Abstract
Objectives
Although radioiodine (131I) is the treatment of choice for feline hyperthyroidism, 131I-dosing protocols commonly induce iatrogenic hypothyroidism and expose azotaemia. A recently reported patient-specific 131I dosing algorithm minimised the risk of 131I-induced hypothyroidism and azotaemia, while maintaining high cure rates. The aim of the study was to report results of 131I treatment in a European population of hyperthyroid cats using this patient-specific dosing algorithm.
Methods
This prospective case series (before-and-after study) evaluated 165 hyperthyroid cats referred for 131I treatment. All cats had serum concentrations of thyroxine (T4), triiodothyronine (T3) and thyroid-stimulating hormone (TSH) measured (off methimazole ⩾1 week). Thyroid volume and percentage uptake of 99mTc-pertechnetate (TcTU) were determined using thyroid scintigraphy. An initial 131I dose was calculated by averaging dose scores for T4/T3 concentrations, thyroid volume and TcTU; 70% of that composite dose was then administered. Twenty-four hours later, percentage 131I uptake was measured, and additional 131I administered as needed to deliver an adequate radiation dose to the thyroid tumour(s). Serum concentrations of T4, TSH and creatinine were determined 6–12 months later.
Results
Median calculated 131I dose was 2.15 mCi (range 1.2–7.5), with only 51 (30.9%) receiving ⩾2.5 mCi. Of 165 cats, 124 (75.2%) became euthyroid, seven (4.2%) became overtly hypothyroid, 27 (16.4%) became subclinically hypothyroid and seven (4.2%) remained hyperthyroid. A higher proportion of overtly (85.7%) and subclinically (26.9%) hypothyroid cats developed azotaemia than euthyroid cats (13.6%; P = 0.0002). Hypothyroid cats were older (P = 0.016) and more likely to have detectable TSH concentrations (P = 0.025) and symmetrical bilateral distribution of 99mTc-pertechnetate uptake (P = 0.0002), whereas persistently hyperthyroid cats had higher severity scores (P = 0.012).
Conclusions and relevance
Our results confirm that 131I dosing with this new algorithm results in high cure rates, with a lowered prevalence of 131I-induced overt hypothyroidism and azotaemia. Age, serum TSH concentrations, bilateral, symmetrical uptake and severity score help predict outcome.
Keywords
Introduction
Radioiodine (131I) is considered the treatment of choice for hyperthyroidism in cats. The goal of 131I therapy is to restore euthyroidism with a single dose of radiation. However, most treatment protocols concentrate on ‘curing’ hyperthyroidism without regard for the development of iatrogenic hypothyroidism, which results in a high prevalence (30–60%) of cats becoming hypothyroid within 6 months of 131I treatment.1–6 Because iatrogenic hypothyroidism can worsen existing azotaemia, enhance progression of kidney disease and shorten survival time in cats with chronic kidney disease, clinicians should attempt to minimise the occurrence of iatrogenic hypothyroidism.7–11
The optimal 131I dose that will maximise the chance of curing hyperthyroidism while simultaneously minimising the risk of developing iatrogenic hypothyroidism is highly variable between individual cats and is therefore difficult to calculate. Peterson et al 12 recently published the results of a large cohort of hyperthyroid cats treated in New York with individualised 131I doses using an objective, novel algorithm. This algorithm calculates the lowest likely curative 131I dose based on results of pretreatment serum thyroid hormone concentrations, quantitative thyroid scintigraphy and percentage thyroid 131I uptake, thereby reducing the risk of iatrogenic hypothyroidism and azotaemia while maintaining a high cure rate (>95%). 12
A follow-up study 13 on the same group of 131I-treated cats identified several pretreatment factors that could help predict which cats develop iatrogenic hypothyroidism or remain persistently hyperthyroid. 13 Cats that developed 131I-induced hypothyroidism were older, female and more likely to have detectable thyroid-stimulating hormone (TSH) concentrations, bilateral and homogeneous 99mTc-pertechnetate uptake on thyroid scintigraphy, milder severity and a higher 24 h percentage 131I uptake. Cats that were more likely to remain hyperthyroid (treatment failure) tended to be younger and more likely to have severe thyroid disease and lower 131I uptake. 13
To examine whether the results obtained in the initial study 12 could be replicated, we evaluated this dosing algorithm for individualised 131I treatment of 165 hyperthyroid cats in Madrid, Spain. Specifically, we used the results of serum thyroid hormone concentrations, quantitative thyroid scintigraphy and percentage thyroid 131I uptake to calculate the lowest possible individual 131I doses, and determined thyroid status (ie, euthyroid, hypothyroid or persistent hyperthyroid), as well as the presence of azotaemia, at 6–12 months after 131I treatment.
