Abstract
Objectives
Total thyroxine (TT4) evaluation is the most commonly used first-line test for the diagnosis and monitoring of cats with hyperthyroidism. Vcheck T4 is a point-of-care immunoassay that measures TT4 using a Vcheck V200 analyser. This study aimed to evaluate the analytic performance of the Vcheck T4 assay in feline sera and the agreement in the classification of normal, high and low TT4 concentrations of Vcheck T4 with those measured by an enzyme immunoassay (EIA).
Methods
Assay precision, reproducibility and linearity were evaluated for the Vcheck T4. For method comparison, TT4 concentrations in 73 serum samples were analysed by both methods.
Results
Vcheck T4 demonstrated good precision, reproducibility (intra- and inter-assay coefficients of variation between 3% and 13.5%) and linearity in the diagnostic range of <0.5 and >8 μg/dl. The correlation coefficient was 0.87, Passing–Bablok regression showed a proportional, but not constant bias, Bland–Altman plots revealed a mean difference of +0.5 μg/dl. The overall inter-rater agreement (K) between TT4 EIA and Vcheck results was substantial (K = 0.69), with 82.2% of concordant results. As a diagnostic test for hyperthyroidism, Vcheck T4 showed a sensitivity and specificity of 88.9% and 84.4%, and a positive predictive value (PPV) and negative predictive value (NPV) of 44.5% and 98.2%, respectively.
Conclusions and relevance
Vcheck T4 assay provided precise and reproducible results in substantial agreement with the EIA. Given the high NPV, Vcheck T4 is useful in ruling out hyperthyroidism when screening cats, but, based on low PPV, samples with results with high TT4 need to be analysed by another reference method. Further analysis with haemolytic, icteric and lipaemic samples is needed to assess the test performance. Finally, the reference intervals provided by the manufacturer require verification, and TT4 intervals specific to this method must be established in future studies.
Introduction
Hyperthyroidism is a common clinical condition in adult and senior cats for which serum total thyroxine (TT4) determination is needed for diagnosis and follow-up. 1 TT4 is abnormally elevated in 90–95% of hyperthyroid cats on a single randomly obtained blood sample, but cats with early or mild hyperthyroidism often have values fluctuating from high-normal to high. A randomly obtained TT4 concentration is the most commonly used first-line screening test for feline hyperthyroidism, and cats with TT4 near decision thresholds require repeated tests or additional diagnostic procedures to determine thyroid status.2–5 Annual TT4 evaluation is strongly recommended by the American Animal Hospital Association (AAHA) 2023 Senior Care Guidelines for Dogs and Cats. 6 The Task Force of Experts in Feline Clinical Medicine recommends that veterinarians strongly consider TT4 testing in healthy mature adult cats. 7 In addition to diagnosing hyperthyroidism, measurement of feline TT4 levels can also help in the monitoring of cats undergoing treatment for thyroid conditions. 1
Several methodologies are used in the measurement of serum TT4, including radioimmunoassay (RIA) (which is considered the gold standard technique), chemiluminescent enzyme immunoassay (CEIA, such as Immulite) and enzyme immunoassay (EIA).4,8–12 Methods such as ELISA are also commercially available as point-of-care tests for in-house evaluation of feline TT4 on feline serum or plasma samples and are useful for in-clinic diagnosis and follow-up of hyperthyroid cats. 5
A new dry-slide point-of-care TT4 quantitative assay (Vcheck T4; Bionote) is currently available for in-clinic testing using a benchtop point-of-care analyser (Vcheck V200; Bionote), which is based on fluorescence immunoassay technology, providing TT4 test results within 20 mins. The objective of this study was to evaluate this new quantitative assay for precision, repeatability and linearity under dilution as a comparison to a previously validated EIA assay, by analysing samples with normal, high and low TT4 concentrations.
Materials and methods
Samples
All samples were leftover feline serum samples remaining after primary diagnostic testing of client-owned cats clinically evaluated by the authors (ES, RP and DP) at the Veterinary Teaching Hospital, Department of Veterinary Medicine and Animal Sciences (DIVAS) of the University of Milan, Italy. Blood samples were collected between January 2022 and February 2024; sera were stored at −20°C and allowed to thaw before analysis with the Vcheck V200.
