Abstract
Objectives
Feline cutaneous squamous cell carcinoma (cSCC) is a malignant tumour arising from squamous epithelium and accounts for 15% of all skin tumours, commonly involving the nasal planum, pinnae and eyelids. Electrochemotherapy (ECT) combines the application of short high-voltage electric pulses with intravenous or intralesional administration of cytotoxic agents to enhance the efficacy of chemotherapy. This study aimed to investigate the efficacy of ECT with intravenously administered bleomycin in the treatment of different stages of cSCC (T1–T4).
Methods
A total of 23 cats with cSCC located on the nasal planum (19/23), lip (1/23), eyelid (1/23), metacarpal pad (1/23) and temporal region (1/23) were included in this retrospective study. Cats were excluded if they did not have a histological or cytological diagnosis of cSCC or if records were incomplete. All cats were treated following a standard protocol with intravenous bleomycin before electroporation of cSCC lesions. Cats were staged according to the World Health Organization staging system: T1 (10/23), T2 (4/23), T3 (1/23) and T4 (8/23). Data on treatment adverse effects and response were collected.
Results
The median follow-up after initial treatment was 136 days (range 7–1763). Common adverse effects were local effects in 13/23 cats consisting of erythema, desquamation (dry/moist), ulceration and crusting. Complete response (CR) was achieved in 65.3% of cases, partial response in 13%, stable disease in 8.7% and progressive disease in 13%. Eight cats received a second ECT treatment, with 4/8 cats achieving CR after the second treatment. The overall response rate was 78.3%. The recurrence rate was 26.7%, with a disease-free interval of 466 days.
Conclusions and relevance
ECT is effective in treating T1 and T2 cSCC and can be considered a relevant treatment alternative for these cases.
Introduction
Feline cutaneous squamous cell carcinoma (cSCC) is a common malignant tumour arising from squamous epithelial cells and accounts for approximately 15% of all skin tumours, commonly involving the nasal planum, the ear pinnae and eyelids. 1 Geriatric cats with a mean age above 10–12 years are at a higher risk of developing cSCC. 2 cSCC is typically associated with a history of chronic ultraviolet (UV) light exposure, with lesions displaying a spectrum of severity both clinically and histopathologically. 3 Histological make-up can range from actinic dysplasia to carcinoma in situ (full-thickness hyperplasia confined to the epidermis) and to invasive cSCC. 3 Clinical presentation generally ranges from plaque-like lesions to more papillary/fungiform and eventually erosive and ulcerated lesions with crust formation. 3 Cats with non-pigmented or lightly pigmented skin with sparse hair coat coverage are at a much higher risk of developing cSCC. 4 cSCCs are usually locally invasive and slow to metastasise, 3 with metastasis to the lungs and draining lymph nodes most commonly reported. 5
In general, treatment success and outcome depend upon clinical stage, invasiveness of the tumour, and location and extent of lesions. Treatment is often more rewarding in the initial onset of disease. Surgical excision in the form of a nosectomy, with margins of at least 4–5 mm, is an effective method to achieve local tumour control and can result in a cure for most cSCCs. 6 This remains difficult for higher-stage cases (Table 1). 7 However, some pet owners may find the postoperative cosmetic outcome challenging. Therefore, alternative treatment pathways have been explored to offer a potentially better postoperative cosmetic outcome for pets. These include radiation therapy, 8 strontium-90 plesiotherapy,9,10 curettage and diathermy, 11 cryosurgery, 12 photodynamic therapy, 13 intralesional chemotherapy 14 and electrochemotherapy (ECT).11,15
World Health Organization staging system 7

