Abstract
Objectives
Squamous cell carcinoma (SCC) is the most common tumour in the nasal planum of cats. Surgery has traditionally been the treatment of choice but might not be feasible in locally advanced scenarios. Electrochemotherapy (ECT) has shown good control in superficial tumours, but there is a lack of robust information about efficacy in locally advanced cases. The aim of this study was to assess the safety and efficacy of ECT in the treatment of locally advanced stage nasal planum SCC in cats.
Methods
The clinical database of a veterinary referral hospital was searched retrospectively for cats diagnosed with a locally advanced nasal planum SCC (T3N0M0 or T4N0M0) that had received ECT. Local response, adverse events and outcome were documented. The data were evaluated by inferential statistics and correlations between response, recurrence, feline immunodeficiency virus/feline leukaemia virus status, number of treatments, voltage and severity of adverse events, with Kaplan–Meier curves and log-rank tests. Statistical significance was set at P <0.05.
Results
In total, 21 cats were enrolled over a 4-year period. Nineteen cats achieved a complete response (CR) and two cats a partial response (PR) for an overall response rate of 100%. Cats achieving a CR had a median disease-free interval of 182 days (range 128–327) and those with a PR had a median progression-free survival of 156.5 days (range 122–191). The median time to progression was not reached. The overall survival was 453 days for a median follow-up of 341 days (range 191–989). Of the cats, 62% had grade 3 or 4 toxicities, but no deaths due to the treatment were documented. Only voltage was correlated with longer survival (P = 0.001).
Conclusions and relevance
ECT appears to be an effective treatment for feline nasal planum SCC and could be considered a first-line therapy for locally advanced cases. Toxicities reported can be severe in the short term and these could be secondary to more invasive lesions and equipment used.
Introduction
Squamous cell carcinoma (SCC) accounts for 15% of cutaneous tumours in cats and is the most common malignancy of the feline nasal planum.1,2 Exposure to sunlight is strongly related to the development of cutaneous SCC, affecting mostly unpigmented or lightly pigmented skin, such as the nasal planum, ears, eyelids and temporal areas.1,3 According to the World Health Organization TNM (tumour, node, metastasis) classification system for feline tumours of epidermal origin, the stage of primary cutaneous SCC lesions is defined by the depth of invasion and by the size of the lesion (Table 1).1,4 In cats, cutaneous SCCs are usually locally invasive with a low metastatic rate to the regional lymph nodes.1,5 Given these characteristics, the main objective of treatment is to achieve the best local control of the tumour, in order to reduce the recurrence rate.1,2 Nasal planectomy is the most successful way of treating invasive nasal planum SCC (stage T3 or T4).1,5,6 However, in this location and in this advanced stage of the disease, achieving a complete tumour excision with proper margins could be challenging. For that reason, other therapeutic modalities are considered when treating locally advanced stage nasal planum SCC.
World Health Organization TNM (tumour, node, metastasis) classification system for feline tumours of epidermal origin 4

Electrochemotherapy (ECT) is a local method for the treatment of solid tumours that combines reversible electroporation and hydrophilic chemotherapy agents, such as bleomycin or cisplatin, which enables the entry of drugs into the cells to increase antitumour activity. 7 In veterinary medicine, ECT has been used as the first therapeutic modality and in adjuvant or neoadjuvant scenarios for specific tumour histotypes, such as cutaneous mast cell tumour, soft tissue sarcoma (STS) and perianal gland tumour, among others. 8 In cases of nasal planum SCC in cats, ECT has been applied to treat both initial and advanced stages of the disease with good responses and tolerance, but, particularly in bigger tumours (stages T3 and T4), ECT has been associated with early recurrence.9 –12 In human medicine, ECT has been used to treat cutaneous or subcutaneous metastasis, non-cutaneous metastases located in bone and liver and STS. It has been used in clinical trials for the treatment of primary tumours, such as ovary or colon cancer. 13 In one study in humans, ECT was found to be a safe and effective local treatment in patients with cutaneous and subcutaneous melanoma metastases and in patients with different primary and secondary skin tumours, including aggressive and larger tumours. 14 Based on these promising results and the lack of robust information about the efficacy and tolerance of ECT in the treatment of locally advanced stage nasal planum SCC in cats, the main aim of this retrospective study was to assess the tolerance and outcome of ECT in cats with locally advanced nasal planum SCC as an alternative to a radical surgical approach.
