Abstract
Objectives
This study undertook a scoping review of research on blood fibroblast growth factor 23 (FGF-23) concentrations in healthy non-azotemic cats and cats with chronic kidney disease (CKD) to describe the volume and nature of existing literature, to determine whether published studies provide adequate evidence to support the use of FGF-23 as a biomarker in clinical practice and to identify any existing gaps in knowledge.
Methods
PRISMA Extension for Scoping Reviews guidelines were used to design and perform the scoping review. Online databases were used to identify observational and clinical studies of blood FGF-23 concentrations in healthy cats and cats with CKD published before December 2022. Study and population characteristics and descriptive data on FGF-23 concentrations were extracted.
Results
A total of 205 publications were reviewed; 17 were retained for inclusion. Most studies were retrospective. Most studies included cats with International Renal Interest Society stage 2–4 CKD, with some variation. Key concepts explored in the literature include FGF-23 concentrations by CKD stage, effect of dietary phosphate restriction on FGF-23 concentrations, relationship between FGF-23 concentrations and blood phosphorus, calcium and magnesium concentrations, and FGF-23 concentrations in cats with progressive CKD. FGF-23 concentrations tended to be higher in cats with CKD compared with healthy cats, with an overlap between healthy and CKD populations, and there was significant variation within stages of CKD.
Conclusions and relevance
FGF-23 is a biomarker of interest for the management and monitoring of phosphate overload in cats. Studies support several potential clinical applications for measuring FGF-23 concentration in practice; however, evidence is limited. Research on FGF-23 in cats with CKD would benefit from longitudinal, prospective studies that standardize CKD diagnosis and categorize cats by stage using current guidelines. Studies should include cats with early-stage, non-azotemic CKD and use commercially available assays so such results are comparable across studies.
Introduction
Renal secondary hyperparathyroidism is common in cats with chronic kidney disease (CKD) and is characterized by elevated parathyroid hormone (PTH) secondary to derangements in phosphorus, calcium and vitamin D homeostasis.1–3 CKD–mineral and bone disorder (CKD-MBD) is a consequence of these hormonal and mineral derangements. In people, CKD-MBD is defined as a systemic disorder manifested by either one or a combination of the following: (1) abnormalities of calcium, phosphorus, PTH, fibroblast growth factor 23 (FGF-23) and vitamin D metabolism; (2) abnormalities in bone turnover, mineralization, volume, linear growth or strength; or (3) vascular or other soft tissue calcification. 4 Clinical manifestations of CKD-MBD in people can include muscle weakness, bone pain, pruritus and skeletal fractures.5,6 Although the hormonal derangements of CKD-MBD in cats and dogs are similar to those in people, the clinical signs are less defined in veterinary medicine. In dogs7–11 and cats,8,12,13 CKD-MBD can manifest as reduced cortical bone quality, dystrophic organ and soft tissue mineralization, and osteopenia and osteodystrophy, most often reported in the skull and mandible.
A key hormone in the regulation of calcium–phosphate homeostasis is the phosphatonin peptide hormone FGF-23. FGF-23 binds to its membrane-bound receptor complexed with α-Klotho co-receptor on target cells to increase renal phosphate excretion and reduce calcitriol production in the kidney. 14 Traditional understanding of the pathogenesis of CKD-MBD suggested that hyperphosphatemia and diminished renal mass resulted in reduced renal production of calcitriol and the subsequent development of renal secondary hyperparathyroidism. 15 In the early 2000s, FGF-23 and its co-receptor Klotho were identified, and the binding of FGF-23 to FGF receptor/α-Klotho complexes was shown to increase fractional excretion of phosphate in the nephron to maintain phosphate homeostasis early in CKD. Thus, blood FGF-23 concentrations increase early in people with CKD and before the development of diminished renal mass or overt hyperphosphatemia. 16 An elevated blood FGF-23 concentration, therefore, has been proposed as an early potential indicator of phosphate dysregulation and trigger of CKD-MBD by inhibiting renal calcitriol synthesis, resulting in hypocalcemia and increased PTH synthesis.
FGF-23 is a biomarker of interest in veterinary medicine, and over the past decade several research groups have published studies of plasma or serum FGF-23 concentrations measured in healthy non-azotemic cats and cats diagnosed with CKD. Recently, two assays to measure serum FGF-23 concentration in cats became commercially available to veterinarians to use as a tool in the monitoring and management of phosphate overload in cats with CKD (IDEXX Laboratories; FUJIFILM VET Systems). Overall, it is unclear what kind of information is available in the literature supporting the measurement of blood FGF-23 concentrations in cats with CKD. This study undertook a scoping review of research on FGF-23 in healthy cats and cats with CKD. The objectives of the study were to conduct a systematic search of databases using published methodology to describe the volume and nature of existing literature on FGF-23 in cats with CKD, to chart and summarize FGF-23 concentrations in healthy cats and according to International Renal Interest Society (IRIS) CKD stage, to review and appraise the literature on the use of FGF-23 in feline medicine, and to chart the study methodology from existing publications to identify knowledge gaps and form recommendations for future research.