ECT was first described in 1991 and has been associated with encouraging results in veterinary medicine. 16 It is an innovative treatment that combines the application of short high-voltage electric pulses (reversible electroporation) with intravenous (IV) or intralesional administration of cytotoxic agents, most commonly bleomycin and cisplatin, to enhance the efficacy of chemotherapy. 17 These drugs are hydrophilic and do not readily enter cells. Electroporation-induced permeabilisation of tumour cell membranes improves bleomycin uptake by up to 700-fold. 17 This process enhances cytotoxic effects within the tumour leading to apoptosis of tumour cells.17,18 Aside from increasing the permeability of tumour cell membranes, two further working mechanisms have been described for ECT that enhance the antitumour effect. ECT induces tumour cell death by decreasing blood supply within the tumour (vascular lock) and by destroying the endothelial cells of small blood vessels (vascular disrupting action). 19 ECT also causes the shedding of tumour antigen in treatment areas, which can result in the activation of the immune system against tumour cells. It may therefore also have an immune-modulatory effect that could enhance the detection and destruction of metastatic tumour cells. 19
Bleomycin has been the drug of choice for use with ECT in the treatment of cSCC in cats according to recent publications.15,18,20 This is because of its ease of use and compliance with health and safety guidelines around administration in small animals. Its main cytotoxic mechanism of action involves the fragmentation of DNA, which arrests the G2-M phase of the cell cycle. 17 Bleomycin is also affordable and well tolerated in cats.15,20
The objective of this study was to investigate the efficacy of ECT in the treatment of feline cSCC. Our hypothesis was that ECT is highly effective in treating low-stage (T1 and T2) cSCC with little to no systemic adverse effects and minimal disruption to quality of life. This publication investigated the use of ECT in a single centre standardising the cytotoxic agent used, limiting application to two people and delivering electric pulses via the same probe each time under a hospital standard protocol.
Materials and methods
Patient selection and staging
A total of 23 privately owned cats with cSCC were included in this retrospective study. All cats were treated for cSCC with ECT at a single veterinary hospital between 2017 and 2024. Cats were excluded if they did not have a histological or cytological diagnosis of cSCC or if their medical records were incomplete. Further staging comprised fine-needle aspiration of the regional lymph nodes, three-view thoracic radiographs or thoracic CT scans. These were performed based on the clinician’s and client’s preference.
Information collected included signalment, coat colour, weight, size of lesion, tumour stage, number of ECT treatments, response to treatment, local and systemic adverse effects, recurrence rate and median survival time. A survey was also sent out to referring veterinarians at the time of this study to collect follow-up histories for all patients in this study.
Treatment
Following a hospital standard protocol, all cats were treated under general anaesthesia with IV bleomycin before electroporation of cSCC lesions. All cases were premedicated with medetomidine (2 μg/kg IV, Domitor 1 mg/ml; Zoetis) and methadone (0.2 mg/kg IV, Methadone 10 mg/ml; Ilium) if temperament allowed or medetomidine 5 μg/kg and methadone 0.3 mg/kg IM before IV catheter placement. IV fluid therapy was provided perioperatively using Hartmann’s solution. Alfaxalone (0.5–1.5 mg/kg IV, Alfaxan Multidose 10 mg/ml; Jurox) was used as an induction agent, titrated to effect. Patients were intubated and maintained with isoflurane (Isoflurane USP; Covetrus). Bair Hugger active warming (Solventum) was provided for all patients. All treatment areas were clipped and aseptically cleaned in preparation for electroporation. The body surface area (in m2) of each patient was calculated based on the formula:
0.1 × (body weight in kg)2/3
IV bleomycin was administered at a concentration of 15,000 IU/m2 8 mins before electroporation was undertaken. Electrocardiogram electrodes were removed before electroporation to minimise the risk of electric pulses being transported through the body. During electroporation, eight biphasic electric pulses each lasting 50 + 50 μs at intervals of 1 ms were delivered with the small needle electrode at a depth of 3–5 mm. The needle electrode was applied circumferentially around the lesion and then overlying the lesion. Ultrasound conductive gel was applied to the treatment area before electroporation to improve contact between the treatment area and the needle electrode. Figure 1 shows a patient undergoing ECT.
