Abstract
Objectives
The aim of this study was to evaluate the expression profile of sex steroid receptors and redox mediators in the uterus of domestic cats with pyometra.
Methods
Twelve cats were used and divided into groups: (1) non-gestational healthy diestrus (n = 7) and (2) pyometra (n = 5). The plasma profiles of estradiol and progesterone (P4) as well as uterine expression levels of estradiol alpha (ERα), progesterone (PR) and androgen (AR) receptors, of the antioxidant enzymes superoxide dismutase 1 (SOD1), catalase and glutathione peroxidase 1 (GPX1), and of the oxidative damage marker 8-hydroxy-2′-deoxyguanosine (8-OHdG) were evaluated.
Results
Cats with pyometra showed higher plasma P4 levels and increased uterine messenger RNA (mRNA) and protein expression of ERα and PR, mainly in the glandular epithelium for ERα and in stromal and myometrial cells for PR. In addition, there was an increase in 8-OHdG immunostaining and GPX1 mRNA and protein expression in cats with pyometra compared with those in non-gestational diestrus, while catalase showed a reduction in endometrial immunostaining in cats with pyometra. There were no differences in uterine AR and SOD1 expression between the groups.
Conclusion and relevance
The findings of this study showed that pyometra is associated with oxidative stress in the uterus of domestic cats and alterations of the profile of sex steroid receptors, especially ERα and PR, and of antioxidant enzymes, suggesting that changes in these mediators may play a role with the etiopathogenesis of this disease.
Introduction
Pyometra is considered the most important inflammatory reproductive disease in dogs and cats and is characterized by the accumulation of intrauterine purulent exudate associated with hyperplasia of the endometrial glands.1,2 Pyometra is frequently reported in intact dogs, and there are still few studies in cats, 1 especially in relation to the molecular mechanisms involved in the pathogenesis of the disease.
Pyometra in cats is more common in diestrous adult females. 1 In young animals, it is most often associated with exogenous administration of high doses of sex steroids, while appropriate doses of progestins do not result in uterine disease.3,4 Studies have hypothesized that increased levels of progesterone (P4) and estrogen are involved in the occurrence of this disease,1,5,6 although no study has demonstrated higher plasma levels of these hormones in cats with pyometra. In dogs, there is an increase in the uterine expression of P4 receptors, suggesting that alterations in uterine expression of this receptor are also involved in the pathogenesis of this disease in this animal species.3,7 In cats, on the other hand, studies are scarce. The only study, carried out by Misirlioglu et al, 8 demonstrated that estrogen receptor alpha (ERα) expression was reduced in the uterus of cats with mild endometrial hyperplasia, while there was an increase in progesterone receptor (PR) in cats with mild and severe endometrial hyperplasia. 8 In addition, there is no knowledge about the androgen receptor (AR) profile in the uterus of cats with pyometra. Studies have already demonstrated the importance of AR in the uterine changes observed in diestrous women,9,10 in addition to the protection of uterine stromal cells against oxidative stress. 11
In that regard, antioxidant enzymes also modulate uterine morphophysiology, not only sex steroids. Alteration of their expression is also involved in pyometra in dogs, 12 as well as in other reproductive diseases in women such as miscarriage, pre-eclampsia and intrauterine growth restriction.13–16 Studies in rats demonstrated that the increase in estradiol (E2) and androgens increased oxidative stress in the uterus and reduced catalase activity, 17 while progesterone stimulated uterine glutathione peroxidase (GPX) activity.17,18 This suggests that changes in the plasma profile of sex steroids in cats with pyometra may also result in an imbalance in the uterine oxidative profile. We have recently demonstrated that the uterine expression of kisspeptin and its receptor Kiss1R, as well as angiogenic and immunological mediators, such as vascular endothelial growth factor, placental growth factor, interleukin-10, tumor necrosis factor, interleukin-6, and macrophage migration inhibitory factor, are altered in the uterus of cats with pyometra, and these mediators also influence tissue oxidative balance.19–21 However, to date, there are no studies that have evaluated the profile of antioxidant enzymes and the occurrence of oxidative stress in the uterus of cats with pyometra.