Materials and methods
Full articles published up to 31 December 2022 were evaluated for inclusion. Articles were considered published if they were available online on the journal website before 31 December 2022, even if the article did not appear in print until 2023. Abstracts without associated manuscripts and unpublished data were excluded. The search was performed using PubMed (https://pubmed.ncbi.nlm.nih.gov), CAB Abstracts and Google Scholar by an investigator (HTM). CAB Abstracts was searched on 17 October 2022. PubMed and Google Scholar were searched on 18 April 2023. For Google Scholar, the first three pages of search results were included in the review. No specialized software was used. Articles identified in the search were then assessed for inclusion by reviewing their abstracts. Only observational or clinical studies that measured serum or plasma FGF-23 concentrations in the context of naturally occurring renal disease in cats were included. Articles published in languages other than English were evaluated for inclusion if they had abstracts translated into English. Article names were searched in Google Scholar, which identified one additional study containing FGF-23 data that was not otherwise identified. Authors were not directly contacted for additional articles. The screening of excluded articles was undertaken by a second reviewer (SS). For the included publications, author names, year of print publication, article title, population description, age of population under study, sample size, inclusion and exclusion criteria, and, if exact values were reported, all available descriptive statistics on FGF-23 (median, first quartile, third quartile, minimum, maximum, range and interquartile range) were extracted. Data were extracted by independent double data extraction (RM, DS) and then further evaluated by other investigators (HTM, SS).
Due to the variations in methodology and lack of clarity around the potential overlap of data points between studies, no meta-analysis or systematic review could be performed, and the authors moved forward with a scoping review. A scoping review was conducted and reported according to the Preferred Reporting Items for Systemic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) guidelines. 17
Results
Study selection
Search terms used in each search engine and the number of items identified by each search term are presented in Figure 1. The search resulted in 205 unique items for evaluation and the number of articles excluded is in Figure 1. A complete report of all items identified from each search term and the reasons for exclusion are presented in Table 1 in the supplementary material. In total, 16 articles were identified from the literature search. A Google Scholar search of individual authors identified one additional article that was not identified in any of the other searches, resulting in a total of 17 articles for inclusion. No single search engine identified all included articles (see Table 2 in the supplementary material). Eight articles were identified by all three search engines, five were identified by two search engines and three were identified by only one search engine. Google Scholar identified the highest number of included articles (n = 15), including one identified in the author search, compared with 11 for PubMed and CAB Abstracts.

Flow diagram showing the number of publications identified and reviewed, and reasons for those excluded from the scoping review of the literature on fibroblast growth factor 23 (FGF-23) in cats with chronic kidney disease (CKD)
Study characteristics
The 17 included studies were published between 2013 and 2022. The sample type and kit used in each study and the study objectives are summarized in Table 1. All studies used a sandwich ELISA to measure intact FGF-23, manufactured by one of four companies: 12 used the Human FGF-23 ELISA Kit (Kainos Laboratories); two used the Cat Fibroblast Growth Factor 23 ELISA Kit (MyBioSource); two used MedFrontier FGF23 (Minaris Medical; MedFrontier); and one used the Feline FGF-23 ELISA Kit (Deyihengda Biotech) (Table 1). Ten of 12 studies that used the Kainos assay and one of two studies that used the MyBioSource assay measured FGF-23 in plasma and the rest used serum. Geddes et al 19 validated the use of the Kainos ELISA to measure intact plasma FGF-23 concentrations in cats, and found the assay to have acceptable precision, reproducibility and specificity for feline FGF-23. The Kainos and MedFrontier assays were reported to use the same primary anti-FGF-23 antibodies with different secondary antibodies. 35 Validation of the MedFrontier assay was presented in the study by Miyakawa et al. 32 Information about the antibodies used in the MyBioSource and Deyihengda kits was not available. Assay validation for the Deyihengda assay was published in the study by Lin et al. 28 At the time of writing, there was no published validation of the MyBioSource assay but information on the precision of the assay was available from the manufacturer. 36 Studies comparing FGF-23 concentration measurements between manufacturers in human and feline samples have found assay-based differences that preclude direct comparison across methodologies.35,37 Therefore, FGF-23 concentrations were not compared across the different kit manufacturers in this study.
