Abstract
Objectives
Phenobarbital (PB) q12h is the most common treatment recommendation for cats with recurrent epileptic seizures. Medicating cats may be challenging and result in decreased quality of life for both cat and owner. The aim of this retrospective study was to evaluate treatment with oral PB q24h in cats with presumptive idiopathic epilepsy.
Methods
Nine cats with presumptive idiopathic epilepsy, receiving oral PB q24h, were included in a retrospective descriptive study.
Results
Seizure remission was achieved in 88% (8/9) of the cats and good seizure control in 12% (1/9) of the cats, treated with a mean dose of oral PB of 2.6 mg/kg q24h (range 1.4–3.8 mg/kg). No cats required an increase of their PB frequency at any time during a mean follow-up period of 3.5 years (range 1.1–8.0 years). No cats displayed side effects or issues with compliance at the last recorded follow-up.
Conclusions and relevance
Once-a-day administration of PB for feline epilepsy was safe and resulted in satisfactory seizure control for the nine cats included in this study. The results of this study justify exploring this topic further in larger prospective studies.
Introduction
Epilepsy is a common neurological condition in cats.1,2 Although the terminology for epilepsy classification has been agreed on in dogs, 3 no consensus has been reached for cats.2,4 –8 Therefore, this paper will use ‘presumptive idiopathic epilepsy’ (PIE) when referring to idiopathic epilepsy in cats. The prevalence of PIE in cats with recurrent epileptic seizures has been reported to be in the range of 22%–75%,1,2,7,9 –12 with the majority having seizure onset at 1–5 years.5,13,14
The use of several antiepileptic drugs for cats with PIE has been reported,15 –17 with phenobarbital (PB) considered the most effective. 18 An important disadvantage of PB is that it can only be given orally by owners, with the recommendation of medicating cats twice daily.6,12,16,19 –21 However, up to 80% of owners have difficulties orally medicating their cat.22 –26 This may negatively affect owner compliance, the human–cat relationship and result in human injury or owners declining treatment.22,24 –31 However, with an elimination half-life of 43–76 h in cats, it has been suggested that treatment q24h may be possible.32 –34 Thus, the aim of the present study was to retrospectively evaluate the treatment response to oral PB once daily in cats with PIE.
Materials and methods
The medical records of cats containing the search terms ‘seizures’ or ‘idiopathic epilepsy’ between January 2017 and June 2021 were retrospectively collected at Anicura Kalmar Small Animal Clinic, Sweden, to identify cats treated with oral PB once daily for recurrent epileptic seizures. The inclusion criteria included two or more unprovoked focal, complex focal or generalised epileptic seizures, occurring at least 24 h apart, with normal neurological examinations, starting once-a-day therapy of PB and a follow-up period of at least 6 months. For all cats, a diagnostic minimum of complete blood work, according to the tier I diagnostic recommendations from the International Veterinary Epilepsy Task Force for dogs, 35 within normal limits, was necessary. To include cats with onset of epileptic seizures under the age of 1 year or over the age of 6 years, an MRI examination of the brain and analysis of cerebrospinal fluid (CSF), or a minimum follow-up period of 1 year after the onset of seizures, without any interictal neurological signs developing during this time, was required.2,5,20 Cats diagnosed with structural or reactive epilepsy were excluded. Cats not treated with PB, receiving PB q12h or treated with other antiepileptic drugs were excluded.
The following information was collected from the clinical records: signalment; classification of seizures; frequency of seizures; initial dose of PB; treatment adjustments; serum concentration of PB; treatment response; compliance; and follow-up. Epileptic seizures were classified as focal, complex focal or generalised. 36
Seizure control and remission were defined as a reduction in seizure frequency by at least 50%4,19 and as a seizure-free period of at least 1 year, 25 respectively. Most follow-up visits consisted of a complete blood cell count, while measurement of serum concentration of PB depended on dosage and seizure control. The therapeutic serum concentration of PB was considered to be 23–30 ug/ml. 37
Results
The study population (n = 9) consisted of five castrated male and four spayed female cats with a mean age and body weight at the onset of epileptic seizures of 4.9 years (range 0.9–11.0 years) and 5.0 kg (range 4.0–7.3 kg), respectively. Domestic shorthairs were the most common breed (n = 4) followed by Norwegian Forest Cat (n = 2), Ragdoll (n = 1), Maine Coon (n = 1) and Siberian Cat (n = 1).
