Abstract
Case series summary
Salivary gland adenocarcinoma, of major or minor salivary gland origin, is an uncommon tumor in cats. This article describes the clinical features, morbidity and survival rates of four cats with salivary gland adenocarcinoma arising from minor salivary gland tissue. Medical records from a private multicenter dentistry and oral surgery practice were reviewed for the period between 2007 and 2021. Four cats were included in this retrospective case series study, with oral masses on either the right or left caudal mandibular labial buccal mucosa. The inclusion criteria included a diagnosis of salivary gland adenocarcinoma in an anatomical location with lack of involvement of a major salivary gland, complete medical history and a follow-up of at least 6 months. The age range of the cats was 9–15 years; three of the cats were castrated males and one was a spayed female. Curative intent surgery was performed in three cats, whereas palliative surgery (debulking) owing to extensive soft tissue invasion was performed in one cat. Survival times were in the range of 210–1730 (mean 787) days. All four cats were euthanized owing to local recurrence and decreased quality of life, regardless of treatment modality.
Relevance and novel information
There are limited documented studies reporting the prevalence of salivary gland neoplasia affecting minor disseminated glands in the oral cavity of feline patients. Salivary gland adenocarcinoma should be a differential in cats presenting with caudal labial masses. Surgical resection has been the recommended treatment for salivary gland neoplasia of major salivary gland origin. According to this current case series, we propose that early aggressive surgical treatment with wide surgical margins should be performed for cats with salivary gland adenocarcinoma of minor salivary gland origin. Surgery increased the quality and duration of life; however, each patient was euthanized owing to local recurrence and morbidity.
Keywords
Introduction
Salivary gland tumors in dogs and cats are uncommon and comprise <0.2% of tumors affecting these species. 1 Primary neoplasms of the salivary gland often arise from the glandular or ductular epithelium resulting in benign adenomas or carcinomas. 2 There are only a few studies documenting the prevalence of primary salivary gland neoplasia of minor disseminated salivary glands in the oral cavity.3,4 There is also one report in the literature of metastatic adenocarcinoma of a minor salivary gland in a cat. 5 In a study of 245 cases of salivary gland disease, cats were shown to have a higher prevalence (1.6 times greater) of salivary gland tumors than dogs. 4 In the study, 33 cats were diagnosed with salivary gland neoplasia. Of the cats, 31 (93.9%) had major salivary gland adenocarcinoma/carcinoma and 2 (6%) had accessory/minor salivary gland adenocarcinoma. 4 In a more recent study, salivary gland tumors more frequently occurred in the mandibular salivary gland (4/6 cases, 66%). 6 In the remaining two cases, one tumor arose from the left buccal cavity and the other tumor was presumed to arise from minor salivary gland tissue on the labial mucosa.
Cats have four pairs of major salivary glands – parotid, sublingual, mandibular and zygomatic 7 – with scattered glandular tissue present in the submucosa of the lips, cheeks and soft palate. 8 In both dogs and cats, the most frequent major salivary glands to develop neoplasia are the mandibular and parotid glands, which account for approximately 75–80% of all salivary gland neoplasia.1,3 Simple adenocarcinoma (tubular or papillary) most frequently affects major salivary glands, whereas complex adenocarcinoma (ie, containing a myoepithelial component) is less common. Other tumor types, including osteosarcoma, mast cell, sebaceous carcinoma, malignant fibrous histiocytoma, oncocytoma, squamous cell carcinoma and adenoma, have been reported in major salivary glands.1–3,9–15 The presenting complaint in most cases of salivary gland neoplasia is halitosis and dysphagia secondary to an oral mass. 16 In cats, morbidity is frequently more advanced,4,10 and distant metastasis is more common when compared with dogs. 10
Historically, the long-term prognosis for patients with malignant salivary gland neoplasia has been poor. Recent reports have shown an increased survival rate (~1 year) if tumors were diagnosed early, wide surgical margins were obtained and adjuvant therapy, such as radiation, had been utilized.17,18 In a more recent case series using external beam radiotherapy for the treatment of feline major salivary gland carcinoma, it was reported that cats with large and invasive primary tumors may have locoregional control with radiotherapy. 6
There are no current data on minor salivary gland neoplasia in cats. This is the first retrospective case series to document the prevalence, treatment and survival rates of cats with salivary gland adenocarcinoma of minor disseminated salivary gland origin.