Sample type, FGF-23 assay kit and study objectives for the 17 articles included in the scoping review
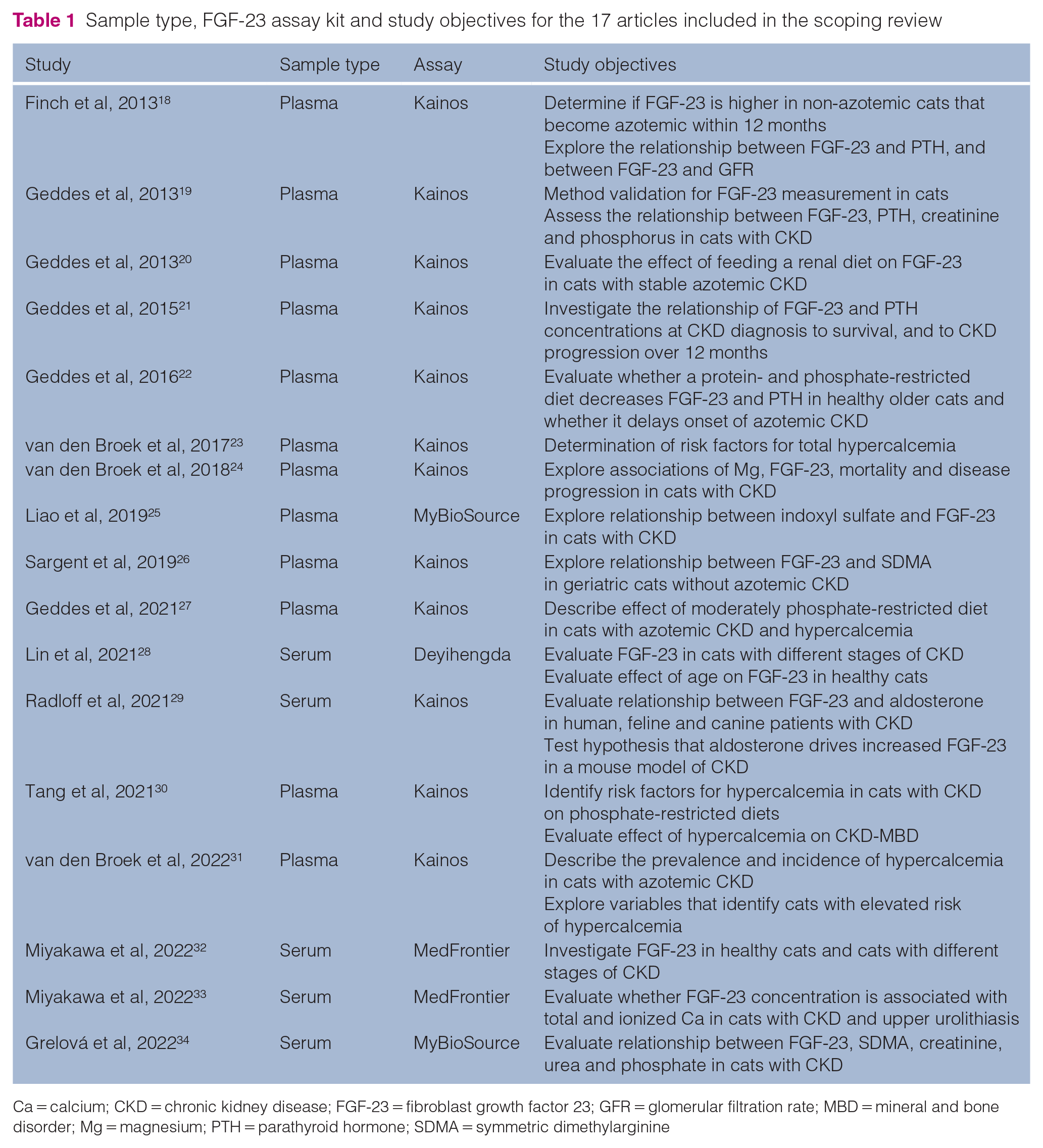
Ca = calcium; CKD = chronic kidney disease; FGF-23 = fibroblast growth factor 23; GFR = glomerular filtration rate; MBD = mineral and bone disorder; Mg = magnesium; PTH = parathyroid hormone; SDMA = symmetric dimethylarginine
Information about the study design, study populations, including geographic location, definitions of health and CKD, and exclusion criteria is provided in Table 2. The included studies were conducted on cats in the UK (11/17), Japan (2/17), and Taiwan, Austria, Slovakia and China (one each). Most studies were retrospective (n = 14), of which eight were cohort studies, five were cross-sectional studies and one included both cross-sectional and cohort components. Three studies were prospective: one was a cross-sectional study; one was a double-blinded, randomized, placebo-controlled clinical diet trial; and one was a cohort study. Seven studies recruited only cats aged 9 years or older and the remaining studies did not state age as a criterion for inclusion.
Study type, country and time frame for sample collection, definition of CKD and inclusion/exclusion criteria for the studies of fibroblast growth factor 23 in cats

People’s Dispensary for Sick Animals (PDSA)
Beaumont Sainsbury Animal Hospital
National Taiwan University Veterinary Hospital
China Agricultural University Animal Teaching Hospital
Teaching Hospital of the University of Veterinary Medicine Vienna
University of Veterinary Medicine and Pharmacy in Kos´ice
Nippon Veterinary and Life Science University
AKI = acute kidney injury; Ca = calcium; CKD = chronic kidney disease; Cr = creatinine; FGF-23 = fibroblast growth factor 23; iCa = ionized calcium; IRIS = International Renal Interest Society; N/A = not applicable; NR = not reported; P = phosphate concentration; P-CL = prospective clinical trial; P-CO = prospective cohort; PTH = parathyroid hormone; PU/PD = polyuria/polydipsia; R-CO = retrospective cohort; R-CX = retrospective cross-sectional; RI = reference interval; SDMA = symmetric dimethylarginine; tCa = total calcium; TT4 = total thyroxine concentration; UPC = urine protein:creatinine ratio; USG = urine specific gravity
Cat population characteristics
The determination of health status (healthy vs CKD) and exclusion criteria varied greatly between studies (Table 2). Most studies (n = 14) included a population of cats with a diagnosis of CKD; the remaining three studies included only healthy cats. All cats included in the studies were client-owned, except for one study 32 that included laboratory cats in the healthy control population. Healthy cats were often defined as non-azotemic, non-diabetic, euthyroid cats (although two studies18,28 did not mention hyperthyroidism as an exclusion criterion, nor did they mention the measurement of total thyroxine concentration in the study population). One study 26 that measured FGF-23 concentrations in apparently healthy older cats (aged >9 years) reported that 25% of included cats had increased concentrations of symmetric dimethylarginine (SDMA; >14 µg/dl). A persistent increase in SDMA concentration is consistent with a diagnosis of early-stage CKD based on the current IRIS CKD staging guidelines. 38 This group of cats could not be classified by the authors of this review as either healthy or CKD since the publication did not report any information about persistence of SDMA concentrations >14 μg/dl. It is therefore plausible that this population of apparently healthy cats contained both healthy cats and cats with stage 1 or 2 CKD based on current IRIS guidelines. A second study by Finch et al 18 included older non-azotemic cats (creatinine <2 mg/dl), and after 12 months grouped cats according to creatinine concentration and, in some instances, urine specific gravity (USG) (groups 1–3) (Table 2). No further detail is provided regarding the diagnosis of CKD, and it is plausible that some cats in group 1 or group 2 were healthy cats without renal dysfunction.