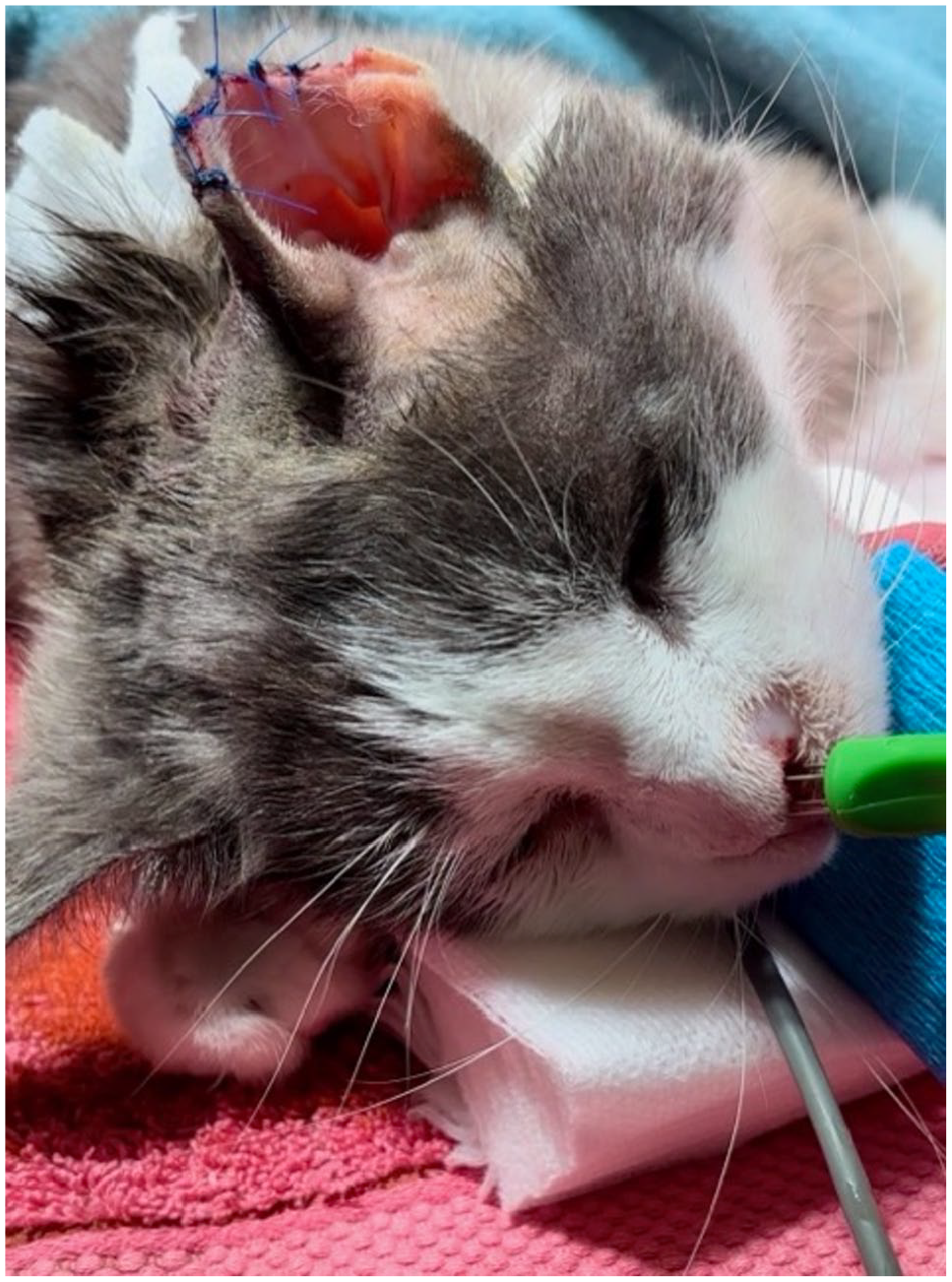
Patient undergoing electrochemotherapy
The electroporator used was an Electrovet S13 (Leroy Biotech) at an amplitude:electrode distance ratio of 1300 V/cm. The electrode consisted of two parallel rows of two needles each measuring 10 mm in length. The diameter of the needles was 0.68 mm, the gap between the needle tips was 3.15 mm and the rows of needles were spaced 4 mm apart.
