Abstract
Objectives
The aim of the present study was to evaluate minimally invasive diagnostic techniques, such as the semi-quantitative indirect IgG antibody enzyme immunoassay (EIA) using blood serum and the urinary lateral flow assay (LFA), for the detection of Histoplasma capsulatum in cats with histoplasmosis.
Methods
Eight client-owned domestic cats diagnosed with histoplasmosis were selected based on cytological, histopathological, mycological, molecular or antigenic techniques. The blood serum of these animals was tested in a semi-quantitative indirect IgG antibody EIA for the detection of H capsulatum. Urine samples were tested for H capsulatum antigen using LFA.
Results
Five cats were seropositive on IgG EIA (5/8, with diagnostic sensitivity equal to 62.5%; 95% confidence interval [CI] 24.5–91.5) and five cats were positive on H capsulatum antigen LFA (5/7, with diagnostic sensitivity equal to 71.4%; 95% CI 29.0–96.3). The combined diagnostic sensitivity when interpreted in parallel was 87.5% (7/8, 95% CI 47.3–99.7). The specificity for the anti-Histoplasma IgG EIA was 100% (95% CI 71.5–100) and for the H capsulatum antigen LFA it was also 100% (95% CI 71.5–100).
Conclusions and relevance
The semi-quantitative indirect IgG antibody EIA for the detection of H capsulatum in blood serum and the urinary LFA for the detection of the same agent emerge as new minimally invasive diagnostic techniques that can assist in the approach to disseminated and pulmonary feline histoplasmosis, especially when both techniques are considered together.
Introduction
Domestic cats can be infected by the dimorphic fungus Histoplasma capsulatum, which causes histoplasmosis. This disease is endemic in the Americas, where it is considered the second most frequent systemic mycosis in cats. Histoplasmosis is acquired through the inhalation of microconidia found in soil, especially that contaminated with bird or bat feces.1–3
Histoplasmosis is considered underdiagnosed in the feline species due to diagnostic difficulties. 4 Conventional tests, such as cytology, histopathology, fungal culture and PCR, are disadvantageous because they require invasive sample collection, which poses potential risks to the animal.5–8 Serology, usually performed using immunodiffusion (ID) and complement fixation (CF) techniques, is considered an unreliable form of diagnosis due to low sensitivity and specificity.5–7,9
Thus, new diagnostic techniques, such as antigenic immunoenzymatic assay for H capsulatum, in serum or urine, have been explored as new approaches for the diagnosis of mycosis. However, despite their usefulness, these techniques have limitations and should not be used as the only diagnostic tool, especially when serum and urine samples are not considered together to evaluate each animal,6,10 which highlights the importance of persistently exploring alternative diagnostic tests.
The semi-quantitative indirect IgG antibody enzyme immunoassay (EIA) for the detection of H capsulatum in blood serum has shown promising results in human medicine. 11 However, its evaluation in cats is limited to a few studies.12,13 This is also true for the lateral flow assay (LFA) for the detection of H capsulatum, which can be performed even at the patient’s side, using urine and without the need to send it to a laboratory, and which, despite being validated in human medicine, has not been used to date in domestic cats. 14
The aim of this study was to evaluate the performance of diagnostic techniques that require minimally invasive sample collection, such as the semi-quantitative indirect IgG antibody EIA in blood serum and urinary LFA, for the detection of H capsulatum, in cats with histoplasmosis.
Materials and methods
Eight domestic cats (Felis catus) with histoplasmosis were selected for this study regardless of their sex, breed or age. These animals were recruited from the medical clinic for domestic cats at the small animal veterinary hospital of the Federal Rural University of Rio de Janeiro (UFRRJ) or from private appointments in Rio de Janeiro, Brazil. All owners whose animals were included in the research provided signed consent. The study was submitted to the Ethics Committee on Animal Research of UFRRJ and was approved under protocol number 4895210619. The research was conducted between August 2019 and May 2022. The cats included in the study were those in which cytology, histopathology, fungal culture, PCR or antigenic immunoenzymatic assay revealed the presence of the fungus H capsulatum. The impossibility of obtaining a blood and/or urine sample was considered an exclusion criterion. Relevant information was collected through medical history, physical examination, imaging studies (thoracic radiography and abdominal ultrasonography) and the diagnostic tests previously mentioned to confirm histoplasmosis.
