Abstract
Objectives
The aim of the study was to determine if cats administered robenacoxib immediately after gastrointestinal surgery have a similar complication rate to cats that were not administered robenacoxib.
Methods
Medical records were reviewed for 154 cats that underwent gastrointestinal surgery between December 2015 and September 2021 in this retrospective study. Data collected included patient signalment, presenting complaint, surgical procedure(s) performed, robenacoxib administration and major postoperative complications. Cats were excluded if they did not have a 2-week postoperative follow-up examination. Two groups were analyzed: group R (postoperative robenacoxib administration) consisted of 43 cats; and group C (no postoperative robenacoxib administration) consisted of 111 cats.
Results
Complications occurred in 10/43 (23.2%) cats in group R and 34/111 (30.6%) cats in group C. Major complications occurred in 1/43 (2.3%) in group R and 14/111 (12.6%) in group C. Minor complications occurred in 9/43 (20.9%) cats in group R and 20/111 (18.0%) cats in group C. One of 43 cats (2.3%) in group R and 10/111 (9.0%) cats in group C were euthanized or died within 0–16 days after gastrointestinal surgery.
Conclusions and relevance
Cats that were administered robenacoxib postoperatively did not have an increase in major postoperative complications after gastrointestinal surgery compared with cats that were administered an alternative analgesic medication. The use of robenacoxib in cats after gastrointestinal surgery may be a safe analgesic option for postoperative pain control.
Introduction
Postoperative pain management can be challenging in cats compared with other species owing to their metabolism, lack of drug availability and difficulty of administration.1,2 Analgesia is a critical component of postoperative recovery, but many analgesics have unwanted side effects. For this reason, multimodal pain management is encouraged. 1 Opioids may be administered postoperatively as constant rate infusion, intermittent intravenous/intramuscular dosing, transdermally or given transmucosally. Transmucosal administration of buprenorphine raises concerns with outpatient opioid prescription and the associated costs of the medication. Tramadol is a synthetic opioid option for postoperative analgesia, but there is speculation as to its effectiveness in cats. 3 Gabapentin is commonly used in cats as a sedative and analgesic, but it is unknown if it provides adequate postoperative pain control.
Non-steroidal anti-inflammatory drugs (NSAIDs) are commonly used after non-gastrointestinal surgical procedures for their analgesic, anti-pyretic and anti-inflammatory properties. 4 However, there is concern that the use of NSAIDs after gastrointestinal surgery may increase the risk of gastrointestinal ulceration and impair wound healing. In humans, several studies found an association between administration of NSAIDs and anastomotic leakage and impaired intestinal healing after gastrointestinal surgery.5–7 NSAIDs inhibit the cyclooxygenase (COX) enzyme, which decreases production of certain prostaglandins. These prostaglandins play a key role in the inflammatory response at sites of tissue damage, but they may also affect homeostasis functions. 8 The COX enzyme has two isoforms that are responsible for these physiologic functions: COX-1 and COX-2. 8 The COX-1 isoform is present in many tissues and is responsible for the production of prostaglandins that are involved with gastric mucosal integrity, regulation of renal blood flow and regulation of platelet activity.9,10 The COX-2 isoform is induced locally and transiently by tissue injury.
Several NSAIDs have been developed that are COX-isoform selective. These NSAIDs are more selective for COX-2 inhibition (termed coxib class drugs) and have the potential advantage of reducing renal injury and gastric ulceration, which are predominantly influenced by the COX-1 isoenzyme inhibition, while still addressing pain, inflammation and pyrexia.11–13 Although there are multiple coxib class drugs available, only robenacoxib is approved for use in cats in the USA.14–16
Robenacoxib is rapidly absorbed after oral, intravenous and subcutaneous administration and has rapid blood clearance. It persists longer at sites of inflammation, providing a longer, more targeted analgesic effect that has been documented to last over 24 h in placebo-control studies.13,14,16 Robenacoxib is approved by the US Food and Drug Administration for 3-day postoperative analgesia in cats. 15 Approved routes of administration and dosages of robenacoxib include 2 mg/kg SC and 1–2.4 mg/kg PO q24h for up to 3 days. Robenacoxib has been approved for longer periods of administration in other countries.14,16
The objective of this retrospective study was to compare major complication rates in cats that were administered robenacoxib immediately after gastrointestinal surgery compared with cats that were not given robenacoxib postoperatively. Our null hypothesis was that cats administered robenacoxib after gastrointestinal surgery would not have increased major postoperative complications compared with cats prescribed other analgesic medications.
