Abstract
Objectives
This study aimed to determine thyroid hormone concentrations in a cohort of healthy kittens due to the paucity of information in the literature, and the potential for congenital hypothyroidism (CH) to contribute to fading kitten syndrome (FKS).
Methods
The serum concentrations of total thyroxine (TT4), free thyroxine (fT4), total triiodothyronine (TT3), free triiodothyronine (fT3) and thyroid-stimulating hormone (TSH) were measured in 19 healthy kittens aged 2–16 weeks.
Results
Mean TT4, fT4, TT3 and fT3 concentrations significantly differed across age groups. Mean TT4 and fT4 concentrations peaked at 6 and 5 weeks of age, respectively. The TT4 concentration exceeded the adult cat reference interval (ACRI) in 54% (32/59) of samples at week 6, with the highest TT4 concentration being 7.1 µg/dl (91 nmol/l). Mean TT3 and fT3 concentrations also peaked at 6 weeks of age. Mean TT3 concentration started below the ACRI until 4 weeks of age, after which it remained within the ACRI. The mean fT3 concentration was within the ACRI at all ages. The mean TSH concentration did not differ across age groups and remained within the ACRI in nearly 100% of samples.
Conclusions and relevance
Peak TT4, fT4, TT3 and fT3 concentrations in healthy kittens at 5 and 6 weeks of age are likely due to changes in the maternal transfer of thyroid hormones after weaning and organ system development. Knowing healthy neonatal and pediatric thyroid hormone concentrations in a cohort of kittens might help a veterinarian interpret thyroid hormone levels when trying to rule out CH in a kitten with FKS.
Introduction
It is well known that many hematologic and biochemical differences exist between kittens and adult cats, and are clinically important when presented with a sick kitten.1,2 Fading kitten syndrome (FKS) is the term used to describe a kitten born healthy, but who subsequently becomes sick and often dies in the first few weeks of life.3–6 The cause of illness and death in kittens is infrequently determined owing to a lack of antemortem diagnostics and postmortem examination. Hypoxemia, prematurity, infection and low birth weight are predictors of illness and mortality in a kitten with FKS.3–7 Some fading kittens survive the neonatal period but persist with vague clinical signs and make a diagnosis and treatment plan challenging.1,3–7
Congenital hypothyroidism (CH) is defined as thyroid hormone deficiency present from birth that results from abnormal thyroid gland development, impaired thyroxine (T4) synthesis, deficiency of thyroid-stimulating hormone (TSH) or a peripheral defect in T4 metabolism, transport or action.8–11 Clinical signs include low birth weight, failure to gain weight, decreased suckling or appetite, hypothermia, decreased gastrointestinal motility and subsequent constipation, and abnormal musculoskeletal and neurologic development, all of which are similar to FKS clinical signs.1,4–7,12–14 As a result of the deleterious developmental effects of CH in humans, neonates are screened in the first week of life.12–15 A diagnosis of CH requires knowing the expected thyroid hormone concentrations at the particular age when testing is performed. However, in human infants with very low birth weights, the diagnosis of CH can be challenging because of a delayed rise in TSH concentration, necessitating serial measurements.12–17
Thyroid hormone concentrations in clinically healthy kittens are not well known. It has been reported that the total thyroxine (TT4) concentration in kittens is 2–3 times higher than that of an adult cat at 5–6 weeks of age, with the free thyroxine (fT4) concentration expected to follow a similar pattern. 18 Unpublished data on thyroid hormone concentrations in kittens aged 2–12 weeks presented over two decades ago demonstrated that the TT4 concentration remained within the adult cat reference interval (ACRI). 19 That study used outdated assays and did not include TSH concentration. The objective of this study was to describe TT4, fT4, total triiodothyronine (TT3), free triiodothyronine (fT3) and TSH concentrations in healthy kittens aged 2–16 weeks using current commercially available assays.