According to the University of Milan regulations, formal approval was not required for sample use because the leftover feline serum used in this study was harvested from blood samples collected for diagnostic purposes or as part of health checks and with the informed consent of the owners (according to the Ethical Committee of the University of Milan decision 29 October 2012, renewed with protocol no. 02-2016).
Procedures
Feline TT4 concentrations were determined using the Vcheck T4 assay, a point-of-care test for the quantitative determination of TT4 in feline serum that uses the Vcheck V200 benchtop point-of-care analyser, and the results were compared to those of an EIA method validated for use in cats.8–11 Both analysers used for TT4 evaluation were cleaned, calibrated, maintained and operated in accordance with the manufacturer’s system operator’s manual.
For diagnostic purposes, all samples had serum TT4 concentration determined at the time of submission at our university hospital laboratory using the Tosoh ST AIA-PACK T4 assay designed for human patients (Tosoh) and run on the automated veterinary analyzer AIA 360 VET (Futurlab). This method utilises a competitive EIA method for serum TT4 determination and it has been previously validated for use in feline serum for TT4 evaluation (mean within- and between-run coefficients of variation [CVs] for feline T4 were ⩽6.5%). 8 The measurement range values for the TT4 with the EIA method provided by the manufacturer is 0.5–24 μg/dl. The previously established reference interval for the EIA method for the feline TT4 in euthyroid adult and senior cats is 0.8–4.7 μg/dl 13 and was verified and transferred to our university hospital laboratory according to Clinical Laboratory and Standards Institute guidelines.14,15 To determine the clinical utility of the Vcheck method, serum TT4 concentration from each cat was classified as normal (TT4 concentration range 0.8–4.7 µg/dl), high (TT4 >4.7 µg/dl) or low (TT4 <0.8 µg/dl) and sera from a mixed population of cats with these TT4 concentrations were analysed.
The Vcheck TT4 test (Bionote) is a fluorescent competitive immunoassay designed for the Vcheck V200 system capable of quantitatively measuring TT4. It consists of a dry-slide point-of-care in-house quantitative fluorescent TT4 immunoassay using monoclonal anti-TT4 antibodies based on a lateral flow assay using europium, a highly sensitive fluorescent material for antigen-antibody binding. When the sample is delivered to an assay diluent tube, the sample and a fixed amount of colloidal gold labelled anti-T4 monoclonal antibody are mixed in the assay diluent buffer using a disposable pipette containing a soluble tablet. T4 in the sample reacts with the gold-labelled antibody. Any unbound gold-labelled antibody in the mixture binds to T4-BSA coated on the membrane of the dry-slide assay. The density of the test line is inversely proportional to the T4 concentration of the sample. The slide is analysed using a benchtop point-of-care analyser (Vcheck V200; Bionote) (Figure 1) that reads the density of the test line and calculates the T4 concentration from the calibration curve data. The control line is a reference line that indicates the test has been performed correctly. The required sample volume is 50 μl of serum. A new test device is used each time a sample is analysed, and the analysis of one sample takes approximately 20 mins (including incubation of 10 mins). To perform the test, after insertion of the test device into the analyser and selecting ‘feline’ species, 50 μl aliquots of serum are added to the assay diluent tube, which is mixed with a disposable pipette until the tablet is completely dissolved (approximately eight times). The incubation countdown is then started on the analyser and an alarm sounds 15 s before the 10 min incubation phase ends. After incubation of the mixture at room temperature for 10 mins, 100 μl of incubated sample is added to the sample well of the test device, using the pipette supplied and start is pressed on the analyser. The reading results are provided after 10 mins. All the reagents need to be stored at 2–8°C and need to reach room temperature before testing. In this study, one of the authors (LB) performed all the Vcheck analyses. The measurement range of TT4 with Vcheck T4 is 0.5–8 μg/dl.