Materials and methods
Case selection
Medical records were reviewed to identify cats that underwent gastrointestinal surgery between December 2015 and September 2021. Data retrieved from the medical records included signalment, presenting complaint, diagnosis, reason for surgery, date of surgery, surgery performed and postoperative complications. Complications were classified as minor or major. Minor complications were defined as those that did not require significant medical intervention, while major complications were defined as those requiring additional surgery, advanced medical intervention or resulting in death or humane euthanasia. A minimum follow-up of 2 weeks was obtained by evaluating medical records. Cats were excluded if they had incomplete medical records, had surgery performed by an emergency clinician, received robenacoxib for a duration shorter than 3 days, had additional surgery performed at the time of gastrointestinal surgery, had colorectal surgery performed or had no 2-week follow-up evaluation.
Cats were divided into two groups: group R (those that received robenacoxib) and group C (control group, no robenacoxib administration nor any other NSAIDs). Cats in group C were administered either gabapentin (Neurontin and generic brands), transmucosal buprenorphine (generic brand) or a combination of the two. Cats in group R received a single injection of robenacoxib (2 mg/kg SC once, Onsior; Elanco Animal Health) immediately postoperatively and were then prescribed 3 or 6 days of robenacoxib (1–2.4 mg/kg q24h, Onsior tablets; Elanco Animal Health) after surgery. The duration of robenacoxib postoperatively was at the discretion of the surgeon. Cats in group R also received gabapentin, transmucosal buprenorphine or a combination of the two for additional analgesia if indicated. All cats included in the study were administered either hydromorphone (generic brand) or morphine (generic brand) intravenously as a premedication followed by a fentanyl continuous rate infusion intraoperatively and continued 4–6 h postoperatively.
Procedure
Surgeries were performed by one of five diplomates of the American College of Veterinary Surgeons or by a veterinary surgical resident under direct supervision by one of the five diplomates.
Statistical analysis
All statistical analyses were conducted using SAS version 9.4 (SAS Institute). A significance threshold of 0.05 was used. Binomial proportions with two-sided Wilson 95% confidence intervals (CIs) were calculated for complication rates. Logistic regressions were used to compare the odds of complications between group R and group C. Log-likelihood ratio P values and profile-likelihood CIs were reported.
Results
A total of 274 cats had gastrointestinal surgery between December 2015 and September 2021. Of them, 98 cases did not meet the inclusion criteria due to a lack of case follow-up. An additional 22 cases were excluded because of various factors, including surgery performed by emergency clinicians (n = 4), robenacoxib used for less than recommended treatment course (n = 1) and additional surgical procedures performed at the time of gastrointestinal surgery (n = 17). In total, 154 cases met the eligibility criteria for the study, and 43 cats were included in group R and 111 cats were included in group C. In the group that received robenacoxib postoperatively (group R), 32 cats received 3 days of therapy and 11 cats received 6 days of therapy. The study population included 93 (60.4%) castrated males, two (1.3%) intact males, 56 (36.4%) spayed females and three (1.9%) intact females. The median age was 7 years (range 1–19.5). The population included 16 different feline breeds, with the most represented breeds including domestic shorthair (n = 89), followed by domestic longhair (n = 14), domestic mediumhair (n = 13), Siamese (n = 11), Ragdoll (n = 6), Maine Coon (n = 5), Siberian (n = 4), Persian (n = 3), Sphynx (n = 2), Singapura (n = 1), Russian Blue (n = 1), Angora (n = 1), Burmese (n = 1), Birman (n = 1), Bombay (n = 1) and Snowshoe cat (n = 1).