Materials and methods
Domestic shorthair cats were raised in the animal colony of the University of Pennsylvania School of Veterinary Medicine under the National Institutes of Health and US Department of Agriculture guidelines for the care and use of animals in research. All procedures were approved by an institutional animal care and use committee (protocol number 804197). Cats in the colony were bred for specific genetic diseases to develop treatments for children with the same disorders. Peripheral blood leukocytes were tested on the first day of life for genetic mutations according to previously published protocols. 20 Cats with no copies of the variant were classified as control cats. The queens were confirmed healthy with serial physical examinations and biochemical evaluation according to the American Association of Feline Practitioners’ recommendations. 21 The queens were housed at 21°C with 12-h light cycles, and given food and water ad libitum. All kittens included were confirmed clinically healthy with weekly physical examinations. Kittens were housed with the queen until weaning at 6 weeks of age. After weaning, kittens were housed with at least one other same-sex kitten under the same conditions previously described and given commercial kitten food ad libitum.
Once weekly, kittens were removed from their enclosures between 7 and 9 AM, weighed and examined. Blood was collected with jugular venipuncture, not exceeding 5% of blood volume, and placed into a glass tube. After clotting for 30 mins, the sample was centrifuged at 674 g for 10 mins. The serum was transferred to a plastic tube and stored at –80°C. All assays were performed in the same analytical run.
The TT4, fT4, TT3, fT3 and TSH concentrations were measured at the Michigan State University Veterinary Diagnostic Laboratory using assays previously validated in adult cats.22–27 The TT4 concentration was measured using a radioimmunoassay (GammaCoat; DiaSorin): ACRI 0.8–3.7 µg/dl (10–47 nmol/l). The fT4 concentration was measured by equilibrium dialysis using a radioimmunoassay (free T4-by ED; Antech Diagnostics): ACRI 0.8–4.1 ng/dl (10–53 pmol/l). The TT3 concentration was measured using a radioimmunoassay: ACRI 39.1–91.1 ng/dl (0.6–1.4 nmol/l). 26 The fT3 concentration was measured using a charcoal separation radioimmunoassay (Clinical Assays GammaCoat Free T3 125I RIA Kit; DiaSorin): ACRI 19.5–188.8 pg/dl (0.3–2.9 pmol/l). The TT4, fT4, TT3 and fT3 concentrations were reported in standard international units and converted to conventional units. The TSH concentration was measured using a canine chemiluminescent immunoassay validated for use in adult cats (IMMULITE 2000 Canine TSH; Siemens Healthcare Diagnostics): ACRI 0.00–0.38 ng/ml.22,27 The TSH concentration was reported in conventional units.
Statistical analyses were carried out using the mixed model procedure of SAS version 9.4 (SAS Institute). The model included the random effects of kitten and the fixed effects of age. The normality of the residuals was checked with normal probability and box plots, and homogeneity of variances with plots of residuals vs predicted values, to ensure no violation of model assumptions. Least square means were compared with a Bonferroni adjustment for multiple comparisons, and an alpha error of 0.05 was considered statistically significant.
Results
A total of 19 healthy kittens aged 2–16 weeks were sampled. The weight and sex were unavailable for three kittens (kittens E, K, L) (Table 1). Of the 16 remaining kittens, seven were male and nine were female (Table 1). Body weight increased as expected for growing kittens (Table 1). 28 Neither sex nor body weight were associated with the thyroid hormone concentrations. Eight kittens were sampled at more than one age (kittens A, B, C, D, I, J, M, N) and the remaining 11 kittens were sampled at one age (kittens E, F, G, H, K, L, O, P, Q, R, S) (Table 2). No kittens were sampled at 7, 9 or 12 weeks of age. There was insufficient serum for the measurement of TT3 and fT3 concentrations in one kitten at 2 weeks of age (kitten C) and fT3 concentration in one kitten at 14 weeks of age (kitten R) (Table 2).