Materials and methods
Animals
All hyperthyroid cats referred to Iodocat in Madrid, Spain for treatment with 131I over the 4.8 year period from September 2016 to July 2021 were evaluated for inclusion in this prospective, consecutive controlled case series (before-and-after study). 14 To be eligible for inclusion, untreated hyperthyroid cats underwent an evaluation that included review of the past medical record, complete physical examination, routine laboratory testing (complete blood count, serum biochemical profile, complete urinalysis), determination of serum concentrations of total thyroxine (T4), triiodothyronine (T3) and TSH (free T4 was also measured in cats with normal serum total T4 and T3 concentrations),15–17 and qualitative and quantitative thyroid scintigraphy.18,19 In cats treated with methimazole, owners discontinued the drug 1–2 weeks before evaluation. Owners feeding a low-iodine diet (Hill’s Prescription Diet y/d Feline) were instructed to feed an iodine-replete diet for at least 4 weeks before treatment.
We excluded hyperthyroid cats with pre-existent azotaemia (defined as serum creatinine >2.0 mg/dl), cats in which methimazole was not discontinued at least a week before 131I treatment and cats with suspected thyroid carcinoma20–22 that were treated with large doses of 131I for multifocal or metastatic tumour ablation.
Protocol for calculating individualised 131I doses
On the day of admission, each cat had blood drawn for determination of serum concentrations of T4, T3 and TSH (Figure 1), using previously described assays validated for use in cats.2,12,15,16 Cats then underwent thyroid scintigraphy by injecting 3–5 mCi (111–185 MBq) of sodium 99mTc-pertechnetate (99mTcO−4) into the saphenous vein and imaging 60 mins later.18,19 Qualitative analysis allowed us to classify cats into unilateral vs bilateral patterns of disease, and helped exclude thyroid carcinoma.18,21 In cats with bilateral disease, we also classified these as having a symmetrical vs asymmetrical distribution of 99mTcO−4 uptake (ie, cats with symmetrical uptake had an equivalent count density or thyroid-to-salivary ratio in both thyroid lobes), where those with asymmetrical distribution had dissimilar count densities in the two lobes.13,18 Quantitative thyroid scintigraphy also allowed calculation of the percent thyroidal uptake of the injected sodium 99mTc-pertechnetate (TcTU) and determination of the volume of each cat’s thyroid tumour (Figure 1), as previously described.12,18,19

Flowchart showing protocol for calculating initial (day 1), composite radioiodine (131I) dose based on three measures of disease severity (serum thyroxine and triiodothyronine concentrations, percentage thyroidal uptake of sodium 99mTc-pertechnetate and thyroid tumour volume). On day 2, thyroid 131I uptake was measured and additional 131I activityadministered as needed
The full details of the 131I dosing algorithm used to treat the cats in our study have been previously reported.12,23 In brief, we calculated individual 131I doses for all cats, based on pretreatment serum T4 and T3 concentrations, estimated thyroid volume (measured from scintigraphic image) and the percentage uptake of 99mTcO4. These four variables contributed to a severity score, from which we calculated the 131I dose (Table 1). On day 1, we administered 70% of this calculated dose subcutaneously. Twenty-four hours later, we measured neck radioactive counts using a survey meter (RM1001-RD Gamma Radiation Monitor and Dosimeter; Lamse) by placing its detector directly on the skin surface over the cats’ hottest thyroid nodule and calculated the percentage 131I uptake by the thyroid gland, as previously described.12,23 Based on this percentage uptake value, we administered additional 131I subcutaneously (as needed) to provide sufficient 131I to meet the final calculated dose (Table 1).
