Abstract
Objectives
The aim of the present study was to describe the distribution of gastrointestinal histopathology findings associated with gastrointestinal obstructions secondary to trichobezoar formation in cats.
Methods
A total of 100 surgical gastrointestinal biopsies were obtained from 44 cats with gastrointestinal obstructions secondary to a trichobezoar. Medical records, including signalment, type and duration of clinical signs, surgical reports and histopathologic analysis, were reviewed for each cat.
Results
Biopsies taken near the site of the trichobezoar were more likely to show neutrophilic inflammation and mucosal erosion/ulceration compared with biopsies taken elsewhere in the small intestine. Lymphoplasmacytic and mixed lymphocytic and eosinophilic populations were the most common histopathologic findings from all biopsies followed by alimentary small cell lymphoma. Biopsy samples were more likely to represent a diagnosis of alimentary lymphoma in cats older than 10 years.
Conclusions and relevance
Gastrointestinal biopsies taken at the time of surgery in cats with trichobezoar obstructions may represent an important diagnostic tool for further evaluation of potential feline chronic enteropathy. Biopsies taken at the site of the obstruction should be interpreted cautiously as the presence of a trichobezoar may induce an acute inflammatory reaction. The resultant histologic interpretation at this site may not represent the chronic state of the intestinal mucosa, supporting the utility of obtaining multiple biopsies orad and aborad to the obstruction.
Introduction
‘Bezoar’ refers to concretions in the gastrointestinal tract that increase in size by continuous accumulation of non-absorbable food or fibers. 1 In cats, grooming can result in ingestion of copious amounts of hair forming ‘trichobezoars’ or ‘hairballs’. This hair usually passes through the intestinal tract and is then expelled in the feces. If hair accumulates in the stomach, vomiting may occur to eliminate the trichobezoar. Some sources consider this a normal physiologic process, though frequent elimination has been cited as evidence of pathologic overgrooming or underlying gastrointestinal pathology.2,3 This lack of consensus regarding normalcy of trichobezoar formation and elimination further contributes to the uncertainty regarding the cause of trichobezoar obstruction. While many veterinarians tend to accept the formation of an obstructive trichobezoar as an unusual, yet inescapable, nuisance (reminiscent of other types of foreign body obstructions), others feel these obstructions are often secondary to gastrointestinal pathology. 3 In the authors’ experience, conservative management with fluid therapy and supportive care may allow passage of trichobezoar obstructions, but a portion of these cats ultimately require surgical intervention. Owing to the paucity of published literature regarding trichobezoar obstructions, the clinical utility of obtaining gastrointestinal biopsies at the time of surgery for relief of trichobezoar obstructions is unknown.2,3
The underlying pathology may result in abnormal intestinal structure and motility, potentially predisposing to trichobezoar obstruction. Many gastrointestinal diseases in cats, including inflammatory bowel disease, diffuse alimentary lymphoma and food-responsive enteropathy, are generally categorized as feline chronic enteropathies (FCE). This term encompasses any gastrointestinal disease causing clinical signs with a duration of more than 3 weeks that cannot be attributed to infectious disease, obstruction or localized neoplasia causing a mass effect. 4 A definitive diagnosis of specific FCE commonly requires advanced diagnostics, including gastrointestinal histopathology, as well as a clinical response to the therapy. However, a recent study suggests that histopathologic results do not always correlate with clinical disease, which complicates a diagnosis strictly from biopsy results. 5 Histopathology results presenting diagnostic ambiguity may benefit from additional testing, such as when PCR for antigen receptor rearrangement (PARR) or immunohistochemistry (IHC) are used to distinguish severe lymphoplasmacytic inflammation from alimentary small cell lymphoma.4,5
The potential for underlying gastrointestinal disease as a precipitating factor for trichobezoar obstructions has led some to the conclusion that cats presenting for surgical intervention should have gastrointestinal biopsies obtained concurrently. However, the decision to take biopsies is not standardized and leads to significant variation in the number and location of biopsies. Some surgeons obtain gastric, duodenal, jejunal and ileal biopsies, while others take just a single biopsy of intestine associated with the obstruction to limit the number of enterotomies performed. Lastly, some surgeons will only obtain biopsies aborad to the obstruction owing to the perceived difficulty of interpreting orad inflammation, which may occur secondarily to trichobezoar passage or a primary enteropathy. To the authors’ knowledge, a distribution of histopathologic findings in gastrointestinal biopsies performed at the time of surgery to relieve mechanical obstructions due to trichobezoars has not been reported.
