Abstract
Objectives
The aim of this study was to evaluate the efficacy of a single dose of oral pregabalin (PGB) for sedation and its impact on physiological and echocardiographic variables in healthy cats.
Methods
This study was a randomised, blinded, crossover trial. Eight cats were randomly assigned to receive PGB or placebo, with a 1-week washout period between each administration. Cats in the treatment group received oral PGB at varying doses (low dose: 2.5 mg/kg, medium dose: 5 mg/kg, high dose: 10 mg/kg). Systolic blood pressure (SBP), pulse rate (PR), respiratory rate (RR) and sedation score were measured at intervals of 30 mins after administration. Echocardiography was performed 120 mins after administration.
Results
Oral administration of PGB 2.5 mg/kg and 5 mg/kg significantly increased sedation scores starting at 150 mins, while 10 mg/kg PGB showed a significant increase in sedation scores starting at 120 mins compared with placebo. PGB 5 mg/kg and 10 mg/kg resulted in a significant reduction in SBP compared with placebo, with minimal impact on PR and RR. In addition, PGB 10 mg/kg resulted in significant changes in the peak velocity of late diastolic transmitral flow (A) and the ratio of peak velocity of early diastolic transmitral flow and A; however, these changes were of marginal clinical significance.
Conclusions and relevance
A single dose of oral PGB could cause mild to moderate sedation. Hypotension was more prevalent in the PGB 5 mg/kg and 10 mg/kg groups among the majority of cats, but it was less frequently observed in the PGB 2.5 mg/kg group.
Introduction
In veterinary practice, veterinary visits are often considered a stressful experience for cats.1,2 Stress in cats can trigger an increase in sympathetic excitability, resulting in elevated systolic blood pressure (SBP), heart rate (HR) and respiratory rate (RR).3,4 An elevated HR has been associated with a decrease in cardiac chamber size and can influence the assessment of left ventricular systolic function.5 –7 Some researchers have proposed preappointment medications to mitigate cats’ fear and anxiety and improve their visit experience.8,9 Commonly used preappointment medications include gabapentin (GBP), 4 trazodone 10 and butorphanol, 11 all of which have documented sedative effects on cats, when used in accordance with established recommended dosage guidelines. Among these medications, GBP, a gamma-aminobutyricacid (GABA) analogue, has demonstrated anti-anxiety effects in cats, with no significant impact on haemodynamics or echocardiographic variables. 5 Therefore GBP has been commonly used as an anti-anxiety medication for cats before veterinary visits. 4
Pregabalin (PGB) and GBP are both structural analogues of GABA and possess the capability to bind with the alpha 2-delta (α2-δ) subunit of voltage-gated calcium channels. This modulation leads to a reduction in the release of neurotransmitters, such as glutamate, norepinephrine and substance P, contributing to the anxiolytic and analgesic effects. 12 Compared with GBP, PGB shows a higher affinity for the α2-δ protein subunit of voltage-gated calcium channels.13,14 Studies have demonstrated that PGB is well tolerated in cats at doses of 4 mg/kg and 7.5 mg/kg, resulting in mild to moderate sedation.13,15 Hence, PGB stands out as a promising candidate for preappointment medication in cats. While PGB may facilitate advanced diagnostics such as echocardiography, additional research is needed to better understand its dose-dependent effects on sedation, and its potential effects on physiological and echocardiographic variables.
The primary objective of this study was to evaluate the sedative effects of oral PGB, and its impact on physiological and echocardiographic variables in healthy cats. We hypothesised that (1) oral PGB has a dose-dependent sedative effect in healthy cats, (2) oral PGB does not significantly alter physiological variables and (3) oral PGB does not significantly affect echocardiographic variables.
Materials and methods
Animals
The study involved a group of eight purpose-bred cats. Among these eight cats, three were castrated males and five were spayed females aged 25–49 months, with a mean ± SD weight of 3.46 ± 0.41 kg. The cats were housed in a comfortable environment, ensuring appropriate conditions, including a controlled temperature of 28°C, access to ample food, sufficient water and daily sanitation. The cats were considered healthy based on physical examination, complete blood count, serum biochemical analysis and echocardiographic examination. Food was withheld from the cats for 12 h before the experiments, while water was provided ad libitum. The study was approved by the Institutional Animal Care and Use Committee of Nanjing Agricultural University (number 20221212235).
