Abstract
Objectives
The aim of the study was to evaluate the effects of trazodone on sedation, and physiological and echocardiographic variables in healthy cats.
Methods
This randomised, blinded, crossover study involved eight healthy adult cats receiving either a placebo or oral doses of trazodone (50 mg, 75 mg, 100 mg), with a washout period of at least 1 week between doses. Sedation, muscle relaxation and analgesia scores were assessed, along with physiological variables including systolic blood pressure (SBP), pulse rate (PR) and respiratory rate (RR) at baseline (T0) and at 30-min intervals after administration (T30–T240). Echocardiographic variables were measured at T0 and T90.
Results
In the trazodone groups, cats’ sedation scores significantly increased compared with T0, with no significant changes in muscle relaxation or analgesia scores. A significant mean reduction of 22 ± 7 mmHg in SBP was observed only at T150 after oral administration of 100 mg trazodone compared with the placebo, but the SBP still remained within the reference interval. Across all trazodone doses, PR showed no significant changes, while RR significantly decreased compared with T0. There were no significant changes in echocardiographic variables after administration of three different doses of trazodone.
Conclusions and relevance
Oral administration of 50 mg, 75 mg or 100 mg of trazodone in cats produces mild sedation but there is a lack of muscle relaxation and analgesic effects. Trazodone has minimal effects on SBP, PR and RR in cats, although the 100 mg dose may cause a slight decrease in SBP within the physiological interval. Furthermore, oral trazodone at the tested doses has no impact on echocardiographic variables.
Introduction
During transportation and veterinary examinations, cats often exhibit stress responses such as fear and aggression, 1 which can affect clinical procedures and physiological variables.2,3 While stress in cats during veterinary visits can be mitigated through gentle handling and training, medication may also be required.1,4 Numerous studies have explored pre-visit medications to alleviate stress in cats. Gabapentin and pregabalin, both gabapentinoid drugs, have been shown to be effective in reducing stress during transportation and veterinary visits. Although oral gabapentin does not significantly impact physiological or echocardiographic variables in cats,5 –7 it can induce notable sedative effects in cats with chronic kidney disease as a result of prolonged kidney metabolism. 8 Dexmedetomidine also induces sedation, facilitating veterinary visits for cats; however, it produces adverse effects, such as vomiting and bradycardia.9,10 Therefore, further research into alternative pre-visit medications for reducing stress in cats during veterinary visits is warranted.
Trazodone is a serotonin (5-hydroxytryptamine, 5-HT2) antagonist/reuptake inhibitor and an alpha (α)-adrenergic receptor and histamine antagonist. It possesses anxiolytic and sedative properties, making it a viable pharmacological option for pre-appointment medication in cats. 11 Trazodone has been demonstrated to improve anxiety disorders in dogs, 12 and its oral administration before veterinary visits can alleviate stress during examinations. 13 In addition, its use in hospitalised dogs helps mitigate stress-related signs and behaviours. 14 However, there is a lack of extensive clinical research supporting the use of trazodone in cats. Sedative effects have been observed in cats after oral administration of 50 mg trazodone, resulting in decreased activity levels, with reports suggesting its facilitation of veterinary visits.15 –17 These studies, however, also demonstrate potential adverse effects such as hypotension and behavioural disinhibition after oral administration of trazodone in cats, with no improvement in aggression.11,16 In addition, although a study has indicated that oral administration of 50 mg trazodone in cats does not clinically alter echocardiographic variables, 15 the influence of higher doses remains unknown. Therefore, further research is needed to validate the clinical effects of oral trazodone in cats.
The primary aim of this study was to evaluate the sedative effects of oral trazodone in healthy cats and its impact on physiological and echocardiographic variables. Our hypotheses were: (1) different doses of oral trazodone induce sedation in cats; (2) oral trazodone has no significant effect on physiological variables; and (3) oral trazodone does not significantly alter echocardiographic variables.
Materials and methods
Animals
The experimental subjects comprised eight adult cats (four castrated males and four spayed females) with a mean age (±SD) of 42 ± 12 months and mean weight of 3.98 ± 0.41 kg. All cats were housed individually in separate spaces, provided with sufficient food and water, and given daily care. Before the study, all cats underwent a thorough physical examination, complete blood count, blood biochemical analysis and echocardiographic assessment to confirm their health status. Ethical approval for the study was obtained from the Institutional Animal Care and Use Committee of Nanjing Agricultural University (20230925143).