Kitten sex, weight and identification for each age group

Values for weight are median (range)
Sex and weight not available
F = female; M = male
Thyroid hormone measurements across age groups

Values are mean ± SD
Superscript letters indicate statistically significant differences between age groups (P <0.05). aMean total T4 concentration at week 6 is greater than at weeks 2, 8, 13, 15 and 16; bmean total T3 concentration at week 2 is less than at weeks 5–16; cmean total T3 concentrations at weeks 3, 4 and 5 are less than at week 6; dmean total T3 concentration at week 4 is less than at weeks 10, 11, 13, 15 and 16; emean free T3 concentration at week 2 is less than at weeks 5, 6, 8, 10, 11 and 14; fmean free T3 concentration at week 4 is less than at week 6
Insufficient quantity of serum to run the assay
ACRI = adult cat reference interval; T3 = triiodothyronine; T4 = thyroxine; TSH = thyroid-stimulating hormone
The mean TT4 and fT4 concentrations were significantly different across age groups (P = 0.001 and P = 0.02, respectively). The mean TT4 concentration at week 6 was greater than the mean TT4 concentrations at weeks 2, 8, 13, 15 and 16 (P <0.035) (Table 2). The TT4 concentration was above the ACRI in 32/59 (54%) samples, with the highest number of samples above the ACRI at weeks 4, 5, 6 and 14 (Figure 1). The highest TT4 concentration was 7.1 µg/dl (91 nmol/l) at week 6. The fT4 concentration was above the ACRI in two samples at week 5 and three samples at week 6 (Figure 2). The highest fT4 concentration was 4.8 ng/dl (62 pmol/l) at week 6. The mean TSH concentration was not significantly different across age groups (Table 2). The TSH concentration remained within the ACRI, except for one sample of 0.39 ng/ml at week 10 (Figure 3).

Serum total thyroxine (TT4) concentrations in each kitten by age. Closed circles represent a single hormone concentration. When hormone concentrations are the same, a single closed circle is shown. The solid curved line represents the quadratic fit. The shaded area represents the adult cat reference interval of 10–47 nmol/l

Serum free thyroxine (fT4) concentrations in each kitten by age. Closed circles represent a single hormone concentration. When hormone concentrations are the same, a single closed circle is shown. The solid curved line represents the quadratic fit. The shaded area represents the adult cat reference interval of 10–53 pmol/l
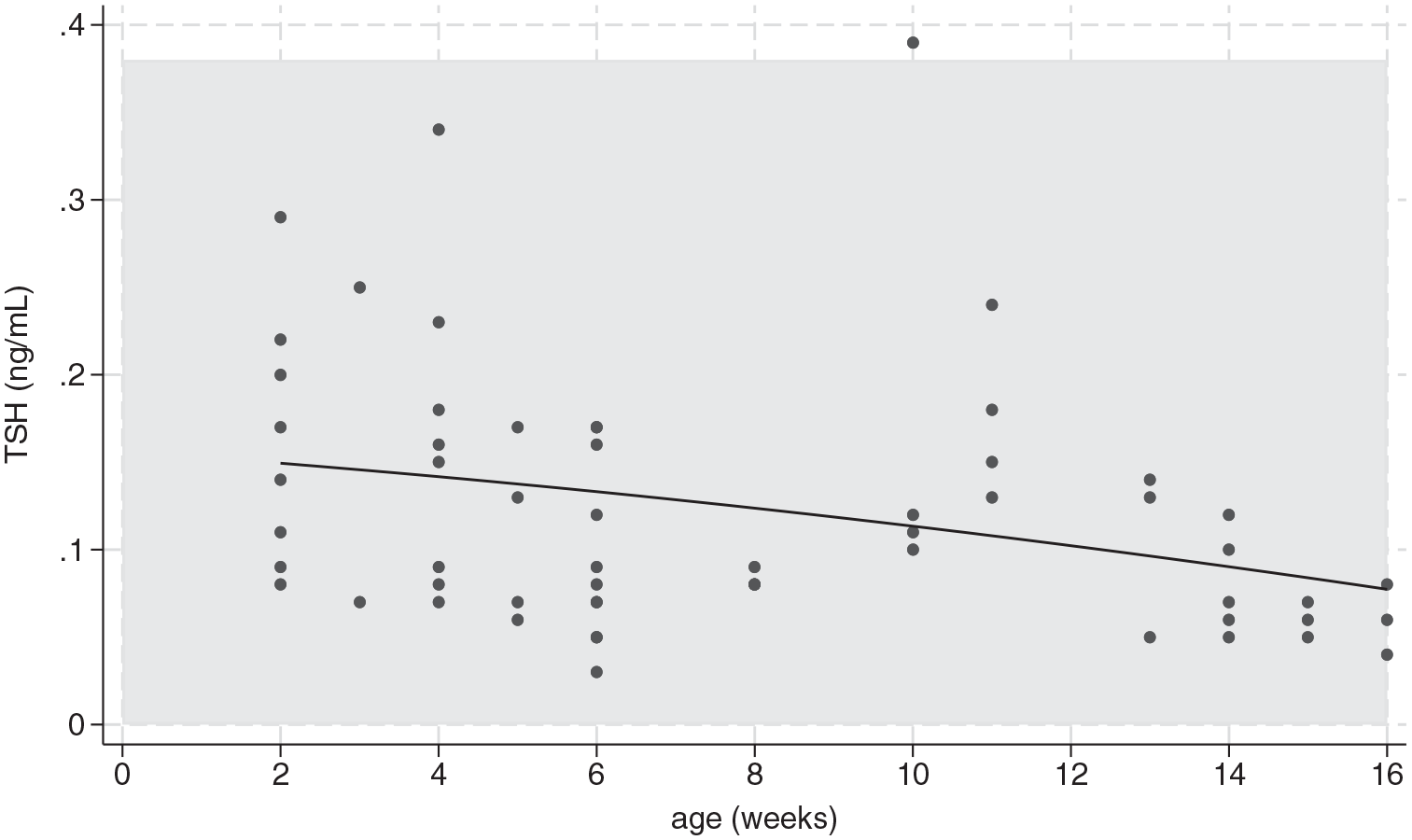
Serum thyroid-stimulating hormone (TSH) concentrations in each kitten by age. Closed circles represent a single hormone concentration. When hormone concentrations are the same, a single closed circle is shown. The solid curved line represents the quadratic fit. The shaded area represents the adult cat reference interval of 0.00–0.38 ng/ml
