Abstract
Objectives
The study aimed to describe the clinical use of chloramphenicol in client-owned cats, examining the patient population, sites of infection, targeted pathogens, prescribed dosing regimen, outcomes and adverse effects.
Methods
Between 2015 and 2023, a retrospective medical record review was undertaken to identify a case series of cats treated with chloramphenicol.
Results
Twelve client-owned cats with complicated infections were treated with chloramphenicol at a median dose of 13.3 mg/kg (range 9.1–34.7) administered orally every 12 h for a median duration of 14.5 days. Within 30 days before starting chloramphenicol, 11/12 cats had received treatment with at least one antibiotic. The sites of infection included skin, urinary tract and multisystemic. Multidrug-resistant pathogens, for which no alternative oral antibiotic therapy was available, were commonly targeted. These included meticillin-resistant Staphylococcus species and multidrug-resistant Enterococcus faecium. Of the 12 cats treated with chloramphenicol, nine effectively achieved either bacteriologic or clinical cure. Chloramphenicol adverse effects were uncommon and limited to gastrointestinal upset, which was self-limiting or manageable with supportive treatment in two cats.
Conclusions and relevance
In these cats, the decision to treat with chloramphenicol was made because an alternative oral antibiotic was not available to target the multidrug-resistant pathogens. Infections of the skin or urinary tract were common. Overall, chloramphenicol was well tolerated, and treatment success was possible for most cats.
Keywords
Introduction
In veterinary medicine, when faced with treating cats with clinical infections associated with meticillin-resistant Staphylococcus pseudintermedius (MRSP), extended-spectrum beta-lactamase (ESBL)-producing Enterobacterales, multidrug-resistant Enterococcus species or other drug-resistant pathogens, chloramphenicol may be the only oral susceptible antibiotic available. Favorable tissue penetration makes chloramphenicol useful for treating susceptible bacteria in various sites, including central nervous system and ocular infections. 1 The potential for cats to be more susceptible to chloramphenicol adverse effects has been hypothesized and attributed to their decreased ability to conjugate drugs with glucuronic acid. 2 However, limited information is available on the clinical use and adverse effects of chloramphenicol in cats to help guide clinicians confronted with treating drug-resistant pathogens.3,4
The risk of irreversible, fatal idiosyncratic aplastic anemia in people has limited the use of chloramphenicol since its discovery in the 1940s.5,6 High doses of chloramphenicol have been associated with bone marrow suppression in experimental studies involving ducks, cats and dogs.7–12 There are no reports of irreversible marrow aplasia resulting from chloramphenicol therapy in these species. Cats administered 120 mg/kg/day of chloramphenicol developed clinical signs of toxicosis that included lethargy, decreased appetite, weight loss and gastrointestinal upset. 1 Within the first week of treatment, the predominate bone marrow changes described include hypocellularity and vacuolation of lymphocytes, as well as myeloid and erythroid precursors.1,7,8 Peripheral hematologic changes detected at 2 weeks include decreased numbers of reticulocytes, platelets and white blood cells.1,7,8,13,14 The mechanism of these hematopoietic changes is not entirely understood, but the severity of bone marrow suppression seems to be dose-dependent and reversible with the discontinuation of chloramphenicol. 1
The literature describes potential clinical applications of chloramphenicol in cats based on case reports and pharmacokinetic or pharmacodynamic data.1,4,13,14 Chloramphenicol has been suggested for treating bacterial infections arising from a range of anatomic sites in cats, including the lungs, biliary system, urinary tract, oral cavity and intra-abdominal area. 1 However, there is currently a lack of published reports to provide information regarding the utilization of chloramphenicol in the clinical setting.
The aim of this retrospective study was to describe the clinical use of chloramphenicol in client-owned cats. The study’s specific objectives were to describe the patient population, sites of infection, targeted bacterial isolates, chloramphenicol dosages, treatment durations, outcomes and adverse effects.