In the 14 studies that included cats with CKD, the definition of renal azotemia varied and included creatinine >2.0 mg/dl (7/14), >1.8 mg/dl (2/14), >1.6 mg/dl (2/14) and was not defined in two studies. The diagnosis of CKD was based on renal azotemia with inadequately concentrated urine (USG <1.030 or <1.035) or persistent azotemia without pre-renal cause in most studies (13/14). One study 34 diagnosed CKD based on clinical signs (including radiographic kidney changes) and either the presence of azotemia (undefined) or persistent elevation in SDMA concentration (>14 µg/dl) without consideration of USG. A few studies included abnormal renal ultrasonographic or radiographic findings (5/14), historical clinical signs of kidney disease (ie, weight loss, polyuria/polydipsia; 3/14) or urine protein:creatinine ratio measurement (one study) in determining the diagnosis of CKD. Only two studies26,34 mentioned measuring SDMA concentrations in their study populations, of which one study 34 used SDMA to diagnose CKD based on the persistence of an elevated value. The other study 26 that measured SDMA concentration included only apparently healthy cats and, as mentioned above, some of the cats had an elevated SDMA concentration.
All the studies from the UK drew patients from the same two clinics with overlapping study periods (Table 2). It was not possible to determine from the information available in the publications if results from individual cats were used in multiple studies or how often or how many overlapping data points may be in the data sets. The two studies performed in Japan32,33 likely also have overlapping data points, but this is similarly unclear. These studies that may have overlapping patient populations included cats with IRIS stage 1 and 2 CKD.
FGF-23 and CKD stage
Five studies subdivided cats by CKD stage, of which four studies provided descriptive information about FGF-23 concentrations by IRIS stage (Table 3; see Figure 1 in the supplementary material).19,25,28,32,33 All these studies used the creatinine concentration to stage. Four studies19,28,32,33 used the 2019 IRIS staging guidelines (stage 2: 1.6–2.8 mg/dl; stage 3: 2.9–5.0 mg/dl; stage 4: >5 mg/dl); however, one study 19 used a creatinine concentration in the range of 2.1–2.8 mg/dl to subclassify cats as stage 2 CKD. One study did not clarify which version of IRIS staging was used. 25 Three studies28,32,33 subclassified cats as stage 1 CKD, of which two studies provided descriptive information about FGF-23 concentrations. Stage 1 CKD was defined as creatinine concentration <1.6 mg/dl (<140 µmol/l) in conjunction with either ultrasonographic renal abnormalities in two studies32,33 or as inadequate urine concentrating ability, radiographic renal changes and/or persistent increase in creatinine concentration within the reference interval (RI) in the past 3 months in one study. 28 No study used SDMA concentrations to stage cats with CKD.
Article characteristics and population information for studies evaluating serum or plasma concentrations of fibroblast growth factor-23 (pg/mL) in healthy cats and in cats with CKD stages 1–4

Age data are presented as median (range) or median (25th, 75th percentiles) in years unless otherwise noted CKD, chronic kidney disease; IQR, interquartile range; Q1, first quartile; Q3, third quartile
Table 3 shows descriptive statistics (median with measure of variability) for FGF-23 concentrations from the seven studies that provided FGF-23 values for healthy cats and/or cats with CKD of different stages. Two studies18,29 did not report exact FGF-23 concentrations and provided only graphical representation. Although FGF-23 concentrations are not directly comparable across methodologies, all studies show a similar trend of increasing median FGF-23 concentration with later stages of CKD. FGF-23 concentrations measured using the Kainos in healthy cats19,22,26 and cats with different stages of CKD 18 are shown in Figure 2. When studies had overlapping sample collection dates, FGF-23 concentrations from only one study are presented to avoid the potential duplication of data. FGF-23 concentrations measured by the MedFrontier 32 and Deyihengda 28 assays in cats with CKD of different IRIS stages are shown in Figure 3.

(a,b) Plasma fibroblast growth factor 23 (FGF-23) concentrations (pg/ml) for healthy controls and cats with chronic kidney disease (CKD) stages 2–4 measured using the Kainos assay. Healthy cats are represented by dark purple boxes 19 , 22 , 26 and subgroups with CKD are represented by green (stage 2), 19 light purple (stage 3) 19 or blue (stage 4) 19 boxes based on stage of disease. Boxes represent the interquartile range (Q1–Q3). The shaded area in panel (a) represents the area that is expanded in panel (b)

Serum fibroblast growth factor 23 (FGF-23) concentrations (pg/ml) measured using (a) the MedFrontier assay and (b) the Deyinhengda assay for healthy cats and cats with chronic kidney disease (CKD) stages 1–4. Healthy cats are represented by dark purple boxes 28 , 32 and subgroups with CKD are represented by dark blue (stage 1), 28 , 32 dark green (stage 2), 28 , 32 light purple (stage 3), 28 light blue (stage 4) 28 or turquoise (stage 3–4) boxes. 32 Boxes represent the minimum to maximum concentration range
Across all studies and assays, the median FGF-23 concentration was lowest in healthy cats and increased with each IRIS CKD stage (Table 3). In addition, there was wide variability in FGF-23 concentrations among within each CKD stage (Table 3; Figures 2 and 3). None of the published studies showed clear differentiation between healthy cats and cats with CKD and between CKD stages 1–4 (Table 3; Figures 2 and 3) based on FGF-23 concentrations. There was overlap in the ranges of FGF-23 concentrations between healthy cats and cats with CKD, particularly cats with stage 1 and 2 CKD, although overlap between healthy cats and cats with stage 4 CKD was reported in two publications (Figure 3).28,32 In Geddes et al, 19 a small number of cats with stage 4 CKD had extremely high FGF-23 concentrations, with a range of 81,963 pg/ml from the first to the third quartile. No similarly dramatic FGF-23 values were reported in cats with stage 4 CKD in other studies.