Protocol for 131I dose calculation, based on measured thyroid tumour volume, serum thyroxine (T4) and triiodothyronine (T3) concentrations, 99mTc-pertechnetate (TcTU) and 24 h thyroid 131I uptake measurements
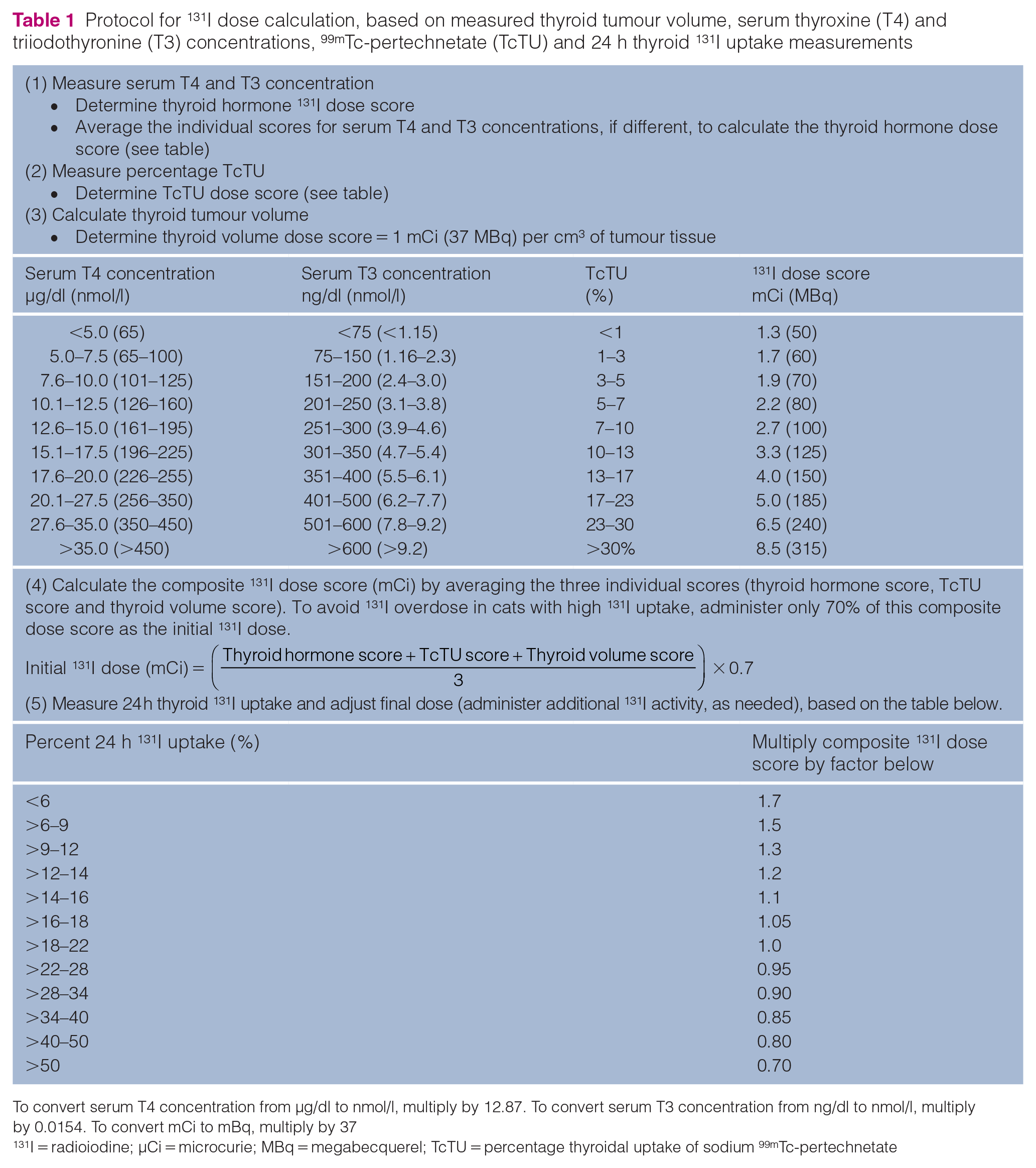
To convert serum T4 concentration from µg/dl to nmol/l, multiply by 12.87. To convert serum T3 concentration from ng/dl to nmol/l, multiply by 0.0154. To convert mCi to mBq, multiply by 37
131I = radioiodine; µCi = microcurie; MBq = megabecquerel; TcTU = percentage thyroidal uptake of sodium 99mTc-pertechnetate
Follow-up monitoring and testing after 131I treatment
After 131I treatment, all cats were scheduled for evaluation at 6 months, with follow-up serum concentrations of T4, TSH and creatinine determined at this visit. To maintain enrolment compliance, the owners of 131I-treated cats could either return to our clinic or have the follow-ups performed by the referring veterinarian, with samples submitted directly to the designated reference veterinary diagnostic laboratory (IDEXX Laboratories, Madrid, Spain).