Epileptic seizures were reported to be focal (n = 1), complex focal (n = 1), generalised (n = 6) and a combination of complex focal with generalised seizures (n = 1). Although seizure frequency varied among individuals, 66% (6/9) of cats had one or several epileptic seizures per month, but not each day or week. One cat underwent an MRI of the head, without any abnormalities. None of the cats had CSF analysed. The mean starting dose of PB (Fenemal; Meda: n = 6, Epirepress; Virbac: n = 3) was 1.8 mg/kg q24h (range 1.4–2.3 mg/kg).
The first follow-up visit (Table 1) was performed within a mean of 35 days (range 19–54 days) after presentation. Side effects were recorded in one cat and consisted of sedation. One cat had one recorded epileptic seizure, while the remaining cats were seizure-free. The mean serum concentration of PB in seven cats was 11 µg/ml (range 6–17 µg/ml). Therapy adjustments consisted of lowering the dose of PB q24h by 50% in one cat, because of sedation, and doubling the dose of PB q24h in one cat, because of seizure activity and a low serum value of PB (6 µg/ml).
Follow-up visits, recorded seizures, treatment and treatment adjustments for cats with recurrent epileptic seizures, treated with oral phenobarbital (PB) once daily
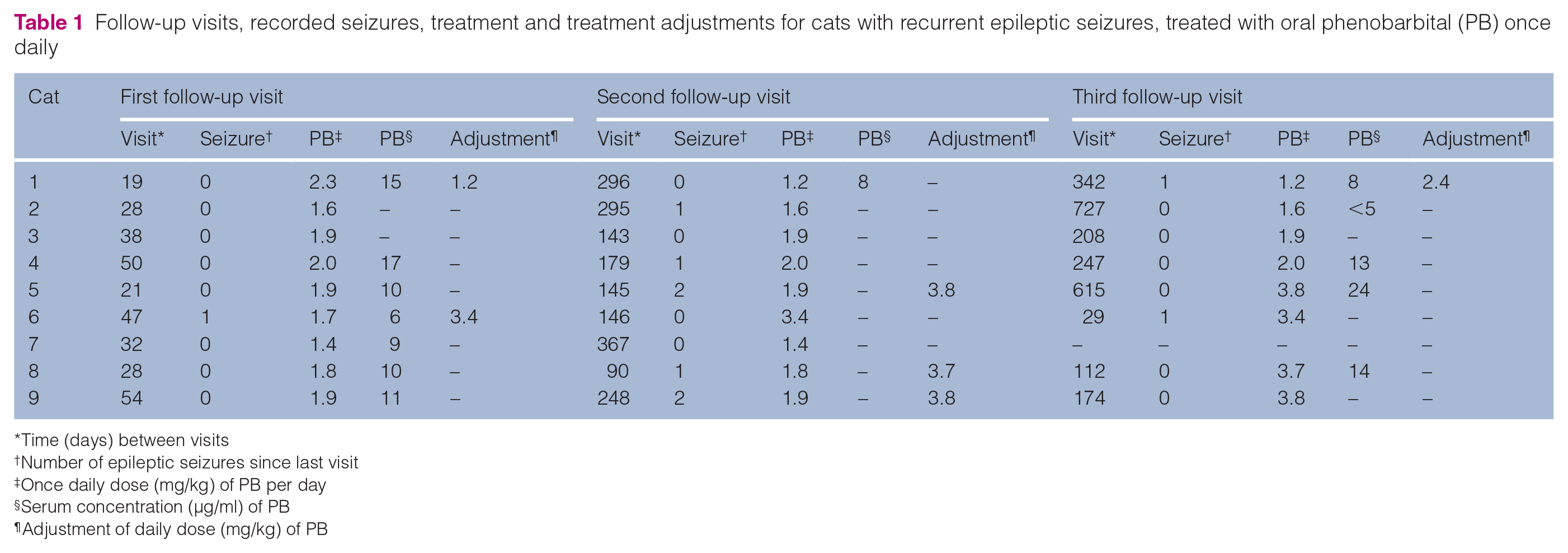
Time (days) between visits
Number of epileptic seizures since last visit
Once daily dose (mg/kg) of PB per day
Serum concentration (µg/ml) of PB
Adjustment of daily dose (mg/kg) of PB
The second follow-up (Table 1) was performed within a mean of 212 days (range 90–367 days) after the first follow-up visit. No side effects were recorded in any cat. The mean dose of PB was 1.7 mg/kg q24h (range 1.2–2.0 mg/kg). Three cats had one epileptic seizure while two cats had two epileptic seizures since the last visit. The remaining cats were seizure-free. One cat had serum concentration of PB measured, with a value of 8 µg/ml. Because of persistent epileptic seizure activity, three cats doubled their dose of PB q24h.