Lin et al 28 compared FGF-23 concentrations in healthy cats subdivided by age (0–2 years, 3–6 years, 7–10 years, 11–14 years, ⩾15 years) and concluded that age was not a determinant of FGF-23 concentrations. In addition, the study found no difference in FGF-23 concentrations based on sex or neuter status.
FGF-23 and phosphate overload
FGF-23 concentrations were found to positively correlate with serum phosphorus concentrations in cats with CKD in some studies20,25,28,32 but some studies showed no correlation.18,26,33,34 Geddes et al 19 found that phosphorus concentration was an independent predictor of FGF-23 in cats with stage 2–4 CKD. The same study subdivided cats with CKD by stage 2 and 3 and by phosphorus concentration (stage 2: ⩽4.5 mg/dl; stage 3: ⩽5.0 mg/dl) and found that FGF-23 concentrations were significantly higher in cats with phosphorus concentrations above the cutoff (Table 4).
Article characteristics and population information for a study evaluating plasma concentrations of FGF-23 (pg/ml) in cats with normophosphatemic and hyperphosphatemic CKD

Stage 2a: phosphate concentration ⩽4.5 mg/dl; stage 2b: >4.5 mg/dl; stage 3a: ⩽5.0 mg/dl; stage 3b: >5.0 mg/dl
CKD = chronic kidney disease; FGF-23 = fibroblast growth factor 23; Q1, first quartile; Q3, third quartile
A prospective diet trial by Geddes et al 22 evaluated the effects of feeding a moderate protein (76 g/Mcal) and phosphate (1.6 g/Mcal) diet and an adult maintenance diet (protein 86 g/Mcal; phosphate 2.6 g/Mcal) to apparently healthy older cats on several markers of MBD, including FGF-23, over an 18-month period. The study found that neither FGF-23 nor phosphorus concentrations changed over time and that neither analyte was affected by diet. In a retrospective study, Geddes et al 20 evaluated the effect of renal diets (dietary phosphate range 0.77–1.12 g/Mcal) on phosphorus and FGF-23 concentrations over approximately 4–6 weeks in cats with stage 2–4 CKD and compared the results with those from cats with CKD without a diet change that remained on various maintenance diets. Cats with CKD fed a renal diet had a significant reduction in phosphorus and FGF-23 concentrations, unlike the comparator cats. In that study, phosphorus and FGF-23 concentrations were compared before and after a renal diet between cats that were categorized as either hyperphosphatemic (stage 2: >4.5 mg/dl; stage 3: >5.0 mg/dl; stage 4: >6.0 mg/dl) or normophosphatemic (stage 2: ⩽4.5 mg/dl; stage 3: ⩽5.0 mg/dl; stage 4: ⩽6.0 mg/dl) based on CKD stage. FGF-23 concentrations decreased in both the hyperphosphatemic and normophosphatemic groups despite phosphorus concentrations decreasing in only the hyperphosphatemic cats.
FGF-23 and calcium and magnesium
Five studies evaluated the relationship between FGF-23 and calcium concentrations (Table 5). Studies reported that total calcium (Geddes et al 19 ) and ionized calcium (Miyakawa et al 32 ) concentrations were independent predictors of FGF-23 concentrations in cats with CKD. Miyakawa et al 33 found that in cats with CKD and upper urolithiasis, FGF-23 concentrations were higher in those cats with ionized hypercalcemia and a normal phosphorus concentration (<4.6 mg/dl). In that study, FGF-23 concentrations positively correlated with both total and ionized calcium. A study by van den Broek et al, 23 which evaluated total calcium concentration in healthy cats and cats with stage 2–4 CKD, found no difference in FGF-23 concentrations between cats with hypercalcemia and those with normocalcemia. On the contrary, a study evaluating ionized calcium in cats with stage 2–4 CKD by van den Broek et al 31 showed higher FGF-23 concentrations in cats with hypercalcemia compared with cats with normocalcemia.
