Abstract
Objectives
Non-steroidal anti-inflammatory drugs (NSAIDs) are infrequently utilized in cats due to concern for renal compromise; however, recent studies demonstrate tolerability of low dose meloxicam. Toceranib phosphate is used to treat several feline cancers and is well tolerated. This study aimed to determine the tolerability and adverse event profile of combined toceranib and low dose meloxicam in cancer-bearing cats. Secondary goals involved assessing anticancer tumor efficacy and impact upon quality of life and analgesia.
Methods
Cats with any cancer not involving the kidneys were eligible. The study adopted a conventional 3 + 3 dose escalation design. Toceranib was administered every other day at a standard dose with meloxicam administered in an escalating fashion in subsequent cohorts, at a starting dose of 0.01 mg/kg on opposite days to toceranib, up to a maximum of 0.02 mg/kg daily, based upon previous safety studies. Laboratory work, blood pressure, tumor measurements, pain score and client-completed quality-of-life surveys were recorded every 2–4 weeks during the 12-week study period.
Results
Twenty-one cats were enrolled. When combined with toceranib, a meloxicam dose of 0.02 mg/kg q24h was safe and well tolerated, with no cats being withdrawn due to adverse events from the drug combination. The majority of cats demonstrated clinical benefit with stable to mildly improved tumor measurements, quality of life and pain scores.
Conclusions and relevance
Low dose meloxicam combined with toceranib is safe and well tolerated in cancer-bearing cats. Continued patient recruitment and data collection are needed to determine the maximum tolerated dose of meloxicam. The results of our study will guide further phase II/III trials.
Introduction
Non-steroidal anti-inflammatory drugs (NSAIDs) are commonly utilized in protocols targeting canine and human neoplasms as supportive medications to mitigate cancer associated pain and inflammation, for direct antineoplastic properties or to treat other chronic inflammatory conditions (such as degenerative joint disease [DJD]) that are common in aged, cancer-bearing populations. 1 NSAIDs inhibit prostaglandin production from arachidonic acid by the cyclooxygenase (COX) enzymes. Upregulation of COX-2 has been identified in many human and canine cancers and has been correlated with increased aggressive potential of tumor cells.2,3
Despite the purported benefits of NSAID use in oncology patients, chronic NSAIDs are infrequently utilized in feline patients due to differences in pharmacologic metabolism and concern for renal compromise, especially given the high incidence of chronic kidney disease in aging cats. 4 In 2010, the Federal Drug Administration released a ‘black box warning’ against repeated use of meloxicam in cats; currently, the only approved use is a one-time injection (0.3 mg/kg) for postoperative pain. 5 Cats’ low capacity for hepatic glucuronidation contributes to increased toxicity, specifically renal toxicity.
Meloxicam at lower doses is licensed in some European countries and Australia, and the International Society of Feline Medicine and the American Association of Feline Practitioners guidelines suggest that the benefits of long-term NSAID treatment in cats outweigh the risks. 6 Low dose meloxicam – 0.02 mg/kg daily, or a range of 0.01–0.03 mg/kg – has recently been evaluated in feline patients with DJD or chronic kidney disease (CKD) and has been shown to be well tolerated without evidence of renal toxicity.7–9
Toceranib phosphate (Palladia; Zoetis) is an orally administered receptor tyrosine kinase inhibitor that has been used to treat a variety of canine and feline neoplasms including mast cell tumors, feline oral squamous cell carcinoma (FOSCC) and other epithelial neoplasms.10–15 Multiple studies have demonstrated that toceranib is generally well tolerated in cats, with toxicity typically limited to mild gastrointestinal, myelosuppressive and hepatotoxic effects.15–17 Renal toxicity does not seem to be common; in various retrospective studies azotemia secondary to toceranib administration has been reported in 0–14.5% of patients, with most cases being mild and requiring no intervention.15–17
No prospective, dose controlled clinical trials exist evaluating the combination of toceranib and meloxicam in cats. The primary aim of this study was to determine the safety and adverse event profile of low dose meloxicam (up to 0.02 mg/kg daily based upon previous studies) in combination with toceranib at standard dose in cancer-bearing cats. While efficacy is not a primary endpoint of safety studies, a secondary goal was to explore the anticancer efficacy of the combination, as well as impact upon quality of life (QOL) and analgesia.