Materials and methods
Case selection criteria
The medical records of client-owned cats prescribed chloramphenicol were reviewed retrospectively. The electronic medical record search included feline cases evaluated at University of Wisconsin Veterinary Care (UWVC) between January 2015 and June 2023. Keywords included chloramphenicol in the context of feline patient discharges, updates, client communications and prescriptions. Retrieved medical records were reviewed, and cases meeting all of the following criteria were included: Evaluation and workup identifying the targeted site of infection; bacterial culture and antimicrobial susceptibility testing performed; chloramphenicol prescription information including dosage and duration of treatment; and a minimum of one follow-up visit after starting chloramphenicol. Cases were excluded if treatment with chloramphenicol was mentioned but not pursued, chloramphenicol prescription information was not available for review, or medical records were incomplete or unavailable. For the cases that met the inclusion criteria, information retrieved from the medical record included signalment, history, clinical signs, physical examination, clinical diagnosis including site of infection, comorbidities, bacterial culture and antimicrobial susceptibility results, concurrent medications including previous antibiotic therapy within 1 month of clinical presentation, chloramphenicol dosing regimen and follow-up, including outcome and adverse effects. Clinical outcome categories were scored as defined in Table 1.
Defined outcome categories used to score each cat’s response to chloramphenicol therapy for the treatment of susceptible infections

The UWVC clinical microbiology laboratory performed all bacterial cultures as routine culture submissions. Microorganisms were identified at the species level using matrix-assisted laser desorption/ionization mass spectrometry. Antimicrobial susceptibilities were performed by broth microdilution according to the manufacturer using Clinical and Laboratory Standards Institute (CLSI) standards for testing. Interpretive categories and their corresponding breakpoints were per CLSI recommendations. Without veterinary-specific CLSI interpretive categories for chloramphenicol, human breakpoints were used as a guide. The CLSI human minimum inhibitory concentration (MIC) breakpoints used were ⩽8, 16 and ⩾32 µg/ml, which were interpreted as susceptible, intermediate and resistant, respectively. Staphylococcus species isolates resistant to oxacillin were reported as meticillin-resistant; further PCR MecA gene testing was not available for the MRSP isolates. Enterobacterales resistant to aminopenicillins, ureidopenicillins, cephalosporins and monobactams, but susceptible to the cephamycins (cefoxitin and cefotetan) and carbapenems, were recorded as ESBL producers. 15 Bacterial isolates reported to be resistant to ⩾3 classes of antibiotics were recorded as multidrug resistant (MDR). 15
Statistical analysis
All continuous variables were summarized using descriptive statistics, and values were reported as medians and ranges.
Results
The electronic medical record search retrieved 57 cases (Figure 1). A total of 45 cases were excluded for the following reasons: chloramphenicol only appeared in medical records as part of a bacterial culture antimicrobial susceptibility report (n = 28); chloramphenicol was mentioned but not prescribed (n = 12); no case follow-up available (n = 3); chloramphenicol prescribed, but the site of infection was not documented (n = 1); and bacterial culture results and chloramphenicol dosing were not available for review (n = 1). Twelve client-owned cats prescribed chloramphenicol met the study’s inclusion criteria, which calculates as 0.69 cats treated with chloramphenicol per 1000 feline patients seen at UWVC over the 8 years reviewed in this retrospective study.

Flow diagram outlining the identification and categorization of feline cases reviewed that were prescribed chloramphenicol. After exclusion, the remaining cases were further categorized based on site of infection
Feline population
The feline population consisted of three spayed females and nine castrated males, with a median weight of 5.05 kg (range 2.47–7.25) and median age of 10 years (range 2–18). Cats included in the study were of the following breeds: Domestic shorthair (n = 9); domestic longhair (n = 1); American Shorthair (n = 1); and Norwegian Forest Cat (n = 1). The comorbidities managed in this clinical population of cats, along with antibiotics prescribed within 30 days and empirical antibiotic therapies pending bacterial cultures, are summarized in Table 2. Of the 12 cats, 11 had at least one comorbidity, with 6/11 cats having a single associated comorbidity and 5/11 having multiple concurrent problems (Table 2). Within the previous 30 days, 11 cats were treated with at least one class of antibiotics; eight cats received more than one class of antibiotics before treatment with chloramphenicol (Table 2). Of the 12 cats, 10 received systemic empirical antibiotic therapy for a median of 6.5 days (range 3–20) before switching to chloramphenicol (Table 2).