The reasons for exploratory laparotomy included gastrointestinal foreign material (84.4%), small intestinal mass/small intestinal thickening (9.7%), small intestinal intussusception (3.2%), septic peritonitis (1.3%) and abdominal trauma (1.3%) (Table 1). Intestinal surgery (enterotomy [single or multiple] or intestinal resection and anastomosis) was the most commonly performed procedure, accounting for 54.5% of all surgeries, followed by gastric surgery (gastrotomy or Billroth 1) at 38.9% and combination surgery (gastrotomy and enterotomy or gastrotomy, enterotomy, and intestinal resection and anastomosis) at 6.5% (Table 2).
Reasons for pursuing exploratory laparotomy (n = 154)
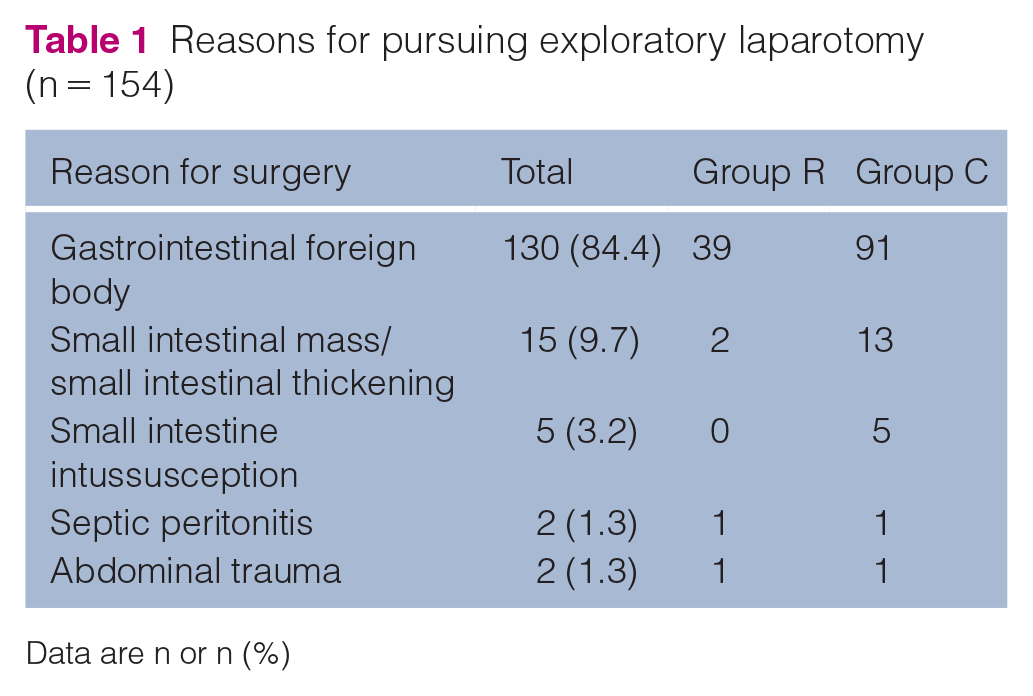