All patients were discharged on the day of the ECT with a course of meloxicam (0.05 mg/kg/day PO 3–10 days, Metacam 0.5 mg/ml; Boehringer Ingelheim) for analgesia.
Response
Response rates were categorised according to Response Evaluation Criteria in Solid Tumours (RECIST) created by the Veterinary Cooperative Oncology Group (VCOG) to provide an objective measurement of tumour burden. 21 Complete response (CR) was defined as complete elimination of the treated lesion. Partial response (PR) was defined as a reduction of more than 30% in size, as defined by the longest diameter, of the gross lesion. Progressive disease (PD) was defined as an increase of more than 20% in the size of the treated lesion or the emergence of one or more new lesions. Stable disease (SD) was defined as a reduction of less than 30% or increase of less than 20% in the size of the treated lesion.
Adverse effects
Adverse effects were categorised according to the Common Terminology Criteria for Adverse Events (CTCAE) created by the VCOG to provide consistency in the assessment of any adverse effects noted after ECT treatment. Grade 1 (mild) was defined as clinical signs or observations not requiring intervention, grade 2 (moderate) was defined as clinical signs requiring outpatient or non-invasive intervention, grade 3 (severe) was defined as clinical signs requiring hospitalisation or prolonged hospitalisation, grade 4 (life-threatening) clinical signs required urgent interventions and grade 5 (death) clinical signs led to euthanasia or natural death.
Statistical analysis
Overall response rate (ORR) was defined by the percentage of cats achieving CR or PR. The recurrence rate (RR) was defined by the number of cats that experienced local recurrence after achieving CR. Disease-free interval (DFI) was calculated from the date of first treatment to the date of local recurrence or evidence of metastasis. The median survival time (MST) was calculated from the date of first treatment to the date of death for all cats that died from cSCC. All statistical analyses were performed with SPSS version 28 (IBM Corp), with the statistical significance level set at P <0.05.
Results
A total of 23 cats were included in this study. There were eight (34.8%) females and 15 (65.2%) males. All cats were desexed. The breed distribution was as follows: domestic shorthair (16/23), domestic mediumhair (4/23), domestic longhair (2/23) and Devon Rex (1/23). All cats with nasal planum cSCCs had white fur coats over their noses with various coat colour combinations for the rest of their bodies. The median age of the patients at presentation was 11 years (range 3–18).
cSCC lesions were located on the nasal planum (19/23), lip (1/23), metacarpal pad (1/23), temporal region (1/23) and eyelid (1/23). cSCCs were confirmed via cytology (4/23) or histopathology (18/23) or both (1/23). Of the 19 cats with nasal planum cSCC, nine had cSCCs with bilateral nasal planum involvement. The nasal planum lesions had a diameter in the range of 5–30 mm, with the bigger lesions extending towards the nasal bridge, the adjacent lip or both. The lip and metacarpal pad lesions both measured 15 mm in diameter, while the temporal and eyelid lesions were 10 mm in diameter.
Cats were staged according to the World Health Organization staging system based on the size and invasiveness of cSCC lesions: T1 (10/23), T2 (4/23), T3 (1/23) and T4 (8/23). Nine cats also underwent further staging in the form of three-view thoracic radiographs (4/9), thoracic CT scans (3/9) and fine-needle aspiration of the regional lymph nodes (5/9). Of the cats that underwent further staging, none showed any evidence of metastatic disease. The remaining cats had no staging but were clinically well and the regional lymph nodes palpated normally.
ECT was the sole therapy performed on all cats in this study. Of the 23 cats, 15 were treated with just one session of ECT while the remaining eight cats underwent two sessions (Table 2). ECT was repeated in two patients with recurrence after initial CR which occurred 16 and 90 weeks after the first treatment, respectively: one cat subsequently achieved CR, the other achieved PD. ECT was repeated a median of 8 weeks (range 7–19) after initial treatment in four cats with PR and in one cat with SD. Of these five cats, three achieved CR after the second treatment. A repeat treatment for a patient with PD was also performed 5 weeks after the initial treatment. This patient continued to have PD.