Cats treated with chloramphenicol (n = 12), summary of clinical cases including working diagnosis that resulted in chloramphenicol treatment, clinical signs, comorbidities, antibiotic therapy within 30 days of presentation and empirical antibiotics pending culture results, including days of therapy

Amoxi/Clav = amoxicillin clavulanic acid; Ampi/Sulbact = ampicillin/sulbactam; CKD = chronic kidney disease; DM = diabetes mellitus; EPI = exocrine pancreatic insufficiency; IBD = inflammatory bowel disease; IRIS = International Renal Interest Society; LUTI = lower urinary tract infection; LUTS = lower urinary tract signs; NA = not applicable; SCC = squamous cell carcinoma; TMS = trimethoprim sulfamethoxazole
All cats had persistent clinical signs at the time chloramphenicol was prescribed. Chloramphenicol was prescribed as either oral capsules or a compounded liquid formation; seven cats received oral capsules and five received the compounded liquid formulation. Chloramphenicol was prescribed at a median dosage of 13.3 mg/kg PO q12h (range 9.1–34.7). The median duration of treatment was 14.5 days (range 10–30). Table 3 provides the individual chloramphenicol treatment durations.
A summary of the site of infection, clinical specimen cultured, bacterial isolate(s) targeted and observed chloramphenicol MIC, duration of chloramphenicol treatment and outcome for the cats (n = 12) treated with chloramphenicol

In cases with mixed bacterial infections, the primary organism(s) targeted are indicated by bold text
BC = bacteriologic cure; BF = bacteriologic failure; CC = clinical cure; CFU = colony-forming units; MDR = multidrug resistant; MIC = minimum inhibitory concentration; MRSP = meticillin-resistant Staphylococcus pseudintermedius
Sites of infection and bacterial isolates
Infection sites targeted in these cats are summarized in Table 3. At the time of bacterial culture submission, 10 cats were receiving at least one antibiotic (metronidazole [n = 3], amoxicillin/clavulanic acid [n = 2], enrofloxacin [n = 2], marbofloxacin [n = 2], doxycycline [n = 1], clindamycin [n = 1]) with three cats receiving more than one antibiotic class. The bacterial cultures in these 12 cats isolated pure growth (n = 7) or mixed bacterial growth (n = 5). Table 3 summarizes the targeted bacterial isolates cultured and the observed chloramphenicol MICs. In 10/12 cats, chloramphenicol was the only oral antibiotic that the targeted bacterial pathogens were susceptible to based on the in vitro-determined MICs.
Outcome
The median follow-up was in the range of 7–49 days (median 35), with 10/12 cats having rechecks/updates available for the entire duration of antibiotic therapy. Follow-up bacterial cultures were available in 6/12 cats. Chloramphenicol treatment outcomes as a function of bacterial isolates and MIC are summarized in Figure 2.