Study design and treatments
The study was conducted using a randomised, blinded, crossover trial design. A randomisation scheme was obtained using the Research Randomizer (www.randomizer.org/). Before each experiment, PGB (Lyrica; Pfizer) capsules were prepared using an analytical balance (Balance XSR204; Mettler-Toledo) according to the administration protocol and the weight of the cats. Empty capsules were prepared for the control group. PGB was administered orally to each cat at low, medium or high doses of 2.5, 5 and 10 mg/kg, or the cat was given an empty capsule; 2 ml of water were subsequently administered to each cat using a syringe. The investigators were blinded to the capsule contents. A 1-week washout period was implemented between each administration. 16
The baseline sedation score (SS),17 –19 SBP, pulse rate (PR) and RR were measured 10 mins after the cat had become accustomed to the experimental environment. After completion of these measurements, the treatment was administered, and the time of administration (T0) was recorded. Sedation score, SBP, PR and RR were measured at intervals of 30 mins after administration, up to 240 mins, with measurement times recorded as T30, T60, T90, T120, T150, T180, T210 and T240. Throughout the experiment, physiological variables and sedation evaluation were conducted and assessed by an investigator who was blinded to the treatment protocol. Echocardiographic measurements were conducted at T120.
Sedation score
The sedation score was derived from previous published studies17 –19 and the specific methodology is detailed in Table 1. The extent of sedation was evaluated based on posture, resistance to positioning in lateral recumbency, resistance to extension of the left hindlimb and left knee, responsiveness to sound stimuli and reactions to air blowing. The sequence of the procedures assessing sedation was consistent and uniform across all experiments.
The 13-point sedation score table

Physiological variables
Non-invasive SBP was measured using an ultrasonic Doppler flow detector (Model 811B; Parks Medical Electronics) following a previously described method. 20 The Doppler crystal was placed over the coccygeal artery, and a cuff with a width of 40% of the tail circumference was utilised. 21 In this study, hypotension was defined as an SBP <110 mmHg. The threshold is based on the established median and interquartile range of SBP for healthy cats, which has been reported as 120.6 mmHg (range 110.4–132.4). 22 The cat’s PR was determined using the Doppler device. Five consecutive measurements of SBP and PR were recorded, excluding the highest and lowest values. Simultaneously, the RR was measured by observing the cats’ thoracic movements for 1 min.
Echocardiogram
A pilot study was performed to establish the timing range for PGB’s peak sedative effect. In this preliminary study, six cats were each given different doses (2.5, 5, 7.5 and 10 mg/kg) of PGB or placebo, following the same procedures as in the full-scale experiment. Based on the findings from this pilot study, an echocardiogram was performed at T120 after the administration of PGB or placebo in the main study. All echocardiography examinations were performed by a single researcher using an ultrasonographic scanner (X5Vet Ultrasound System; Esaote). The thoraxes of all cats were shaved 3 h before the examination. Each cat was first positioned in right lateral recumbency, followed by left lateral recumbency, on a specifically designed table for echocardiographic examination with minimal restraint needed. Each measurement was conducted three times and the resulting average values were used for the statistical analysis.
In the standard right parasternal long-axis and short-axis views, and left apical parasternal long-axis view, the size and function of the left ventricle (LV) were evaluated. The measured variables included interventricular septal thickness at end diastole and end systole, LV internal dimensions at end diastole and end systole, and LV posterior wall thickness at end diastole and end systole. The LV fractional shortening was calculated, and the ejection fraction was calculated using the Simpson method.
In the standard right parasternal short-axis view, the size of the left atrium (LA) was evaluated. The measured variables included LA diameter (LAsax) and aortic diameter (Ao) in diastole. The ratio between LAsax and Ao (LA:Ao) was calculated.
In the standard left apical parasternal long-axis view, the transmitral and aortic flow velocities were recorded using pulsed-wave Doppler. Pulmonary artery flow velocity was recorded from a right parasternal short-axis view. The measured variables included peak velocity of early diastolic transmitral flow (E), peak velocity of late diastolic transmitral flow (A), peak velocity of left ventricular outflow tract and peak velocity of right ventricular outflow tract. The ratio between peak E and peak A (E:A) was then calculated.