Summary of signalment, method of diagnosis, tumour location, tumour staging, number of electrochemotherapy (ECT) treatments, response and outcome for all cases included in the study
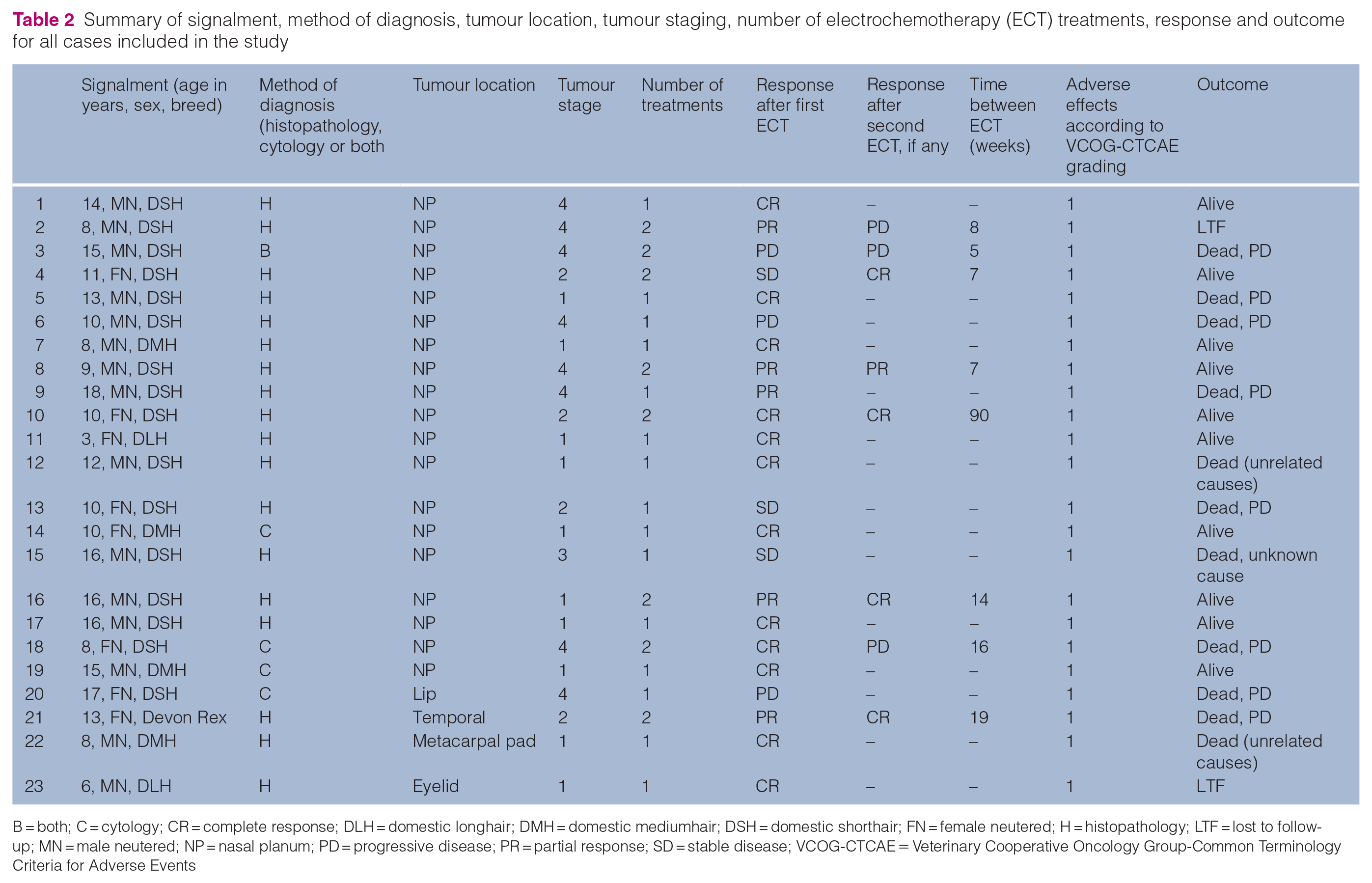
B = both; C = cytology; CR = complete response; DLH = domestic longhair; DMH = domestic mediumhair; DSH = domestic shorthair; FN = female neutered; H = histopathology; LTF = lost to follow-up; MN = male neutered; NP = nasal planum; PD = progressive disease; PR = partial response; SD = stable disease; VCOG-CTCAE = Veterinary Cooperative Oncology Group-Common Terminology Criteria for Adverse Events
The median follow-up after initial treatment was 136 days (range 7–1763). At the time of data analysis for this study, 11 cats had died: eight of these cats died as a result of their cSCC based on our records or records from the referring veterinarian, two cats died from chronic kidney disease and the cause of death for the remaining cat was unrecorded. The MST for the eight cats that died because of cSCC was 447 days (range 46–993). Of the 10 cats that were verified still alive, nine were in CR and one was in PR. Two cats were lost to follow-up at 7 and 66 days. At the time of the last follow-up, one cat was in CR and the other was in PD.
Common adverse effects were local effects in 13/23 cats, including erythema (2/13), desquamation (dry/moist) (7/13), ulceration (4/13) and crusting (4/13). All cats presented with some degree of skin abnormality as part of the disease process; adverse effects of the ECT were therefore recorded when new or more severe signs were noted after ECT. Those subsided after a median of 10 days (range 7–14). None of the cats required additional