Study design and treatments
The study utilised a randomised, double-blind, crossover trial design. The drug administration schemes were randomised using Research Randomizer (https://www.randomizer.org/), and the researchers were blinded to the dosing protocols. A 1-week washout period was implemented between each administration. Cats were fasted for 12 h prior to each trial but had free access to water. Thirty mins before each trial, the cats underwent shaving and acclimatisation to the environment. Baseline measurements (T0) were then recorded, including systolic blood pressure (SBP), pulse rate (PR), respiratory rate (RR), sedation score, muscle relaxation score, analgesia score and echocardiographic examination. Subsequently, cats were administered 50 mg, 75 mg or 100 mg of trazodone (Meishiyu; Hemony), designated as TRA-50, -75 or -100, or magnesium–aluminium hydrotalcite chewable tablets (Talcid; Bayer) as a placebo according to a predetermined protocol. Assessments excluding echocardiography were repeated every 30 mins after administration, up to 240 mins, designated as T30, T60, T90, T120, T150, T180, T210 and T240. In addition, echocardiographic measurements were performed at T90, with each echocardiogram completed within 15 mins.
Clinical effects
The clinical effects encompass the evaluation of sedation, muscle relaxation and analgesia scores in cats after oral administration of either a placebo or various doses of trazodone. The sedation scores are outlined in a previous study and detailed in the supplementary material. 18 The total sedation score, in the range of 0–13, is a cumulative assessment of posture, positioning in lateral recumbency, extension of the left hindlimb, extension of the left knee, response to a sound stimulus (clap) and response to a blow of air in the ear. A higher score indicates a deeper level of sedation. Muscle relaxation scores and analgesia scores are presented in Table 1. These scoring systems are derived from previously published studies.18,19 All assessment procedures adhere to a sequence from mild to intense.
Muscle relaxation score and analgesia score

Physiological variables
A cuff width covering 30–40% of the tail circumference was chosen. The Doppler crystal was placed over the coccygeal artery and an ultrasonic Doppler flow detector (Model 811B; Parks Medical Electronics) was used to monitor the sound. SBP and PR values were recorded five times consecutively. After excluding the highest and lowest values, the average of the remaining three was calculated. Concurrently, RR was determined by observing the chest wall movements of the cats for 1 min.
Echocardiogram
Based on prior studies,15 –17 echocardiographic examinations were conducted at both T0 and T90, maintaining the same sequence for all assessments, and completed within 15 mins. These examinations were performed by the same researcher who was blinded to the dosing protocols. Each variable was measured three times and the resulting average was used for analysis.
Echocardiographic examinations were performed using an ultrasonographic scanner (Mylab W70; Esaote). Initially, the cats were positioned in right lateral recumbency. From the standard right parasternal long-axis view, several dimensions of the left ventricle (LV) were evaluated, including LV internal dimensions at end diastole (LVIDd) and the LV posterior wall thickness in diastole (LVPWd). LV fractional shortening (FS) was calculated using the built-in function of the ultrasonographic scanner, and ejection fraction (EF) was calculated using the Simpson method. In addition, in the standard right parasternal short-axis view, the left atrium diameter (LAsax) and aortic diameter (Ao) in diastole were measured to obtain the ratio between LAsax and Ao (LA:Ao).
Subsequently, the cats were placed in left lateral recumbency. Using the standard left apical parasternal long-axis view, pulsed-wave Doppler was used to measure the transmitral flow. The peak velocity of early diastolic transmitral flow (E) and the peak velocity of late diastolic transmitral flow (A) were obtained to calculate the ratio between E and A (E/A). Tissue Doppler imaging was also utilised to measure the velocity of mitral annular motion, including mitral annular motion during early diastole (Ea) and late diastole (Aa). Ratios between E/Ea and Ea/Aa were subsequently calculated.
Statistical analysis
The data were analysed using SPSS Statistics (version 29; IBM) and data visualisation was performed using GraphPad Prism version 10.1 (Graphpad). The normality of the data was assessed using the Shapiro–Wilk test. For continuous variables that followed or approximately followed a normal distribution, descriptive statistics were presented as mean ± SD. A two-way repeated measures ANOVA was utilised for paired measurements. After the ANOVA, sphericity was tested; in cases where the assumptions of sphericity were violated, the Greenhouse–Geisser correction was applied. The main effects of time and dose, as well as their interactions, were separately analysed. Bonferroni correction was employed for multiple comparisons to analyse the simple effects of time and dose.
For continuous variables and qualitative data (sedation score, analgesia score, muscle relaxation score) that did not adhere to a normal distribution, descriptions were provided using the median (lower quartile–upper quartile). Generalised estimating equations were used to assess the main effects and interactions of time and dose, utilising an exchangeable working correlation matrix. In addition, the Kruskal–Wallis test (with pairwise comparisons using the all-pairwise method), the Friedman test and the Wilcoxon signed-rank test were conducted to analyse the simple effects of time and dose.