Data are n or n (%)
Surgical procedures performed (n = 154)
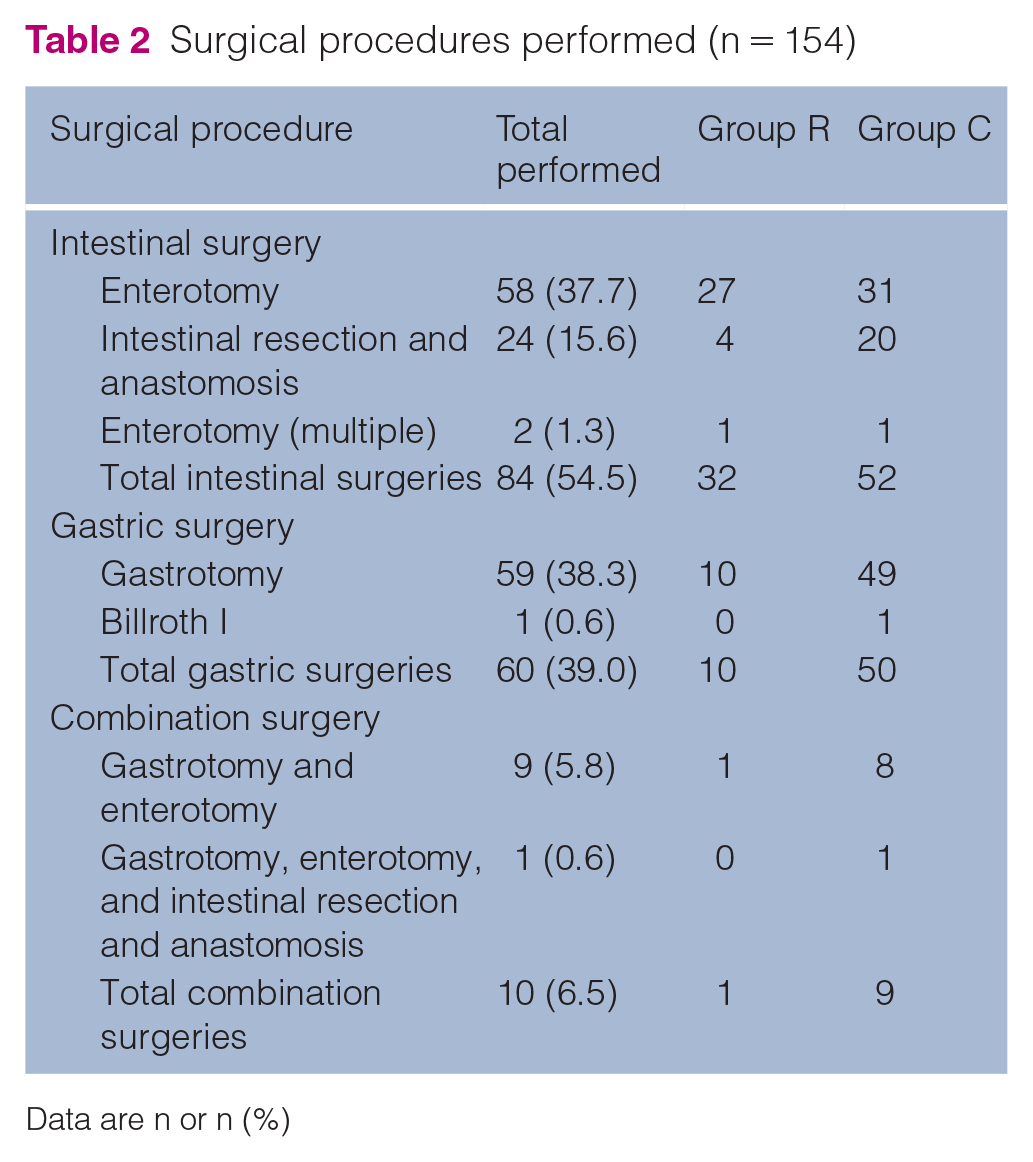
Data are n or n (%)
Complications occurred in 44 cases. Major complications included septic peritonitis, newly acquired respiratory disease (aspiration pneumonia, pneumonia, acute respiratory distress syndrome), cardiac disease (congestive heart failure, cardiac arrest), intracranial disease (altered mentation, vestibular disease), hypoglycemia, neutropenia and esophageal stricture. Minor complications included transient anorexia, surgical site infection, vomiting/nausea, pyrexia, mild electrolyte derangements (hyponatremia, hypochloremia [uncorrected]), mild anemia and subcutaneous bleeding or bruising (Table 3).