Article characteristics and population information for studies evaluating serum or plasma concentrations of FGF-23 (pg/ml) and calcium or magnesium in cats with CKD
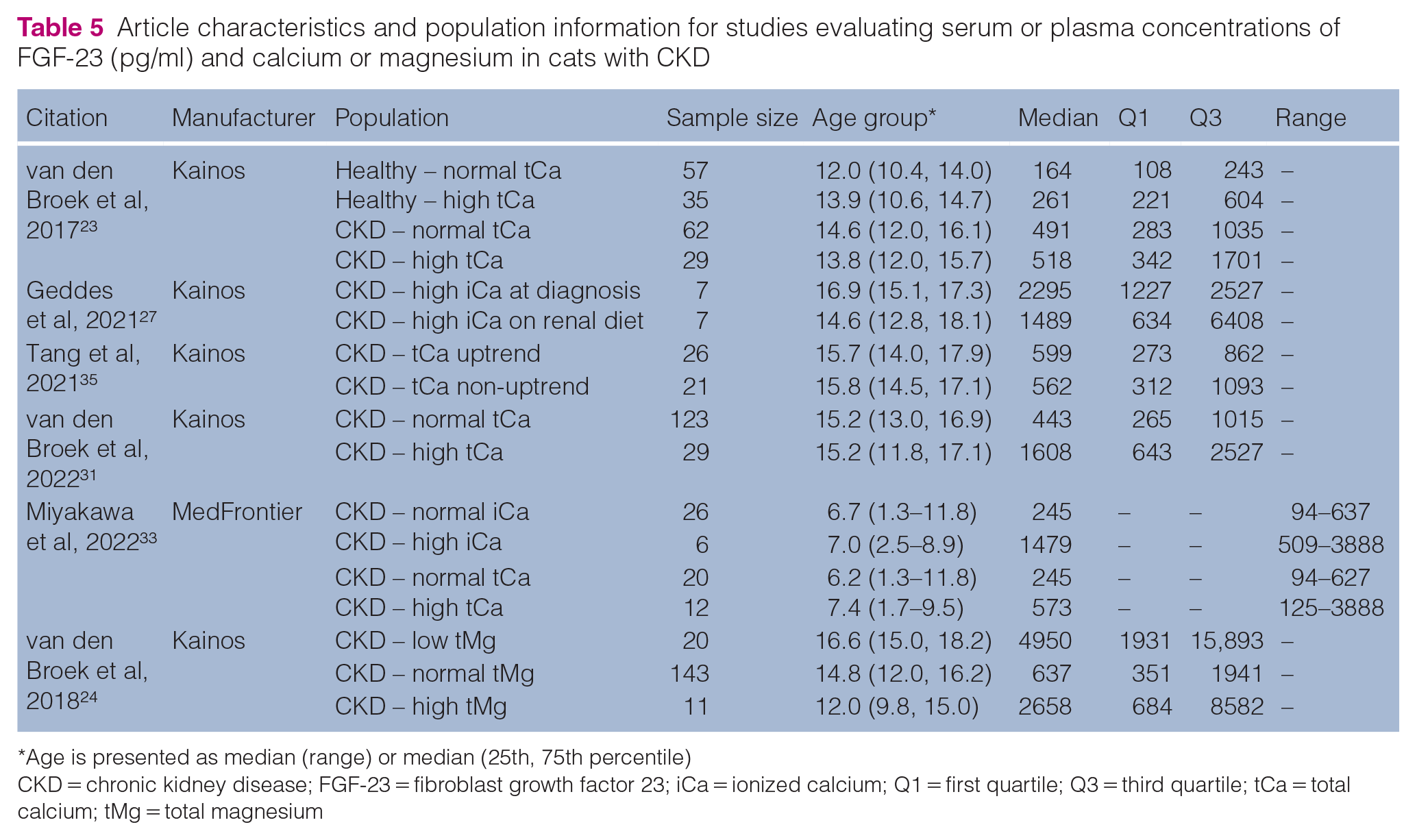
Age is presented as median (range) or median (25th, 75th percentile)
CKD = chronic kidney disease; FGF-23 = fibroblast growth factor 23; iCa = ionized calcium; Q1 = first quartile; Q3 = third quartile; tCa = total calcium; tMg = total magnesium
Tang et al 30 evaluated the risk factors associated with the development of total hypercalcemia in cats with stage 2 and 3 CKD within the first 200 days of transitioning to a phosphate-restricted renal diet. No significant difference in FGF-23 concentrations at baseline (ie, before transitioning to the renal diet) was found between those cats with an uptrend in total calcium concentration and those without (Table 5). Furthermore, unlike phosphorus concentrations, baseline FGF-23 concentrations were not found to be an independent risk factor for increasing total calcium concentration after transitioning to the renal diet. van den Broek et al 31 evaluated FGF-23 concentrations in cats with CKD based on ionized calcium concentrations at the time of diagnosis and over a 1-year follow-up period. There was no difference in FGF-23 concentrations at the time of CKD diagnosis between those cats that developed persistent ionized hypercalcemia and those cats that continued to have normocalcemia over a 1-year follow-up period. Diet was not controlled in that study; however, there was no difference in the proportion of daily food intake consisting of a renal diet between cats that became hypercalcemic and cats that remained normocalcemic over time. Geddes et al 27 looked at whether transitioning cats that develop ionized hypercalcemia on a renal diet to a moderate-protein, moderate-phosphate diet improved their calcium–phosphate homeostasis. The dietary change reduced ionized calcium concentrations without changing either the FGF-23 or phosphorus concentrations.
One study by van den Broek et al 24 investigated the relationship between total magnesium and FGF-23 concentrations and found that higher FGF-23 concentrations were independently associated with hypomagnesemia and that cats with hypomagnesemia had higher FGF-23 concentrations across all CKD stages (Table 5).
FGF-23 and development of azotemic CKD
Finch et al 18 measured FGF-23 concentrations in healthy non-azotemic (creatinine <2.0 mg/dl) older cats (aged >9 years) over a 12-month period. Baseline screening FGF-23 concentrations were higher in the healthy cats that developed azotemic CKD (creatinine >2.0 mg/dl) within 12 months than in those that did not. A weak correlation between FGF-23 and creatinine was found at baseline and at the 12-month follow-up. Sargent et al 26 found that FGF-23 concentrations were higher in apparently healthy cats with an SDMA concentration above the RI (>14 mg/dl) and creatinine <2.0 mg/dl compared with cats with a normal SDMA concentration (Table 3) despite no difference in phosphorus and PTH concentrations.