Thus, the present study evaluated the expression profile of sex steroid receptors and redox status in the uterus of cats with pyometra.
Materials and methods
Experimental design
Nulliparous cats without a defined breed, aged 6 months to 2 years, were used in the present study. All the animals were subjected to hemogram and urinalysis laboratory examinations, and healthy cats and those presenting with pyometra were subjected to ovariohysterectomy at the Veterinary Hospital of Universidade Estadual de Santa Cruz (UESC). All procedures were approved by the Ethics Committee on the Use of Animals (CEUA) (Process no. 029/21).
The genital tracts of 12 cats were collected. Macroscopic analysis of the genital system was performed and uterus fragments were collected for histopathology and immunohistochemistry (IHC). Uterine fragments were also stored in cryotubes with Trizol, frozen in liquid nitrogen and stored at −80°C for evaluation of gene expression by quantitative real-time polymerase chain reaction (qPCR). Blood samples were collected to measure P4 and E2.
After clinical, macroscopic and histological analyses and evaluation of the plasma profile of P4 and E2, the animals were divided into the following experimental groups: (1) non-gestational healthy diestrus (n = 7) and (2) pyometra (n = 5).
Ovariohysterectomy
To perform the ovariohysterectomy, acepromazine (0.05 mg/kg IM) (acepromazine 2%; Syntec) and morphine (0.3 mg/kg IM) (morphine sulfate 10 mg/ml; Hipolabor + Sanval) were used as pre-anesthetic medication for healthy cats in diestrus, while meperidine (3 mg/kg IM) (Petidina; Uniao Chemical Laboratory) was used for cats with pyometra. For anesthetic induction, propofol (6 mg/kg IV) (Propotil; Biochimico) was used, with subsequent continuous infusion of Ringer’s lactate (6 ml/kg/h). The cats were intubated and anesthetic maintenance was performed with isoflurane (Isoforine; Cristália) in 100% oxygen (300 ml/kg/min) in an open circuit (Baraka) using an anesthesia machine (WATO EX-65; Mindray). Before the surgical procedure, the animals received sodium cephalothin (30 mg/kg IV) as prophylactic antibiotic therapy, even with all the proper aseptic conditions. Ovariohysterectomy was performed using the three-clamp technique after laparotomy through a midline incision.
Tissue processing and histopathological evaluation
The uterus samples were fixed with 10% formaldehyde for 24 h. Then, the material was processed by the paraffin embedding technique and subsequent cleavage. Tissues were serially dehydrated in a 70% alcohol solution to absolute alcohol (100%), with subsequent diaphanization in xylene and embedding in paraffin. Histological sections of 4 µm were obtained on histological slides. After this process, the sections were stained with hematoxylin and eosin for histopathological analysis. Sections of 4 µm were also obtained on polarized silanized slides (StarFrost Polycat) to perform the IHC.
Female cats were considered diestrous when the uterus was dilated, with endometrial gland hypertrophy and hyperplasia associated with secretory activity, and increased plasma levels of E2 (>10 pg/ml) and P4 (>4 ng/ml).22–24 For animals with pyometra, previously described parameters were used5,25–27 and only cats with grade 3 or 4 pyometra were considered. Cats with grade 3 or 4 pyometra displayed cystic endometrial hyperplasia associated with inflammatory infiltrate in the uterine lumen and endometrial glands, with or without myometrial hypotrophy.
Hormone determination
Blood samples were collected in EDTA tubes immediately after ovariohysterectomy by puncturing the cephalic or jugular vein, between 09:00 am and 11:00 am. The blood was centrifuged at 3000 rpm for 20 mins and the plasma obtained was stored at −20°C for later hormonal determination of E2 and P4 by ELISA using commercial kits (Enzo Life Sciences).