Classification thyroid subgroups and azotaemia after 131I treatment
Based on the serum concentrations of T4 and TSH measured at a median time of 6 months after treatment with 131I, we classified the cats’ thyroid status into one of four categories: euthyroid (T4, 1.0–3.8 µg/dl; TSH ⩽0.30 ng/ml), overtly hypothyroid (T4 <1.0 µg/dl; TSH >0.30 ng/ml), subclinically hypothyroid (T4, 1.0–3.8 µg/dl; TSH >0.30 ng/ml) and persistently hyperthyroid (T4 ⩾ 3.9 µg/dl; TSH <0.03 ng/ml), as previously defined.2,9,12,24 We also classified cats as azotaemic or non-azotaemic based on the serum creatinine concentration, with azotaemia defined as a serum creatinine concentration above our institution’s reference interval (>2.0 mg/dl). We excluded cats lost to follow-up or only tested sooner than 6 months after 131I treatment, to prevent misclassification of cats that might still be recovering from TSH suppression secondary to the previous hyperthyroid state (thereby missing a subsequent diagnosis of iatrogenic hypothyroidism).12,23 In addition, as it can take 3–6 months for masked azotaemia to develop after a 131I-treated cat becomes euthyroid,10,25,26 our minimum follow-up time of 6 months provided a period long enough for azotaemia to develop if the cat did indeed have underlying kidney disease.
Data analysis
Data were assessed for normality by the D’Agostino–Pearson test and by visual inspection of graphical plots. 27 Data were not normally distributed; therefore, all analyses used were performed using non-parametric tests. Results for continuous data (eg, serum T4, T3, TSH and creatinine concentrations) are expressed as median (interquartile range [IQR]) and represented graphically as box and whisker plots (Tukey method). 28 We compared continuous variables between groups by the Mann–Whitney U-test or the Kruskal–Wallis test. We compared categorical variables between groups using the χ2 test or Fisher’s exact test, where appropriate, followed by the Holm–Bonferroni correction procedure for within-group comparison.
For further analyses, the untreated hyperthyroid cats were categorised into mild-to-moderate disease (131I dose <2.5 mCi) and severe disease (dose scores >2.5 mCi), based on their composite 131I dose (severity) scores.12,13