The mean TT3 and fT3 concentrations were significantly different across age groups (P = 0.0001 for both hormones). The mean TT3 concentration at week 2 was less than the mean TT3 concentrations at weeks 5–16, the mean TT3 concentrations at weeks 3, 4 and 5 were less than the mean TT3 concentration at week 6 and the mean TT3 concentration at week 4 was less than the mean TT3 concentrations at weeks 10, 11, 13, 15 and 16 (P <0.05) (Table 2). The TT3 concentration was less than the ACRI in 8/57 (14%) samples, and the highest TT3 concentration was 65.1 ng/dl (1.0 nmol/l) at weeks 6, 10 and 16 (Figure 4). The mean fT3 concentration at week 2 was less than the mean fT3 concentrations at weeks 5, 6, 8, 10 and 14, and the mean fT3 concentration at week 4 was less than the mean fT3 concentration at week 6 (P <0.05) (Table 2). The fT3 concentration was within the ACRI in all samples (Figure 5). The highest fT3 concentration was 169.2 pg/dl (2.6 pmol/l) at week 6 (Figure 5).

Serum total triiodothyronine (TT3) concentrations in each kitten by age. Closed circles represent a single hormone concentration. When hormone concentrations are the same, a single closed circle is shown. The solid curved line represents the quadratic fit. The shaded area represents the adult cat reference interval of 0.6–1.4 nmol/l

Serum free triiodothyronine (fT3) concentrations in each kitten by age. Closed circles represent a single hormone concentration. When hormone concentrations are the same, a single closed circle is shown. The solid curved line represents the quadratic fit. The shaded area represents the adult cat reference interval of 0.3–2.9 pmol/l
Discussion
The TT4, fT4, TT3, fT3 and TSH concentrations were measured in healthy kittens aged between 2 and 16 weeks. The mean TT4 concentration started above the ACRI at 2 weeks, peaked at 6 weeks and decreased to the ACRI at 15 weeks. At 6 weeks, 50% of the TT4 concentrations were above the ACRI, with the highest value nearly twice the upper limit of the reference interval (RI). The mean fT4 concentration was within the ACRI at all ages and peaked at 5 weeks, with only five kittens exceeding the ACRI at 5 and 6 weeks. There was no difference in mean TSH concentration across age groups and it remained within the ACRI in all but one kitten at 10 weeks. The mean TT3 concentration was below the ACRI at 2–4 weeks, after which it remained within the ACRI. The mean fT3 concentration was within the ACRI at all ages. Both the mean TT3 and fT3 concentrations had a similar peak to TT4 at 6 weeks. The increases in TT4, fT4, TT3 and fT3 concentrations are likely due to the changing demand for T4 in growing, healthy kittens.