In the standard left apical parasternal long-axis view, the velocity of lateral mitral annular motion was recorded using tissue Doppler imaging. The measured variables included lateral mitral annular motion at early diastole (Ea lat) and late diastole (Aa lat). The ratio between E:Ea lat and Ea:Aa lat was calculated.
Statistical analysis
The statistical analysis was performed using Excel 2016 and SPSS Statistics 26.0. GraphPad Prism 9.2.0 was used for visualisation. The Shapiro–Wilk test was employed to assess the normality of the variables. Data following a normal distribution are presented as mean ± SD. A two-way repeated measures ANOVA was employed to examine both the main effects and interaction effects of treatment and time. Pairwise comparisons were conducted using Bonferroni adjustment. For non-normally distributed data and qualitative data, particularly sedation scores, values are presented as median (range). Generalised estimating equations were applied to analyse the main effects and interaction effects of treatment and time, and the working correlation matrix was selected as an exchangeable correlation structure. The Kruskal–Wallis test, Friedman test and signed-rank test were employed to analyse the effects of treatment and time. Pairwise comparisons were specifically conducted for placebo and T0. For the analysis of differences in echocardiographic variables among treatments, normally distributed data were subjected to one-way ANOVA, while non-normally distributed data were analysed using the Kruskal–Wallis test. The significance level was set at P <0.05.
Results
Sedation score
Sedation scores at different time points for each group are presented in Figure 1. PGB-2.5 exhibited a significant increase in sedation score from T120 to T240 compared with T0 (P <0.05), and at T150, it was significantly higher than the placebo (P <0.05). Both PGB-5 and PGB-10 demonstrated a significant elevation in sedation score from T60 to T240 compared with T0 (P <0.05). Specifically, in comparison with the placebo, PGB-5 showed a significant increase in sedation score from T150 to T240 (P <0.05), while PGB-10 exhibited a significant elevation in sedation score from T120 to T240 (P <0.05).
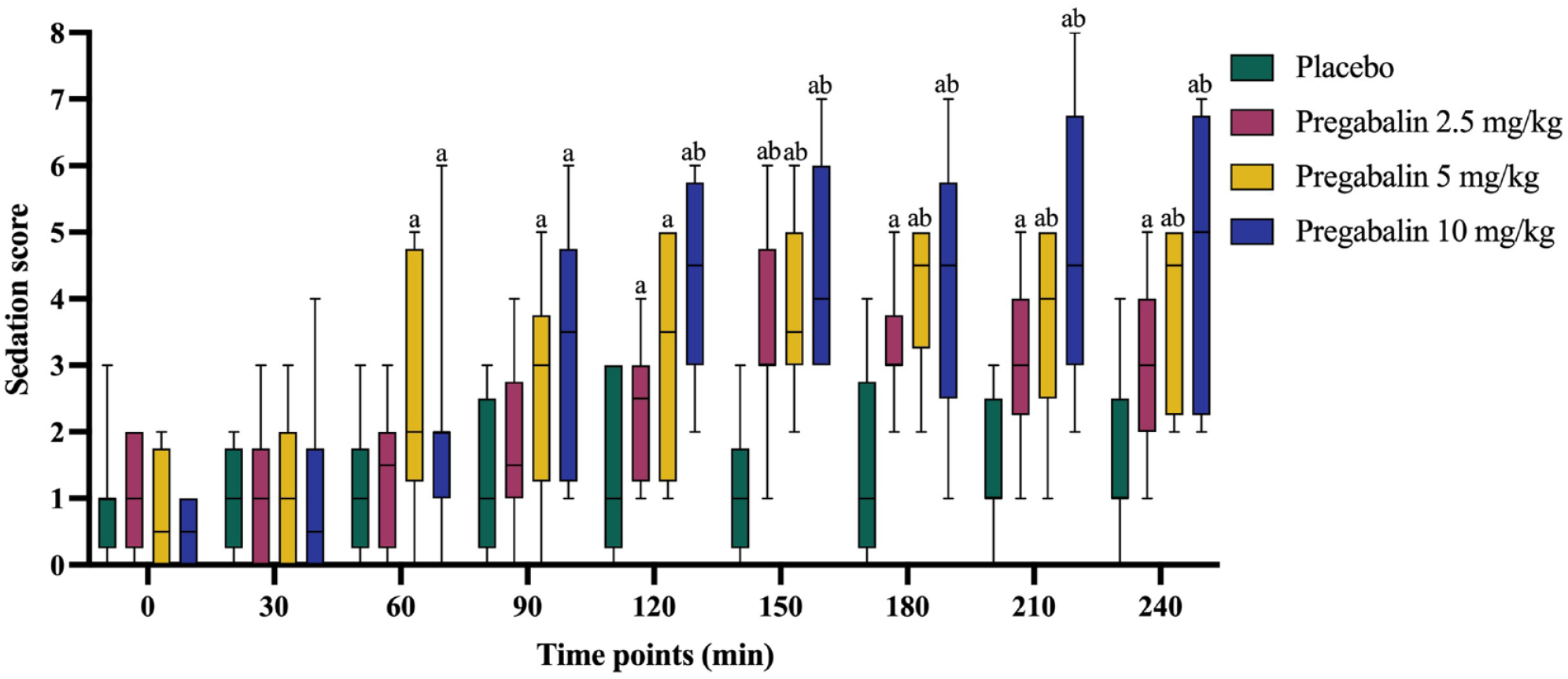
Box-and-whisker plots of sedation score at various time points after oral administration of placebo or different pregabalin doses in eight cats. aSignificantly different from baseline (T0) (P <0.05). bSignificantly different from placebo at the same time point (P <0.05)