Case series description
Medical records from a private multicenter dentistry and oral surgery specialty practice were reviewed for cases of feline patients presenting with caudal mandibular oral masses using the query terms ‘oral mass,’ ‘labial mass’ or ‘oral tumor’. The inclusion criteria included biopsy-diagnosed salivary adenocarcinoma and minor salivary gland location. Specifically, the inclusion of masses was based on specific location in the caudal labial mandibular buccal mucosa, caudal to the mandibular third premolar tooth. This anatomical location is not consistent with a known location of a major salivary gland in feline patients. A description of the presenting anatomical location of the oral masses was imperative to categorize these tumors as arising from minor salivary gland tissue vs major salivary gland tissue owing to their histological similarities. Cases without histological diagnosis or not definitively originating from minor salivary gland tissue were excluded. In addition, cats that received additional histopathology post mortem owing to recurrence of their oral mass and were histologically confirmed as oral minor salivary gland adenocarcinoma were also included in the study.
Of the 4574 cats seen at the private multicenter specialty practice between 2007 and 2021, 85 cats presented with caudal mandibular oral masses. Of these 85 cats, four (4.7%) met the inclusion criteria for the case series. Other diagnoses for the remaining 81 cats consisted of benign cysts of salivary origin, benign fibromatous gingival tumor, carcinoma, fibrosarcoma, chronic inflammation, squamous cell carcinoma and stomatitis (Table 1).
Morphological diagnoses for 85 cats seen between 2007 and 2021 with caudal mandibular oral masses out of a total of 4574 feline patients

The four cats in the study included two domestic shorthairs, one Oriental Shorthair and one Russian Blue cat. Three of the cats were castrated males and one was a spayed female. The median age was 11 years (range 9–15 years).
The most commonly reported clinical complaint was a notable mass associated with the caudal mandibular labial buccal mucosa (3/4). The masses had been present for a period of 1–6 months before treatment. Three of the four cats had blood-tinged saliva. One cat presented with a history of hyporexia and weight loss. Comorbidities included diabetes mellitus, seizures, hypertrophic obstructive cardiomyopathy, heart murmur (echocardiogram declined), microphthalmia, elevated liver enzymes and inappropriate urination (Table 2).
Data summary for four feline patients diagnosed with minor salivary gland adenocarcinoma

DSH = domestic shorthair; FS = female spayed; m = months; MN = male neutered; PD = progressive disease; y = years
Complete blood count (CBC) and serum biochemical analyses were performed on all patients, revealing only minor non-clinically significant abnormalities. Of the four cats, three had three-view chest radiographs performed with no evidence of distant metastasis (patients 2, 3 and 4) at the time of diagnosis. One of the four cats had an evaluation of local draining lymph nodes (ipsilateral to the oral mass), revealing metastatic disease.
On oral examination under anesthesia, 4/4 cats had a solitary caudal mandibular labial buccal mucosal mass with variable location within the labial mucosa (Figures 1 and 2). All masses were caudal to the mandibular third premolar tooth. All masses were greater than 1 cm, with the largest dimension being 3.4 × 4 × 4 cm in patient 3. Other descriptions of the oral masses from the medical record included pedunculated (n = 1/4), ulcerated (n = 2/4), firm on palpation (n = 2/4) and active bleeding (n = 3/4) (Table 3).

Patient 2 with presentation of a left mandibular buccal mucosal mass

Patient 3 with presentation of a right mandibular buccal mucosal mass
Oral mass inclusion criteria

hpf = high power field; L = left; R = right
All cats underwent general anesthesia, and full mouth intraoral dental radiographs were obtained. There were no radiographic osseous changes associated with the oral masses. Advanced imaging (CT, cone-beam CT or MRI) was not performed at the time of the procedures. Only one cat had dental anomalies associated with the mass (type 3 tooth resorption).