During mammalian gestation, there is minimal maternal transfer of T4 to the fetus, no maternal transfer of TSH to the fetus and no fetal transfer of T4 and TSH to the mother. 29 These findings demonstrate that the fetal hypothalamic-pituitary-thyroid axis develops autonomously in utero. 29 In humans, breast milk contains high levels of T4 and TSH until the age of approximately 2 months. 30 This is thought to allow the neonate’s peripheral tissue deiodination and utilization of T4 to mature despite a functional hypothalamic-pituitary-thyroid axis. 30 In dairy cows, the amount of T4 in the milk accounts for only 4–5% of the calf’s T4 requirement, with no detectable T4 in the milk at 12 weeks after parturition. 31 Although the amount of T4 in the queen’s milk is unknown, there is likely a small amount similar to other mammals. A kitten should produce their own T4 once they stop nursing, resulting in an increase in measured TT4 and fT4 concentrations. In addition, appropriate tissue utilization of T4 should occur, causing an increase in measured TT3 and fT3 concentrations. In the studied kittens, there was a steady increase in the fT4 and TT4 concentrations up to the ages of 5 and 6 weeks, respectively, paralleled by a rise in the TT3 and fT3 concentrations. These findings could be due to neonatal utilization of maternal T4 while nursing, followed by a transition to using their own T4 once they stop nursing.
Thyroxine is necessary for many aspects of fetal and pediatric development. Neonatal kittens are poikilothermic, with their body temperature closely matching environmental temperature until approximately 5 weeks of age. 32 Between 5 and 6 weeks of age, a kitten’s metabolic rate increases to maintain body temperature, requiring an increase in thyroid hormone production to do so.32,33 Although it is unknown in kittens, gastrointestinal motility in puppies is primarily dependent on luminal pressure changes until the enteric nervous system begins to develop at 5 weeks of age. 32 Abnormal gastrointestinal motility can lead to decreased appetite and constipation, clinical signs commonly seen with FKS and CH.3–11 In addition, the kitten’s spinal cord develops quickly, with myelination of the lateral fibers starting at approximately 5 weeks of age and maturing by 12 weeks, and the postural and balancing system reaches maturity at 6–8 weeks of age. 34 Although many specifics of neonatal and pediatric kitten development are yet to be determined, the increases in TT4, fT4, TT3 and fT3 concentrations between 2 and 6 weeks of age in this study could be due to the increased demand for T4 in developing organ systems.
There was no difference in the TSH concentration across age groups. The TSH concentration is expected to decrease or increase with increasing or decreasing thyroid hormone concentration, respectively; however, this was not documented in this cohort of kittens. In the first 6 weeks of life in human infants, the TSH concentration is considered variable because it does not demonstrate changes consistent with an intact hypothalamic-pituitary-thyroid axis, in that it does not respond via negative feedback to changes in the TT4, fT4 and TT3 concentrations.29,35 Although it is unknown if neonatal and pediatric kittens have a similar immature hypothalamic-pituitary-thyroid axis that lacks the ability to respond to negative feedback, this could potentially, if present, contribute to the lack of significant change in TSH concentration across age groups. An additional cause for the lack of significant change in TSH concentration could come from the assay used. The canine TSH assay has been validated and is commonly used in adult cats, with 0.3 ng/ml as the limit of quantification for the assay. 36 Although all kittens sampled had detectable TSH concentrations using the canine TSH assay, many of them were less than 0.3 ng/ml (Table 2), introducing the potential for increased intra-assay variability. 36 In humans, the initial screening for primary hypothyroidism is often a heal-prick test, requiring only a drop of blood applied to a special filter paper to detect TSH.14,17 If elevated, additional confirmatory tests are subsequently performed.14,17 Future studies should include measuring serum TSH concentrations in neonatal and pediatric kittens using the commercially available feline-optimized TSH assay and potentially creating a diagnostic test similar to the heal-prick test in infants, which would allow for easier screening of neonatal kittens with only a drop of blood.