For echocardiographic variables, differences between various treatment regimens at T0 and T90 were compared. Inter-group comparisons of parametric echocardiographic data, which followed a normal distribution, were analysed using a one-way ANOVA, with pairwise comparisons conducted using the least significant difference least significant difference method. Non-parametric data were analysed using the Kruskal–Wallis test. For intra-group comparisons within the same treatment regimen, differences between T0 and T90 were assessed using the paired samples
Results
Among the eight cats included in this study, the ranges of oral trazodone doses were 11.29–14.04 mg/kg for the TRA-50 mg group, 16.30–21.19 mg/kg for the TRA-75 mg group and 21.60–27.62 mg/kg for the TRA-100 mg group.
Clinical effects
Changes in sedation scores over time, after administration of different doses of trazodone, are illustrated in Figure 1a. For the TRA-50 mg group, sedation scores significantly increased from T60–T240 (

Changes in (a) sedation score, (b) muscle relaxation score and (c) analgesia score at different time points after oral administration of a placebo or various doses of trazodone in eight cats. a = Significantly different from baseline (T0) (
Figure 1b illustrates the variations in muscle relaxation scores over time after the oral administration of different doses of trazodone. Compared with T0, muscle relaxation scores in the TRA-50 mg group increased significantly at T30 (
There were no significant differences in analgesia scores between the different TRA treatment groups compared with T0 and placebo (
Physiological variables
Among the measured variables, only TRA-100 mg produced a significant effect, with SBP demonstrating a reduction of 22 ± 7 mmHg at T150 compared with placebo (

Changes in systolic blood pressure (a), pulse rate (b) and respiratory rate (c) t different time points after oral administration of a placebo or various doses of trazodone in eight cats. a = significantly different from baseline (T0) (
RR variations over time after oral administration of different trazodone doses are illustrated in Figure 2c. Specifically, RR in the TRA-50 mg group significantly decreased from T30–T240 relative to T0 (
Echocardiogram variables
Echocardiographic variables measured at baseline and after administration of a placebo or various doses of trazodone are presented in Table 2. Only LV-FS and E/Ea in the placebo group showed a significant increase compared with pre-administration measurements (
Echocardiographic variables of eight cats before and after administration of different doses of TRA or a placebo

Values significantly different from baseline (T0) (
*LVIDd = left ventricular internal dimensions at end diastole; LVPWd = left ventricular posterior wall thickness at end diastole; LV-EF = left ventricular ejection fraction; LV-FS = left ventricular fractional shortening; LA/Ao = ratio between left atrial and aortic diameter in diastole; E = peak velocity of early diastolic transmitral flow; A = peak velocity of late diastolic transmitral flow; Ea = peak mitral annular velocity during early filling; Aa = peak mitral annular velocity during late filling
Adverse effects
Among the eight cats, two exhibited marked constriction of the pupils to pinpoint size after administration of any dose of trazodone (Figure 3a). In addition, one cat displayed protrusion of the third eyelid at T90–T120 after oral administration of 75 mg and 100 mg of trazodone (Figure 3b). In the TRA-75 group, one cat demonstrated unprovoked aggression at T120–T150. Similarly, in the TRA-100 group, two cats exhibited sudden aggression at T60.