The objective of the present study was to report histopathological findings in surgically obtained gastrointestinal biopsies from cats with an obstructive trichobezoar. The second aim of the study was to report the location of the changes relative to the obstruction and clinical characteristics of the study cats.
Materials and methods
The medical records of cats undergoing enterotomy or gastrotomy for gastrointestinal foreign body obstructions between January 2016 and July 2022 at six private specialty hospitals were evaluated. The following inclusion criteria were applied: medical records describing a trichobezoar (‘hair’, ‘hairball,’ ‘trichobezoar’); at least one biopsy of the gastrointestinal tract obtained at time of surgery; and histopathologic evaluation by a diplomate of the American College of Veterinary Pathologists. Cats previously diagnosed with any enteropathy (based on review of available medical records) were excluded.
All surgeries were performed by diplomates of the American College of Veterinary Surgeons (ACVS) or residents within an accredited ACVS residency program. Biopsies obtained at the site of obstruction were obtained via initial enterotomy with a #11 blade and excision of a variable strip of tissue from one side of the enterotomy using Metzenbaum scissors. Biopsies were submitted as formalin-fixed samples to either Antech Diagnostics or the Texas A&M Veterinary Medical Diagnostic Laboratory. Histopathology reports were completed by the attending pathologist at the time of original submission.
Data obtained from the medical record included age, sex, breed, clinical history and histologic findings. Surgical reports were evaluated for location of the obstruction and site of gastrointestinal biopsy.
Medical records were reviewed by the authors for histopathologic diagnosis, location within the gastrointestinal tract and location in relation to the obstruction at the time of surgery. Histopathologic diagnosis was based on the predominant cell type. When more than one predominant cell type was noted, all predominant cell types were used to classify the sample. Gastrointestinal biopsy sites included the stomach, duodenum, jejunum and ileum. The location of the biopsy in relation to the obstruction was classified as orad to the obstruction (pre-Trichobezoar or pre-T), at the site of the obstruction (at-Trichobezoar or at-T) and aborad to the site of obstruction (post-Trichobezoar or post-T).
Biopsies were categorized by the severity of inflammation and presence of mucosal erosion or ulceration as described on the original pathology report, in addition to patient history of acute or chronic gastrointestinal clinical signs before surgery. The severity of inflammation, documented for biopsies that did not have evidence of alimentary small cell lymphoma, was categorized as none, mild, moderate or severe. Chronicity of clinical signs was divided into acute (less than 3 weeks) and chronic (greater than 3 weeks) as defined by the American College of Veterinary Internal Medicine (ACVIM) consensus statement. 6
All statistical analyses were performed using SAS 9.4 (SAS). A significance threshold of 0.05 (P <0.05) was used. Two-sided Wilson confidence intervals were calculated for binomial proportions. An analysis of variance and Tukey-Kramer’s test were used to compare age between biopsy result groups.
Results
An initial evaluation of medical records identified 667 cats that had undergone gastrotomy or enterotomy for foreign body obstruction between January 2016 and July 2022. Of the initial 667 cats, 44 (6.6%) met the inclusion criteria. The population consisted of 18 (41%) spayed female and 26 (59%) castrated male cats. Eight different breeds were represented in the population (Table 1), including domestic longhair (n = 20, 45.5%), Maine Coon (n = 9, 20.5%), domestic shorthair (n = 7, 15.9%) and domestic mediumhair (n = 4, 9.1%). The remaining breeds (Himalayan, Norwegian Forest Cat, Ragdoll, Bobtail) were all represented by one (2.3%) cat.