Major and minor complications in group R and group C combined (n = 154)
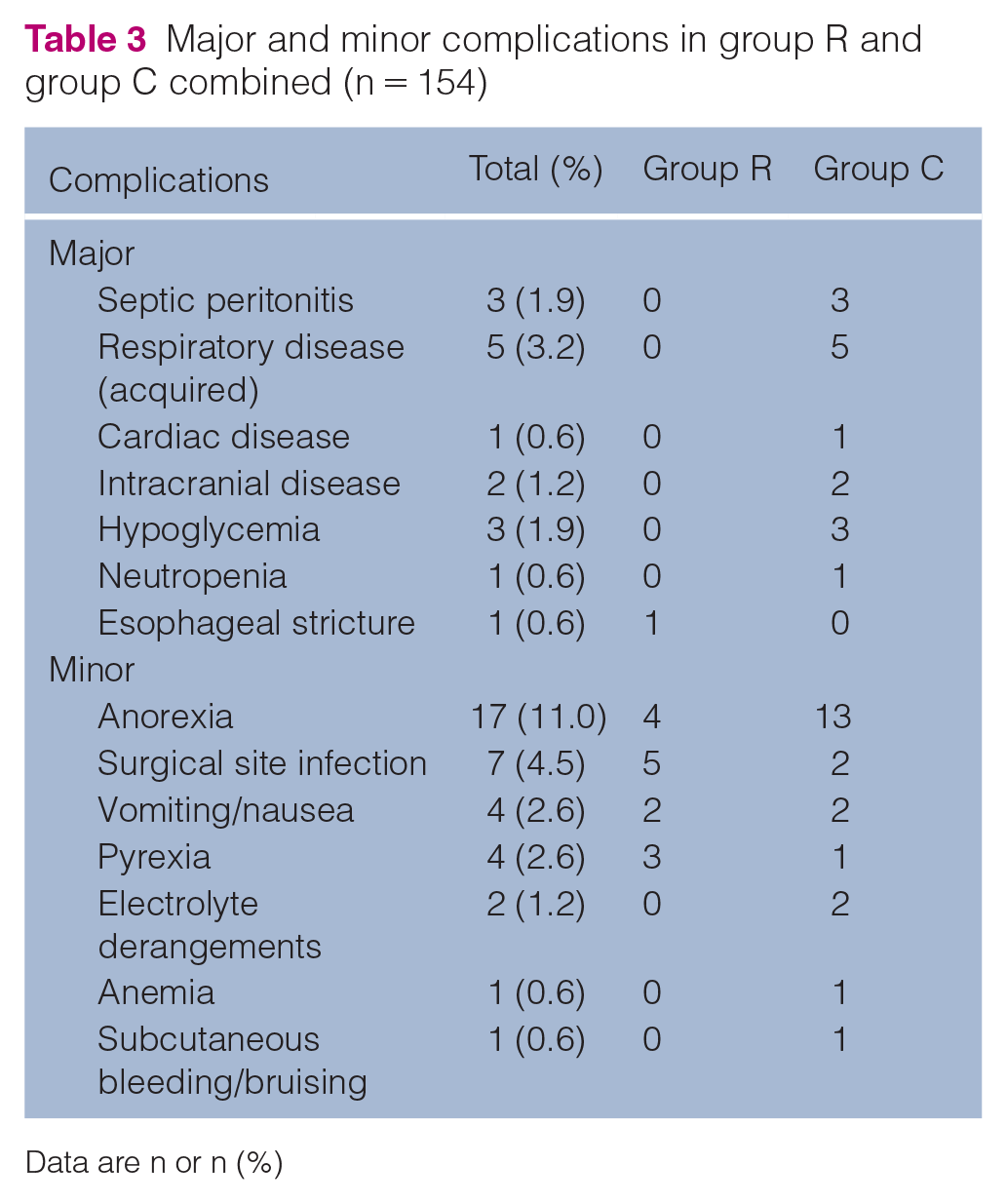
Data are n or n (%)
The overall complication rate in group R was 23.2% (10/43; 95% CI 13–38) compared with 30.6% (34/111; 95% CI 22–40) in group C (P = 0.357; odds ratio [OR] 0.69; 95% CI 0.3–1.55) (Table 4). The major complication rate in group R was 2.3% (1/43; 95% CI 0.4–12) compared with 12.6% (14/111; 95% CI 8–20) in group C (P = 0.030; OR 0.2; 95% CI 0.02–1.3). The minor complication rate in group R was 20.9% (9/43; 95% CI 11–35) compared with 18.0% (20/111; 95% CI 12–26) in group C (P = 0.681; OR 1.2; 95% CI 0.5–2.9). The overall major and minor complication rates were not significantly different between group R and group C.