For all analyses, statistical significance was defined as P ⩽0.05. Statistical analyses were performed using proprietary statistical software (GraphPad Prism, version 9.0; GraphPad Software).
Results
Patient characteristics
Over the 5.5 year study period, we treated 193 hyperthyroid cats that were eligible for inclusion and enrolled in the study; 28 cats were lost to follow-up after <6 months of 131I treatment and were excluded from study (Figure 2). The remaining 165 cats were re-examined and retested at a median of 6 months (IQR 6–6) after 131I treatment. Almost all cats (n = 158/165) were rechecked at 6 months, whereas the remining seven (4.2%) were re-evaluated at 12 months after 131I treatment.
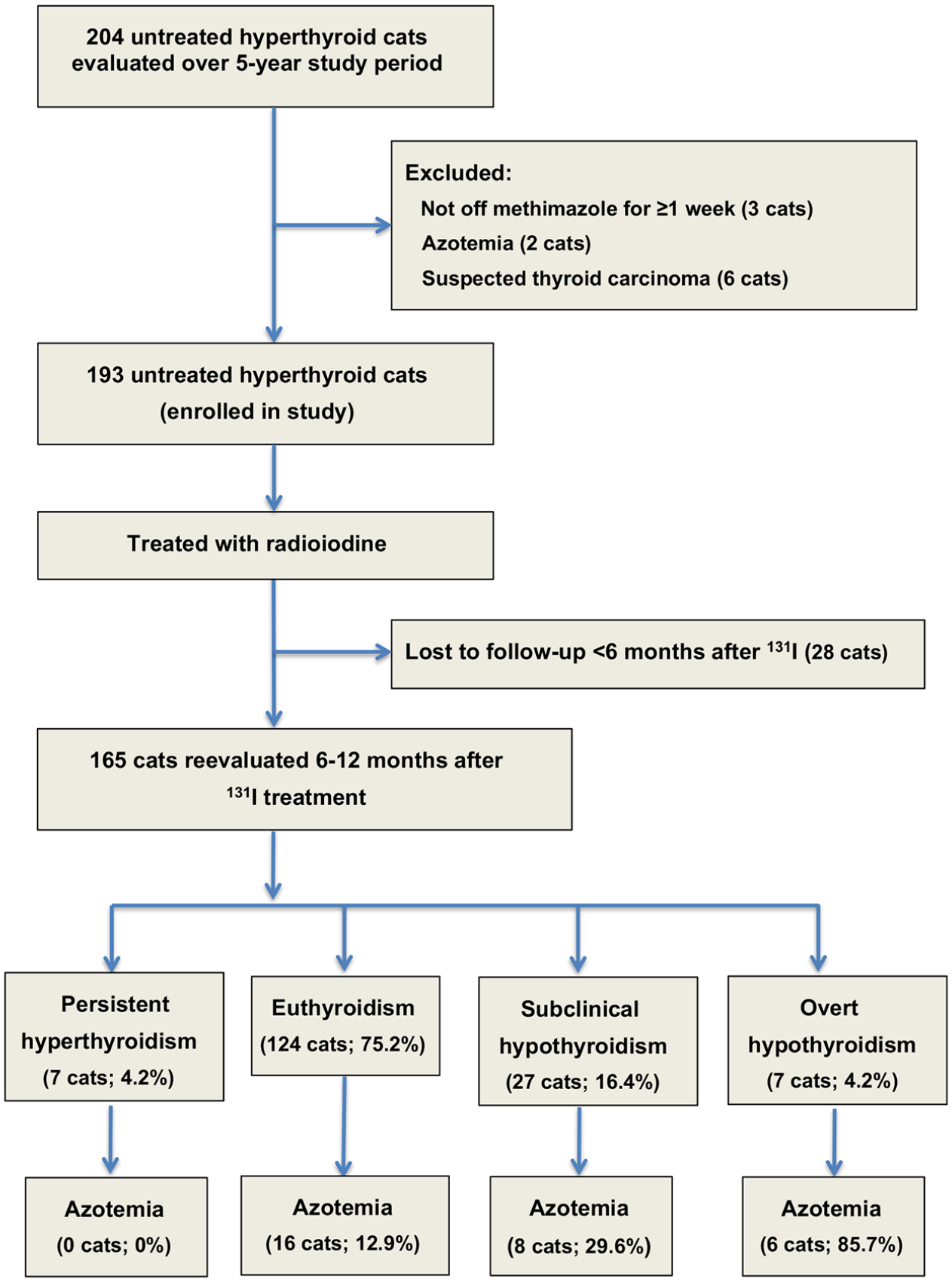
Flowchart for enrolment of hyperthyroid cats, separated into four thyroid outcome groups, as well as the development of azotaemia after treatment with radioiodine
The 165 hyperthyroid cats ranged in age from 3 to 20 years (median 12; Table 2). Cats that developed 131I-induced overt and subclinical hypothyroidism were older than the euthyroid cats (Table 2).
Pretreatment signalment data and serum thyroid hormone concentrations in 165 radioiodine (131I)-treated cats, divided into in three thyroid outcome groups

All continuous data (age, body weight, time from diagnosis, serum thyroxine [T4], thyroid-stimulating hormone [TSH] and creatinine) are expressed as median (interquartile range). All qualitative data are expressed as ratio (breed, sex) or n (%) of cats (detectable TSH concentration).
Reference intervals: T4 = 1.0–3.8 μg/d; triiodothyronine (T3) = 30–80 ng/dl; TSH <0.03–0.3 ng/ml. Values with the same superscript letters are significantly different to one another
Kruskal–Wallis test, followed by Dunn multiple comparisons test
χ2 test, followed by the Holm–Bonferroni correction procedure for within-group comparison
Breeds included domestic longhair and shorthair (n = 143; 86.7%), Persian (n = 7), Siamese (n = 6), Ragdoll (n = 4 ), Norwegian Forest Cat (n = 3), Maine Coon (n = 1) and British Shorthair (n = 1). Seventy-eight (47.3%) were male and 87 (52.7%) were female; all had been neutered (Table 2). Body weight ranged from 1.9 to 8.3 kg (median 4.0; Table 2); 58 (35.2%) cats were considered underweight, 77 (46.7%) had an ideal body condition score and 30 (18.1%) were considered overweight.