Physiological variables
The changes in SBP for each group at different time points are illustrated in Figure 2. At T210, PGB-2.5 significantly reduced SBP compared with the placebo (P <0.05). PGB-5 exhibited significantly lower SBP at T180–T240 than at T0 and significantly lower than placebo at T180–T210 (P <0.05). PGB-10 demonstrated a significant reduction in SBP at T90–T240 compared with T0 and significantly lower than placebo at T120–T210 (P <0.05). The incidence of hypotension varied with the dose of PGB. At a dose of 2.5 mg/kg, hypotension was observed in 3/8 cats. When the dose was increased to 5 mg/kg, 6/8 cats experienced hypotension. The highest dose tested, 10 mg/kg, resulted in hypotension in 7/8 cats, with three of these cats showing particularly low SBP values, under 100 mmHg.

Box-and-whisker plots of systolic blood pressure at various time points after oral administration of placebo or different pregabalin doses in eight cats. aSignificantly different from baseline (T0) (P <0.05). bSignificantly different from placebo at the same time point (P <0.05)
The PR and RR results are shown in Table 2. No significant differences in PR were found among any of the treatment groups when compared to placebo at any time point examined. PGB-2.5 had a significantly lower PR at T120 and T240 compared with T0 (P <0.05). At T120 and T180–T240, PGB-5 exhibited a significant reduction in PR compared with T0 (P <0.05). Similarly, PGB-10 demonstrated a significantly lower PR at T90–T240 compared with T0 (P <0.05). In terms of RR, only PGB-5 showed a significant decrease compared with T0 from T90–T240 (P <0.05).
Results of pulse rate (PR) and respiratory rate (RR) in eight cats across placebo and pregabalin (PGB) treatment groups

Data are presented as mean ± SD
Significantly different from baseline (T0) (P <0.05)
Echocardiographic variables
The echocardiographic variables recorded for the different groups are shown in Table 3. Among all variables tested, only peak A and E:A in the 10 mg/kg PGB group showed significant differences from the placebo group. Compared with the placebo group, the 10 mg/kg group exhibited a significant decrease of 0.2 in peak A and a significant increase of 0.4 in E:A (P <0.05). No significant differences in echocardiographic variables were observed between the 2.5 mg/kg and 5 mg/kg groups when compared with other groups at any time points.