Immunohistochemistry
Histological sections of the uterus were subjected to IHC analysis using anti-PR (1:2000, sc-130071; Santa Cruz Biotechnology), anti-ERα (1:100, 6f11; Thermo Fisher Scientific), anti-AR (1:50, PG-21; EMD Millipore Corp), anti-catalase (1:200, sc-271803; Santa Cruz Biotechnology), anti-superoxide dismutase 1 (SOD1) (1:1000, sc-101523; Santa Cruz Biotechnology), anti-GPX1/2 (1:1000, sc-133160; Santa Cruz Biotechnology), and anti-8-hydroxy-2′-deoxyguanosine (8-OHdG) (1:200, sc-393871; Santa Cruz Biotechnology) antibodies.
The streptavidin-biotin-peroxidase technique was used. Antigen retrieval was performed by heating in a water bath at 98°C for 20 mins and then for 20 mins at room temperature, using EnVision FLEX Target Retrieval solution (DM828; EnVision FLEX+, Mouse, High pH; Agilent DAKO) with pH 6.0. Slides were incubated in a humid chamber for 5 mins in the endogenous peroxidase blocking stage (EnVision FLEX Peroxidase-Blocking Reagent [SM801]), followed by 30 mins in the serum blocking stage (Ultra vision Block; Lab Vision Corp). The slides were incubated with the primary antibodies in an overnight humid chamber for 18 h or 40 h and for 30 mins in the stabilizing protein stage (EnVision FLEX + Mouse LINKER [SM804]) and then with the streptavidin-conjugated secondary antibody (EnVision FLEX HRP [SM802]) for 30 mins. The chromogen used was diaminobenzidine (EnVision FLEX DAB+ Chromogen [DM827]). Sections were counterstained with Harris hematoxylin. The negative control was obtained by replacing the primary antibody with phosphate-buffered saline. 20
Descriptive and quantitative evaluation of the immunohistochemical staining of PR, ERα, AR, catalase, SOD1, and GPX1/2 in the uterus was performed. In the descriptive analysis, luminal epithelium, glandular epithelium, stromal and myometrial cells were evaluated in the uterus. For quantitative analysis, images of three random fields of the superficial endometrium and myometrium of each animal were obtained using a Leica DM 2500 microscope, with a 40× objective, using a Leica DFC 295 digital camera (Leica Microsystems). The area of immunostaining was determined using WCIF ImageJ software (Media Cybernetics Manufacturing). Color deconvolution and thresholding of the images were performed to analyze the marked area. Data from each tissue were archived, analyzed and expressed as immunostaining area in pixels. 20