Benchtop point-of-care analyser (Vcheck V200; Bionote) for Vcheck T4 dry slide analysis (on the left of the analyser)
Precision and repeatability of the Vcheck T4 were assessed by evaluating intra- and inter-assay CV for serum samples with normal, high and low serum TT4 concentrations. For intra-assay precision, five replicates from one sample from each of the concentration groups were evaluated within the same run on the same day. For interassay precision, one thawed aliquot of one serum from each TT4 concentration group was evaluated for five separate runs over 10 days. Within- and between-run precisions were expressed as CV (%) following the calculation of the mean ± SD for each set of results.
The stability of TT4 in feline serum samples was investigated on three feline serum samples with normal (2.0 µg/dl), high (6.0 µg/dl) and low TT4 values (0.6 µg/dl), evaluated after 6 months of storage at −20°C.
Linearity was determined by diluting high (with TT4 concentration not above 8 µg/dl) and normal TT4 concentrations samples with a saline solution to obtain the 3:1, 1:2, 1:3 and 1:4 dilution samples prepared by mixing. The linearity was assessed by comparing the observed TT4 concentrations following dilution with the expected (calculated) TT4 concentrations.
Statistical analysis
Data were assessed for normality by the D’Agostino–Pearson test. Because data were not normally distributed, all analyses used non-parametric tests, and results were reported as the median and interquartile range (IQR, ie, 25th to 75th percentile) and represented graphically as scatter dot plots.
Samples with a Vcheck TT4 >8 μg/dl (upper limit of the dynamic range of Vcheck assay) and samples with a Vcheck and EIA values <0.5 µg/dl (lower limit of both assays) were excluded from the continuous analyses.
Statistical analyses included the calculation of the correlation coefficient (r) and the Passing–Bablok linear regression to measure constant and proportional bias. 16 Bland–Altman difference plot analysis was performed to determine the degree of agreement between the two analysers, by comparing the difference in measured serum TT4 concentration with the average concentration for the Vcheck T4 vs values of the EIA method. 17
An inter-rater agreement (kappa, K) was calculated to evaluate the agreement between EIA and Vcheck T4 in the classification of TT4 value in normal, high and low TT4 concentration groups, with the K value interpreted as: 0–0.20, slight; 0.21–0.40, fair; 0.41–0.60, moderate; 0.61–0.80, substantial; and 0.81–1.00, almost perfect agreement. 18
Finally, sensitivity, specificity, positive and negative predictive values (PPV and NPV) for diagnosing hyperthyroidism were calculated. For the calculation of predictive values, we used a pretest probability (eg, population disease prevalence) of 12.3% from a feline population from Southern Germany. 19
All statistical analyses were performed using commercially available software (MedCalc Statistical Software, version 22.021). P <0.05 was considered statistically significant.
Results
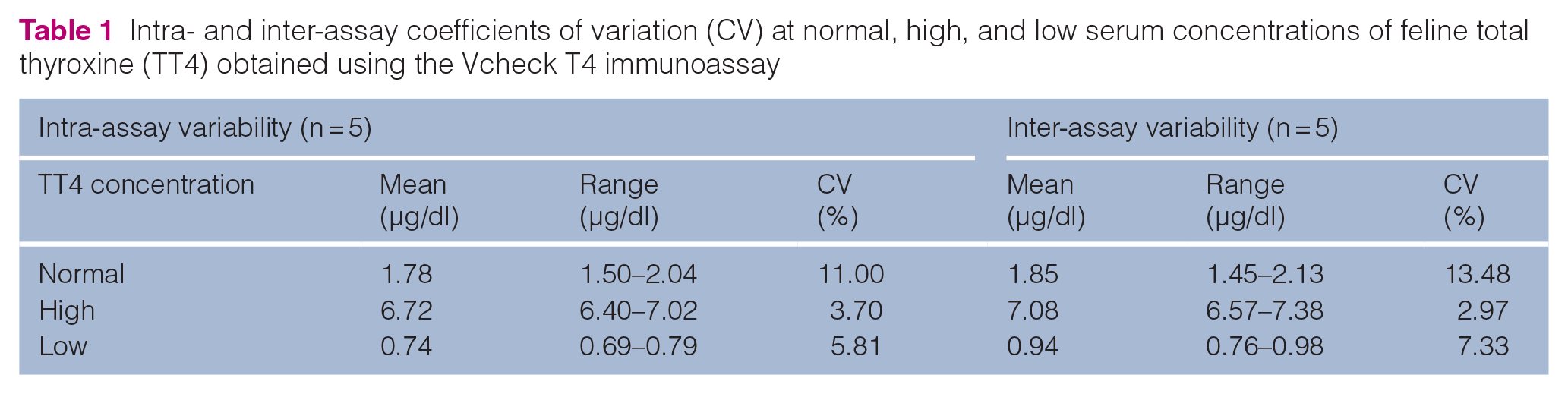
Resultant CVs for the intra-assay and inter-assay measurements for TT4 are reported in Table 1, while linearity results are reported in Table 2.
