Abstract
Objectives
The objectives of this study were to investigate the c.1024G>T SNP in the TRPV4 gene in Scottish Straight and Fold cats, and to evaluate the pattern of skeletal phenotype and the evolution of radiological signs of Scottish Fold osteochondrodysplasia (SFOCD) over time in heterozygous subjects.
Methods
DNA was obtained from blood samples of 17 cats (Scottish Fold: n = 12; Scottish Straight: n = 5) and subsequently genotyped by sequencing in a 249 bp region of the TRPV4 gene (exon 6), including the known c.1024G>T causative mutation for osteochondrodysplasia. Orthopaedic and radiographic analyses were performed on animals carrying the mutant allele.
Results
Genotyping by sequencing confirmed that all and only the Scottish Fold cats carried the mutant allele in a heterozygous asset. Furthermore, two other exon variants, already described in the literature as silent variants, were found in some of the sampled cats. Comparative orthogonal radiographic views of the shoulder, elbow, carpus, hip, stifle and tarsus were obtained. A mediolateral projection of the thoracic and lumbar column was also performed. Three out of four cats were clinically and radiographically examined again 1.5 years later.
Conclusions and relevance
Although the presence of the mutant allele in all the tested Scottish Fold cats was confirmed, only 1/12 showed clinical signs of SFOCD. Furthermore, no cats in the 1.5-year follow-up showed skeletal changes. Although significant, the c.1024G>T mutation in the TRPV4 gene, supposedly, is not the only cause or risk of developing SFOCD.
Introduction
Scottish Fold is a purebred cat breed characterised by typical ears folded forward on the head. This trait develops from 3–4 weeks of age and usually is set by the age of 3–4 months. 1 The breed was developed in 1961 by mating a Fold queen (carrying a de novo mutation) with a British Shorthair stud. 2 The first deformities were mentioned in 1971, when progressive bone abnormalities and crippling lameness were described. 3 The breed was no longer recognised in England from 1974, while in the USA, imported cats from the UK were outcrossed with normal eared breeds, and in later generations, fold-to-fold mating was carefully avoided. Thus, only 50% of kittens will have folded ears, while kittens with normal ears are known as Scottish Straight. 4
Scottish Fold osteochondrodysplasia (SFOCD) is an inheritable disorder (dominant trait displaying incomplete dominance) 5 characterised by skeletal deformities. 2 In homozygous individuals, joint lesions progress until the cats are unable to walk. On radiography, distorted metaphyses of metatarsal and metacarpal bones are evident. This results in the decreased length and abnormal shape of bones and distal limbs. Shorter caudal vertebrae, widened endplates and gross plantar exostoses of tarsal and metatarsal bones are clinically and radiographically evident. 6
In 2016, a genome-wide association study revealed a significant association between fold phenotype, osteochondrodysplasia and a missense variant in TRPV4. 7 Located on chromosome D3, TRPV4 encodes for the Transient Receptor Potential Vanilloid family member 4 protein, is expressed in a wide range of tissues and is considered the strongest candidate marker due to its mechanosensory action on chondrocytes. 8 In total, 11 variants were detected in TRPV4: five were responsible for a change in the amino acidic sequence, but only one was associated with the folded ear phenotype (exon 6, c.1024G>T). In 2008, Masuyama et al demonstrated that the lack of TRPV4 expression in mice increases bone mass by impairing bone resorption. 9 Furthermore, a genome-wide screening carried out on two murine cell lines showed that TRPV4 is involved in the regulation pathway of SRY-Box Transcription Factor 9 (SOX9), a transcription factor for chondrocyte differentiation. 10 Lastly, the skeletal phenotypes reported in the literature in heterozygous cats for the c.1024G>T ranged from mild to severe, with a high variability of the severity of skeletal abnormalities among affected cats, 11 although the age of onset of clinical signs, as well as the severity and the progression of the secondary bone formation, are highly variable. So far, no information is available about the evolution of the SFOCD over time. The aim of the present study was to investigate the c.1024G>T variation in TRPV4 in Scottish Straight and Fold cats, to evaluate the pattern of skeletal phenotype in heterozygous subjects and to determine the progression of SFOCD radiological signs over time.
Materials and methods
Animals
A total of 17 cats from two catteries were selected: five Scottish Straight and 12 Scottish Fold. The mean age of the cats at blood sampling was 2.3 ± 1.3 years (age range 0.9–6.5). The cats were identified during routine visits at the university veterinary hospital where samples were collected in K3-EDTA blood vacutainers. Fold-eared cats were then submitted for orthopaedic examination by specialised veterinarians; the degree of lameness, pain and joint thickening were recorded and the mobility of each joint was evaluated by a ‘range of motion’ test. A CT examination was performed on 5/12 Scottish Fold cats (ie, Cat_03, Cat_06, Cat_15, Cat_16 and Cat_17) to assess whether signs of osteochondrodysplasia were present. The mean age of the cats at the first radiographical examination was 3.7 ± 1.9 years (age range 1.1–6.5) (Table 1). Thanks to the cooperative nature of the subjects, neither general anaesthesia nor mild sedation was required. Manual restraint was applied for the positioning of the patients.