qPCR
For the qPCR technique, the extraction of total RNA from the uterus was performed using Trizol (Invitrogen, Life Technologies) following the kit protocol. Then, 1 µg of RNA was used for complementary DNA (cDNA) synthesis using the GoTaq 2-Step RT-qPCR System (A6010). Target gene transcripts were quantified by qPCR using SYBR Green on Applied Biosystems 7500 Fast Real-Time PCR System equipment (Applied Biosystems, Life Technologies). For qPCR reactions, 1.5 µl of cDNA, 100 nM of each primer and 12.5 µl of GoTaq qPCR Master Mix reagent 2× were used in a final reaction volume of 20 µl. As a negative control, the DNA amplification mix was used, in which the cDNA sample was replaced with water. Amplifications were performed under the following conditions: enzymatic activation at 95°C for 2 mins, then 40 cycles of denaturation at 95°C for 15 s and annealing/extension at 60°C for 60 s. Standard curves of all transcripts were used to evaluate the linearity and efficiency of the amplification, as well as to evaluate the melting curve of the amplification products. The primers for Sod1, Catalase, Gpx1, Ar, Pr, and Erα were designed based on the messenger RNA (mRNA) sequence of Felis catus (Table 1). Gene expression was analyzed by the 2−∆∆CT method, in which the results obtained for each group were quantitatively compared after normalization based on the expression of Felis catus 40S Ribosomal protein S7 (Rps7). 28
List of genes and nucleotide sequence of primers for qRT-PCR
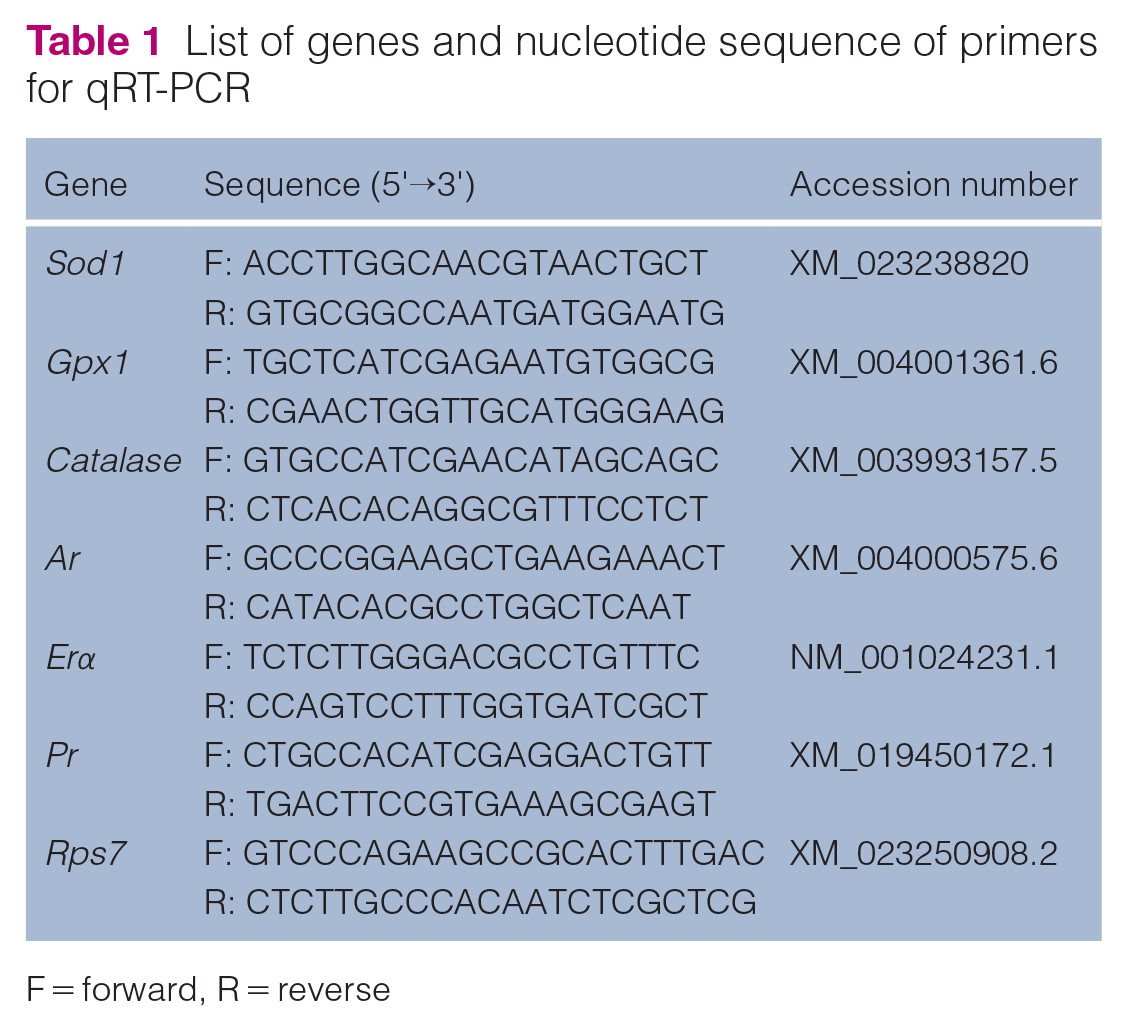
F = forward, R = reverse
Statistical analysis
Data are presented as mean ± SEM. The unpaired Student’s t-test was performed for comparisons between two groups, using GraphPad Prism 8.0.2 software. Analyses were considered significant when P <0.05 (*P <0.05; **P <0.01; ***P <0.001).