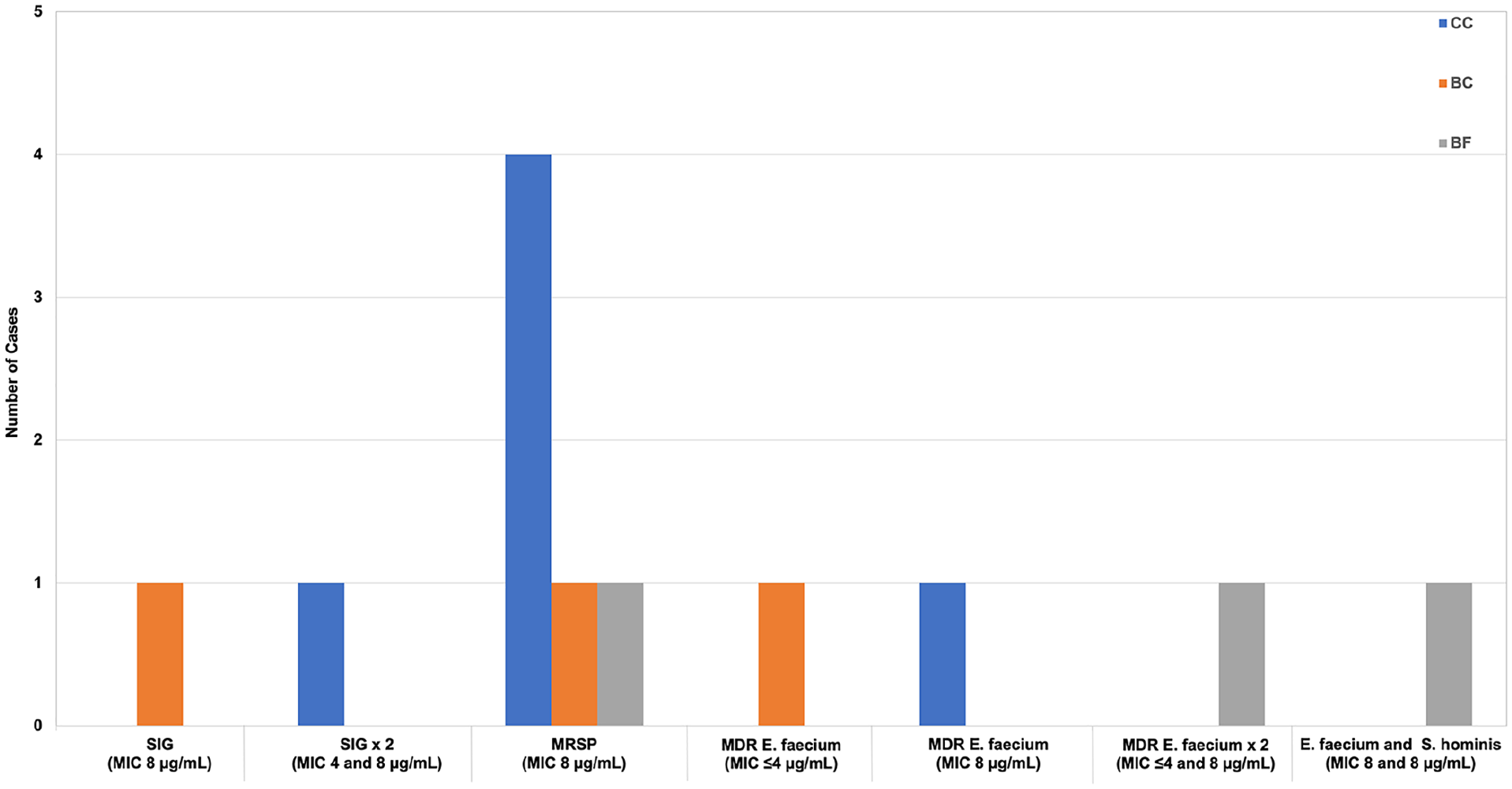
Outcome data for the cats (n = 12) treated with chloramphenicol. The number of bacterial isolates is plotted vs the reported chloramphenicol minimum inhibitory concentration (MIC) overlayed by the outcomes, clinical cure (CC), bacteriologic cure (BC) or bacteriologic failure (BF). MDR = multidrug resistant; MRSP = meticillin-resistant Staphylococcus pseudintermedius; SIG = Staphylococcus intermedius group
Of the 12 cats, nine had successful outcomes (clinical cure [CC], n = 6; bacteriologic cure [BC], n = 3) (Table 3 and Figure 2). The three cats that achieved BC comprised cats diagnosed with a lower urinary tract infection (n = 1), recurrent pyelonephritis (n = 1) or draining cutaneous wound (n = 1). In the cat with recurrent pyelonephritis, along with the resolution of bacteriuria, the cat’s creatinine decreased from 3.9 to 2.6 mg/dl after chloramphenicol therapy. Treatment failure was associated with bacteriologic failure [BF] in three cats. The cat diagnosed with pyelonephritis and cholangitis had persistent clinical signs and bactibilia that resulted in a cholecystectomy despite resolution of the Enterococcus faecium bacteriuria with concurrent improvement in creatinine, from 6.7 to 5 mg/dl. Another cat that scored as a BF was a cat with recurrent otitis that underwent a total ear canal ablation after remaining bacterial culture-positive with persistent clinical signs. The third cat that failed treatment was a cat with progressive chronic kidney disease that was persistently bacteriuric due to being repeatedly Enterococcus faecalis urine culture positive. That cat’s owner elected euthanasia because of its progressive clinical decline.