Wide soft tissue excision, including 10 mm from grossly abnormal tissue, was performed in 3/4 patients. A deep margin was not attempted. One of the four cats had marginal excision performed. All surgical sites were closed using a combination of simple interrupted and/or horizontal mattress suture patterns with 5-0 Monocryl (Figure 3).
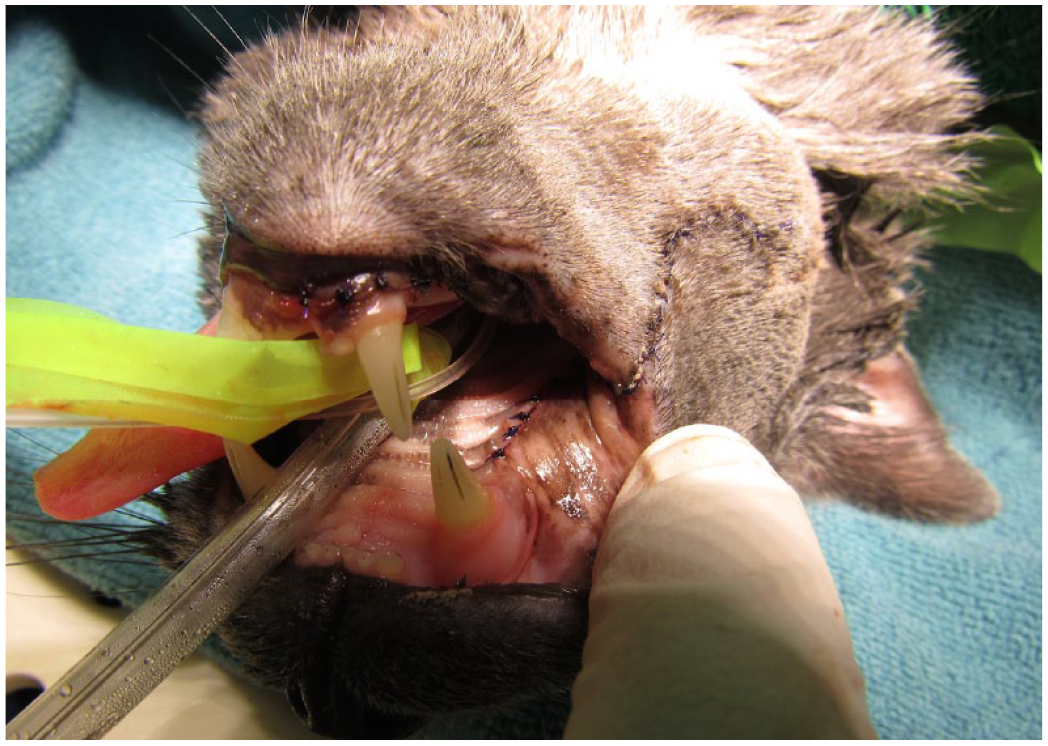
Surgical closure in patient 4 after performing an excisional biopsy of the right mandibular buccal mucosal mass
Histopathology revealed a neoplastic cellular proliferation forming acini, tubules, duct-like structures or solid sheets (Figure 4). The neoplastic cells ranged from cuboidal to polygonal, with moderate amounts of eosinophilic cytoplasm and round to oval nuclei. The mitotic index was in the range of 4–22 (number of mitotic figures in 10 400× microscope fields). Margins were evaluated as clean, narrow or incomplete, with narrow margin status interpreted as <1.0 mm. Clean margins >5 mm were obtained for the three cats with wide excisional biopsy and incomplete margins were obtained for the patient with palliative surgical debulking. All masses were located in the mucosa; therefore, the deep margins measured did not involve bone. Bone was not sampled.

Neoplastic cells arranged in acini, tubules and solid sheets. The neoplastic cells are polygonal to cuboidal with round to oval nuclei. The lobules of neoplastic cells are supported by a collagenous stroma, and there are occasional central areas of necrosis (hematoxylin and eosin, 20×)
All four cats were re-evaluated 2 weeks postoperatively. In all four cases, the surgical sites had healed without complications. Owners noted increased appetite, increased energy and activity level, subjectively decreased pain and overall increase in quality of life. No patients underwent adjuvant chemotherapy or radiation.