Echocardiographic variables in eight cats across placebo and pregabalin (PGB) treatment groups

Data are presented as mean ± SD or as median (range)
Significantly different from placebo (P <0.05)
Ao = aortic diameter; AV Vmax = peak aortic flow velocity; E:A = ratio between peak E to peak A; E:Ea lat = ratio between peak E to peak Ea lat; Ea:Aa lat = ratio between peak Ea lat to peak Aa lat; IVRT = isovolumetric relaxation time; IVSd = interventricular septal thickness at end diastole; IVSs = interventricular septal thickness at end systole; LA = left atrium diameter; LA:Ao = ratio between left atrial and aortic diameters in diastole; LV-EF = left ventricular ejection fraction; LV-FS = left ventricular fractional shortening; LVIDd = left ventricular internal dimensions at end diastole; LVIDs = left ventricular internal dimensions at end systole; LVPWd = left ventricular posterior wall thickness at end diastole; LVPWs = left ventricular posterior wall thickness at end systole; MV Dec T = deceleration time of early diastolic transmitral flow; Peak A = peak velocity of late diastolic transmitral flow; Peak Aa lat = peak late diastolic velocity of the lateral mitral annulus; Peak E = peak velocity of early diastolic transmitral flow; Peak Ea lat = peak early diastolic velocity of the lateral mitral annulus; PV AT = acceleration time for flow across the pulmonic valve; PV Vmax = peak pulmonic flow velocity
Discussion
To the best of our knowledge, this study represents a pioneering investigation into the impact of different oral PGB doses on sedation, as well as the assessment of physiological and echocardiographic variables in healthy cats. The findings of the study indicate that oral administration of PGB in normal cats yields a significant and clinically effective sedative effect, albeit with a potential risk of hypotension.
In our study, the administration of a single dose of PGB 2.5 mg/kg showed a significant difference in sedation score compared with the placebo only at T150. Similarly, when administering PGB at 5 mg/kg, significant differences in sedation score, relative to placebo, were observed between T150 and T240. This observed pattern aligns closely with the mean expected peak plasma concentration time frame for PGB (2.8 ± 1.2 h). 13 However, the 10 mg/kg group exhibited a significant sedative effect with an earlier onset at T120 and a longer duration compared with the 2.5 mg/kg group, persisting until the end of the T240 observation period. This observed variance might be attributed to the linear pharmacokinetics of PGB in normal cats, characterised by low interindividual variability, which enables a reliable dose-dependent effect, as outlined in a prior study. 15 In addition, the elimination half-life of PGB in normal cats is approximately 10–12 h.13,15 Additional research could be performed to further investigate the impact of dosing intervals on the sedative effects to optimise clinical outcomes.
The dose range of PGB chosen in this study was determined based on recent studies.13,15,16,23 In a study aimed at alleviating transportation anxiety in normal cats, oral administration of PGB at doses of 5 mg/kg and 10 mg/kg yielded reduction in anxiety and fear levels at T90, with only a few adverse events. 23 A separate pharmacokinetic study in normal cats revealed that PGB at 4 mg/kg had a moderate sedative effect, resulting in mild obtundation while maintaining interactive behaviour. 13 Yet another study reported no clinical signs or sedative effects after single oral doses of PGB at 2.5 mg/kg and 5 mg/kg. 15 The varying sedative effects of PGB within the dose range of 4–5 mg/kg, and the absence of information regarding its effects at lower doses such as 2.5 mg/kg, necessitate further investigation. Thus, our study incorporated a range of doses to explore potential dose-dependent patterns in sedative effect.
Throughout the experiment, all investigators dressed casually to mitigate the white-coat effect on cat physiological variables.24,25 PGB at doses of 5 and 10 mg/kg resulted in significant reductions in SBP compared with T0 at certain time points during the experiment. The incidence of hypotension was notably higher in cats receiving 5 mg/kg and 10 mg/kg doses of PGB, affecting the majority of cats in these groups. In contrast, hypotension was less frequently observed in cats treated with the 2.5 mg/kg dose of PGB. Between T120 and T150, slight increases in PR were observed in all groups, but were not statistically significant. These minor fluctuations may be attributed to operational factors during the echocardiography measurements conducted from T120 to T150. The results for PR and RR indicate a significant time effect, while the treatment effect was not significant. Over the course of this experiment, RR showed an overall decreasing trend, likely reflecting cats adapting to the experimental environment. Environmental changes in a clinical setting can impact a cat’s physiological variables. 26 Despite allowing time for the cats to adapt to the environment before the experiments, it may be difficult to achieve a totally relaxed state.