Materials and methods
This prospective dose escalation clinical trial assumed a conventional 3 + 3 cohort design. 18 All cats received toceranib every other day by mouth (PO); given the limitations of the toceranib capsule size (10 or 15 mg) along with the typical size of feline patients, cats received a target dose of 2.5–3.75 mg/kg. The dose of meloxicam was escalated, with a starting dosage in cohort 1 of 0.01 mg/kg PO every other day, administered on opposite days to toceranib. If no dose-limiting toxicities (DLTs) were noted in a cohort of three cats, then the dose of meloxicam was escalated in subsequent cohorts until the maximum tolerated dose (MTD) was established or a dose of 0.02 mg/kg daily was reached. A DLT was defined as any grade 3 or higher adverse event (according to the Veterinary Comparative Oncology Group’s common terminology criteria for adverse events, VCOG-CTCAE v1.1) 19 that was clinically relevant except for hematopoietic events (ie, neutropenia, thrombocytopenia), for which grade 4 was defined as dose-limiting.
Four cohorts were initially planned (0.01 mg/kg every other day, 0.01 mg/kg daily, 0.02 mg/kg every other day and 0.02 mg/kg daily). Owing to decreased hospital caseload and study recruitment in the light of the COVID-19 pandemic, along with discussion with the consulting pharmacologist that the cohorts would have similar pharmacology characteristics and dose intensity, the middle two cohorts were collapsed into one. Thus, cohort 2 included cats that received meloxicam 0.01 mg/kg daily or 0.02 mg/kg every other day. If a DLT was noted, then the cohort was expanded to include six cats. The MTD was the highest dose level at which no more than one of six cats developed a DLT. Blood and urine samples and blood pressure measurements were collected on days 0, 14, 28, 56 and 84 (Table 1). Cohorts were also expanded if any cat died, was euthanized or withdrew from the study prior to the 56-day recheck in order to obtain adequate laboratory and safety data before enrolling cats in subsequent cohorts.
Study timetable

Every other day toceranib dosing began on day 0. Meloxicam dosing began on day 0 or day 1 dependent upon cohort
(X) = tumor measurements were performed if accessible via physical examination and caliper measurements. If measurements were only obtainable via imaging, the frequency was recommended at minimum at baseline and day 84
SDMA = symmetric dimethylarginine; QOL = quality of life
Adverse events (AEs) were graded according to VCOG-CTCAE v1.1. 19 Given concern for meloxicam-induced kidney damage, creatinine values were monitored closely. In cats experiencing a grade 3 or higher acute kidney injury (defined as a >3 × increase in creatinine over baseline, or >2 × upper limit of normal), and urinary tract infection and urolithiasis had been ruled out with culture and imaging, then meloxicam treatment was discontinued and the patient was withdrawn from the study. Hypertension was managed with amlodipine as recommended by the University of Wisconsin Veterinary Medical Teaching Hospital cardiology service.
Client-owned cats were eligible for the study if they had a cytologically or histologically confirmed, naive or previously treated malignant tumor of any histology that did not directly involve the kidneys. Eligible cats were required to have a VCOG performance status of 0 (normal activity) or 1 (mild lethargy over baseline). Appropriate washout periods from previous therapy (1 week from glucocorticoids, 2 weeks from previous chemotherapy, surgery or radiation therapy) were required. Further eligibility was based on International Renal Interest Society (IRIS) CKD guidelines, with cats with CKD stage 1 or 2 (those with creatinine ⩽2.8 mg/dj when clinically stable and hydrated) being eligible for enrollment. Hydration status was determined via assessment of mucous membrane tackiness, skin turgor and bloodwork values (blood urea nitrogen [BUN], total protein, hematocrit or packed cell volume). Use of pain medications (buprenorphine, gabapentin) and gastrointestinal supportive medications (maropitant, ondansetron, metronidazole, famotidine, omeprazole, mirtazapine, capromorelin) at the time of enrollment or added during the study period was allowed. Cats were excluded if they had received toceranib or chronic NSAIDs at any time prior to enrollment.