interventions to help with their breathing or ability to eat. Systemic adverse effects, such as inappetence (1/23), decreased defecation (1/23) and lethargy (1/23), were also reported. All systemic adverse effects were considered to be grade 1 (mild) according to the VCOG-CTCAE grading system. 22 These systemic effects lasted for a median of 5 days (range 3–7) and are likely attributed to general anaesthesia and stress in hospital rather than true chemotherapy-associated adverse effects.
CR was achieved in 15/23 (65.3%) cases, PR in 3/23 (13%), SD in 2/23 (8.7%) and PD in 3/23 (13%). The ORR was 78.3%. Of the 15 cases that achieved CR, four cases subsequently experienced local recurrence. The RR was therefore 26.7% (4/15), with a DFI of 466 days (range 111–815). The overall MST was 211 days (range 30–909).
The breakdown of responses by tumour stage is listed in Table 3. Of the 10 cats with T1 tumours, six were still alive at the time of this study. One cat was euthanased after tumour recurrence 563 days after initial ECT, two died from chronic kidney disease and one cat was lost to follow-up 7 days after treatment. Of the four cats with T2 tumours, two were still alive at the time of this study with no visible recurrence; the other two cats were euthanased 869 and 993 days after initial ECT because of disease progression. The death of the only cat with a T3 tumour was unrecorded. Of the eight cats with T4 tumours, five died because of cSCCs, with the MST for this subset of cats being 132 days (range 30–230). Two of the remaining cats with T4 tumours were still alive and one was lost to follow-up 66 days after treatment. Figures 2 and 3 show CR and PR, respectively, in patients with T4 tumours.