The patients’ follow-up data are shown in Table 4. At the time of writing this retrospective study, patient 1 was the only cat alive and additional follow-up information was provided by its primary care veterinarian. The cat was re-evaluated 60 days after the excisional biopsy owing to a pedunculated pink lesion, measuring approximately 4.0 mm in the area of the previous surgical site. Histopathology results from an incisional biopsy of this lesion were consistent with scar/fibrotic tissue and no evidence of neoplastic cells. At 280 days postoperatively, interstitial changes on thoracic radiographs were suggestive of metastatic disease, but multiple soft tissue nodules were not noted. Signs of regrowth of the oral mass at the original surgery site were noted 850 days postoperatively; the cat was asymptomatic. Patient 1 was eventually euthanized 1730 days after the excisional biopsy owing to progression of disease and decreased quality of life. The cat’s owners approved diagnostic imaging and additional histopathology of the oral mass post mortem, which had grown to surround the ventral aspect of the left mandible (Figure 5). Thoracic radiographs showed lesions compatible with metastatic nodules present within the lungs (Figure 6) and a post-mortem incisional biopsy of the mass confirmed recurrence of the adenocarcinoma.
Survival data for feline patients after oral surgery

N/A = not applicable; mm = millimeters
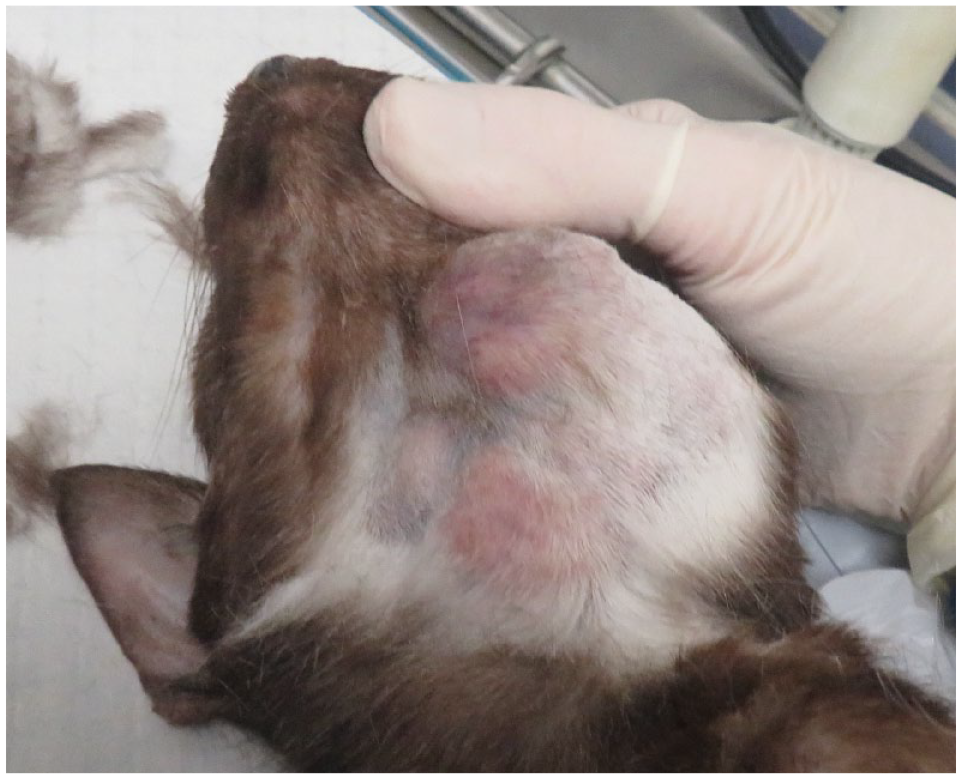
Patient 1 post mortem, on dorsal recumbency, with regrowth of the left mandibular buccal mucosal mass extending towards the ventral aspect of the mandible; 1730 days after excisional biopsy

Post-mortem radiograph of patient 1 showing diffuse bilateral pulmonary nodules involving all lung segments without airspace consolidation or atelectasis. Differential diagnosis favoring metastatic disease
Patient 2 received multiple recheck examinations postoperatively for a period of 5 weeks, with no signs of oral mass regrowth (Table 4). Follow-up via phone call revealed that the patient was euthanized at the time of local regrowth of the mass at 910 days postoperatively.