While not a primary endpoint of safety trials, preliminary antitumor efficacy of the drug combination was assessed when available, as well as impact upon QOL and analgesia. Tumor response was assessed with VCOG response evaluation criteria for solid tumors (RECIST) for patients presenting with gross disease. 20 Measurements were performed via caliper measurement of longest tumor diameter or with image-guided measurements if available, although imaging was not required for the study. Cats that developed progressive disease were withdrawn and owners were offered alternative treatment options. Time to progression was defined as the number of days from trial enrollment to disease progression or death from disease. Impact upon QOL was assessed via owner survey at baseline and subsequent evaluations (see the supplementary material). The QOL survey focused on behaviors at home including food and water intake, grooming behavior, litter box use and interaction with the owner. Effect upon analgesia was evaluated using the Glasgow pain scoring system (see the supplementary material) assigned by the overseeing clinician at baseline and at each visit based upon physical examination findings.
The clinical protocol was approved by the Animal Care and Use Committee and written informed consent was obtained from participating clients. Owners were permitted to withdraw consent at any time.
Results
In total, 21 cats met the inclusion criteria and were enrolled in the trial between August 2019 and March 2021. The median age of these cats was 11.7 years (range 5.7–17) and median weight was 4.44 kg (range 2.70–7.77). There were nine castrated males, one intact male and 11 spayed females. The majority (n = 15) of cats were domestic shorthair, with the remainder consisting of domestic longhair (n = 2) and one each of domestic mediumhair, Maine Coon, Bengal and Norwegian Forest Cat. The most common cancer diagnosis was FOSCC in six cats, although several other cancer types were represented (Table 2).
Tumor types and outcome

Cats were still alive at the time of manuscript preparation
Two cats were lost to follow-up 66 and 83 days after enrollment; one cat was still alive at the time of manuscript preparation
Seven cats were enrolled in the first cohort, eight in the second cohort and six in the third and final cohort (Table 3). Eleven cats were withdrawn early due to disease progression (n = 7), difficulty administering medications (n = 2), owner-perceived reduction in QOL (n = 1) or sudden death (unknown cause and necropsy declined, but unlikely due to the therapy, n = 1) (Table 4). Thus, cohort sizes were larger than three given the need for replacements. A meloxicam dose of 0.02 mg/kg daily was reached, with no clinically relevant DLTs.