Reported cat breeds and their distribution based on length of clinical signs and histologic diagnosis of alimentary small cell lymphoma

All cats were presented for acute vomiting or worsening of chronic vomiting. In total, 20 (45.5%) cats had only acute clinical signs, while 24 (54.5%) cats had chronic clinical signs. Additional presenting complaints included hypo-/anorexia (27/44, 61.4%), lethargy (10/44, 22.7%), acute wheezing/gagging (1/44, 2.3%) and laterally recumbent/obtunded (1/44, 2.3%). Chronic clinical signs included vomiting (23/24, 95.8%), weight loss (5/24, 20.8%) and previous trichobezoar obstruction (5/24, 20.8%).
The mean age of the study cats was 14.2 ± 4.3 years (range 1.0–18.8 years) (Table 2). The mean age in cats with acute clinical signs (n = 20) was 6.4 ± 3.7 years; in cats with chronic clinical signs (n = 24), the mean age was 9.3 ± 4.3 years; and in cats with lymphoma (n = 10), the mean age was 12.5 ± 3.4 years.
Age distribution among groups reported in years

In total, 1–5 biopsies were collected from the cats in the study, with one (13/44) and three (13/44) biopsies being obtained most commonly (Table 3). In cats with a single biopsy, samples were most often obtained from the site of the obstruction (10/13). From the 44 study cats, a total of 100 biopsies were obtained and subsequently evaluated by various pathologists. The most common histopathologic diagnoses were lymphoplasmacytic inflammation (25/100), lymphoplasmacytic/eosinophilic inflammation (25/100) and small cell lymphoma (18/100) (Table 4). Of the 100 samples, eight (8%) were considered non-diagnostic as they did not contain mucosa.
Number of biopsies taken at the time of surgery per cat

Histopathologic diagnosis distribution based on biopsy location

Of the biopsy locations, 26% were noted to be pre-T, 41% at-T and 33% post-T (Table 5). The most common histopathologic finding orad and aborad to the obstruction was lymphoplasmacytic inflammation (10/26, 38.5% and 10/33, 30.3%, respectively). Mixed lymphoplasmacytic and eosinophilic inflammation was the most common finding at the site of obstruction (at-T: 11/41, 26.8%). Of the 17 biopsies with evidence of neutrophilic inflammation, 14 (83.3%) were from at-T.
Histopathologic diagnosis based on predominant cell population in relation to trichobezoar obstruction site
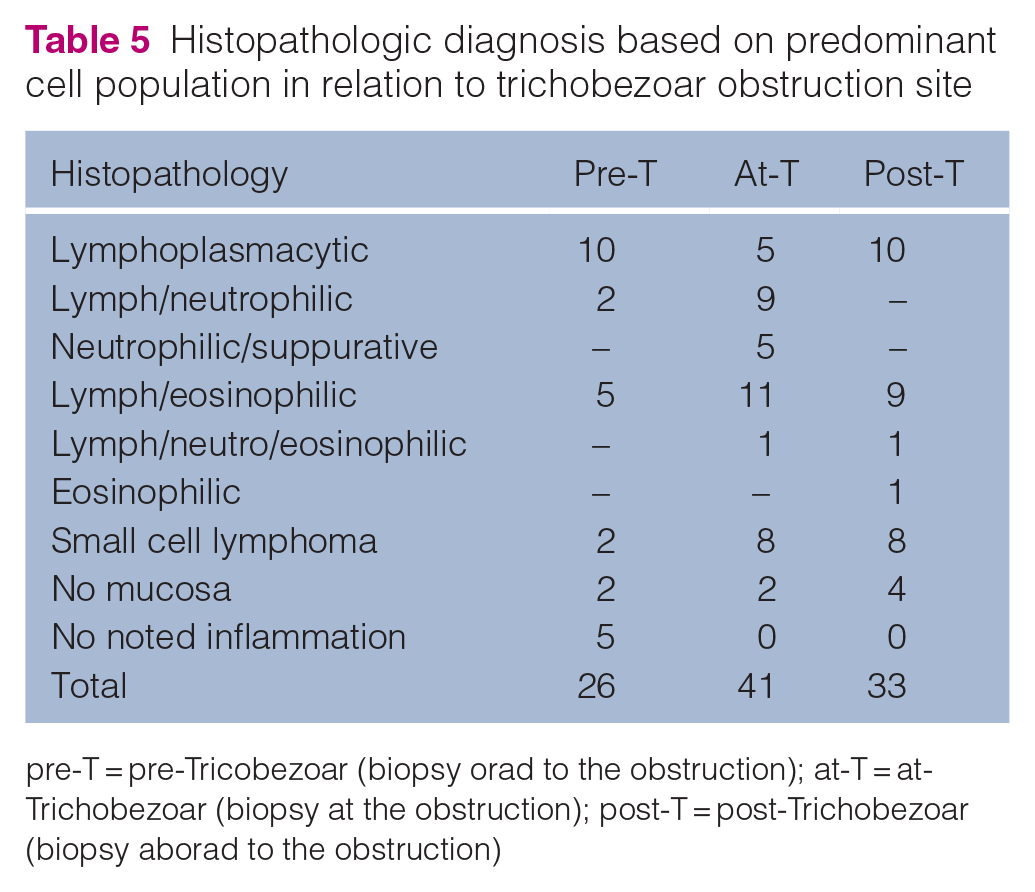
pre-T = pre-Tricobezoar (biopsy orad to the obstruction); at-T = at-Trichobezoar (biopsy at the obstruction); post-T = post-Trichobezoar (biopsy aborad to the obstruction)