The time from diagnosis of hyperthyroidism to 131I treatment ranged from 2 days to 6.5 years (median 80 days; Table 2). Of the 165 cats, 101 (61.2%) had never received methimazole treatment and 64 (38.8%) had been treated with methimazole for a median time of 60 days. In all methimazole-treated cats, the drug was discontinued ⩾1 week (median 14 days; range 7–400 days) before treatment with 131I. Eight (4.8%) of the 165 cats had been fed a low-iodine diet (Hill’s Prescription Diet y/d Feline), but this food was changed to an iodine-replete diet 4 weeks prior to evaluation.
Pretreatment serum T4, T3, TSH and creatinine concentrations
Almost all untreated hyperthyroid cats (n = 160/165; 97%) had high serum T4 concentrations (Figure 3a). All five cats with normal serum T4 concentrations had high serum-free T4 concentrations, as well as increased radionuclide uptake on thyroid scintigraphy (ie, one or more hot thyroid tumour nodules). Cats that remained hyperthyroid after 131I had higher pretreatment serum T4 concentrations than cats that became euthyroid or hypothyroid (Table 2).

Boxplots of serum thyroid hormone and thyroid-stimulating hormone (TSH) concentrations in 165 hyperthyroid cats before treatment with radioiodine (131I). (a) Thyroxine (T4); (b) triiodothyronine (T3); and (c) TSH. Boxes represent the interquartile range (IQR; 25th to 75th percentile). The horizontal bar in each box represents the median value. The whiskers indicate the range of data values unless outliers are present, in which case the whiskers extend to a maximum of 1.5 times the IQR. 28 Such outlying data points are represented by open circles. The shaded area indicates the reference interval
Before treatment, 132 cats (80%) had high serum T3 concentrations (Figure 3b). Cats that remained persistently hyperthyroid after 131I had higher pretreatment serum T3 concentrations than cats that became euthyroid or hypothyroid (Table 2).
Serum TSH concentration was below the limit of detection (<0.03 ng/ml) in 161/165 cats (97.6%; Figure 3c). Cats that developed 131I-induced hypothyroidism had a higher prevalence of detectable serum TSH concentrations (⩾0.03 ng/ml) than the cats that became euthyroid (Table 2). None of the cats with persistent hyperthyroidism had a detectable serum TSH concentration.
Thyroid scintigraphy findings
On qualitative scintigraphy, 85 (51.5%) hyperthyroid cats had bilateral disease, whereas 80 cats had unilateral thyroid nodules. Seventy-seven (90.6%) of the 85 hyperthyroid cats with bilateral disease had an asymmetrical distribution of 99mTcO−4 uptake, but cats that developed 131I-induced hypothyroidism were more likely to have bilateral, symmetrical uptake of the radionuclide (Table 3).
Thyroid scintigraphy variables and measurements in 165 radioiodine (131I)-treated cats, divided into three thyroid outcome groups

Qualitative data are expressed as ratio. Continuous data are expressed as median (interquartile range). Reference intervals: thyroid:salivary ratio 0.5–1.5; percentage thyroidal uptake of 99mTc-pertechnetate (TcTU) 0.05–0.8; thyroid volume 0.2–1.0 cm3. Values with the same superscript letters are significantly different to one another
Kruskal–Wallis test, followed by Dunn’s multiple comparisons test
χ2 test, followed by the Holm–Bonferroni correction procedure for within-group comparison
The thyroid nodules of all hyperthyroid cats had increased intensity of uptake, as evidenced by high thyroid: salivary gland ratio (Table 3) and a high TcTU (Figure 4a). Most untreated hyperthyroid cats (n = 149/165; 90.3%) had an increased thyroid tumour volume (Figure 4b). Cats with persistent hyperthyroidism had higher TcTU than cats that became euthyroid or hypothyroid (Table 3). Likewise, cats with persistent hyperthyroidism also had higher thyroid tumour volumes than cats that became euthyroid or hypothyroid (Table 2).

Boxplots of quantitative scintigraphic results and 24 h thyroidal radioiodine (131I) uptake used in our algorithm for calculating individual 131I doses in 165 hyperthyroid cats. (a) Percentage thyroidal uptake of 99mTc-pertechnetate (TcTU); (b) thyroid volume; and (c) percentage 131I uptake. See Figure 3 for key
Individualised 131I dose calculations
As calculated by averaging the thyroid hormone score, the TcTU score and thyroid volume score (Table 1), the composite 131I dose score for the 165 cats ranged from 1.1 to 7.1 mCi (median 2.1; Table 4). On day 1, cats received approximately 70% of this composite dose score (median 1.5 mCi; IQR 1.2–1.7). After adjusting the composite 131I dose for the 131I uptake value (Table 1, Figure 4c), the 156 cats with 131I uptake values <50% received a second 131I dose on day 2; the nine cats with values >50% received no additional 131I.