Treatment cohorts
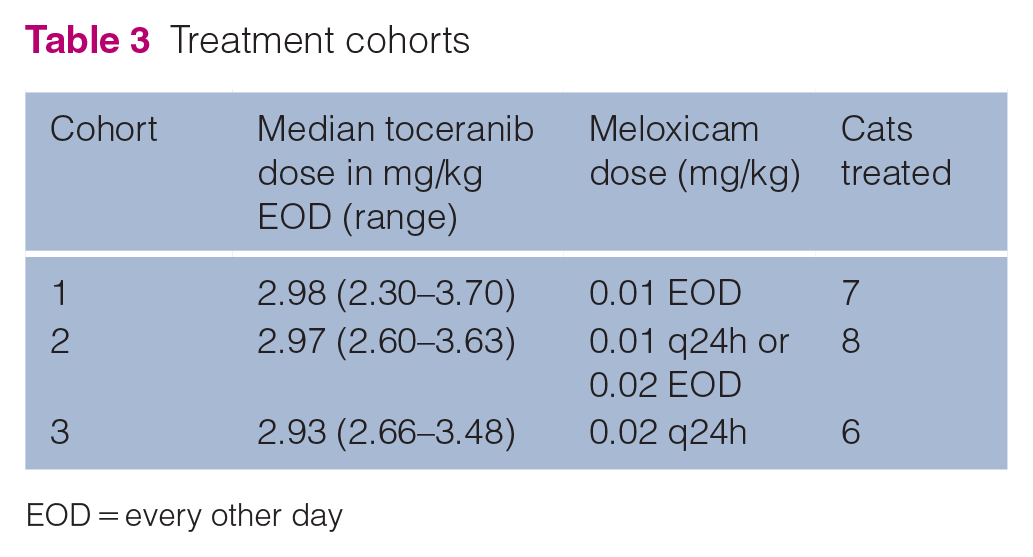
EOD = every other day
Withdrawn cats

FOSCC = feline oral squamous cell carcinoma
The most common adverse events were weight loss (n = 13) and anorexia (n = 9) (Table 5). Eight cats experienced a mild (grade 1 or 2) hematologic adverse event; the most common was grade 1 thrombocytopenia (n = 7), although platelet clumping in four cats may have falsely decreased the reported value. Fourteen cats experienced a biochemical adverse event, most commonly involving mildly elevated (grade 1–2) liver or kidney values, potassium or creatine kinase (CK). Elevated creatinine and BUN were noted in 2/21 (9.5%) and 3/21 (14.3%) cats, respectively. The cat with grade 2 elevated creatinine had metastatic urothelial carcinoma of the urinary bladder, which likely contributed to the elevation. The cat with grade 1 elevated creatinine was treated with subcutaneous fluids, and creatinine subsequently decreased from 2.8 mg/dl to 2.4 mg/dl upon recheck examination; the elevated value was likely secondary to early CKD. Elevated glucose levels were noted in four cats and were likely secondary to a stress response rather than the drug combination as hyperglycemia has not been a reported side effect from either toceranib or meloxicam. Glucosuria was not noted on urine dipstick in these cats.
Adverse events (AEs) by grade and cohort

AST = aspartate amonotransferase; ALP = alanine aminotransferase; BUN = blood urea nitrogen; CK = creatine kinase: EOD = every other day
Hematologic, biochemical, gastrointestinal and/or constitutional side effects were noted in 20/21 cats (95.2%). Adverse events were generally mild (grade 1 or 2). No cats needed to be withdrawn from the study from adverse events, although a 2-week toceranib holiday and a reduction in toceranib frequency to Monday-Wednesday-Friday dosing was recommended in one cat each in cohort 3 due to hyporexia (grade 2) and weight loss (grade 3), respectively. Notably, three cats experienced grade 3 weight loss, although two of these cats had previously documented weight loss and the other cat had been diagnosed with carcinomatosis at the time of trial enrollment, which was the likely cause of the weight loss. Continued enrollment was elected given physical examination and owner survey findings. In all three, the cats’ cancer was likely the cause of the decreased weight rather than the therapy, but imaging of the chest and abdomen was declined by the owners and thus drug-related toxicity cannot be ruled out.
Other grade 3 and above adverse events were rare and not clinically significant, and thus the meloxicam dose did not increase the frequency of DLTs in any one cohort over another. The cat with grade 3 alanine aminotransferase (ALT) elevation was diagnosed with concurrent hyperthyroidism, which likely contributed to the abnormality. The ALT value improved with methimazole treatment. The grade 4 ALT elevation in another cat was transient and resolved with no change in therapy, making a drug-related toxicity less likely. Seven cats experienced an aspartate aminotransferase (AST) elevation, two of which were grade 3. AST elevation has been noted in another feline toceranib study 13 and the grade 3 AST elevation noted in the present study was not clinically relevant given adequate physical examination findings, QOL scores, pain scores and unremarkable values for the remaining liver enzymes.