Percentage of major and minor complications in group R and group C

Data are n (%)
Complication rates (major and minor) were compared between type of surgery performed (gastric vs intestinal surgery) for group R and group C (Tables 5 and 6). The overall complication rate for intestinal surgery was 18.8% in group R (6/32) and 44.2% in group C (23/52) (P = 0.014). The overall complication rate for gastric surgery was 40% in group R (4/10) and 12% in group C (6/50) (P = 0.048). The major complication rate for intestinal surgery was 0% in group R (0/32) and 19.2% in group C (10/52) (P = 0.001). The major complication rate for gastric surgery was 10% in group R (1/10) and 4% in group C (2/50) (P = 0.468). The minor complication rate for intestinal surgery was 18.8% in group R (6/32) and 25% in group C (13/52) (P = 0.502). The minor complication rate for gastric surgery was 30% in group R (3/10) and 8% in group C (4/50) (P = 0.077). For combination surgery (gastrotomy and enterotomy or gastrotomy, enterotomy, and intestinal resection and anastomosis), too few cases were included in this category for comparison. No major or minor complications occurred in group R for combination surgery.
Major and minor surgical complications in group R
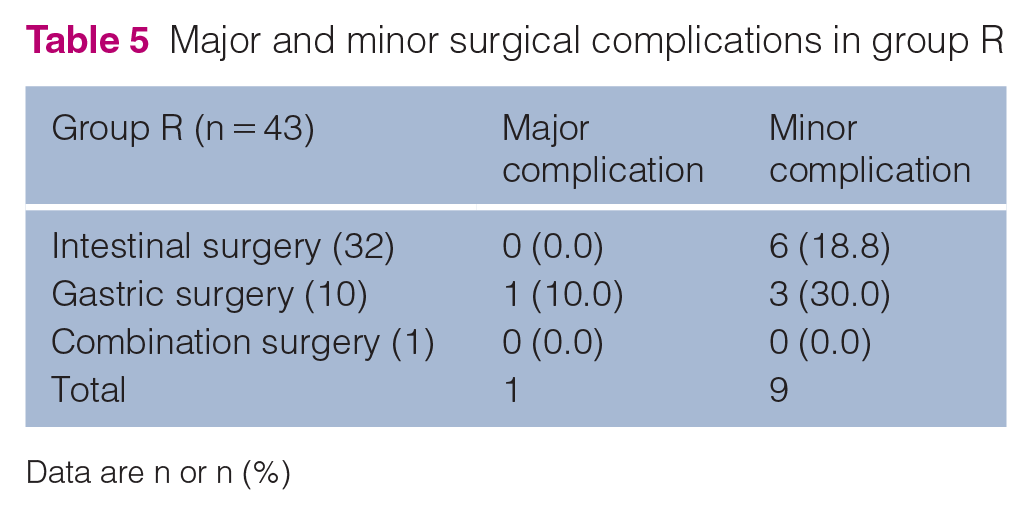
Data are n or n (%)
Major and minor surgical complications in group C

Data are n or n (%)
Short-term follow-up was available for all 154 cats. In total, 143/154 (92.8%) cats recovered well and were healthy 2 weeks postoperatively. The remaining cats (11/154, 7.1%) were euthanized after surgery. One cat from group R was euthanized because of the development of an esophageal stricture 16 days postoperatively. A total of 10 cats from group C were euthanized or died a mean of 2.7 days (range 0–9 days) postoperatively owing to respiratory disease (n = 4), septic peritonitis (n = 2), hypoglycemia (n = 2), arrest immediately postoperatively (n = 1) and neurologic disease (n = 1) (Table 7).