The degree of inflammation was evaluated in 73 biopsies (excluding non-diagnostic biopsies or those reported as alimentary lymphoma) (Table 6). Among these, 6.85% had no inflammation, 46.6% had mild inflammation, 20.5% had moderate inflammation and 26% had severe inflammation. Of the biopsies, 22 were taken pre-T, 30 were taken at-T and 21 were taken post-T. Mild inflammation was noted most commonly in pre-T (10/22) and post-T biopsies (13/21). Conversely, severe inflammation was noted most frequently in at-T biopsies (14/30). Mucosal erosion or ulceration was noted in 14/73 biopsies, all of which were at the site of the obstruction.
Distribution of severity of inflammation and mucosal damage within biopsy samples in relation to the trichobezoar obstruction site*

pre-T = pre-Tricobezoar (biopsy orad to the obstruction); at-T = at-Trichobezoar (biopsy at the obstruction); post-T = post-Trichobezoar (biopsy aborad to the obstruction)
Biopsies carrying a diagnosis of lymphoma and those without mucosa were omitted
For cats with multiple small intestinal biopsies (27/44), histopathology reports were evaluated for consistency between the predominant type of inflammation. In total, 11/27 cats had two small intestinal biopsies with 9/11 (81.8%) demonstrating the same type of inflammation in both biopsies. One cat (9.1%) had a sample with no mucosa, so agreement could not be evaluated.
Of the 27 cats, 13 had three intestinal biopsies, with 7/13 (53.8%) demonstrating the same inflammation type within all three biopsies. Of these seven cats, three also had gastric biopsies, all of which showed a predominant inflammation type consistent with that found within the corresponding intestinal biopsies. Three of the 13 cats had one or more samples without mucosa so agreeance could not be evaluated. All of the cats with agreement between all intestinal biopsies (16/44, 36.4%) had a chronic history of gastrointestinal clinical signs.
All 10 cats diagnosed with lymphoma were presented with a history of chronic gastrointestinal signs, re-presenting 41.7% of cats with a chronic history. Diagnosis was based on a single small intestinal biopsy in three cats, two biopsies in three cats and three biopsies in four cats. Five cats had agreement between all small intestinal biopsies, while five cats showed various inflammation in one or more sites. In addition, samples from five mesenteric lymph nodes were taken in these cats and lymphoma was identified in two lymph nodes.
Discussion
This study demonstrates the variability in histopathologic findings in cats with obstructions secondary to trichobezoars. Cats included in this study exhibited both acute and acute-on-chronic gastrointestinal signs.