Primary hypothyroidism is diagnosed based on finding a low TT4 or fT4 concentration and a concurrently high TSH concentration.14,17 In humans, the incidence of hypothyroidism in very low birth weight infants is 14-fold higher than in normal birth weight infants; however, low birth weight infants can have normal to low TT4 and fT4 concentrations with normal TSH concentrations that do not increase until 5–8 weeks of age. These circumstances can make an early diagnosis of primary hypothyroidism very difficult.12–17 Some of these infants with a delayed rise in the TSH concentration have transient hypothyroidism that requires supplementation with levothyroxine for only a few months, a phenomenon referred to as atypical hypothyroidism.14,17 Additional studies would need to be performed to determine whether kittens could experience a syndrome similar to atypical hypothyroidism in humans and whether it could be a contributor to FKS.
Thyroid hormone concentrations measured in this cohort of kittens support the previous statement that the TT4 concentration in kittens is above the ACRI; however, this finding was not consistent in all kittens, nor was it more than twice the upper limit of the ACRI in those kittens with an increased TT4 concentration. 18 It was also thought that the fT4 concentration in healthy kittens would follow a similar pattern to the TT4 concentration. 18 Although the changes in fT4 did mirror those of TT4 in this study, only five fT4 concentrations were above the ACRI. In addition, the results of this study contrast with previously unpublished data stating that healthy kitten thyroid hormone concentrations remain within the ACRI up to 12 weeks of age; however, this could be a consequence of the different hormone assays used between studies. 19 Additional studies with larger numbers of healthy kittens are needed to establish an RI for thyroid hormone concentrations in both neonatal and pediatric kittens.
This study has several important limitations. First, the small number of samples at each time point introduces a type II error and precludes any speculation on the RIs for thyroid hormones in kittens. To determine this, at least 120 samples at each time point would be required. 37 Unfortunately, the availability of neonatal and pediatric kittens and the cost of the assays limit the practicality of such a study. Second, the sex and weight of three kittens were unavailable at the time of statistical analysis. It is unknown whether this information would have led to any relevant effect of sex or weight on the thyroid hormone concentrations. Third, the kittens were not screened with a complete blood count and a serum chemistry panel before obtaining blood for measurement of serum thyroid hormone concentrations. Without this, the presence of illness not detected on routine physical examination cannot be excluded. Finally, there were a few time points (7, 9 and 12 weeks) when no samples were collected. The addition of results at these time points may have changed the trend in thyroid hormone concentrations seen in these kittens.
Conclusions
In a cohort of 19 healthy kittens, the mean TT4 concentration peaked at 6 weeks, was no more than twice the upper limit of the ACRI and returned to the ACRI at 15 weeks. The mean fT4 concentration peaked at 5 weeks, with concentrations only exceeding the ACRI at 5 and 6 weeks of age. The TT3 and fT3 concentrations did not exceed the ACRI at any age; and, despite peaks in TT4, fT4, TT3 and fT3, the TSH concentration did not significantly change across age groups. CH in kittens is one of the most overlooked disorders in veterinary medicine (ML Casal, unpublished data) and FKS has many clinical signs that overlap with CH in neonatal and pediatric kittens. Additional studies evaluating thyroid hormone concentrations in kittens and the creation of diagnostic tests that could be used to screen kittens for CH are needed. The small blood volume that can be safely obtained in these kittens often dictates the diagnostic tests that can be performed, requiring the veterinarian to choose a single diagnostic test in a sick kitten. If the veterinarian obtains a TT4 concentration in a sick kitten, an understanding of normal thyroid hormone concentrations in a cohort of healthy kittens may help with the interpretation and the decision to submit further thyroid hormone function tests, such as fT4 and TSH, to rule out CH in a kitten with FKS.
Footnotes
Acknowledgements
The authors thank Dr George Moore DVM, PhD, Dip ACVIM-SAIM, Dip ACVPM-Epi for his assistance with statistical analysis. The authors would also like to thank the entire staff in the Section of Medical Genetics at the University of Pennsylvania School of Veterinary Medicine for their assistance with animal care and venipuncture.
Author note
The results of this paper were presented in part at the 2016 ACVIM Forum.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclose receipt of the following financial support for the research and/or publication of this article: The study was financed by the Society for Comparative Endocrinology, Jack Oliver Graduate Research Award.
Ethical approval
The work described in this manuscript involved the use of experimental animals and the study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