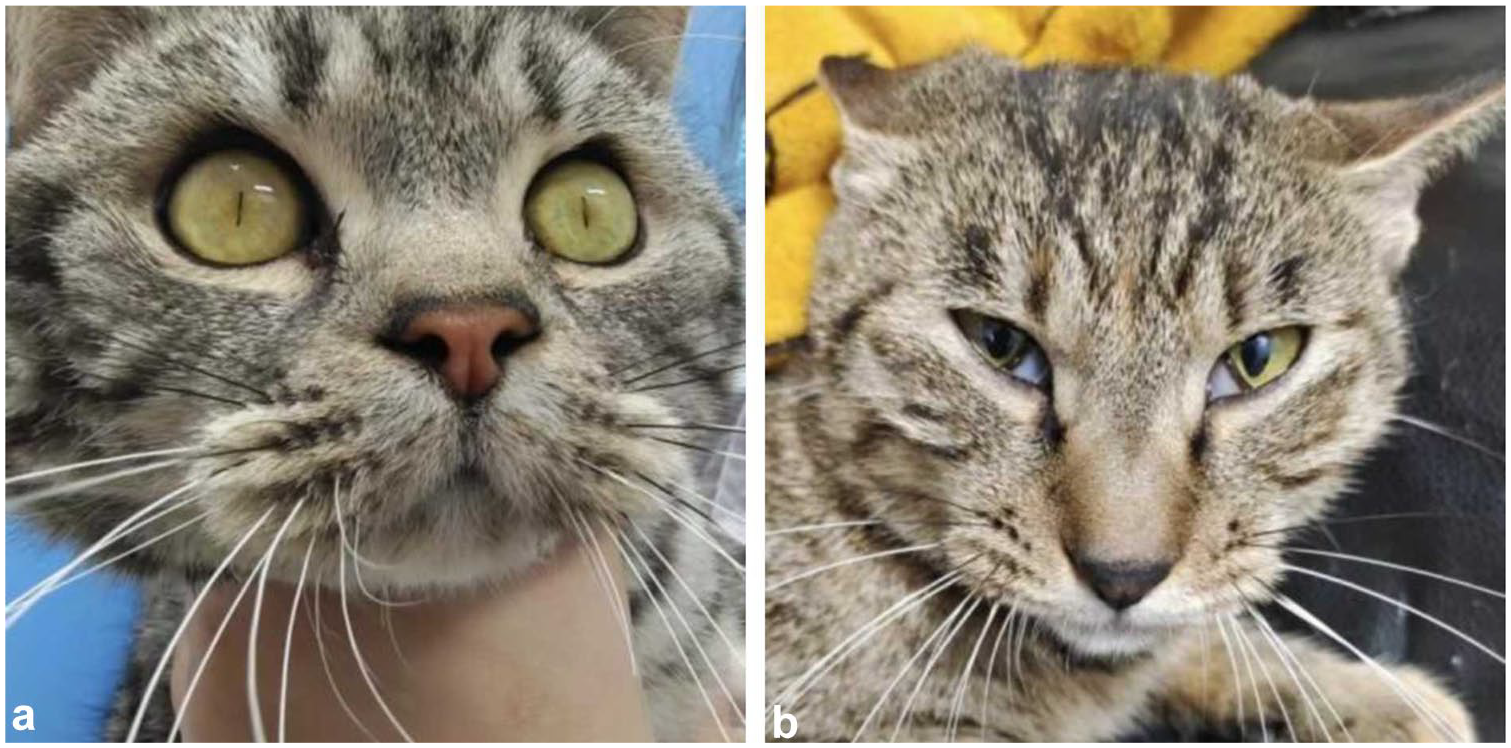
(a) Miosis and (b) protrusion of the third eyelid observed in cats after oral administration of trazodone
Discussion
This study presents the first comparison of the clinical effects of different doses of trazodone in cats. The findings indicate that oral administration of trazodone in healthy cats results in mild sedation, without causing significant changes in physiological or echocardiographic measurements. However, trazodone does not appear to provide muscle relaxation or analgesic effects.
Trazodone has a mild sedative effect in cats, which does not show a dose-response relationship. There were no significant differences in sedation scores among doses at 90–120 mins after oral administration of trazodone. This study observed potential differences in the sedative level of cats compared with other studies, which may be influenced by various factors. For example, Orlando et al 16 assessed sedation using accelerometers and video observations, with behavioural responses during physical examinations scored 90 mins after trazodone administration. 16 Accelerometer data showed that a 100 mg dose of trazodone significantly reduced activity and video observation scores compared with placebo; however, there were no significant differences between groups in behavioural responses during physical examination. This suggests that reduced activity and sedation scores in their study may not correlate with greater compliance during veterinary examinations. 16 Unlike Orlando et al’s, study our assessment involved more human interaction during sedation assessment, which could have influenced the cats’ responses. Our approach may better reflect cats’ responses to external stimuli in a clinical setting, though further validation is needed. In addition, the previous study revealed notable individual variation in activity levels among the cats; while there was a general reduction in activity across the studied cats after trazodone administration, some cats demonstrated increased activity. 16
Another study found that trazodone improves cats’ transportation anxiety and increases their ease of handling during veterinary examinations. 17 However, unlike our approach, that study primarily relied on owner observations during the entire transportation process to assess the cats’ stress scores. 17 A 2021 review focusing on pre-appointment medications highlights the inconsistent terminology in current veterinary research on trazodone, noting that studies do not clearly distinguish between its sedative and anxiolytic effects, leading to ongoing uncertainty over recommendations for trazodone use. 1 By drawing on studies of trazodone in dogs that have distinguished between sedative and anxiolytic effects, 20 future research can refine assessment criteria to better differentiate trazodone’s effects in cats.