The third follow-up for 8/9 cats (Table 1) was performed within a mean of 307 days (range 29–727 days) after the second follow-up visit. No side effects were recorded. The mean dose of PB was 2.7 mg/kg q24h (range 1.2–3.8 mg/kg). One cat had one recorded epileptic seizure since the last visit and doubled the dose of PB q24h. The remaining cats were seizure-free. The mean serum concentration of PB in five cats was 12 µg/ml (range <5–24 ug/ml).
Further follow-up visits were performed during the remainder of the follow-up period. The included cats had a mean follow-up period of 3.5 years (range 1.1–8.0 years) (Table 2), without any interictal neurological signs developing. The cats had a recorded seizure-free period with a mean of 3.0 years (range 0.5–5.3 years) to the last follow-up, treated during this period with a mean dose of PB of 2.6 mg/kg q24h (range 1.4–3.8 mg/kg). Eight cats were considered to have achieved remission and one had good seizure control. No cats displayed side effects or compliance difficulties at the last recorded follow-up.
Age, follow-up period, treatment and treatment response for cats with recurrent epileptic seizures, treated with oral phenobarbital (PB) once daily

Age (years) at onset of epileptic seizures
Time since onset of epileptic seizures without interictal neurological signs developing
Once daily dose (mg/kg) of PB per day
Duration of seizure-free period (years) until last follow-up
Discussion
This study describes a population of cats with PIE, where treatment with oral PB q24h was well tolerated and resulted in satisfactory seizure control. Our results show that all cats treated q24h had good seizure control at each follow-up visit, with 88% (8/9) of cats demonstrating seizure remission at the last follow-up. Interestingly, though, despite good seizure control, 4,19 the serum concentration of PB was considered subtherapeutic (<23 µg/ml) 37 in 80% (4/5) of samples analysed during the third follow-up visit (Table 1). Unfortunately, the serum value of PB was not routinely analysed at each follow-up visit. In addition, it is not known at which time point relative to medication administration the sampling of the cases was performed. The serum concentration of PB quickly reaches a steady state approximately 10 h after oral administration in cats.32,33 However, which level of serum concentration of PB is therapeutic in cats has not been evaluated, with current recommendations being extrapolated from dogs.4,30,37 It is therefore possible that lower serum concentrations in cats can result in good seizure control, but this needs to be further investigated.
It has been suggested that cats with epilepsy may have a reduced quality of life (QOL). 38 The animal’s QOL has also been reported to affect the caregiver burden for owners, a subject that is well recognised in human healthcare, where providing care for a chronically sick family member may cause psychosocial distress for the caregiver.29,39 –42 Although good seizure control leads to higher QOL for cats with epilepsy, thereby decreasing caregiver burden for owners, the cat owner relationship is another important aspect that affects these parameters in the same manner. 29 Medicating the cat orally has been reported to affect the cat–owner relationship negatively in most cases, illustrating that a combination of multiple factors, including ease of treatment administration, should be considered when evaluating QOL.24,26
In this study, where only one cat had an MRI examination performed and no cats had CSF analysed, it cannot be excluded that one or more cats had structural epilepsy. However, most cats were followed over multiple years, with no additional neurological signs recorded, which makes structural aetiologies unlikely. 4 Furthermore, the necessity to strictly include cats with PIE can be debated, since seizure control with PB treatment for structural epilepsy and PIE has been reported in 91% and 93% of cats, respectively. 4 Therefore, if one or more of our included cats had structural disease, it may not have directly affected the conclusions of our study.
Conclusions
Once-a-day administration of oral PB for feline epilepsy was safe and resulted in satisfactory seizure control for the nine cats included in this study. Although the cats in this study were well controlled with oral PB q24h, it is possible that some cats will need treatment q12h. Prospective studies are necessary to compare the efficacy between both dosing regimens.
Footnotes
Acknowledgements
We thank Tycho Carlssons Foundation and Ulla Yards Foundation for their financial contributions to the publication of this study.
Author note
The results from this study were orally presented as an abstract at the Nordic Veterinary Neurology Meeting in Riga, Lithuania, on 5 May 2023.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This publication was funded by Tycho Carlssons Foundation and Ulla Yards Foundation.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