Materials and methods
Patients
Between November 2018 and May 2022, 27 cats were diagnosed with a nasal planum SCC in our institution (AniCura Glòries Veterinary Hospital), of which 21 fulfilled the inclusion criteria and were enrolled in the study. The inclusion criteria were as follows: patients treated with ECT for nasal planum SCC (patients with recurrence after nosectomy or imiquimod use were accepted); a minimum of 6 months of follow-up after the first ECT treatment; and patients with SCC staged as T3N0M0 or T4N0M0 following the World Health Organization TNM classification system for feline tumours of epidermal origin (Table 1). Patients with concurrent grave morbidities were excluded.
Information recorded included breed, sex, age, presenting clinical signs, location of the tumour, pre- and postoperative pictures, previous local or systemic treatments applied, feline immunodeficiency virus (FIV)/feline leukaemia virus (FeLV) status, complete blood cell count (CBC) and biochemistry profile, urinalysis, staging procedures, clinical stage, histological or cytological diagnosis, ECT treatment details and additional adjuvant or rescue therapies.
Electrochemotherapy treatment procedures
Electroporation of the tumour was performed with an electric pulse generator (OnkoDisruptor) under general anaesthesia. Cats were premedicated with a combination of medetomidine (Sedator 1 mg/ml; Dechra), midazolam (Midazolam Normon 5 mg/5 ml; Normon S.A.) and buprenorphine (Bupredine Multidose 0.3 mg/ml; Dechra), butorphanol (Torphadine 10 mg/ml; Dechra) or methadone (Metasedin 1 mg/ml; Esteve) at standard dosages. General anaesthesia was induced with propofol (Propofol Lipuro 10 mg/ml; Braun) and maintained with isoflurane (Isospire; Dechra). The target surface to be treated was shaved as needed and scabs and necrotic material were removed. The region was cleaned with chlorhexidine 1% (Desinclor 1%; Imark). An intravenous bolus of 15,000 IU/m2 of bleomycin (Bleomicina; Almirall) was then administered. Five minutes later, the clinician proceeded with the application of electric pulses (eight biphasic pulses of 100 µs) with plates and/or needles (parallel position, 1 cm apart). Conductive gel was used in every pulse. The relationship between voltage and distance used was in the range of 1000–1200 V/cm. All patients were provided with oral meloxicam (Metacam 0.5 mg/ml; Boehringer Ingelheim) and additional analgesia with buprenorphine (Bupredine Multidose 0.3 mg/ml; Dechra). Cephalexin (Convenia; Zoetis) or amoxicillin clavulanic acid (Synulox 140/35 mg/ml; Zoetis) were prescribed at the discretion of the clinician in charge based on the macroscopic aspect of the tumour. All the drugs were prescribed at standard dosages.
Treatment response and toxicity
The response to treatment was assessed visually by the primary clinician based on the clinical appearance of the treated surface and was evaluated according to the Veterinary Cooperative Oncology Group Response Evaluation Criteria in Solid Tumors (VCOG RECIST, version 1.0). The evolution of the treated surface was documented photographically on each reexamination. A complete response (CR) was defined as the complete eradication of the treated tumour with reepithelialisation of the skin. A partial response (PR) was defined as a notable decrease (>30%) of the macroscopic tumour. Progressive disease (PD) was defined as either the appearance of one or more new lesions or at least a 20% increase of the target lesion. Stable disease (SD) was defined as the presence of minor changes in the target lesion; less than 30% reduction or 20% increase. The definitive response was determined 4–6 weeks after the treatment, when healing should have been completed. Follow-up was performed weekly for the first month and then every 1–3 months by emails with photographs, phone calls or face-to-face consultation.