After the 2-week postoperative recheck, patients 3 and 4 were lost to follow-up. Follow-up via phone call of patient 4 revealed that the cat was alive with recurrence of an oral mass visible at approximately 120 days postoperatively.
The survival times for all four patients were in the range of 210–1730 days from surgical resection (Table 4). The three cats that had an excisional biopsy procedure, with 1.0 cm attempted margins, had a higher mean survival time than patient 3, which was treated with palliative surgery and lymph node removal. All four cats were eventually euthanized owing to recurrence of the oral mass regardless of surgical treatment modality. The owners of each cat perceived a decrease in quality of life when the oral tumor recurred. The most common observations after oral mass recurrence included oral pain and dysphagia.
Discussion
To the authors’ knowledge, this is the first feline case series of salivary gland adenocarcinoma of minor salivary gland origin. Diseases of the salivary glands are various and include inflammatory, obstructive, ischemic and neoplastic processes. 2 A study of 245 cases of salivary gland disease in dogs and cats found that the major salivary glands are usually affected by malignant neoplasia, sialoadenitis, sialocele and salivary gland infarction. 4
Primary neoplasms of major (parotid, mandibular, sublingual, zygomatic) and minor (palatine, lingual, labial, gingival) salivary glands are infrequent in animals.19–21, 22 Adenocarcinoma is the most common type of malignant salivary gland tumor in animals, with a reported 39% rate of lymph node metastasis at the time of diagnosis. 10
Historically, the distinction between minor and major salivary glands has not been well established in the veterinary literature 23 and some of the definitions have varied depending on the number of ducts and size/amount of glandular tissue. Owing to the similar histopathologica structures, documenting the anatomical site of the biopsy, as well as providing a substantial biopsy sample, can help identify these tumors as originating from minor vs major salivary gland tissue. In a more recent study, the precise location of salivary gland tumors in the majority of feline cases, 16/20 (80%) cases, was undetermined owing to the lack of a specific location of the tumor. 24
Both the major and minor salivary glands comprise tubules/ducts and are commonly differentiated depending on the complexity of the tubuloacinar glands. This is currently an accepted classification albeit there is continued debate about the correct way to classify these lesions. Major salivary glands are categorized as compound tubuloacinar glands since they are branched with a more complex system of ducts and acini. 21 Minor salivary glands are simple tubuloacinar glands measuring 1–2 mm in diameter. 21
Besides the four pairs of major salivary glands – parotid, sublingual, mandibular and zygomatic – cats also have minor salivary glands on the lingual and labial aspects of the mandibular first molar tooth, referred to as lingual molar and labial molar glands (or buccal glands).23,25 The buccal molar salivary gland empties into the buccal oral cavity by several small ducts. 26 The membranous molar pad just lingual to the mandibular first molar tooth has numerous small salivary gland openings directed towards the tongue. 27 The minor mucosal labial glands in cats are scattered throughout the submucosa of the lips, with numerous small excretory ducts. 26 Cats also have minor mucosal buccal glands, which similarly have numerous small excretory ducts. 27
Histological changes with adenocarcinoma of the salivary gland can present as neoplastic epithelial cells forming acini, ducts, trabeculae, nests or solid sheets. 2 Tumor cells can have different morphologies such as cuboidal, columnar, polygonal, clear, mucinous, oncocytoid and plasmacytoid. 21 The histopathology of the cats of this series showed acini, tubules and duct-like structures. The histopathology findings combined with the location of the oral masses support the conclusion that salivary adenocarcinoma was arising from the labial (buccal) molar minor salivary gland in all four cats.