Reason for euthanasia or death in group R and group C

Data are n
Discussion
To the authors’ knowledge, there is no literature evaluating the use of NSAIDs in cats immediately after gastrointestinal surgery. This retrospective study found that there was no statistically significant difference in overall complication rates in cats that received robenacoxib postoperatively compared with those that did not (P = 0.357). When evaluating type of surgery (intestinal vs gastric) between cats, the control group had a significantly higher rate of intestinal surgery complications (44.2% [23/52] vs 18.8% [6/32] in group R; P = 0.014) and the robenacoxib group had a significantly higher rate of gastric surgery complications (40% [4/10] vs 12% [6/50] in group C; P = 0.048). It is difficult to determine if the differing results between types of surgery are due to low case numbers or are truly representative. With the study data, upper gastrointestinal complications may be a consideration with robenacoxib; however, further investigation is needed. Overall, it is challenging to conclude that robenacoxib had no meaningful effect on complications compared with cats that did not receive the mediation postoperatively.
The use of NSAIDs after gastrointestinal surgery has been documented to increase the risk of gastrointestinal leakage and delay tissue healing in humans.5, 6, 17 A large meta-analysis evaluating NSAID usage in humans after gastrointestinal anastomoses documented a significant increased risk of postoperative anastomotic leakage associated with NSAID administration (7.5% for NSAID use vs 5.3% for cases that did not receive postoperative NSAIDs). 18 In the same study, the administration of non-COX selective NSAIDs had a significantly higher rate of anastomotic leakage compared with those patients who received a selective COX-2 inhibitor. The patients who received selective COX-2 inhibitors had a comparable anastomotic leakage rate with those who had not received NSAIDs. This suggests that a selective COX-2 inhibitor may be a safer alternative for postoperative pain management compared with non-selective COX inhibitors for this population of patients.
Non-selective NSAIDs have several mechanisms that may result in reduced intestinal integrity. These include enterocyte mitochondrial dysfunction leading to increased epithelial permeability and decreased production of protective prostaglandins.19–22 NSAIDs in a neutral pH environment (cytoplasm) will become trapped and accumulate inside cells, potentially leading to cellular injury. 23 In addition, non-selective NSAIDs affect the submucosal collagen layer, which may result in decreased tensile strength at the anastomosis site. 24 Mastboom et al 24 evaluated the effect of NSAIDs on collagen degradation during the initial phase of wound healing in rats that had undergone experientially induced anastomoses. They concluded that the administration of NSAIDs, specifically the non-selective NSAIDs piroxicam and ibuprofen, resulted in postoperative degradation of collagen in colonic anastomoses sites and increased the rate of surgical infections. As robenacoxib is a COX-2 selective NSAID, we hypothesized that the risk of anastomotic leakage would be no different from that in non-NSAID treated controls.
COX-2 selective NSAIDs may effectively reduce pain and inflammation while minimizing negative side effects, including gastric ulceration and renal injury. Because COX-1 is expressed constitutively and is involved in normal physiological hemostasis, its inhibition may lead to gastric ulceration and bleeding disorders. 25 Conversely, COX-2 is only expressed in response to external stimuli, such as inflammation, pain and fever. COX-2 is also thought to play a role in wound repair. 25 De Hingh et al evaluated healthy and postoperative ileal and colonic tissue from rats using immunohistochemistry. 26 In that study, it was noted that COX-1 was almost absent while COX-2 was abundantly present in the anastomotic tissues, most notably 3 days postoperatively. 26 One theory behind this upregulation of the COX-2 isoform at the anastomotic sites was that prostaglandins help with early collagen formation and this increases anastomotic strength. However, study results vary and the location of the anastomosis (small intestinal vs colonic) shows different results.25,27 A second theory is that prostaglandins may aid in mucosal barrier repair after injury; therefore, a reduction in mucosal prostaglandin concentration may increase the risk of leakage at an incisional site postoperatively. 25
We chose to evaluate robenacoxib in cats after gastrointestinal surgery because of its COX-selective properties and its approval for use in cats in the USA. King et al 12 documented its high safety index. When robenacoxib was administered at 20 times (10 mg/kg q12h for 42 days) the recommended dose, no toxic effects were observed. 9 Lees et al 28 also confirmed this wide safety margin when robenacoxib was administered at up to eight times (2, 4, 6 mg/kg SC and 2.4, 4.8 and 7.2 mg/kg PO) the recommended dosage in cats over a 37-day period. No adverse effects were observed in general health, hematological or biochemical variables, visible intestinal injury (ie, gastric or intestinal ulceration) and histopathology results in healthy cats. 28 These studies demonstrated that even when robenacoxib was prescribed in excess of the manufacturer’s recommended dosage of 1 mg/kg for a duration of 3 days in healthy cats, no significant complications were observed. Given robenacoxib’s wide safety margin in healthy cats, we speculated that it could be an attractive analgesic choice in cats with prior gastrointestinal compromise.