The infection was categorized according to its manifestation as disseminated, pulmonary or gastrointestinal. Histoplasmosis was classified as disseminated when the fungus was identified in the lung tissue and at least one organ outside the thoracic cavity was affected by H capsulatum. Histoplasmosis in cats with the infectious organism restricted to the lungs was classified as pulmonary, whereas in cats with the agent restricted to the gastrointestinal tract, it was classified as gastrointestinal. For these classifications, imaging tests such as radiography and ultrasonography were used to assess the presence of the fungus in the body systems, along with conventional diagnostic tests. However, when the histopathology of all organs suspected of involvement was performed after necropsy, this approach was considered superior to the findings of radiography and/or ultrasonography.
Collection of biological samples
Blood samples were collected from all cats diagnosed with histoplasmosis. After restraining the animal and disinfecting the collection site, a 5 ml syringe with a 23 G butterfly needle was used. Blood samples (1 ml) were collected through either cephalic, saphenous or jugular venipuncture, depending on the cat’s temperament and hydration level. The blood samples were placed in tubes containing a separator gel and clot activator (Vacutube separating gel; Biocon) and then centrifuged at a speed of 8000 rpm for 8 mins to obtain serum.
Serum from eight cats were stored frozen until shipment to MiraVista Diagnostics (MiraVista Veterinary Diagnostics) where they were subjected to the semi-quantitative indirect IgG antibody EIA for detection of H capsulatum.
The cats diagnosed with histoplasmosis underwent urine collection by cystocentesis with a 25 mm × 7 mm needle and a 10 ml syringe, or through natural micturition directly into a sterile vial; for this purpose, a minimum volume of 1 ml was obtained. The urine samples were stored in a universal urine collection container with a maximum capacity of 50 ml. Subsequently, the urine from seven cats was stored frozen until shipment to MiraVista Diagnostics for LFA analysis for the detection of H capsulatum.
Anti-Histoplasma IgG enzyme immunoassay
A commercially available assay was used, in which 96-well microplates were coated with H capsulatum antigens and blocking buffer. Patient serum, standards and controls were added to the wells. After incubation at 37°C for 1 h, the wells were washed, and then biotinylated anti-feline IgG was added to each well. The microplate was incubated once again at a temperature of 37°C for 1 h. The wells were washed again, and the streptavidin–peroxidase conjugate from horseradish was added to each well. The wells were then incubated at 37°C for 1 h. Thereafter, the wells were washed, and the chromogenic solution containing peroxidase substrate was added. The microplate was then incubated at room temperature (22°C) for 10 mins. The enzymatic reaction was halted by the addition of 2 N sulfuric acid. The plate was read at a dual wavelength of 450/620 nm. IgG levels <8.0 enzyme units (EU) were considered negative, 8.0–9.9 EU were considered indeterminate, and ⩾10.0 EU were considered positive.
Histoplasma antigen lateral flow assay
A commercially available LFA was used. The immunoassay is based on a qualitative methodology, using lateral flow, where polyclonal antibodies are used for the direct detection of H capsulatum galactomannan antigen in urine. This was performed by adding 100 µl of urine to 25 µl of sample diluent using a pipette provided in the kit. The diluted specimen was then placed in the sample port of the LFA device. The test was read after incubation at room temperature (22°C) for 30 mins, where the visible presence of the control line confirmed a valid assay and verification of the test line indicated the presence of the Histoplasma antigen. The test was considered invalid if the control line was not observed. 15
Control group
In addition, 11 domestic cats were recruited, which were presented to the veterinary service for check-up or preoperative examinations for sterilization; these cats had no associated clinical complaints. Blood samples from these animals were collected by venipuncture to reserve an aliquot of the obtained serum (minimum of 2 ml). During the same appointment, urinary specimens (minimum 1.5 ml) from these cats were obtained by natural urination or cystocentesis.