Dosing score variables, 24 h percentage radioiodine (131I) uptake and final calculated 131I dose in 165 hyperthyroid cats, divided into three outcome groups

Data are expressed as median (interquartile range). Values with the same superscript letters are significantly different to one another
Kruskal–Wallis test, followed by Dunn multiple comparisons test
T4 = thyroxine; T3 = triiodothyronine; TcTU = percentage thyroidal uptake of 99mTc-pertechnetate
The total 131I dose administered to the 165 cats ranged from 1.2 to 7.0 mCi (median 2.15 mCi). Of the 165 cats, only 51 (30.9%), 30 (18.2%) and 11 (6.7%) received a 131I dose ⩾2.5 mCi, ⩾3 mCi and ⩾4 mCi, respectively.
Cats remaining persistently hyperthyroid had higher thyroid hormone scores, TcTU scores and thyroid volume scores than did cats that became euthyroid or hypothyroid (Table 4). None of these three individual scores differed between the euthyroid or hypothyroid cats. Similarly, persistently hyperthyroid cats had higher composite 131I dose scores than did cats that became euthyroid or hypothyroid. Again, the composite 131I dose score did not differ between euthyroid and hypothyroid cats (Table 4).
Thyroid and renal outcome status after 131I treatment
After 131I treatment, 124 (75.2%) cats became euthyroid, seven (4.2%) developed overt hypothyroidism, 27 (16.4%) cats had subclinical hypothyroidism and seven (4.2%) remained hyperthyroid (Figures 2 and 5a,b). A higher proportion of cats with severe hyperthyroidism (dose score >2.5 mCi) failed treatment and remained persistently hyperthyroid (n = 6/48; 12.5%) than cats with mild-to-moderate disease (n = 1/117 [0.9%]; P = 0.003).

Boxplots of serum thyroid (T4) hormone and creatinine concentrations in 165 hyperthyroid cats, treated with individual radioiodine (131I) doses calculated with our algorithm, divided into four thyroid outcome groups. (a) T4; (b) thyroid-stimulating hormone (TSH); and (c) creatinine. See Figure 3 for key
New azotaemia (serum creatinine >2.0 mg/dl) developed in 30 (18.2%) of the 165 cats. The prevalence of post-treatment azotaemia was higher in the cats with overt (n = 6/7; 85.7%) and subclinical (n = 7/26; 26.9%) hypothyroidism than in the cats that remained euthyroid (n = 17/125; 13.6%) or cats remaining persistently hyperthyroid (n = 0/7; 0%) after 131I treatment (Figure 5c; P = 0.0016).
Discussion
Using a recently reported patient-specific 131I dosing algorithm 12 to treat hyperthyroid cats in Spain, we replicated the original findings, curing almost all cats, with a relatively low prevalence of both 131I-induced hypothyroidism and azotaemia. Our results substantiated that this dosing method (which calculates the ‘lowest possible’ individual 131I dose provides consistent, repeatable results at different treatment sites when used by different investigators.
Using this 131I dosing algorithm, we induced euthyroidism in 75% of our cats (vs 74.8% in the original study), with 15% developing mild (subclinical) hypothyroidism (17.1% in the original study). Fewer than 5% of our cats developed overt hypothyroidism (low T4 with high TSH concentration) or remained persistently hyperthyroid after 131I treatment (vs 4.1% and 4%, respectively, in the original study). 12 Because of the lowered prevalence of overt hypothyroidism, fewer cats developed post-treatment azotaemia (18% vs 19% in the original study 12 ) than in most other studies (26–41%), where investigators administered higher 131I doses.1–5
Although this study confirms that the approach is generalisable beyond the one centre where it originated, both Spanish investigators received limited training at the Animal Endocrine Clinic in New York to learn how to measure the quantitative scintigraphic variables (eg, thyroid volume and TcTU) and preform the 131I thyroid uptakes. That said, the Spanish investigators examined the 165 cats in this study, performed all the thyroid scintigraphic scans and 131I thyroid uptakes, calculated all of the results for the 131I dosing algorithm and treated all of the cats without the direct input from the senior author (who developed the algorithm).