Histopathologic findings from at-T samples were noted to have an increased severity of inflammation compared with biopsy samples obtained elsewhere in the gastrointestinal tract. When excluding biopsies with histopathological diagnosis suggestive of alimentary small cell lymphoma, 42.4% of the at-T biopsies demonstrated a severity qualifier of ‘marked’ or ‘severe’ inflammation. This contrasts significantly to the pre-T and post-T groups, which had severe inflammation noted in 8.3% and 12% of samples, respectively (P = 0.002). Similarly, mucosal erosion or ulceration was more common at-T. This finding, along with the increased severity of inflammation, is suspected to be secondary to shearing forces and direct trauma from attempted peristaltic advancement of the trichobezoar. The presence of severe inflammation and/or mucosal damage could obscure underlying intestinal pathology and thus confound evaluation for underlying chronic enteropathies.
A neutrophilic component of inflammation was present in 18/100 (18%) biopsy samples, and when present, was significantly more likely to be obtained from at-T (P <0.001). Furthermore, all five histopathological diagnoses of predominantly neutrophilic/suppurative enteritis were obtained from the at-T. This neutrophilic component is suspected to be secondary to acute, traumatic injury of the intestinal mucosa from the trichobezoar. In fact, 66.7% of biopsies with a neutrophilic inflammatory component were obtained from cats with acute clinical signs only. This was significantly more common than cats with chronic clinical signs (P <0.001). Bacterial enteropathogens, such as Campylobacter species, some Enterobacteriacea, Bacteroides species and Clostridium species, have been shown to result in neutrophilic enteritis and gastrointestinal clinical signs.7,8 However, enteric bacteria can also be present in clinically healthy animals. 8 Additional testing for enteric bacteria was not pursued in our study, likely because the clinical signs were attributed to the obstructive trichobezoar.
A comparison of pre-T and post-T samples showed similar frequencies of both type and degree of inflammation between groups (Tables 5 and 6). None of these samples showed evidence of mucosal erosion/ulceration. No post-T biopsies exhibited predominantly neutrophilic inflammation, which was anticipated given the lack of mucosal exposure to the trichobezoar aborad to the obstruction. However, only 2/26 intestinal biopsies obtained orad to the trichobezoar obstruction and were described as having a neutrophilic component that would commonly occur with acute inflammation. The unexpectedly low incidence of neutrophilic inflammation in pre-T samples may be associated with the quick regenerative nature of the small intestinal mucosa, which fully regenerates within 2–3 days. 9 Although the duration of time elapsed between trichobezoar exposure of the intestinal mucosa and biopsy collection cannot be determined, it is possible the intestinal mucosa orad to the obstruction had gone through complete regeneration by the time of surgical intervention.
It is worth noting that only 5/100 biopsies in this study showed no evidence of inflammation, and all of these were obtained pre-T. In contrast, no biopsies obtained aborad to the trichobezoar were diagnosed as ‘normal’ as all demonstrated histopathological changes. This may lead some to the conclusion that all cats in this study had some level of underlying enteropathy that may result in secondary intestinal obstruction. This confusion may stem from lack of understanding of ‘normal’ amounts of inflammatory cells within the gastrointestinal tract in cats. The most widely used gastrointestinal histopathology standards, released by the World Small Animal Veterinary Association (WSAVA) Gastrointestinal Standardization Group in 2008, were based on full-thickness biopsies obtained from specific pathogen-free colony cats aged 5–18 months. 6 However, a recent study used WSAVA guidelines to evaluate endoscopic gastric and duodenal biopsies from clinically normal cats aged over 3 years, which were thought to more accurately represent the population demographics of cats presenting for evaluation of chronic enteropathies. All 20 cats in that study had histopathological abnormalities, including 2/20 that were diagnosed with small cell lymphoma. While these two cats later developed clinical signs and were subsequently euthanized, 17/20 never showed any gastrointestinal clinical signs after a median time of 709 days. 4 These findings highlight the need to evaluate biopsy results cautiously and in conjunction with clinical signs and response to treatment.