Our results indicate that oral sedative doses of PGB have few effects on echocardiographic measurements in normal cats. Significant changes were observed only at the 10 mg/kg dose of PGB, with a significant decrease of peak A and significant increase of the E:A. Peak E did not show significant changes at any dose, suggesting that the increased E:A was primarily due to the decreased peak A. The changes in peak A suggest a reduction in the maximum blood flow velocity during atrial contraction into the left ventricle; however, it remains within the normal range for cats. 17 Furthermore, when taking the changes of other variables into consideration, the reduction in peak A alone is not clinically significant.
Isovolumetric relaxation time (IVRT) can also be used to assess ventricular diastolic function. PGB demonstrated a dose-dependent effect on the prolongation of IVRT in this study. A slower HR could lead to a prolonged IVRT, 17 although several studies suggest that HR and IVRT may not be clinically relevant.13,15 In addition, mitral annular motion was assessed using pulsed-wave tissue Doppler, and no abnormalities were observed in left ventricular diastolic function. The echocardiographic variables representing left ventricular systolic function did not change significantly across all tested PGB doses. Furthermore, PGB did not exhibit a significant impact on the thickness of the interventricular septum and left ventricular free wall. Hence, the findings of this study indicate that the administered dose of PGB did not lead to clinically significant alterations in echocardiographic variables. Therefore, PGB is considered appropriate for use in healthy cats undergoing echocardiographic examinations. However, given the limited research on the peak sedative effect of PGB, we opted, following a pilot study, to perform echocardiographic examinations 120 mins after oral administration of treatment or placebo. Based on the sedation score results, it was observed that 10 mg/kg of PGB exhibited a significant increase in sedation scores compared with the placebo at T120, while a significant difference in sedative effect for PGB at 2.5 mg/kg and 5 mg/kg was identified at T150. This indicates the potential need to refine the timing of echocardiographic examinations for various PGB doses in future research.
Other sedative drugs can lead to considerable haemodynamic changes that impede the accurate interpretation of physiological and echocardiographic variables. Oral administration of 50 mg of trazodone does not affect echocardiographic variables but significantly reduces blood pressure (25 ± 16 mmHg) in cats. 16 Reports of sedation protocols using dexmedetomidine-butorphanol or dexmedetomidine-buprenorphine in cats show significant effects on physiological and contractile function variables, such as HR and fractional shortening.11,27 The sedative effects of PGB offer a potential option for clinical use; however, the impact on SBP cannot be ignored, warranting further investigation with a larger sample size.
Pharmacokinetically, GBP has a smaller distribution volume and a slower clearance rate than PGB, with a terminal half-life of approximately 3 h after the oral administration of 10 mg/kg in normal cats.24,28 –30 In contrast, PGB has an average systemic bioavailability of 94%, 31 and a mean half-life of 14.7 h after oral administration of a 5 mg/kg dose. 15 A preappointment medication tailored to an individual patient needs to be selected based on the drug’s characteristics and clinical requirements.
The present study has some limitations. The observation period after oral administration lasted only up to 240 mins. However, at T240, both the moderate and high doses of PGB still showed adequate sedative effects compared with the control group and baseline. The remaining duration of the sedative effect of high-dose PGB is uncertain, and further investigation should be carried out by appropriately extending the observation time. In addition, echocardiographic measurements were conducted only at T120 after PGB administration, lacking baseline values before PGB administration. This lack of baseline data may introduce discrepancies in the comparison between pre- and post administration results, which is an area that future research could improve upon. Furthermore, because of the limited sample size, the statistical power was reduced. It is important to highlight that both the experimental cats in this study, as well as those in the related studies, were healthy, without apparent illnesses. Consequently, these findings may not be applicable to cats with specific diseases.
Conclusions
The results of this study indicate that a single oral administration of PGB can induce sedation in cats without significantly affecting echocardiographic variables. Doses of 5 and 10 mg/kg PGB resulted in hypotension in the majority of cats. Meanwhile, a 2.5 mg/kg dose of PGB appears to induce sedation in healthy cats; however, caution should be exercised because of the potential negative impact on systolic blood pressure.
Footnotes
Acknowledgements
We thank Dr Xianqi Wang for the manuscript review and Dr Danchen Yang for statistical support.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by Meng Li’s startup fund from Nanjing Agricultural University (804127).
Ethical approval
The work described in this manuscript involved the use of experimental animals and the study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal (s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