Results
Cats with pyometra have higher plasma P4 levels and higher uterine expression of ERα and PR
First, to confirm the animals in the non-gestational healthy diestrus and pyometra groups, we evaluated the plasma profile of E2 and P4, as well as the macroscopic appearance and histopathology of the uterus. Cats in non-gestational diestrus showed high secretion activity of the uterine glands and high plasma levels of E2 and P4 (Figure 1a,b). For the pyometra group, animals also presented high plasma levels of E2 and P4, in addition to purulent exudate in the uterine lumen (Figure 1c,d) and interstitial lymphohistiocytic inflammatory infiltrate with neutrophils in the interior of the uterine glands (Figure 1e,f). Interestingly, higher plasma P4 levels were observed in cats with pyometra compared with those in non-gestational healthy diestrus (P <0.05) (Figure 1b).

Uterine macroscopic and microscopic appearance and hormonal profile of estradiol (E2) and progesterone (P4) in healthy cats in diestrus and cats with pyometra. (a,b) Plasma levels of (a) E2 and (b) P4 in healthy cats in diestrus and cats with pyometra (mean ± SEM; n = 4–7). (c,d) Macroscopic appearance of the uterus of a cat with pyometra. (e,f) Uterine photomicrographs showing inflammatory infiltrate in the interstitium (asterisk) and inside the uterine glands (arrowhead) (staining: hematoxylin and eosin; bar = 200 µm (e), 100 µm (f)). Significant differences were determined using Student’s t-test; *P <0.05
Because studies have also shown changes in the expression of sex steroid receptors in the uterus of dogs with pyometra 3 and because there is only one study of cats in this regard, 8 we evaluated the gene and protein expression of ERα, PR, and AR in the uterus of cats with pyometra. Endometrial immunostaining of ERα and PR was higher in cats with pyometra compared with healthy cats in diestrus (P <0.05; P <0.001) (Figure 2a,b), while in the myometrium, cats with pyometra also showed higher PR staining compared with healthy cats in diestrus (P <0.05). In the analysis of gene expression, similar to immunostaining, cats with pyometra also showed a higher expression of transcripts of Erα and Pr than healthy cats in diestrus (P <0.05; P <0.01) (Figure 2d). Regarding AR, there was no significant difference in the immunostaining area or mRNA expression between the groups (P >0.05) (Figure 2b–d).

Expression of estradiol receptor alpha (ERα), progesterone receptor (PR) and androgen receptor (AR) in the uterus of healthy cats in diestrus and cats with pyometra. (a) Photomicrographs of immunohistochemical staining of ERα, PR and AR in the endometrium and myometrium (Streptavidin-biotin-peroxidase; Harris hematoxylin; bar = 50 µm). (b,c) Area of immunostaining in pixels of ERα, PR and AR expression in (b) the endometrium and (c) the myometrium (mean ± SEM; n = 5–7). (d) Relative gene expression of Erα, Pr and Ar in the uterus (mean ± SEM; n = 5–7). Significant differences were determined using Student’s t-test; *P <0.05; **P <0.01; ***P <0.001
Uterine expression of catalase and GPX1 is altered in the uterus of cats with pyometra
Considering that dogs and mares with pyometra display reduced plasma and/or uterine activity of antioxidant enzymes,12,26,29 we sought to evaluate the expression profile of SOD1, catalase, and GPX1 in the uterus of cats with pyometra. Regarding SOD1 expression, there was no significant difference in uterine immunostaining between the groups, while a trend of increased mRNA expression was observed in the uterus of cats with pyometra compared with those in non-gestational healthy diestrus (P = 0.05) (Figure 3d). Regarding catalase expression, it was observed that the endometrial immunostaining was reduced in cats with pyometra compared with healthy cats in diestrus (P <0.05) (Figure 3a,b). On the other hand, myometrial immunostaining of GPX1/2 and mRNA expression of Gpx1 were increased in the uterus of cats with pyometra compared with those in non-gestational diestrus (P <0.05; P <0.01) (Figure 3a,c,d).