The cats’ serum and urine were initially subjected to an immunoenzymatic antigenic assay for Histoplasma to verify negativity for the fungal agent in question. Subsequently, the EIA for IgG antibodies in serum and urinary LFA were performed on samples from animals considered negative for mycosis. All tests performed were performed in the MiraVista Veterinary Diagnostics Laboratory.
Statistical analysis
For the descriptive analysis, frequencies and relative frequencies along with minimum and maximum values were reported. In addition, diagnostic sensitivity and specificity, as well as associated 95% confidence intervals (CIs; exact Clopper–Pearson) were calculated. For these analyses, the open-source statistical software R (version 4.2.2, 2022-10-31 ucrt) was used.
Results
Of the eight cats whose blood samples were tested for IgG EIA against H capsulatum, five tested positive, with concentrations in the range of 13.2–70.1 EU. Regarding urinary LFA, out of the seven cats that underwent this test, five tested positive (Table 1). It is worth noting that for the two cats negative on urinary LFA, new analyses were carried out based on the same methodology; however, dilutions of the samples of 1:2, 1:4 and 1:8 were used. The results were negative for both cats in all additional dilutions performed. Results obtained from the urinary LFA are shown in Figure 1.
Results of the semi-quantitative indirect IgG antibody EIA for the detection of Histoplasma capsulatum in blood serum and urinary LFA for the detection of H capsulatum in domestic cats previously diagnosed with histoplasmosis

IgG antibody EIA of blood serum (EU): IgG antibody enzyme immunoassay of blood serum in enzymatic units
IgG antibody EIA: reportable range: results <8.0 are considered negative, results 8.0–9.9 are considered intermediate and results ⩾10.0 are considered positive
EIA = enzyme immunoassay; LFA = lateral flow assay
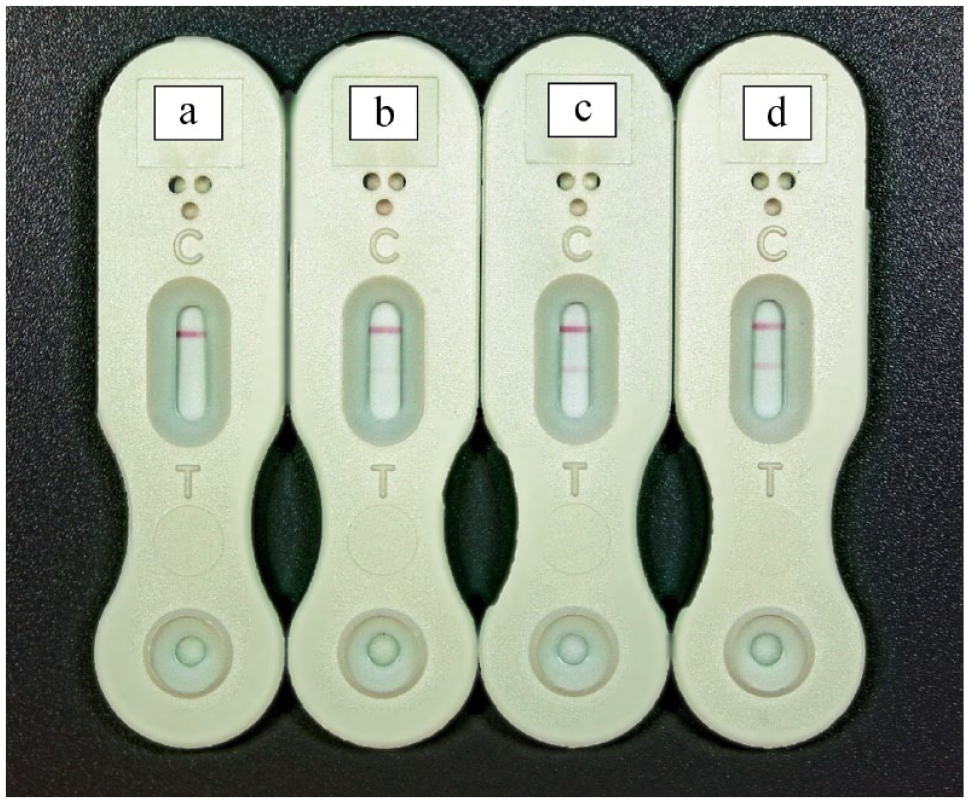
Results of the lateral flow assay urine test in cats previously diagnosed with histoplasmosis. The upper pink line refers to the control and the lower pink line indicates the test result. (a) Negative; (b) low positive; (c) moderately positive; and (d) highly positive
The diagnostic sensitivity for anti-Histoplasma IgG EIA was 62.5% (95% CI 24.5–91.5), and for the H capsulatum antigen LFA it was 71.4% (95% CI 29.0–96.3). The combination of both tests (interpreted in parallel) provided a diagnostic sensitivity of 87.5% (95% CI 47.3–99.7).