ECT toxicity was graded according to the VCOG-Common Terminology Criteria for Adverse Events (VCOG-CTCAE, version 2). Dyspnoea, pain, anorexia and lethargy were assessed. An additional adverse event titled erythema and scabs was created at our discretion based on the general aspects defined in VCOG-CTCAE (Table 2). Mild adverse events were considered grade ⩽2, and severe adverse events were considered grade >2. The classification of adverse events was done retrospectively by reviewing medical records, as assessed by the clinician in charge of the case at the time of each re-examination. Follow-up information was acquired from the medical records of referring veterinarians. The surgical management of adverse events was recorded.
Grading system used to assess electrochemotherapy toxicity

Based on Veterinary Cooperative Oncology Group-Common Terminology Criteria for Adverse Events (VCOG-CTCAE, version 2)
ADL = activities of daily living (eating, drinking, sleeping, defecating and urinating); IV = intravenous; NSAID = non-steroidal anti-inflammatory drug
Statistical analysis
CR and PR rates were defined as the number of cats achieving CR or PR expressed as a percentage of the total number of cats treated and for which response information was available from the medical records. Overall response rate (ORR) was defined as the percentage of cats that showed partial or complete response to the treatment in the study period. Overall survival (OS) was defined as the time between the start of treatment and the date of death by any cause. Patients that were lost to follow-up or were alive by the end of the study period were censored for the purpose of survival analysis. Progression-free survival (PFS) was defined as the time from the start of treatment to the date of disease progression. This term was used in those patients that achieved a PR. Disease-free interval (DFI) was defined as the time from treatment initiation to the date of relapse. It was applied to those patients that achieved a CR. Time to progression (TTP) was defined as the time from treatment initiation to the date of relapse or progression. All patients achieving a PR or CR were included. For all three categories, censorship was applied for those patients that were lost to follow-up, died for a different cause without evidence of progression or patients that were still alive without relapse or progression.
A Kaplan–Meier product-limit method was used to estimate survival and the log-rank test was applied to test differences between the following variables: retroviral status; response to the treatment; recurrence; number of treatments; voltage used; and severity of adverse events. Statistical significance was set at P <0.05. Statistical analyses were performed using commercial software (SPSS version 28.0; IBM Corp).
Results
A total of 21 cats fulfilled the inclusion criteria and were enrolled in the study. Individual data for each patient are summarised in Table 3. Six cats were excluded due to the presence of another life-threatening disease or by the multifocal presentation of the SCC. The median age at diagnosis was 10 years (range 5–13). Twenty cats were domestic shorthairs and one was a Turkish Angora; 12 cats were neutered males and nine were neutered females.
Summary of signalment, clinical stage, diagnosis, staging, therapy, response and survival for all cases included in the study

BC = biochemistry; C = cytology; CBC = complete blood count; CR = complete response; CT = computed tomography; Cx = surgery; DFI = disease-free interval; DSH = domestic shorthair; Dx method = diagnostic method; ECT = electrochemotherapy; FeLV = feline leukaemia virus; FIV = feline immunodeficiency virus; FN = female neutered; FNA = fine-needle aspiration; H = histopathology; M0 = no distant metastasis; MN = male neutered; N0 = no lymph node metastasis; PFS = progression-free survival; PR = partial response; OS = overall survival; T3 = tumour >5 cm or with invasion of the subcutis irrespective of size; T4 = tumour invading other structures such as fascia, muscle, bone or cartilage; TOC = toceranib phosphate; TOC-M = toceranib phosphate-meloxicam; UA = urine analysis; WHO = World Health Organization; XR = radiograph
Information on presenting clinical signs was available for all cases but some information was missing in cases referred from shelters. Presenting signs included abnormal upper respiratory tract sounds (n = 7), pain (n = 6), lethargy (n = 3), hyporexia (n = 1) and sneezing (n = 1). Five patients were receiving medication at the time of the first consultation: antibiotics (n = 3); meloxicam (n = 1); and prednisolone (n = 1).