FGF-23 and CKD progression
Two studies21,25 looked at initial FGF-23 concentration at CKD diagnosis and progression of CKD based on increasing creatinine concentration (Table 6). Liao et al 25 classified progression as an increase of >0.5 mg/dl over 3 months or death, and Geddes et al 21 classified progression as an increase of >25% in creatinine concentration over 12 months after the date of diagnosis of azotemic CKD. Both studies found that FGF-23 concentration was independently associated with disease progression. Specifically, Liao et al 25 found that FGF-23 concentrations were significantly higher in cats with stage 2 and 3 progressive CKD than in cats with non-progressive disease at the same stage. Cats with stage 1 CKD were not included in that study, and there were too few cats with stage 4 CKD enrolled to include in the evaluation of progression. Geddes et al 21 included cats with stage 2–4 CKD and similarly found that FGF-23 concentrations were higher in cats with progressive disease than in those with stable disease.
Article characteristics and population information for studies evaluating serum or plasma concentrations of FGF-23 (pg/ml) in cats with progressive or non-progressive CKD

Age is presented as median (range) or median (25th, 75th percentile)
CKD = chronic kidney disease; FGF-23 = fibroblast growth factor 23
Discussion
This scoping review describes the current literature on FGF-23 in the context of kidney disease in cats and was intended to provide evidence for veterinarians to make informed decisions about the use of measuring FGF-23 concentrations in clinical practice during the management and treatment of cats with CKD. In addition, we aimed to identify knowledge gaps and opportunities for future research. The review identified 17 studies published between 2013 and 2022. Most studies were retrospective and included cats with CKD. Most studies subcategorized cats with CKD by stage, with some variation between studies in the creatinine concentrations used to determine stage. FGF-23 concentrations tended to be higher in cats with CKD compared with healthy cats and trended higher with later IRIS CKD stages, albeit with significant variation within CKD stages and overlap between stages. These are expected findings given that FGF-23 is excreted by the kidneys and similar findings have been reported in people.39–41
This review found several key concepts within the literature regarding measuring FGF-23 concentrations in cats with CKD. First, several studies evaluated the relationship between phosphate overload and FGF-23 concentrations, with some studies, but not all, showing a positive correlation between phosphorus and FGF-23 concentrations. The onset of CKD-MBD and of hyperphosphatemia and hyperparathyroidism varies between cats, especially those with early-stage CKD, and is most common in cats with late-stage disease.1,3 Therefore, IRIS treatment guidelines for cats with IRIS stage 1 and 2 CKD recommend monitoring blood phosphorus concentrations to identify phosphate overload and reducing phosphate intake if needed to maintain normal phosphorus concentrations in the range of 2.7–4.6 mg/dl. This recommendation is supported by a study indicating improved renal biomarkers and short-term outcomes with phosphate reduction in cats with early CKD before the onset of hyperphosphatemia. 22 In this review, only one study 19 explored FGF-23 as an early marker of CKD-MBD before the development of overt hyperphosphatemia in cats with stage 2 and 3 CKD. Based on the findings of that study, measuring FGF-23 concentration may help identify cats with early-stage CKD that have total body phosphate overload but have not yet developed a phosphorus concentration outside the target range. Unfortunately, most studies in this scoping review included cats with stage 2 and 3 CKD, with a paucity of research in cats with non-azotemic CKD (stage 1). Further study of cats with stage 1 CKD and early (non-azotemic) stage 2 CKD would be valuable to define the expected FGF-23 concentrations in these cats and to determine the clinical decision limits to identify early CKD-MBD.
Two commercial laboratories now offer an FGF-23 ELISA assay using serum to measure FGF-23 concentrations in practice. IDEXX Laboratories offers a feline-only FGF-23 ELISA at reference laboratories globally using the Kainos assay. Fujifilm offers a chemiluminescent FGF-23 assay at reference laboratories in Japan (CL-Jack; Minaris Medical) for dogs and cats. The CL-Jack assay uses the same antibodies as the MedFrontier assay but has been shown to yield relatively lower concentrations compared with the MedFrontier ELISA at higher FGF-23 concentrations. 42 The IDEXX test suggests measuring an FGF-23 concentration in cats with a diagnosis of stage 1 or 2 CKD to help veterinarians decide which cats may benefit from phosphate reduction therapy. The RI for the assay is <300 pg/ml. If a cat with stage 1 or 2 CKD has an elevated FGF-23 concentration (⩾400 pg/ml), then the commercial laboratory suggests initiating targeted therapy to reduce phosphate overload, such as a reduction in dietary phosphate or adding a phosphate binder at mealtimes. While this is a guide for veterinarians in practice, research is needed to determine if these therapeutic targets are appropriate in client-owned cats with early-stage CKD.
A second concept explored in the literature was the effect of dietary phosphorus restriction on FGF-23 concentrations. Dietary phosphate restriction followed by the addition of a phosphate binder as needed is the recommended practice in veterinary medicine to slow the progression of renal secondary hyperparathyroidism and CKD-MBD. 43 A variety of prescription therapeutic renal diets marketed for cats are commercially available, and they have different formulations with variation in phosphate and protein concentrations. This review included two studies that evaluated the effect of a phosphate-restricted diet on FGF-23 concentrations in healthy older cats 22 and cats with stage 2–4 CKD. 20 The study of cats with CKD, 20 but not that of healthy older cats, 22 showed that feeding a moderately phosphorus-restricted diet can decrease FGF-23 concentrations. Some phosphate binders have been found to reduce FGF-23 concentration in human patients; 44 however, to date, no study has evaluated the effect of phosphate binders on FGF-23 concentrations in veterinary medicine. Whether measuring FGF-23 concentration is useful to guide the treatment of phosphate overload in cats with CKD is worthy of further investigation.