Cats in this study had more severe thyroid disease than the cats in the original study. 12 Cats were thinner (median body weight 4.0 kg vs 4.4 kg; P <0.01), had been hyperthyroid for longer before 131I treatment (median 80 days vs 65 days; P = 0.02), had higher serum concentrations of both T4 (10.5 µg/dl vs 8.9 µg/dl; P = 0.02) and T3 (156 ng/dl vs. 133 ng/dl; P = 0.04) and had a higher cumulative 131I dosing (or severity) score (2.07 mCi vs 1.87 mCi; P = 0.03). As might be expected, this led to a higher 131I dose being administered to the cats in this study (2.15 mCi vs 1.87 mCi; P <0.01). Despite these differences, most cats still had relatively mild disease, with >70% needing a relatively small 131I dose (<2.5 mCi) to achieve a cure. Furthermore, despite these differences, the cats responded appropriately to this 131I dosing algorithm, demonstrating that the tailoring of dosing to severity achieves the desired result in most cats.
The cats in the current study showed many of the risk factors for iatrogenic hypothyroidism and persistent hyperthyroidism (treatment failure) identified previously. 13 In both studies, cats that developed 131I-induced hypothyroidism were older than the euthyroid cats. In the previous study, 13 female sex was a risk factor for hypothyroidism; although we found that 62% of hypothyroid cats were female vs 51% of euthyroid cats, these proportions did not differ (P = 0.33). In both studies, cats that developed 131I-induced hypothyroidism had a higher prevalence of detectable serum TSH concentrations (⩾0.03 ng/ml) and were more likely to have bilateral thyroid nodules, with symmetrical uptake of the radionuclide into both lobes than the cats that became euthyroid. In both studies, cats that failed treatment tended to be more severely hyperthyroid, as reflected by higher serum T4 and T3 concentrations, higher thyroid tumour volumes and TcTU, and higher composite dosing or severity scores. These findings suggest that these factors should be considered when treating cats with 131I, at least when using this individualised 131I dosing algorithm, calculated to administer the lowest effective dose possible. 12 Slightly decreasing or increasing the algorithm’s final calculated dose in cats at risk for hypothyroidism or treatment failure, respectively, might improve treatment outcomes and result in a higher rate of euthyroidism.
The 131I dosing protocol used in this study has some distinct disadvantages. Firstly, it requires that the radioiodine facility has a gamma (scintillation) camera to perform thyroid scintigraphy (and therefore determine thyroid volume and TcTU); a dose calibrator to accurately measure the 131I doses administered to the cats, as well as the dose standard for 131I uptake studies; and a survey meter/probe (Geiger counter) to count the cat’s neck and thigh 24 h after initial 131I treatment, as well as the dose standard to calculate the percentage 131I uptake. 29 In virtually all instances, the survey meter used to measure the cats’ thyroid 131I uptake can be the same meter used for radiation safety monitoring purposes, as well as to determine when the radiation emitted from the cat has reached a level that poses no radiation safety threat to the general public (allowing the cat to be discharged).30–32 Secondly, our protocol is more time-consuming that most other 131I dosing methods. Finally, performing post-therapeutic 131I uptake studies does exposes veterinary staff to radiation, but this can be kept to a minimum with a short duration of exposure (ie, time needed to count most cats for thyroid uptake is generally <3 mins, thereby limiting one’s exposure).
Conclusions
This study confirms that the use of the recently reported algorithm for calculating individual 131I doses (based on serum thyroid hormone concentrations, thyroid scintigraphy and 131I uptake measurement) 12 shows repeatable results in curing a large proportion of hyperthyroid cats, while reducing the risk of overt hypothyroidism. This algorithm also reduces the 131I dose for most cats, as compared with other commonly used dosing protocols,1–6 thereby reducing radiation exposure to veterinary staff and owners. By lowering the prevalence of iatrogenic hypothyroidism, this low-dose algorithm also lowers the rate of azotaemia that commonly develops after 131I treatment.
Footnotes
Acknowledgements
The authors thank Belén Jimenez BVSc, Eva Carmona BVSc and Ms Irene Saavedra for their assistance with management of the hyperthyroid cats, and Dr Mark Rishniw for assisting with manuscript preparation.
Author note
This paper was presented, in part, as an oral research report at the XXI Veterinary Specialties Congress AVEPA and at the Vetmadrid 2022 Veterinary Congress.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