All patients had the tumour located in the nasal planum with a locally advanced stage of the disease. Seven were T3N0M0 and 14 were T4N0M0. At presentation, two patients had had recurrence of the disease after a previous nosectomy, one had been treated with topical imiquimod and 18 were naive to therapy. In 12 patients, both nostrils were affected, and in nine patients, only one nostril was affected or the lesion was located dorsally. The diagnosis of SCC was based on cytology in four cats and on histopathology in 17 cats. Staging methods included CBC and biochemistry in 21 cases, urine analysis in three cases, one nasal culture, submandibular lymph nodes cytology in 21 cases, thoracic radiographs in 10 cases and two head CT scans (one included neck and chest). No cases had documented SCC spread to other locations based on available staging. Twelve cats tested negative for FeLV and FIV, five tested positive for FIV and three positive for FeLV. One cat was not tested.
By the end of this study, updates were available for all patients through communication with either the referring veterinarian or the owner, as was a photographic document for assessment. None of the patients that originally met the inclusion criteria were lost to follow-up or died in the first 6-month period. In total, 17 patients had a single ECT session and four patients had two sessions 17–31 days apart. The bleomycin dose was 15,000 IU/m2 for all patients. The relationship between voltage and distance used was 1200 V/cm in two cats, 1100 V/cm in 12 cats and 1000 V/cm in seven cats. Nineteen (90.5%) cats achieved a CR and two (9.5%) cats achieved a PR for an ORR of 100%. Figures 1 and 2 show the CR in two patients. Of the four cats receiving two sessions, three had a CR and one had a PR. Those cats with a CR had a median DFI of 182 days (range 128–327) and those with a PR had a median PFS of 156.5 days (range 122–191). The median TTP was not reached (Figure 3).

(a) Patient 3 (stage T3N0M0) at presentation; and (b) the patient in complete remission 96 days after treatment
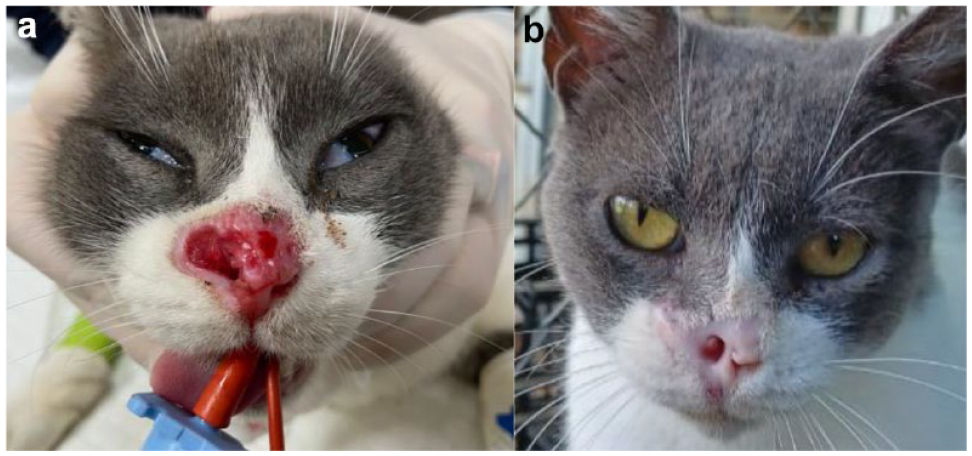
(a) Patient 18 (stage T4N0M0) at presentation; and (b) the patient was in complete remission 223 days after treatment

Kaplan–Meier curve for time to progression (TTP). The censored cats were those that were lost to follow-up, died from a different cause without evidence of progression or patients still alive without relapse or progression at the time of the analysis
Six cats died during the study, four from tumour-related causes and two from other causes, including chronic kidney disease, cardiac disease and primary hepatic epithelial tumour (diagnosed by cytology). Of the 15 cats still alive at the end of the study, four had local recurrence documented by cytology. Staging was determined with submandibular lymph node cytology in all four patients; thoracic radiographs in one cat and a CT scan (head, neck and thorax) in one cat were deemed metastasis free. Of these four cats, one was treated with an additional ECT session and had a relapse documented 154 days later; two cats had cytoreductive surgery and intraoperative ECT and showed a relapse 31 and 142 days later; and one cat was treated with toceranib phosphate and meloxicam as rescue therapies, which were discontinued due to gastrointestinal adverse events. All these patients were receiving symptomatic medication, showing a good quality of life with mild upper airway signs and local discomfort at the time of writing. OS for all patients was 453 days (95% confidence interval [Cl] 286–620) for a median follow-up of 341 days (range 191–989).