Transient grade 1 hypertension was noted in one cat in cohort 3 at the 8-week recheck. Grade 2 hypertension was noted in four cats (two in cohort 1, one each in cohorts 2 and 3) at the 4-week recheck so amlodipine treatment was recommended; one of these cats also developed simultaneous grade 1 creatinine elevation and another had grade 1 BUN elevation with grade 2 proteinuria, but no other evidence of target organ damage was noted. The remaining urinalysis and dipstick data were unremarkable.
QOL was evaluated at baseline and each recheck examination via owner surveys. Among cats whose owners filled out at least one survey beyond baseline (n = 17), three had improved overall QOL, eight had stable QOL and six had decreased QOL. Three of the six cats with decreased QOL had progressive disease documented at the time of the reduced QOL scores, and one of the six resented being medicated, which likely contributed to the lower score. The other two cats – diagnosed with mammary carcinoma and adenocarcinoma of suspected nasal origin – had stable measurable disease on physical examination but additional imaging (abdominal ultrasound, head CT scan) was declined; thus, progression of the cancer may have contributed to the lower scores.
Glasgow pain scores improved in six cats over the treatment period, remained stable in 12 and worsened in three. Among the three cats with worsening pain scores, one had progressive disease, one was withdrawn due to difficulty administering medications and one was likely stressed in the hospital environment during the final recheck examination, which may have resulted in a falsely elevated pain score.
Although evaluation of anticancer efficacy was not a primary aim of our study, Table 2 depicts outcome data. Fourteen cats died or were euthanized due to their disease, five cats remain alive at the time of manuscript preparation and two cats were lost to follow-up. In patients with measurable disease, two cats (one with mammary carcinoma, one with FOSCC) achieved a partial response that was maintained throughout the trial period. Ten cats had progressive disease (seven of which were documented during the 84-day study period), and the remaining cats had stable disease.
Discussion
Meloxicam at a dose of 0.02 mg/kg daily in combination with toceranib at standard dose was well tolerated in cats with cancer. Additional studies are needed to determine the MTD of meloxicam in this drug combination. Our study supports previous findings suggesting the role of NSAID therapy in feline cancer patients. COX-2 expression has been reported in several feline cancers, including transitional cell carcinoma (TCC) and FOSCC.21,22 Long-term safety of COX inhibitors has also been examined. Cats with DJD that received 0.01–0.03 mg/kg meloxicam daily exhibited no adverse changes to renal function as determined by monitoring creatinine values over 1 month of therapy. 7 Another study examining cats with DJD demonstrated less progression of renal disease in patients with CKD treated with low dose meloxicam (median dose 0.02 mg/kg daily) compared with untreated controls. 8 Additional studies revealed no decline in renal excretory function or lifespan in cats with or without pre-existent stable CKD that were treated with meloxicam (median dose 0.02 mg/kg daily) for at least 6 months, even for those classified as IRIS stage 2 (creatinine 1.6–2.8 mg/dl when clinically stable and hydrated) or 3 (creatinine 2.8–5.0 mg/dl) disease.9,23
Given the previously published studies evaluating low dose meloxicam in cats, and the concern for renal compromise, we did not evaluate doses higher than 0.02 mg/kg daily in this study. However, no noteworthy renal adverse events were noted, and no cats were withdrawn from the trial due to side effects attributable to the medications.
The majority of cats had stable to improved QOL scores (11/17) and pain scores (18/21) throughout enrollment, with worsening scores likely due to progression of the cancer, difficulty administering medications leading to both patient and owner stress, or secondary infections developing at tumor sites.