Adverse effects
Suspected chloramphenicol-associated adverse effects occurred in two cats. One cat was treated with chloramphenicol (34.7 mg/kg q12h) for 10 days and experienced a decreased appetite and lethargy, which improved with supportive care. The cat’s decreased appetite resolved with the discontinuation of chloramphenicol. The other cat experienced one episode of vomiting during chloramphenicol treatment (14 mg/kg q12h for 29 days).
Of the 12 cats, seven had follow-up complete blood counts (CBCs) within a median of 12.5 days (range 4–26) after starting chloramphenicol. The CBC findings included a median hematocrit (HCT) of 30% (range 21–41), platelet count of 459,000/µl (range 177,000–608,000) and white blood cell count of 11,500/µl (range 4700–17,300). Only one cat experienced a decline in any of its cell lines while being treated with chloramphenicol. This cat’s previously documented anemia was progressive but regenerative (HCT 18% to 16%) 4 days after starting chloramphenicol. The cat’s progressive anemia was prioritized as secondary to blood loss due to previous surgery, incision infection and hospital blood sampling rather than chloramphenicol bone marrow suppression. The cat was given a blood transfusion and the dose of chloramphenicol was reduced from 20 to 10 mg/kg q12h. The cat completed 21 days of chloramphenicol therapy and maintained a stable post-transfusion HCT (28%).
Discussion
This retrospective study describes a diverse population of cats treated with chloramphenicol. All cats had complicated infections due in part to comorbidities. The most common sites of infection were associated with the skin or urinary tract in all but one cat treated with at least one class of antibiotics within 30 days of their clinical evaluation. MDR infections were common and included meticillin-resistant Staphylococcus species and MDR E faecium. Of the 12 cats treated with chloramphenicol, nine effectively achieved either BC or CC. Chloramphenicol adverse effects were uncommon and limited to gastrointestinal upset in two cats.
There was a wide age range in this study population, with most cats being neutered males and aged ⩾10 years. In this small study, 4/9 male cats were treated for urinary tract infections as a sequelae of a lower urinary tract obstruction and associated perineal urethrostomy. The male cats in this study population represent a clinical population at risk for lower urinary tract obstruction and subsequent infections. 16
All cats were evaluated and treated for complicated bacterial infections with a range of complicating factors, with all but one cat receiving antibiotic therapy within 30 days of their evaluation and culture. Chloramphenicol was prescribed to target the MDR pathogen(s) isolated from skin, urine or bile cultures with oral antibiotic susceptibility limited to chloramphenicol; MICs were in the range of ⩽4–8 µg/ml. The chloramphenicol-targeted pathogens included meticillin-resistant Staphylococcus species and MDR E faecium. In all cases, these MDR pathogens were likely a result of opportunistic infections resulting from compromised natural defenses of the skin, urinary tract or biliary tract. In addition, previous antibiotic administration is a known risk factor for the development of clinical infections associated with MDR pathogens.17–19 For example, meticillin resistance of Staphylococcus species has been linked to previous fluoroquinolone and cephalosporin therapy.17,18 Interestingly, 5/8 cats with a meticillin-resistant Staphylococcus species infection had been treated with cephalosporins or fluoroquinolones within the previous 30 days.