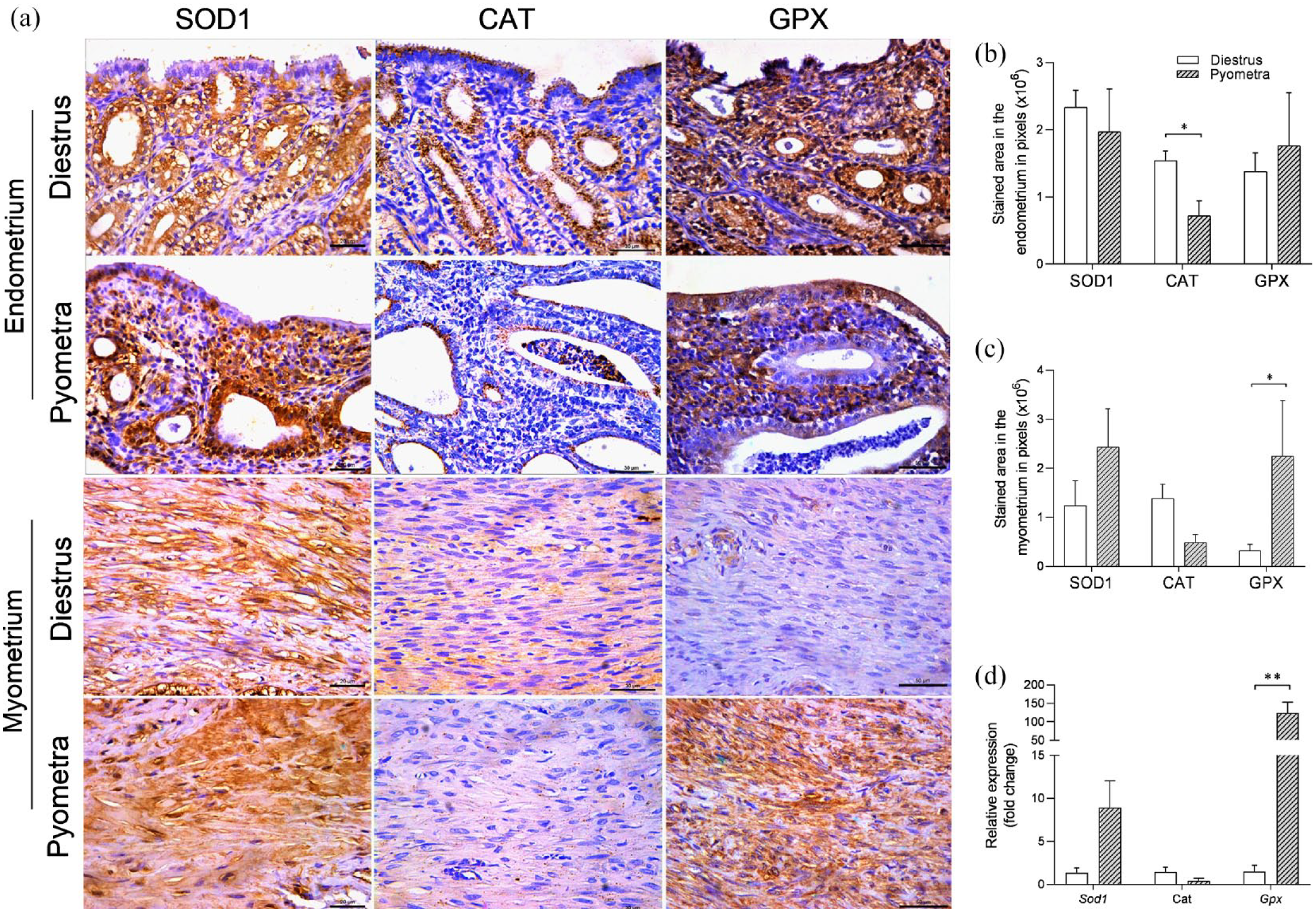
Expression of superoxide dismutase 1 (SOD1), catalase and glutathione peroxidase (GPX) in the uterus of healthy cats in diestrus and cats with pyometra. (a) Photomicrographs of immunohistochemical staining of SOD1, CAT and GPX1/2 in the endometrium and myometrium (Streptavidin-biotin-peroxidase; Harris hematoxylin; bar = 50 µm). (b,c) Area of immunostaining in pixels of SOD1, CAT and GPX1/2 expression in (b) the endometrium and (c) the myometrium (mean ± SEM; n = 5–7). (d) Relative gene expression of Sod1, Cat and Gpx1 in the uterus (mean ± SEM; n = 5–7). Significant differences were determined using Student’s t-test; *P<0.05; **P <0.01
Pyometra causes oxidative stress in the uterus of cats
Since there was an imbalance in the uterine expression of GPX1 and catalase in the uterus of cats with pyometra, we evaluated whether this disease causes oxidative stress in the uterus by evaluating the immunostaining of 8-OHdG, a biomarker of endogenous oxidative DNA damage.21,30 The analysis showed that endometrial and myometrial immunostaining of 8-OHdG was higher in cats with pyometra compared with healthy cats in diestrus (P <0.05) (Figure 4a–c).

8-Hydroxy-2′-deoxyguanosine (8-OHdG) immunostaining in the uterus of healthy cats in diestrus and cats with pyometra. (a) Photomicrographs of immunohistochemical staining of 8-OHdG in the endometrium and myometrium (Streptavidin-biotin-peroxidase; Harris hematoxylin; bar = 50 µm). (b,c) Area of immunostaining in pixels of 8-OHdG expression in (b) the endometrium and (c) the myometrium (mean ± SEM; n = 5–7). Significant differences were determined using Student’s t-test; *P<0.05
Discussion