A third concept explored in the literature is the relationship between FGF-23 and calcium concentrations. Both ionized and total calcium concentrations were found to be independent predictors of FGF-23 concentrations in cats with CKD.18,20 Since hypercalcemia can directly increase blood FGF-23 concentrations in cats independent of glomerular filtration rate and phosphate status, 33 calcium concentrations should be evaluated in cats with a normal phosphorus concentration but elevated FGF-23 concentration before dietary phosphate restriction. In addition, recent evidence supports that feeding a phosphate-restricted diet (<1.3 mg/Mcal) may put cats with CKD at risk of developing hypercalcemia, particularly those with a phosphorus concentration within the IRIS target.27,30 Two studies30,31 included in the scoping review measured FGF-23 concentrations in cats with CKD that developed hypercalcemia and both found that FGF-23 concentrations did not differ between cats that developed hypercalcemia and those that continued to have normocalcemia on a renal diet. Thus, FGF-23 concentration does not appear to be able to predict which cats will develop hypercalcemia after the transition to a phosphate-restricted diet.
Lastly, some studies evaluated the prognostic value of FGF-23 concentrations in healthy cats and cats with CKD. In healthy geriatric cats, FGF-23 concentrations predicted the development of azotemia. 18 In addition, the findings of a few studies support the potential use of FGF-23 as a biomarker of disease progression and survival in cats with CKD, similar to in people.46–48 In the two studies21,25 that defined progression as increasing creatinine concentration, cats with CKD that progressed had higher baseline FGF-23 concentrations, and FGF-23 was an independent predictor of disease progression to a higher IRIS stage. Whether the association between FGF-23 and disease progression in cats is causal or correlative remains unknown.
This scoping review revealed several knowledge gaps in the literature worthy of further research. First, there is still missing information about the expected range of FGF-23 concentrations in healthy cats. Based on current IRIS definitions, it is possible that some studies included cats with early-stage, non-azotemic CKD in their healthy cat population. Several studies were published before and/or included cats with data collected before the commercial availability of a feline SDMA test in 2015. Of the four studies that enrolled cats starting in 2015 or later, only one study used SDMA in the criteria to diagnose CKD. The serial measurement of SDMA concentrations could assist in identifying some cats with stage 1 CKD and better identify and stage cats with CKD with muscle atrophy. Second, little is known about the biological variability of FGF-23 concentrations in healthy cats. A recent study of cats using the MedFrontier assay showed that FGF-23 was relatively stable within a 24 h period with no significant circadian rhythm or postprandial effect, supporting that the timing of sample collection is of little importance. 49 Documentation of the degree of day-to-day variability and the biological variation of FGF-23 concentration in cats is needed to better understand the potential use of FGF-23 measurement in clinical practice. Finally, our results are up to date only as of 31 December 2022, and additional studies investigating FGF-23 in the context of renal disease may have since become available.
Studies should be undertaken prospectively, with cats diagnosed and staged with CKD using current IRIS guidelines and the collection of longitudinal data. SDMA concentrations should be measured, especially in healthy cats and in those with muscle atrophy, to help differentiate between healthy cats and those cats with early-stage, non-azotemic CKD, as well as to appropriately stage cats with late-stage disease. The methodology of FGF-23 measurement should be standardized among studies to allow comparison of results. Consideration should be given to blood phosphorus and calcium concentrations and to study population size to ensure that a minimum number of patients are enrolled to reach statistical significance.
Conclusions
This scoping review identified and described the available literature relating to FGF-23 in cats with CKD. Most studies were retrospective and varied in their methodologies for measuring FGF-23 concentration and for categorizing cats by disease state (healthy vs CKD) and CKD stage. Although research into FGF-23 in cats with CKD is sparse, FGF-23 shows potential as a useful measurement in feline medicine to help improve the identification and understanding of CKD-MBD, and to improve monitoring and guide therapeutic choices in cats with CKD. There are some clear areas for further clinical study in the role and clinical significance of FGF-23 in early-stage CKD. There is a need to understand the timing and clinical significance of increased FGF-23 concentrations and CKD-MBD in early-stage CKD, and whether early dietary intervention can improve clinical outcomes for those cats with increased FGF-23. There is also a need for further investigation into the role of FGF-23 in identifying or predicting the progression of CKD.
Supplemental Material
Supplementary Table 1:
Complete search results and reasons for exclusion for all articles identified in the literature search.
Supplemental Material
Supplementary Table 2:
The search engines that identified the 17 articles included in the scope review.
Supplemental Material
Supplementary Figure 1:
The median fibroblast growth factor 23 concentration (pg/ml) by IRIS stage (1−4) and in healthy cats measured using one of four assays (Deyinhengda, Kainos,MedFrontier, MyBioSource).
Footnotes
Supplementary material
The following files are available as supplementary material: Supplementary Table 1: Complete search results and reasons for exclusion for all articles identified in the literature search.Supplementary Table 2: The search engines that identified the 17 articles included in the scope review.Supplementary Figure 1: The median fibroblast growth factor 23 concentration (pg/ml) by IRIS stage (1–4) and in healthy cats measured using one of four assays (Deyinhengda, Kainos, MedFrontier, MyBioSource).
Conflict of interest
SS is a consultant of IDEXX Laboratories and her previous work has been supported by IDEXX Laboratories. RM, HM and DS are full-time employees of IDEXX Laboratories.
Funding
IDEXX Laboratories funded the project.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