Intra- and inter-assay coefficients of variation (CV) at normal, high, and low serum concentrations of feline total thyroxine (TT4) obtained using the Vcheck T4 immunoassay
Effect of dilution with saline solution on feline total thyroxine (TT4) concentrations measured by Vcheck T4 (observed: value obtained analysing the sample by Vcheck; calculated: 75%, 50%, 33% and 25% of the initial high and normal TT4 values, based on the relative dilutions)

Storage of serum at −20°C for up to 6 months did not significantly alter the measured TT4 concentration and post-storage TT4 values fell in the same TT4 concentration group as the fresh serum sample, resulting in TT4 concentration in normal, high and low ranges of 2.3 µg/dl, 5.6 µg/dl and 0.4 µg/dl, respectively.
For the method comparison studies, a population of 73 cats was analysed (37 males and 36 females), all neutered except for one intact male, with a mean age of 13 ± 3.5 years (range 5–19), 56 domestic shorthair (DSH) cats, 11 domestic longhair cats and six purebred cats (two Ragdolls and one of each: Maine Coon, Norwegian Forest Cat, Siamese and Chartreux).
Based on EIA results, 40 samples had normal TT4 concentration, 22 samples had high TT4 concentration and 11 had low TT4 concentration. Vcheck T4 classified 37 samples with normal TT4 concentration, 28 samples with high TT4 concentration and eight with low TT4 concentration. Compared with the TT4 results obtained from the EIA method, the Vcheck T4 results agreed in 32/40 cats with normal values, 21/22 cats with high values and 7/11 cats with low values, for a total of 60/73 (82.2%) samples classified with the same EIA TT4 concentration group by Vcheck T4 (Figure 2). The overall inter-rater agreement (K) between EIA and Vcheck T4 in classifying the TT4 in the same concentration group was 0.69 (substantial agreement), with K in samples with normal, high and low TT4 concentration groups resulting, respectively, at 0.64, 0.75 and 0.69. Most discordant results in classifying TT4 in the same concentration groups were in samples with TT4 concentrations near the high or low decision threshold values (4.7 µg/dl and <0.8 µg/dl, respectively) (Figure 2).

Feline serum samples with normal (a) high (b) and low (c) total thyroxine (TT4) concentrations evaluated by enzyme immunoassay (EIA) and Vcheck T4 methods. Horizontal lines represent the interquartile range, red squares represent the median for each group and the shaded box represents the reference interval
Out of these 73 samples, Vcheck identified five samples with a TT4 concentration <0.5 μg/dl and 13 samples with TT4 concentrations >8 μg/dl, respectively the lowest and highest limit of TT4 detection of the Vcheck, while EIA identified one more sample than Vcheck as <0.5 μg/dl, for a total of 19 samples excluded from continuous analysis and of 54 samples available for continuous analysis.
The correlation coefficient was r = 0.87 (95% confidence interval [CI] = 0.79–0.92, P <0.0001). No significant deviation from linearity was detected by the Passing–Bablok regression (P = 0.40, Cusum test for linearity). The regression equation reveals intercept = 0.106 (95% CI = 0.124–0.423) and slope = 0.797 (95% CI = 0.694–0.906) (Figure 3). Therefore, a proportional, but not constant error was present since the slope is different from 1.