Electrochemotherapy response rate by squamous cell carcinoma (SCC) tumour stage

Data are n (%)
CR = complete response; PD = progressive disease; PR = partial response; RR = recurrence rate; SD = stable disease

Patient with stage T4 squamous cell carcinoma at (a) 8 weeks, (b) 9 weeks, (c) 12 weeks and (d) 3.5 months after treatment

Patient with stage T4 squamous cell carcinoma at (a) treatment, (b) partial response at 5 weeks and (c) recurrence at 13 weeks
Discussion
The present study investigated the efficacy and adverse effects of ECT in the treatment of feline cSCC and presented a hospital standard treatment protocol with a single electroporator at a set amplitude:electrode distance ratio. Our study demonstrated that ECT was an effective procedure with low morbidity, achieving a CR rate of 65.3%. This CR rate was not significantly different from previously reported CR rates in the range of 65.6–81.8 %.15,18,23 Our MST of 211 days and RR of 26.7% were also not significantly different from previously reported MSTs of 193 23 and RRs in the range of 22.2–22.5%.18,23
However, our finding of a DFI of 466 days was much longer than previously reported DFIs of 136–182 days.23,24 This difference may be due to differences in tumour stages, with either only DFI for higher-stage patients reported 24 or information regarding breakdown of tumour stages being unavailable. 23 There was also variation in terms of the number of ECT treatments used, electroporators, probe and amplitude:electrode distance ratio used between studies. As such, it was impossible to make a direct comparison.
The CR rates for tumour stage T1 and T2 cSCCs were 100% and 75%, respectively. When compared with other treatment options, ECT provided added benefits. Strontium-90 plesiotherapy has previously been shown to produce CR rates in the range of 75–88%.9,10,25 However, strontium plesiotherapy treats cSCCs up to a depth of only 2 mm, with the dose delivered at a 2 mm depth only approximately 30% of the surface dose.23,25 In ECT, the depth of treatment is determined by the length of the needle probe, with a reported treatment depth of up to 7 cm. 26 At the hospital where the present study was based, superficial tumours with a depth in the range of 5–20 mm were treated. ECT also provided subjectively better cosmetic outcomes than surgical excision. Compared with radiation therapy, anaesthetics were required less frequently, reducing the need for regular visits to the veterinary clinic.
ECT was associated with low morbidity. Common adverse effects were local effects consisting of erythema, desquamation (dry/moist), ulceration and crusting. These adverse local effects were self-limiting and did not require further intervention. We suspected that the reported systemic adverse effects, such as inappetence, decreased defecation and lethargy, were likely due to the effects of general anaesthesia and stress encountered in a hospital environment. These systemic effects were also self-limiting and short-lived, required no intervention and therefore would have been classified as VCOG grade I (mild) toxicities had they been bleomycin induced.
Even though the ORR for T4 tumours was lower at 62.5% in the present study when compared with lower stage lesions, ECT may still be an option to reduce the size of the initial lesion in preparation for surgical excision of the remaining tumour lesion. In a recent report, stage T3 and T4 cases had a reported CR of 90.5% and ORR of 100%, adding to the confidence in this treatment option. 24
Some previously published papers18,23 utilised monophasic pulses for their ECT sessions and documented similar results to our study using biphasic pulses. It could be concluded that both monophasic and biphasic pulses are equally effective in treating cSCC. However, biphasic pulses have been shown to enhance the efficiency of cell permeabilisation 27 and elicit a reduced pain response. 28
There was justification for repeat ECT sessions for higher-staged cSCCs if a PR or SD was recorded or recurrence was noted. Between two and four sessions of ECT have been reported to date.15,18,23 In our study, 8/23 cats underwent two ECT sessions, with 4/8 cats achieving CR and 1/8 cats achieving PR after the second ECT session. Given the low morbidity associated with ECT and the improved response rates after additional ECT, a repeat ECT session warranted consideration. The potential need for repeat ECT sessions should be discussed during initial conversations with owners to help manage expectations.
The main limitations of our study are its retrospective nature and low number of cases. The assessment of local adverse effects was subjective and up to the interpretation of the two experienced treating clinicians. Most cats presented with some degree of local tissue changes due to the locally destructive nature of cSCC, thus assessment of local adverse effects attributed to ECT specifically can be challenging; any differences from the original presentation were noted in this study.
Conclusions
ECT was effective in treating T1 and T2 cSCCs. Given that ECT was minimally invasive with low morbidity, it may be considered a relevant treatment alternative for lower-stage cSCC cases.
Footnotes
Acknowledgements
Special thanks to Dr Irina Gramer for her supervision and guidance in this study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental animals (owned or unowned animals). Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