Regarding the control group, the serum and urinary samples of the 11 domestic cats submitted to the antigenic immunoenzymatic assay for Histoplasma were negative; from these cats known to be negative, the results were that all 11 cats did not show seropositivity in the anti-Histoplasma IgG EIA and all 11 cats did not have positive urinary LFA for Histoplasma.
The specificity for the anti-Histoplasma IgG EIA was 100% (95% CI 71.5–100) and for the H capsulatum antigen LFA it was also 100% (95% CI 71.5–100).
Histopathology and cytopathology (n = 2) and cytopathology alone (n = 2) were the most common means of diagnosis, followed by antigenic enzyme immunoassay for the detection of H capsulatum in serum, fungal culture, histopathology alone, and histopathology and PCR, which were used once each (Figure 2).

Diagnostic methods used to detect histoplasmosis in cats that were subsequently tested using a semi-quantitative indirect IgG antibody enzyme immunoassay for the detection of Histoplasma capsulatum in blood serum and a urinary lateral flow assay for the detection of H capsulatum
The organ with the greatest fungal tropism in this research was the lung, where the fungus was found in 4/8 (50%) cats; this was followed by the skin, eyes, bone marrow, pancreas, kidneys, spleen and liver, where, in each of these, the mycotic agent was found in 12.5% of recruited cats (1/8 cats).
Histoplasmosis was classified as disseminated in five (62.5%) cats and as pulmonary in three (37.5%) cats.
Discussion
A high correlation was observed between seropositivity for histoplasmosis by IgG antibody EIA and previous diagnosis of histoplasmosis in our group of eight cats. Previous studies have advised against using serology by ID or CF as a diagnostic tool for cats with mycosis. These tests have been found to be unreliable due to the occurrence of false-positive results caused by previous exposure and recovery, as well as cross-reactivity with other fungal agents. In addition, there is a possibility of false-negative results due to the time required for seroconversion.5–7,9
However, the serological study conducted in the present research was carried out using a semi-quantitative methodology, which is also employed in human and veterinary medicine, where it has been shown to be superior to ID and CF techniques.13,16 Furthermore, it is worth noting that the anti-Histoplasma IgG EIA proved to be superior to the detection of urinary antigens in cats. 13 The possibility of cross-reactivity of the IgG in the Histoplasma antibody test in patients with Cryptococcus has been verified. 17 However, this does not undermine the efficacy of the test in the present research, since the cats had been previously diagnosed with histoplasmosis using other diagnostic tools.