Eleven cats were withdrawn before completing the trial (day 84) as noted in Table 4. Thus, cohort sizes were expanded frequently despite no clear evidence of DLTs. The cat from cohort 1 that was withdrawn following sudden death was originally diagnosed with cutaneous carcinoma caudal to the left pinna; it had experienced grade 3 AST elevation, grade 2 hypertension, grade 1 anorexia and grade 1 BUN elevation prior to collapse and sudden death 53 days following study enrollment. The remainder of the most recent bloodwork was unremarkable, and no imaging was performed. Necropsy was declined, so the cause of the sudden death remains unknown. The cat with urothelial carcinoma was withdrawn due to progressive azotemia (increase of creatinine from 2.2 mg/dl to 5.6 mg/dl; increase of BUN from 46 mg/dl to 99 mg/dl) 14 days after enrollment; progression of the cancer was suspected, although a recheck abdominal ultrasound was not performed so toxicity from the medications, as well as infection or urolithiasis, could not be ruled out. Cohort 2 consisted of eight cats, with three surviving beyond the 56-day recheck. Given the lack of clinically relevant DLTs, it was determined that the drug combination in cohort 2 was safe and thus recruitment for cohort 3 commenced.
Our findings build upon previous studies, which report tolerability and activity of NSAIDs and/or toceranib in cats with cancer. Potential clinical benefit has previously been shown in retrospective studies of cats with FOSCC and TCC receiving NSAIDs.21,22,24 One study evaluating toceranib in cats with FOSCC demonstrated an association between concurrent NSAID use and improved survival, although given the retrospective nature of the study, the specific NSAID used and dose were not consistent. 10 Several additional reports document activity and/or safety of toceranib in cats with FOSCC, mast cell tumor and feline injection site sarcoma.13–16 Most of these studies were retrospective, and follow-up time varied. The present study is, to our knowledge, the first to prospectively examine the safety and tolerability of meloxicam in combination with toceranib.
Several limitations of the study exist. Imaging was not required at any recheck examination, so internal tumor progression or pulmonary or abdominal metastasis may have been missed. Thus, disease progression cannot be ruled out as the cause for adverse events, or worsening QOL or pain scores. The high number of cases of FOSCC (n = 6), an aggressive cancer with most reported median survival times of less than 3 months,25,26 as well as other advanced cancers in our study population often resulted in early withdrawal from the trial given the rapid disease progression. Also, patients were allowed to receive additional supportive care medications, which may have confounded QOL and pain scores and potentially could have influenced adverse events. In addition, the inherent subjectivity of the owner QOL surveys and Glasgow pain scoring, along with observer variation, may have introduced bias into these scores. Rarely, a urine sample was unobtainable in the clinic due to small bladder size; there was no evidence of azotemia in these instances and urine was collected at subsequent appointments. Even so, transient abnormalities may have been missed. Finally, although the dose intensities of meloxicam within cohort 2 were identical (0.01 mg/kg once daily vs 0.02 mg/kg once every other day), the peak drug concentrations may be different.
Conclusions
Our data reveal that cats with cancer can safely tolerate meloxicam 0.02 mg/kg once daily in combination with standard dose toceranib. Additional studies are needed to determine if even higher doses of meloxicam are safe. While there is concern for meloxicam-induced renal compromise in cats, this low dose may provide owners with a reasonable option when other treatments are declined or if a standard of care does not exist. The anticancer efficacy of the drug combination cannot be definitively evaluated with the current data given the lack of imaging, lack of a control population and the small sample size. Therefore, phase II/III clinical trials with larger groups of cats with similar tumor types should occur.
Supplemental Material
Supplemental Material
Feline quality of life survey
Supplemental Material
Supplemental Material
Glasgow feline composite measure pain scale
Footnotes
Acknowledgements
We thank Dr Lauren Trepanier DVM, PhD, DACVIM, DACVCP for her assistance with study design and pharmacology consultation. We also thank the entire UW Veterinary Medical Teaching Hospital Medical and Radiation Oncology and support services for their help with patient recruitment and patient care.
Author note
Preliminary findings from this study were presented in a poster at the 2020 American Association of Feline Practitioners annual conference, and as an oral abstract at the 2020 Veterinary Cancer Society annual conference.
Supplemental material
The following files are available online:
Feline quality of life survey
Glasgow feline composite measure pain scale
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Veterinary Cancer Society Resident Research Grant.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animals described in this work (experimental or non-experimental animals, including cadavers) for all procedures undertaken. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