MRSP infections encountered in small animal practice are therapeutically challenging, as the isolates express the MecA gene, resulting in resistance to all beta-lactam antibiotics, and some isolates are concurrently resistant to other important antibiotic classes.20,21 Meticillin-resistant Staphylococcus species was the most common pathogen targeted in this population of cats, with the isolates demonstrating resistance to oxacillin, as well as fluoroquinolones, clindamycin, tetracyclines and trimethoprim-sulfamethoxazole. The meticillin-resistant isolates included Staphylococcus intermedius group and MRSP that were isolated from common sites (urine and skin) associated with meticillin-resistant Staphylococcus infections. 22 The MRSP skin infections were mixed infections and the Staphylococcus isolates drove chloramphenicol treatment because of their limited antibiotic susceptibility, with the co-isolates also susceptible to chloramphenicol.
In small animals, the most common enterococcal species associated with disease are E faecium and E faecalis. 23 E faecium isolates are often classified as MDR isolates. 24 Enterococcal isolates can be a primary pathogen resulting in clinical disease but can also be cultured as a single subclinical isolate or as a contaminant in mixed infections.20,24 Skin, urine and bile are common sites for MDR enterococcal infections in people and animals with complicated or mixed infections.24–27 In cats, E faecalis has been associated with urinary tract infections. 24 The enterococcal infections associated with clinical disease in this study population included E faecium infections of the skin complicated by urinary extravasation or being non-ambulatory. In another cat, a multisite E faecium infection was cultured in the urine as a single isolate and in the bile as a mixed infection with Proteus mirabilis.
Despite the complicated nature of the infections and underlying comorbidities, 9/12 cats achieved either CC or BC when treated with chloramphenicol. The chloramphenicol MICs of the targeted MDR bacterial isolates were in the range of ⩽ 4–8 µg/ml, which, at the time of writing, was interpreted as susceptible based on the CLSI’s human breakpoint interpretation and clinically effective based on case outcomes. Interestingly, for Staphylococcus species isolated in animals, the CLSI chloramphenicol MIC breakpoint is anticipated to be updated to ⩽2 µg/ml. 28 Thus, clinicians need to remember that as breakpoint interpretations continue to be updated, current interpretations of susceptible may be interpreted as resistant in the future, demonstrating that bacterial culture and antibiotic susceptibility testing will continue to be critically important in the clinical management of MDR pathogens. Of the cats considered chloramphenicol treatment failures, 2/3 cases ultimately required concurrent surgical management to assist in resolving their persistent infections. These cases highlight the importance of appropriate antibiotic treatment guided by in vitro antimicrobial susceptibility testing; however, in some cases, concurrent surgical management may be necessary to optimize outcomes. As reported here, the cats treated with chloramphenicol classified as treatment failures were all scored as BF because of persistent clinical signs and remaining bacterial culture positive; they comprised a cat with persistent cholangitis leading to a cholecystectomy, a cat with recurrent otitis secondary to an aural ceruminous adenocarcinoma leading to a total ear canal ablation, and a cat with chronic kidney disease with persistent bacteriuria and progressive clinical signs resulting in the owner electing humane euthanasia. In these three cats, their comorbidities also required treatment or management, making it impossible to know if the limitations associated with antibiotic susceptibility testing, including the lack of specific bug-drug MIC breakpoint interpretations, contributed to their treatment failures. In all three cats, BF was likely multifactorial. Further research would be required to investigate if breakpoint interpretations for chloramphenicol need to be tailored to the bacterial isolates targeted or sites of infection.