In human medicine, the primary treatment of salivary gland tumors involves surgical excision, with radiation reserved for inoperable masses or adjuvant therapy after incomplete removal. 28 In a recent study of feline major and presumed minor salivary gland carcinoma treated with radiotherapy after surgical excision of the primary tumor, the role of radiation therapy was unclear and its outcome was variable. 6 The survival times of our study were an average of 980 days for cats with excisional biopsies with clean (>5.0 mm) margins (patients 1, 2 and 4), and 210 days for patient 3 which had palliative surgery performed. The survival times in our study surpass previous survival rates in the study by Hammer et al, 10 where cats with adenocarcinoma in both major and minor salivary glands, with variable treatment (surgery alone, surgery and radiation, or surgery and chemotherapy), had a median survival time of 516 days, regardless of treatment type. These results suggest that complete, aggressive resection of adenocarcinoma of minor salivary gland tissue could potentially offer an increased survival time and decreased morbidity in cats.
Owing to the small number of cases in our study, no conclusions can be made regarding risk factors for minor salivary gland adenocarcinoma. In a previous study in 2001, Siamese or Siamese-cross cats represented 30% (9/30) of affected cats with salivary gland neoplasia, indicating a possible breed predisposition. 10 In a more recent study of 56 dogs and 24 cats diagnosed with salivary gland neoplasia, a feline breed predilection was not determined. 24 In the 2001 study, a 2:1 predilection ratio for male cats was found in 30 cats with salivary neoplasia. 10 In the current study, the mean age of cats diagnosed with salivary gland adenocarcinoma was 11 years (range 9–15). These results are similar to those of the 2001 previous study in which the median age for affected cats with salivary gland neoplasia was 12 years. 10
The retrospective nature of this case series resulted in limitations in the information able to be obtained and evaluated. Minor salivary gland tumors are less frequently reported than major salivary gland tumors, 24 and additional data are necessary to determine the presence of metastatic disease at the time of initial diagnosis. Metastatic disease occurred in 2/4 cats of this study (mandibular lymph node in patient 3 and pulmonary metastasis in patient 1). This supports the previous literature, which found that salivary gland adenocarcinoma has the potential to metastasize and thus recommends preoperative staging.10,29
The prevalence of salivary gland adenocarcinoma of minor salivary glands is low, representing only 4.7% of all caudal oral masses documented over a 14-year period. Other differential diagnoses for cats with caudal labial buccal mucosal mandibular masses should be considered. Feline chronic gingivostomatitis can present with generalized or localized areas of ulceration or proliferation on the alveolar and buccal mucosa of the caudal oral cavity.30,31 A focal proliferative mass-like lesion of the caudal buccal mucosa in cats, referred to as a pyogenic granuloma, is a chewing or traumatic reactive lesion (inflamed granulation tissue).32,33 Pyogenic granulomas most often occur on the mucosa that is buccal and sometimes distal and/or lingual to a mandibular molar tooth. 34 Additional granulomatous inflammatory lesions can also develop secondarily to allergies, chronic infection and even embedded foreign material.
Conclusions
This retrospective case series describes the survival times of four cats with minor salivary gland adenocarcinoma treated with either wide excisional resection or palliative surgery. The survival time was greater for these cats compared with previously reported literature. Based on this current case series and a literature review over the past four decades, we suggest that regional control with wide excisional biopsy can increase the survival time and quality of life in cats presenting with adenocarcinoma of minor salivary gland origin. Salivary gland neoplasia should be a differential for masses located in the caudal labial buccal mandibular mucosa of a cat. Further research is necessary in order to identify other treatment options, such as radiation therapy for feline patients with large and invasive primary tumors that do not qualify as surgical candidates or have had incomplete margins obtained.
The primary cause of death in the cats in this study was from local recurrence rather than distant metastasis. Yet, our study supports previous reports of metastatic potential of minor salivary gland adenocarcinoma in cats, 5 in particular to the regional lymph nodes and pulmonary parenchyma as noted in two cases (patients 1 and 3) in this study.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