The presence of lymphoplasmacytic and lymphocytic/eosinophilic inflammation is potentially relevant in this study as these patterns of inflammation are commonly seen in FCE (idiopathic inflammatory bowel disease (IBD), dietary hypersensitivities) and has been reported with intestinal lymphoma,5,10,11,12 all of which may predispose a cat to an intestinal obstruction. These inflammatory patterns are not specific, however, and the retrospective nature of this study precludes definitive diagnosis in our cats. Interestingly, long-haired cats have been described as over-represented in cases of feline gastrointestinal eosinophilic sclerosing fibroplasia, possibly due to increased ingestion of hair and associated allergens. 13 While no masses were noted in these cats, ingestion of hair and the associated allergens could be considered an etiology for eosinophilic enteritis.
Alimentary small cell lymphoma was diagnosed in 10/44 (22.7%) study cats and our findings suggest both chronicity of clinical signs and age should be considered when discussing diagnostic utility of additional gastrointestinal biopsies during surgical intervention for trichobezoar obstruction. Cats diagnosed with alimentary lymphoma represented 41.7% of cats with chronic clinical signs (10/24) and cats diagnosed with alimentary lymphoma were also significantly older, with an average of 12.9 years (vs 6.4 years for cats with inflammatory changes, P = 0.001). This finding is consistent with several studies that have noted the mean age in cats with alimentary lymphoma to be 12.5 years.14 –16
Histopathological diagnoses can vary between biopsies obtained from different anatomic locations, as evidenced in our study. A total of 26 cats had biopsies from two or more anatomic segments of the small intestine, and seven of these cats were diagnosed with alimentary lymphoma. Interestingly, 6/7 (85.7%) cats diagnosed with alimentary lymphoma showed complete agreement among a diagnosis of lymphoma between all small intestinal biopsies, which contrasts a recent study in which a diagnosis of lymphoma within both duodenal and ileal biopsies occurred in only 17% of the population. 17 While the small population of this study was a potential limitation, it does not substantially differ from the number of cats diagnosed with lymphoma in the present study (18 and seven cats, respectively).
Of 26 cats with two or more small intestinal biopsies, 19 had histopathology consistent with various inflammation, and complete histologic agreement occurred in 9/19 (47.4%) cats. Although our study demonstrated relatively high agreement between biopsies, the sample size and lack of standardization with biopsy location and number are considered potential limitations to this finding. Furthermore, biopsies from each cat were submitted and evaluated together, which could bias histopathologic interpretation owing to lack of blinding among pathologists. 18
It has been estimated that long-haired cats are twice as likely as short-haired cats to have frequent elimination of hairballs. 3 This is consistent with the findings of our study, as most cats were considered long- or medium-haired (84.1%). Interestingly, 66% of the short-haired cats in this study were diagnosed with alimentary lymphoma. These findings identify a higher incidence of obstructions in long-haired cats, which may suggest that cats with trichobezoars and short hair are more likely to have underlying gastrointestinal disease as a predisposing factor. However, the small study population hinders our ability to draw definitive conclusions, warranting further studies.
This study’s retrospective design introduces inherent limitations. The subjective nature of biopsy interpretation and lack of blinding among pathologists to the age, clinical signs and additional histopathology results within the same cat may cause considerable bias affecting the interpretation of samples. In addition, there is a lack of consistency among both biopsy quantity and specific location of biopsies, making accurate comparison difficult. Owing to the lack of follow-up in this study, there is no comparison between histopathologic diagnosis and response to treatment, thus preventing a definitive diagnosis of specific forms of FCE.
Conclusions
Our findings suggest that the presence of a trichobezoar may alter the degree and/or type of inflammation present at the level of the obstruction, potentially limiting the ability to interpret biopsies from this site. Furthermore, histopathology changes are commonly found both orad and aborad to the obstruction, emphasizing the need to biopsy multiple sites of the small intestine. Interpretation of these findings should always be performed with regard to the individual’s chronicity of clinical signs and response to therapy. A prospective study with a standardized number of biopsies and biopsy technique, with subsequent evaluation by blinded pathologists, is warranted to further characterize the findings of our study in an effort to develop clinical recommendations moving forward.
Footnotes
Acknowledgements
We would like to thank Dr Deborah Keys for her expertise in running our statistical analysis.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