In the cats treated with chloramphenicol for a lower urinary tract infection or suspected pyelonephritis, 4/5 cats achieved BC based on follow-up urine culture and resolution of bacteriuria. The two cats with acute kidney injury and suspected pyelonephritis had an improvement in their creatinine in association with chloramphenicol therapy. The remaining cat treated for a lower urinary tract infection achieved a CC with the resolution of its lower urinary tract signs, as a follow-up urine culture was not performed. In this small group of cats, chloramphenicol appears to be clinically effective in treating chloramphenicol-susceptible bacteriuria in cats with complicated urinary tract infections due to meticillin-resistant S intermedius group or MRSP, suggesting chloramphenicol may achieve adequate tissue and/or urinary drug concentrations. Chloramphenicol is a lipophilic drug that achieves high kidney concentrations in most species, with approximately 25% of the absorbed chloramphenicol excreted as an active drug in the urine of cats.1,14,29,30 Higher urinary concentrations of the active drug are estimated in the urine of cats compared with other species because of the cat’s decreased capacity to metabolize chloramphenicol via glucuronidation, the major route of hepatic metabolism in most species.1,14,29,30
In this small group of cats, chloramphenicol was well tolerated at dosages in the range of 8–20 mg/kg q12h. There was no evidence of hematologic adverse effects in the seven cats with follow-up CBCs during or after chloramphenicol treatment. The cat with progressive regenerative anemia was given a blood transfusion. Despite the decision to decrease the chloramphenicol dose, chloramphenicol bone marrow suppression was unlikely based on the cat’s regenerative response to its progressive anemia. The cat’s blood loss was suspected to be secondary to a previous surgery, incision infection and hospital blood sampling rather than chloramphenicol bone marrow suppression. Gastrointestinal upset, a commonly described adverse effect associated with chloramphenicol, occurred in two cats. The cat treated with a higher dosage of chloramphenicol (36 mg/kg q12h) developed a decreased appetite that was self-limiting and resolved once the chloramphenicol treatment was completed. In cats treated with standard dosage recommendations for chloramphenicol (50 mg/cat or 10–20 mg/kg q12h), treatment for a median duration of 14 days seemed well tolerated and did not result in clinical adverse effects or require drug discontinuation. Not all cats had CBC monitoring during treatment to rule out subclinical hematologic adverse effects. However, CBC monitoring is recommended, especially when treatment durations are longer than 14 days. 29 Cats are reported to be at higher risk of chloramphenicol adverse effects, primarily bone marrow suppression, because of their limited ability to metabolize chloramphenicol via glucuronidation.1,29,31
The present study has some limitations. One limitation is the small number of cases included, likely attributable to limited clinical and microbiological indications for the use of chloramphenicol, a lack of familiarity with this antibiotic and clinicians’ reservations regarding potential adverse effects. The retrospective nature of the study presents limitations on the case details available for review and non-standardized decisions made based on individual patient care. Clinical and diagnostic evaluations were clinician-dependent and reliant on information documented in the medical record to assess comorbidities, response to treatment and adverse effects. For example, standardized CBC monitoring does not provide an objective assessment of the hematologic adverse effects associated with chloramphenicol therapy in cats. The complexity of the individual cases in this study population may represent a limitation in evaluating the use of chloramphenicol in cats. However, this diverse population of cats with comorbidities may represent a realistic and applicable clinical situation in which veterinarians may prescribe chloramphenicol in cats.
Conclusions
Chloramphenicol is uncommonly used in feline practice. As described in this clinical population of cats, it may be the only oral antibiotic effective in treating MDR-complicated skin and urinary tract infections. Overall, chloramphenicol was well tolerated, and clinical signs resolved in most (9/12) of the treated cats. Adverse effects occurred in a few (2/12) cats. Larger populations of cats need to be studied prospectively to fully describe the clinical use of chloramphenicol in cats and to document chloramphenicol’s clinical, hematologic and biochemical adverse effects.
Footnotes
Acknowledgements
The authors thank Marianthi Geltos for her assistance in identifying feline cases treated with chloramphenicol for inclusion in this retrospective study.
Author note
This work was performed at University of Wisconsin Veterinary Care through the Department of Medical Science, School of Veterinary Medicine, University of Wisconsin-Madison, 2015 Linden Drive, Madison, WI 53711, USA.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