Voltage administered was associated with better outcome (P = 0.001; median 453 V/cm, 95% Cl 286–619). The clinical response (P = 0.79), number of treatments (P = 0.78), recurrence rate (P = 0.61), FIV+ status (P = 0.321), FeLV+ status (P = 0.127) and side effects (P = 0.54) were not statistically significant for survival.
Regarding toxicities, mild adverse events were seen in eight (38%) cats and severe adverse events were seen in 13 (62%) cats. Of the 12 cats with both nostrils affected, 11 (91%) experienced severe adverse events and only one (8%) had mild complications. For the remaining nine cats, seven (77%) experienced mild adverse events and two (22%) had severe events. The most common event was local discomfort associated with erythema and the formation of scabs, both present in all patients. Of the 12 cats with both nostrils affected, seven presented with significant obstruction before treatment. All patients presented grade 4 and grade 3 adverse events associated with erythema and scabs, respectively. Those with grade 4 toxicity required intervention. Two had regional debridement and four had nasal reconstruction with sutures (Figure 4) and nasal stents placement (Figure 5), with the aim of permeabilising the airways. A feeding tube was placed in four patients. No deaths due to the treatment were documented, and all patients recovered completely without sequelae once the nasal planum was healed.
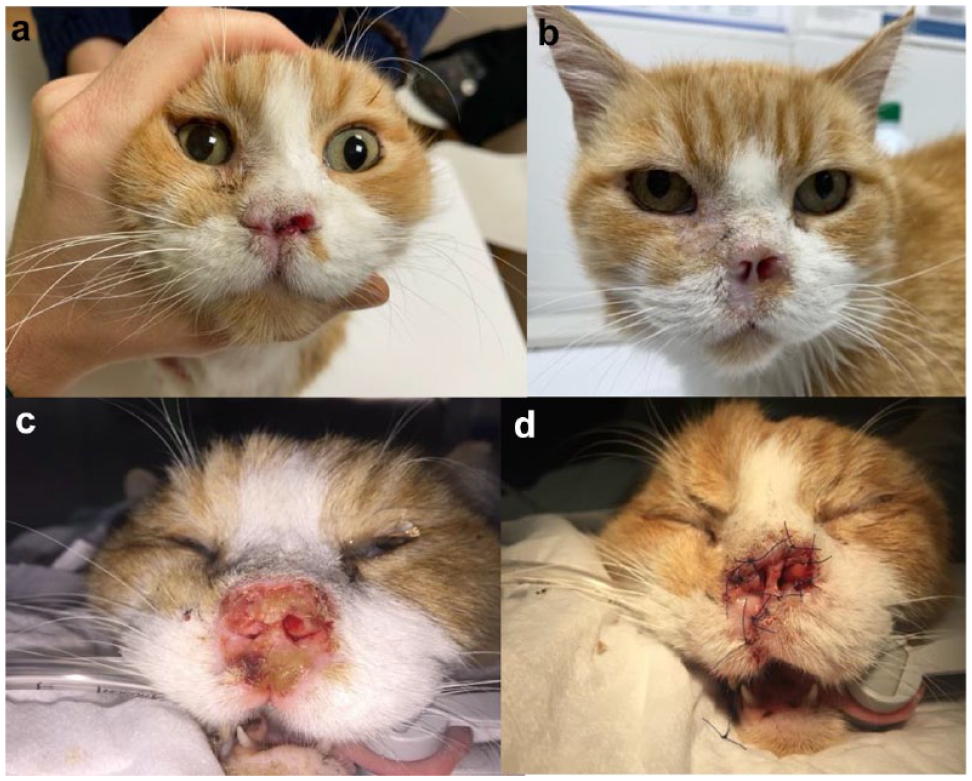
(a) Patient 4 (stage T3N0M0) at presentation; (b) patient 4 at 260 days after presentation; (c) 22 days after the electrochemotherapy session, the patient was completely obstructed; and (d) a nasal plasty was performed

(a) Patient 9 (stage T3N0M0) at presentation; (b) patient 9 at 33 days after the treatment; and (c) nasal stent placement in patient 9
Discussion
This study suggests that ECT is effective for the treatment of locally advanced nasal planum SCC in cats, allowing long-term control of the disease. Only cats with the tumour located at the nasal planum and stages T3N0M0 or T4N0M0 were included in this study; in comparison, previous studies have commonly included cats with tumours in different locations and at earlier stages of disease.5,6,9 –11 The ORR in this study was 100% and in previous studies, it has been greater than 89%.11,15 In this study, the majority of the patients achieved a CR, similar to other reports if the population is assessed as a whole. However, contrary to our findings, previous studies have reported only short-term control in cats with locally advanced disease due to a poor response to the treatment.6,10
No objective measurements were taken to assess the response to the treatment since these evaluate the ulcerated surface only and ignore deep infiltration, which is, most likely, the reason for therapy failure. Advanced imaging is usually of little help in ulcerated skin lesions in cats; therefore, follow-up rechecks and photographic documentation were considered the best methods to evaluate response and relapse events. A CT scan was performed in only two cases where deep nasal cavity infiltration was suspected. This tool could help in ECT procedure planning.