Minimal changes were observed over time in SBP and PR in the trazodone treatment groups. At T150, the SBP in the TRA-100 group significantly decreased compared with placebo, but it remained within the reference interval for cats. 21 However, this effect was not observed with doses of 50 mg and 75 mg. This finding aligns with previous reports,16,17 but a study by Fries et al 15 showed a significant decrease in SBP after the administration of 50 mg of trazodone in cats. Thus, further investigation is needed to confirm changes in SBP after oral administration of trazodone in cats. It is worth noting that the current study did not directly measure heart rate, but instead indirectly assessed PR through Doppler measurements; in most cases, there were no significant differences between the two in numerical values. 22 Specifically, our data showed that during the intervals T0–T30 and T90–T120, although the increases were not statistically significant, a slight elevation in PR was noted across trazodone treatment groups. These time intervals corresponded to the first and second echocardiographic examinations, respectively, which may have induced a state of stress in cats, potentially resulting in the observed elevation in PR. 2 However, all the SBP and PR data were considered to be within the normal physiological interval for cats.15,21,23
Compared with the typical resting RR of normal cats reported in other studies, 24 both the placebo group and each treatment group at T0 displayed elevated RR. After the administration of trazodone, the RR transitioned from rapid to more regular breathing, resembling the resting RR in healthy cats. 25 This may reflect environmental adaptation and a reduction in stress in cats after oral administration of trazodone. However, there were no significant differences in RR among the different trazodone dosing groups, suggesting minimal impact of dose on RR.
A pharmacokinetic study conducted in cats revealed that oral administration of trazodone suspension at a dose of 5 mg/kg resulted in peak plasma concentrations approximately 0.17–0.5 h after administration, with peak sedative effects observed around 2 h. 24 However, compared with the current study, there were differences in the formulation and dosage, which may result in pharmacokinetic discrepancies. In previous studies, cats were administered oral doses of 50 mg or higher (75 mg and 100 mg) of trazodone, with assessments conducted 90–120 mins after administration.15 –17 Therefore, in this study, echocardiographic examinations were performed at T0 and T90. There were no statistically significant differences in echocardiographic variables compared with placebo and T0 after trazodone administration. Significant differences in systolic and diastolic function, specifically in LV-FS and E/Ea, were observed only in the placebo group before and after administration, with these variables remaining within the normal physiological interval reported in other studies.26 –28 Variations in some measurements may be attributed to true differences, systematic measurement or recording error, imaging artifact in the near field and so on. 27 This study’s findings are consistent with those of Fries et al, 15 who reported that 50 mg of trazodone does not significantly alter cardiac variables in a clinically relevant manner in cats.
In this study, after oral administration of trazodone, marked pinpoint pupil constriction (2/8 cats) and protrusion of the third eyelid (1/8 cats) were observed. Notably, there were no adverse effects reported, such as vomiting or salivation. The occurrence of pinpoint pupil constriction can be attributed to the α-adrenergic blocking properties of trazodone. 29 A separate study noted that protrusion of the third eyelid, in combination with other signs such as dilated pupils, ‘hunting’ invisible objects in the cages and growling without handler interaction after intravenous administration of 3 mg/kg trazodone, may indicate a transient excitatory response. 24 However, protrusion of the third eyelid alone is not a definitive indicator of excitation.
There are certain limitations to consider. First, the sample size in this study was constrained by the limited availability of experimental animals. Caution should be exercised in generalising results from studies with small sample sizes to the entire population. In addition, the scoring criteria for sedation, muscle relaxation and analgesia involved some level of subjectivity. The measurement of echocardiographic variables and the sedation assessment were conducted in the same experimental process, and may have influenced each other to some extent. Moreover, this study was limited to healthy adult cats, excluding juvenile or geriatric cats. Given that trazodone is primarily metabolised by the liver, 24 caution is advised when considering its use in cats with liver disease. Lastly, the cats used in this study were experimental cats, which may have some adaptation to medical procedures, potentially differing in response from household cats in clinical settings.
Conclusions
This study demonstrates that oral administration of 50 mg, 75 mg or 100 mg of trazodone induces mild sedation in cats, but with no improvement in muscle relaxation or analgesic effects. Notably, administration of 100 mg trazodone was associated with a mild decrease in SBP, which remained within the reference interval. Trazodone can be considered in clinical settings as a mild sedative agent in cats when no significant muscle relaxation or analgesia is required.
Supplemental Material
Supplementary material 1
Sedation score.
Footnotes
Supplementary materials
The following file is available as supplementary material:
Supplementary material 1: Sedation score.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by Meng Li’s startup fund of Nanjing Agricultural University (804127).
Ethical approval
The work described in this manuscript involved the use of experimental animals and the study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