King et al 12 documented the selectivity of robenacoxib with approximately 98% inhibition of COX-2 and only 26.8–58.3% of COX-1 inhibition at peak effect in cats. Other studies have confirmed robenacoxib’s isoenzyme selectivity based on IC50 COX-1:IC50 COX-2 ratios.12,28 Because COX-1 isoform helps maintain gastrointestinal mucosal integrity, a low COX-1 to COX-2 selectivity ratio (ie, inhibiting predominantly COX-2) suggests the potential for reduced gastrointestinal tract complications. 29
Independent of route of administration, robenacoxib is rapidly cleared from blood, with a terminal half-life of 0.6–1.4 h. 13 In comparison, in exudate or sites of inflammation, high concentrations of robenacoxib are maintained for close to 24 h. 13 A possible explanation for the significance difference in the persistence of robenacoxib in inflamed tissues is the drug’s high degree of binding to plasma proteins and the leakage of these robenacoxib-bound proteins into sites of inflammation. Collectively, all of these attributes suggest that robenacoxib could provide analgesia and anti-inflammatory effects at sites of gastrointestinal injury while still maintaining gastrointestinal mucosal integrity.
There are some limitations to this study. This is a retrospective study, which limited the evaluation of overall patient health before surgery, as well as any peri-anesthetic complications known to impact postoperative complications. Some medical records lacked adequate postoperative documentation, thus limiting the assessment of patient recovery and potential complications, including asymptomatic ulceration, delayed healing or gastrointestinal leakage at the surgery site. There were also a significant number of cats excluded due to the lack of follow-up. This may have affected our reported postoperative complication rates. There were multiple surgeons performing surgery, which limits the consistency between procedures. The type of surgery performed (gastrotomy, enterotomy vs resection and anastomosis) could influence recovery and affect documented complications. The duration of robenacoxib administered postoperatively was not consistent. Most cats received 3 days of robenacoxib; however, some were administered 6 days of therapy. We were unable to compare complication rates between the 3- and 6-day groups because of smaller numbers. Other factors that were not directedly compared between groups that may have influenced postoperative complication rates included intraoperative complications, total anesthesia time, total surgery time, albumin concentrations and postoperative gastrointestinal motility. Lastly, our small sample size of 154 cats, with only 43 of the cats administered robenacoxib, may have resulted in a type 1 statistical error.
Conclusions
Future prospective studies are needed to more clearly evaluate potential benefits and adverse effects of robenacoxib use after gastrointestinal surgery in cats. Important factors to be evaluated would include the effect of pre- and postoperative renal and hepatic function on complication rates as well as more objective markers of gastrointestinal healing and postoperative integrity. Although repeat surgery in client-owned animals is impracticable, these data could be achieved with less invasive approaches, such as endoscopy or advanced imaging. Our data suggest that robenacoxib may be safe to use in cats after gastrointestinal surgery, but caution is still advised and further studies are needed.
The use of robenacoxib in cats after gastrointestinal surgery did not result in a statistically significant difference in overall complications compared with cats that were not administered NSAID therapy postoperatively. Additional studies are needed to evaluate the role of robenacoxib in the management of postoperative pain in cats after gastrointestinal surgery.
Footnotes
Acknowledgements
The authors thank Deborah Keys, PhD, Kaleidoscope Statistics, for assistance with the statistical analysis of the data in this study.
Conflict of interest
AHJ was a previous key opinion leader for Novartis and Onsior, before Onsior was sold to Elanco.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