None of our patients presented with metastatic disease; however, in some patients, the chest was not assessed because of the low distant metastatic rate reported for SCC of the nasal planum in the absence of nodal infiltration. 15 That is why local control of the disease correlates better with prognosis, as suggested in our study.
In our study, DFI and PFS were calculated from the first day of ECT treatment rather than from the response endpoint evaluation 4–6 weeks after therapy. We reasoned that, while the treatment day was set at a specific time, the date of the evaluation of the response could depend on the compliance of the owners, the idiosyncratic speed of response and healing in each case, and, most of all, the difficulty of evaluating when the maximum response was achieved vs second intention healing. This was also a problem we faced when trying to evaluate response, but within the first 6 months of follow-up, it could be definitively defined if the response achieved was partial or complete but very difficult to define the specific time that this could have happened, and we could have set a starting date weeks or months apart from the real biological date.
No lethal toxicities were reported in this study; however, the toxicities documented were more severe than previously reported.5,6,9 –11 The location of the tumour and the locally advanced stage of the disease could be the main reasons for these marked adverse events. However, the equipment used was also considered important in terms of adverse events. In our patients, we used plates or needles depending on the macroscopic aspect of the tumour. The needles were thick and reusable, producing more tissue damage. The use of thinner single-use needles has shown less tissue damage, reducing anorexia and providing a better recovery with equally good results in terms of efficacy. 12 The use of plates could lead to a higher volume of healthy tissue being included in the field but also more superficial treatment, which could explain the marked inflammatory reactions and recurrence rate seen in our study.
The present study has some limitations. It was retrospective in nature with a relatively low number of cases; a significant proportion of them were censored as they were still alive at the time of writing the manuscript. The minimum follow-up period was 6 months so the response rate and disease-related mortality may be underestimated. Adverse event evaluation was also subjective, sometimes based on photographs or owners’ explanations.
Conclusions
Our results reinforce the growing evidence that ECT is the treatment of choice for the management of locally advanced stage nasal planum SCC in cats. Toxicities reported can be severe in the short term; however, with medical and/or surgical management, a complete resolution without any sequelae is expected. Specific equipment could reduce the severity of local adverse events.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