Passing–Bablok regression of feline TT4 Vcheck T4 vs the enzyme immunoassay (EIA) method in 54 feline serum samples. The regression equation for the Passing–Bablok regression was y = 0.106 + 0.797x. The solid line represents the data regression line, with dashed lines representing the confidence intervals
The Bland–Altman difference plot revealed a Vcheck T4 bias of +0.5 μg/dl in measuring TT4 and three results were identified as outliers, with 95% of measurements (51/54) within the mean ± 1.96 × SD difference between the methods (Figure 4).

Bland–Altman plot showing the difference between the measured feline serum total thyroxine (TT4) concentration by the Vcheck T4 and the enzyme immunoassay (EIA) method against the average TT4 measured by the Vcheck T4 and EIA in 54 feline serum samples. The solid line indicates the mean difference between the methods (+0.5 μg/dl) and the dashed lines represent limits of agreement, which are defined as the mean of the differences ±1.96 SD (lower limit, –1.40; upper limit, +2.50 μg/dl)
When evaluated as a diagnostic test for hyperthyroidism, the Vcheck T4 method showed a sensitivity and specificity of 88.9% and 84.4%, and a PPV and NPV of 44.5% and 98.2%, respectively (Table 3).
Diagnostic performances for Vcheck T4 immunoassay compared with the enzyme immunoassay method as a diagnostic test for hyperthyroidism

AUROC = area under the ROC curve; CI = confidence interval; NPV = negative predictive value; PPV = positive predictive value
Discussion
To our knowledge, this is the first study in which the performance of a fluorescent competitive immunoassay designed for the Vcheck system capable of quantitatively in-clinic measurement of TT4 has been evaluated and compared with the AIA 360 analyser using the EIA method for the measurement of feline TT4. The Vcheck system was evaluated in feline TT4 measurement only in a previous study that compared the results with the Immulite 2000 method showing a strong correlation (r = 0.81) and a good linearity between the two methods in evaluating TT4 concentrations in 44 feline sera. It was concluded that TT4 concentrations obtained by the Vcheck and Immulite 2000 methods were highly comparable across the physiological and pathological TT4 range of values. 20 However, that study evaluated only 10 feline samples with high TT4, three with low TT4 concentration and 21 samples with normal TT4 concentration.
In the present study, the intra-assay (range 3.7–11.0%) and inter-assay (range 2.9–13.4%) CVs indicate good precision of Vcheck T4 because they are lower than 17.2%, the maximum CV reported for dogs, 21 and lower than 20%, the Clinical Laboratory Improvement Amendments (CLIA) criteria for acceptable analytical performance for thyroxine in human patients. 22 In addition, the CVs for T4 using Vcheck were similar to those reported for the measurement of T4 in cats with the reference method used in this study: EIA (range intra-assay, 3.3–5.6%; inter-assay, 4.0–11.2%). 8
The AIA 360 analyser used in our study as the reference method uses the EIA method for serum TT4 determination and, compared with Vcheck, our study demonstrated that the Vcheck T4 assay provides valid diagnostic results in most samples with normal, high and low TT4 concentration. However, the Vcheck T4 concentrations do not agree perfectly with those measured by the EIA method; indeed, 13/73 (17.8%) samples gave discordant results and were categorised incorrectly by Vcheck T4 in normal, high and low TT4 concentration groups. In addition, method comparison with EIA showed that with Vcheck T4, a proportional systematic error was found, which means that the differences between the two methods were proportionally related to the level of measurements or that the magnitude of systematic error changes as the analyte concentrations change. Finally, Bland–Altman analysis revealed a Vcheck T4 bias of +0.5 μg/dl in measuring TT4 and identified three outliers between Vcheck T4 and EIA. The first sample (for EIA with a normal TT4 concentration of 2.15 μg/dl, with Vcheck with a high TT4 concentration of 5.11 μg/dl) was from a 7-year-old female spayed DSH cat which had severe head scratching for a suspected allergic disease, bilateral enlargement of thyroid lobes, weight loss with normal appetite and hypertrophic cardiomyopathy that we evaluated only for a second-opinion consultation with no possibility of follow-up and no additional tests for thyroid function were performed. The other two samples, classified respectively with normal and high TT4 concentration with EIA (TT4 3.67 and 5.16 μg/dl, respectively) and with high and normal TT4 concentration with Vcheck (TT4 6.45 and 2.26 μg/dl, respectively) were from senior DSH male hyperthyroid cats under long-term medical treatment with methimazole. It would have been interesting to assay these samples against a ‘gold standard’ diagnostic testing for hyperthyroidism as the RIA method; 4 however, due to the low sample volume, this could not be carried out.