List of cats with phenotypes, radiographic examinations and age at the time of radiographical examination

Comparative orthogonal radiographic views of the shoulder, elbow, carpus, hip, stifle and tarsus were obtained, along with a lateral view of the thoracic and lumbar column. A second clinical and radiographic examination on 3/5 cats (ie, Cat_03, Cat_06 and Cat_15) was performed 1.5 years later with an identical approach, and the images were evaluated by the same radiologist. The mean age of the cats at follow-up was 4.2 ± 1.4 years (age range 2.6–5.0).
DNA extraction and genotyping
DNA was extracted using a commercial DNA extraction kit (NucleoSpin Blood; Macherey Nagel) according to the manufacturer’s instructions. Genotyping for the c.1024G>T SFOCD-associated variant was performed by sequencing a 249 bp fragment on TRPV4 exon 6 using two primers (5’-TGACAGAGAACCCGCACAA-3’ and 5’-CACTCACCCCAATCTTGCC-3’), designed with Primer3 software (http://biotools.umassmed.edu/bioapps/primer3_www.cgi) to also include two other mutations, identified by Gandolfi et al. 7 The PCR was performed in a thermal cycler (2720 Thermal Cycler; Applied Biosystems) in a total volume of 25 μl (HotStarTaq DNA Polymerase; Qiagen). Amplification occurred at 95° for 15 mins, 35 cycles at 94° for 30 s, 54° for 30 s and 72° for 1 min, and a final extension step at 72° for 10 mins.
Results
Genetic analysis
No Scottish Fold cats were found homozygous for the mutant allele and no mutant alleles were found in Scottish Straight cats. The DNA sequence of the 249 bp fragment of TRPV4 exon 6 investigated in all cats revealed three mutations, namely c.963A>C, c.1024G>T and c.1104C>T. As detailed in Table 2, the point mutation c.963A>C was detected in 13/17 cats. The point mutation c.1024G>T was detected only as heterozygous and only in all the Scottish Fold cats. Lastly, the point mutation c.1104C>T was detected in 16/17 cats.
Cat genotypes for the three mutations in TRPV4 exon 6: c.963A>C, c.1024G>T and c.1104C>T

Both c.963A>C and c.1104C>T point mutations were labelled as silent by Gandolfi et al, 7 and, consistently, were not related to either phenotype in our sample. In contrast, the c.1024G>T mutation was found in all the animals with folded ears.
Clinical and radiographic evaluation
One Scottish Fold cat (Cat_17) showed clinical signs related to SFOCD. For the other cats in the study, no gait alterations, lameness or pain was reported. Only Cat_17 showed radiographical alterations attributable to SFOCD at first radiographic evaluation. Specifically, there was periarticular smooth bone remodelling with a narrowing of the joint spaces (both tarsometatarsal joints) with elongated osteophytes formation. Periarticular ill-defined soft tissue swelling was noted, likely indicative of synovial hyperplasia or non-aggressive synovitis. Mild periarticular smooth bone remodelling was noted at the caudal aspect of the right elbow joint with two-pinpoint mineralisation located caudal to the supratrochlear foramen and visible on the mediolateral view. This mineralisation was likely indicative of feline synovial osteochondromas or fractured osteophytes. The radiographic findings were indicative of multifocal bilateral degenerative joint disease and early ankylosis of the tarsometatarsal joints, associated with right elbow osteoarthrosis with possible synovial osteochondromatosis. Radiographic images of the alterations observed at this first radiographic evaluation are shown in Figure 1.