Thus, based on the results obtained, it is noted that the IgG antibody EIA is effective as a complementary diagnostic method for feline histoplasmosis, in accordance with what was verified in the only two studies that evaluate the use of such a technique in cats, with a sensitivity of 81.1% and a specificity of 94.7%.12,13 However, it is worth highlighting that this is the only study that evaluates the use of the serological test in question in a group of cats with confirmed disease. It is not advisable to rely solely on this test for the verification of histoplasmosis in cats owing to the occurrence of false-negative results. 13
Notably, serological tests are not suitable for immunocompromised patients, those on immunosuppressive drugs or those that have recently undergone transplantation, as their ability to produce antibodies may be impaired.11,18 In the present study, out of the three animals that tested negative on serology, two (66.67%) had received systemic corticosteroid therapy before sample collection. Therefore, further evaluation is needed to determine the influence of corticosteroid use on the results of this assay.
The LFA for the detection of H capsulatum in urine samples from animals with histoplasmosis demonstrated high sensitivity and specificity. The LFA, marketed by MiraVista Laboratory, is a diagnostic tool used in human medicine to detect histoplasmosis from urine samples, and is associated with a diagnostic sensitivity of 79–96% and specificity of 92–100%. This method offers several advantages in terms of speed and convenience, allowing prompt diagnosis and timely treatment with minimal requirements in terms of resources and infrastructure, and it can even be used at the point of care.14,15,19,20 The best-evaluated diagnostic methodologies in cats, which are based on collecting samples in a non-invasive way, such as the immunoenzymatic antigen assay for H capsulatum, require sending the collected sample for testing in reference laboratories, which are concentrated in the USA. Thus, the H capsulatum antigen LFA is extremely important when considering speeding up the diagnosis of mycosis, especially in countries outside the USA, given the possibility of previously purchasing the test and carrying it out at the point of care in less than 40 mins. 15
Therefore, urinary LFA can be used as a diagnostic tool for feline histoplasmosis, but caution should be exercised due to the possibility of false-negative results, observed in a minority of animals in the present study (2/7, 28.57%). This tool had not previously been evaluated in cats, making this the first study to use this methodology for diagnosing fungal infections in this animal species.
The LFA can also be performed on blood serum, which has already been validated in human medicine, with high performance. 21
Cross-reactions in human patients with cryptococcosis, paracoccidioidomycosis, blastomycosis, talaromycosis and coccidioidomycosis are likely,15,19,21 and should be prospectively evaluated in cats.
Thus, this research indicates that Histoplasma antigen detection by LFA and anti-Histoplasma IgG detection by EIA are potentially useful for supporting the diagnosis of histoplasmosis in cats, since at least one of these tests was positive in all but one cat with histoplasmosis.
This way, it was found that the combined execution of both tests conducted in this research could provide greater sensitivity than when tests were performed separately. Thus, the paired analysis of urinary LFA alongside IgG antibody EIA is associated with enhanced diagnostic performance in cats with histoplasmosis.
The main limitation of this study is the small number of animals evaluated. However, it is still relevant considering the diagnostic challenges encountered in feline histoplasmosis. In addition, it creates opportunities for conducting large-scale prospective studies in the future.
Conclusions
The IgG antibody EIA and urinary LFA for H capsulatum enable sample collection in a minimally invasive way and exhibit considerable efficacy in the diagnosis of histoplasmosis in cats. Thus, such techniques have the potential to enhance the safety of disease management for cats and enable veterinarians to confidently diagnose histoplasmosis. Furthermore, testing for the Histoplasma antigen in urine via a point-of-care LFA might be clinically useful in cats with suspected histoplasmosis. However, it is important to note that relying solely on either of these techniques will lead to false-negative results, although the techniques can be used in conjunction to maximize diagnosis.
Footnotes
Conflict of interest
Andrew Hanzlicek is employed by MiraVista Diagnostics, which offers commercially the diagnostic tests for histoplasmosis used in this study. The other authors reported no potential conflict of interest.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