Our results underline that Vcheck T4 cannot be used interchangeably with the EIA method in serum feline TT4 evaluation. In particular, based on the high NPV (98.2%, the probability that hyperthyroidism is not present when the test is negative), the usefulness of the Vcheck T4 test is in screening cats and ruling the disease out in adult and older cats, while, based on its low PPV (the probability that hyperthyroidism is present when the test is positive) of 44.5%, samples identified by Vcheck T4 as having high TT4 concentration need to be analysed and the result confirmed by another standard test. This underlines the utility of Vcheck in the initial screening of adult and old cats for the absence of hyperthyroidism because it is very improbable that a cat with normal TT4 concentration is hyperthyroid. In addition to its low PPV, another important limitation of this method is that it cannot differentiate between various degrees of elevated TT4 because the upper limit of the assay is fairly low (8 μg/dl). The endpoint value of high TT4 is useful to categorise hyperthyroid cats into severity groups (mild, moderate or severe hyperthyroidism) 23 and the severity of hyperthyroidism based on TT4 concentration is one of the factors influencing the initial methimazole dosage and the time needed to reduce circulating thyroid hormone concentrations to within normal range. 24 For this reason, values >8 μg/dl obtained with Vcheck need to be evaluated by another reference method.
There are several limitations to this study. First, we compared the results from Vcheck T4 with an EIA method previously validated for use in cats, but not with a ‘gold standard’ diagnostic testing for hyperthyroidism (the RIA method), which provides results that most accurately classify the cats’ thyroid status. 4 However, using a non-gold standard test can still provide valuable information about the performance of a new test. The EIA method, although not considered a ‘reference standard’, is used by most reference veterinary laboratories, and has been previously validated for feline TT4 evaluation.8–10 This test is routinely used at our teaching hospital, with samples for comparison testing being easily available, without the need to ship the samples to an external laboratory. Therefore, the evaluated agreement with the EIA method, even compared with a non-gold standard test, can provide insights into new Vcheck T4 test performance. Another important limitation of our study is that new reference intervals were not derived for Vcheck T4. However, conducting a validation and methods comparison study before establishing a reference interval is still useful, primarily to ensure that the new method generates accurate and consistent results. That said, feline TT4 reference intervals specific to this method must be established in future studies for the population in which the test is to be performed and the limits provided by the manufacturer require verification, particularly because they are based on a comparative study with Immulite technique 20 and reference intervals from other methods cannot be used interchangeably with those generated by a new method. 8 Finally, we did not investigate the effect of heamolysis, lipaemia or icterus to assess the Vcheck T4 performance across a range of diseases that might cause interference with the results.
Conclusions
The Vcheck T4 assay provided precise and reproducible TT4 concentration results in substantial agreement with EIA results, and it was able to correctly classify most samples with normal, high and low TT4 concentrations. This test is useful for screening adult and old cats and ruling out the presence of hyperthyroidism, but samples with high TT4 concentrations using this method need to be analysed and the result confirmed by another reference method. Further analysis with haemolytic, icteric and lipaemic samples is needed to broadly assess the test performance across a range of substances and diseases that could cause interference. The reference limits provided by the manufacturer require verification and feline TT4 reference intervals specific to this method must be established in future studies for the population where the test is to be performed.
Footnotes
Conflict of interest
Bionote played no role in the study design, in the collection, analysis and interpretation of data, in writing this article, nor in the decision to submit the manuscript for publication. None of the authors have any other financial or personal relationships that could inappropriately influence or bias the content of the paper.
Funding
This work was supported by the Department of Veterinary Medicine and Animal Sciences – Università degli Studi di Milano. The study was funded by Bionote USA, Big Lake, MN, USA.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