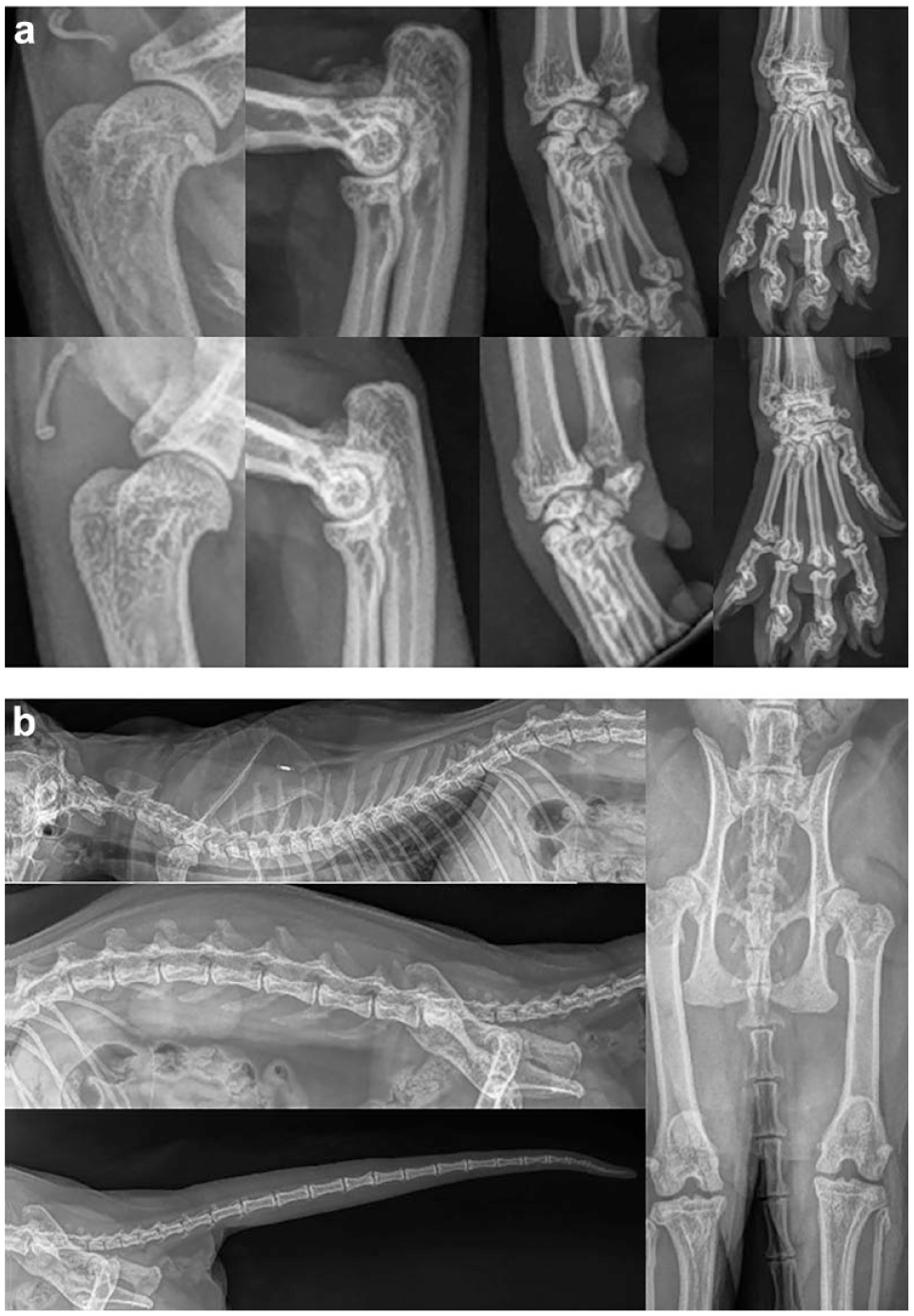
Radiographic images of the alterations observed in Cat_17: (a) limbs and paws (right and left side in upper and lower images, respectively); (b) lateral and ventral view of the column, hips and tail
No skeletal changes were detected at the follow-up radiographic evaluation, performed 1.5 years later on 3/5 cats.
Discussion
Our findings confirm the association between c.1024G>T missense mutation and the folded ears phenotype, whereas the other two mutations (namely c.963A>C and c.1104C>T) detected in exon 6 are silent and not associated with fold phenotype. 7
Previous studies suggested that SFOCD in heterozygous animals has a milder and slower development in comparison with homozygous animals. 3 Other studies reported cases of lesions radiographically evident starting from 17 months of age. 12 This variance in the manifestations and severity of clinical signs is justified by the incomplete dominant pattern of inheritance of the disease 5 and could explain why, in the first radiographical examination, only one (ie, Cat_17, aged 6.5 years at radiographic examination) of the Fold sampled population showed clinical signs of the disease. The radiographical follow-up 1.5 years later showed no progression of the bone changes. This result is consistent with the scenario of a milder and slower development of SFOCD in heterozygous cats.
Additional unknown factors are supposed to exert an effect on skeletal phenotypes and could explain the observed variability. Among them, age is a risk factor. With SFOCD being a progressive disorder, changes could be very subtle in cats aged younger than 1 year. On the other hand, regardless of breed, 61% of cats aged older than 6 years demonstrate osteoarthritis in at least one joint. 13 Further studies will be required to expand the knowledge on these other factors affecting the development of SFOCD.
Conclusions
SFOCD is a disorder influenced by various factors, including genetic ones. Furthermore, this variance is also found in the manifestation and severity of clinical signs, which is justified by the incomplete dominant pattern of inheritance of the disease. Although the effects of the c.1024G>T substitution have been investigated, the progression of this disorder in heterozygous cats is still partly unknown. In this study, heterozygous cats showed minor to no clinical signs of SFOCD, and after a 1.5-year follow-up, no progression of bone changes was observed. Further studies will be needed to evaluate the development of SFOCD, in order to assess risks and other causative effects.
Footnotes
Acknowledgements
Thanks to the two cat breeders, Vincenzo Pagliarello (‘Dei reali’ cattery) and Taisia Sevets (‘Agorà Artemide’ cattery), for providing the animals for this study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the University of Turin (MORR_RILO_23_01: Ricerca Locale 2023 – Linea B – Moretti Riccardo).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
