owner preferences, clinical improvement with dietary or medical management alone, or concurrent medical conditions precluding general anaesthesia.
The prevalence of high-grade lymphoma in our study was higher than previously reported, where LGITL is typically more commonly diagnosed. This discrepancy may be due to the limited number of histopathological diagnoses and potential selection bias inherent in retrospective studies. Many cats did not undergo full diagnostic workups, including histopathology, PARR or IHC, potentially leading to an underdiagnosis of LGITL cases. In addition, cats with more advanced clinical signs might have been more likely to receive definitive diagnoses, further contributing to this imbalance.
The most common clinicopathological findings identified in hypocobalaminaemic cats included mild non-regenerative anaemia, leucocytosis, neutrophilia, monocytosis and hypoalbuminaemia. The haematological changes are non-specific and suggestive of inflammatory response, while the anaemia likely reflects chronic disease. Hypoalbuminemia is most likely secondary to increased intestinal losses and reduced absorption. An unexpected finding was hypercholesterolaemia, which was most prevalent in hypercobalaminaemic cats. This could be related to cholestasis or kidney disease, although diet and post-prandial influence could play a role. Hypercobalaminaemic cats also exhibited increased ALKP levels, likely indicative of secondary hepatopathy (such as secondary to gastrointestinal disease).
Furthermore, both groups had a high prevalence of lymphadenomegaly and thickened intestines. These findings are not unexpected and reflect the large proportion of cats diagnosed with gastrointestinal disease or neoplasia. A previous study 8 identified intestinal ultrasound changes in 65% cats with signs of gastrointestinal disease and hypocobalaminaemia.
Cobalamin supplementation can be administered effectively either parenterally or orally.3,24 –26 In this study, most cats received injectable supplementation; however, as supplementation was not standardised, definitive conclusions regarding the most efficacious modality cannot be drawn. Nevertheless, only half of the supplemented cats had cobalamin levels above 1000 pg/ml, suggesting that supplementation alone may be insufficient to replenish cobalamin body stores if the underlying disease causing hypocobalaminaemia is not adequately managed.
Multiple studies1,9,10,14,16,17,27 have highlighted the therapeutic and prognostic importance of cobalamin supplementation. In support to the latter, our study found that hypocobalaminaemic cats had an increased risk of mortality. Specifically, the median survival time was 274 days, with more than twice the risk of shorter survival compared with normo- and hypercobalaminaemic cats. This risk increased five-fold in cats with cobalamin concentrations below 200 pg/ml. Thus, early identification and supplementation of cobalamin deficiency are crucial for improving clinical outcomes. To the authors’ knowledge, this is the first study to evaluate hypocobalaminaemia as a risk factor for negative outcomes in cats.
Although statistically significant differences in clinical signs were observed across the three groups, their overlapping nature suggests limited diagnostic utility. In addition, it is noteworthy that cats with normal cobalamin concentration also had a history of gastrointestinal signs and were frequently diagnosed with gastrointestinal disease, hepatobiliary disease or neoplasia. This further indicates that cobalamin is not a specific diagnostic marker. The prognostic value of cobalamin status, particularly its association with survival, remains the most clinically impactful finding.
The present study has some limitations. First, paired methylmalonic acid measurement, a marker for cellular cobalamin status, was not performed because of unavailability in our diagnostic laboratory. To address this limitation, further studies should measure both serum cobalamin and methylmalonic acid to better assess cellular cobalamin status. Second, the retrospective data collection may have resulted in incomplete or inaccurate records. Many cats lacked histopathological confirmation of CIE, potentially leading to misclassification bias. It is also possible that CIE cases are overrepresented because of selection bias at our referral centre. The recent literature 23 suggests that LGITL and CIE represent a diagnostic continuum rather than discrete categories and it is well known that a differentiation between LGITL and CIE is particularly challenging in feline patients; as such, it is not excluded that some cats may have been misdiagnosed, and a proportion of cases classified as LPE may have actually represented LGITL, especially considering that IHC or PARR were not consistently performed in all cases. Equally, the definitive diagnosis was not confirmed in all cats, and some conditions were diagnosed in a small number of cats, preventing statistical analysis. For conditions such as triaditis, cholangitis, hepatic lipidosis and pancreatitis, a retrospective diagnosis might be inaccurate given the lack of gold standard testing (histopathology); as such, misclassification is possible and might have impacted our results. Future prospective studies with larger sample sizes and standardised diagnostic protocols, including histopathology, IHC and PARR, could help address these limitations. Finally, despite recruiting a large cohort of cats, the number of cats in the hypercobalaminaemic group fell short of the calculated sample size, potentially limiting the study’s power to detect a difference between groups.
Conclusions
This study suggests that cobalamin has limited diagnostic utility in differentiating between underlying disease processes. In contrast with the previous literature, chronic diarrhoea and CE were more common in hypercobalaminaemic cats. The clinical significance of hypercobalaminaemia remains uncertain, warranting further investigation into its underlying mechanisms and prognostic implications. Hypocobalaminaemia was associated with reduced survival in this cohort of cats, suggesting the importance of early cobalamin supplementation to improve clinical outcomes.
Footnotes
Acknowledgements
The authors would like to thank Tim Spark (Waltham Petcare Science Institute) for his invaluable assistance with the statistical analysis and Hannah Sargent (Linnaeus Clinical Research) for her precious support during the application for the RCVS ethical approval.
Author note
This manuscript was presented in part as an abstract at the 2024 European College of Veterinary Internal Medicine (ECVIM) congress in Lyon, France.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Linnaeus Veterinary Limited supported the costs of the open access publication charges.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