This is the first study to concurrently evaluate the modulation of antioxidant enzymes and sex steroid receptors in the uterus of cats with pyometra. The findings demonstrated that cats with pyometra have uterine oxidative stress and higher plasma P4 levels, and these changes are associated with increased uterine expression of ERα, PR and GPX1 and reduced expression of catalase.
In cats with pyometra, an increase in plasma P4 levels and in uterine gene and protein expression of ERα and PR was observed. ERα showed higher immunostaining, mainly in the endometrial glands, while PR showed higher expression in glandular epithelium and in stromal and myometrial cells. Similar to the present study, Misirlioglu et al 8 also observed greater immunostaining of PR in the uterus of cats with pyometra, as was also demonstrated in dogs. 7 However, different from the ERα results of this study, Prapaiwan et al 3 observed a reduction in endometrial immunostaining of ERα in dogs with pyometra, suggesting that there may be species-specific differences in uterine ERα expression in the pyometra condition. Together, our results suggest that the higher uterine gene and protein expression of ERα and PR in cats with pyometra, as well as higher plasma levels of P4, are involved in the pathogenesis of this disease in cats, since the development of pyometra in dogs depends on a prior hyperprogesteronic state. 29 This is the first study to demonstrate a higher plasma concentration of P4 in cats with pyometra compared with cats in healthy diestrus, reinforcing the importance of the hyperprogesteronic state for the development of the disease. However, whether the higher P4 levels observed in cats with pyometra is the result of higher luteal steroidogenic activity or reduction of luteolytic enzymes, such as 20alpha-hydroxysteroid dehydrogenase, 31 needs further study. Furthermore, not only high luteal activity may be associated with the occurrence of pyometra in cats, but also the presence of ovarian cysts, which should also be included in the differential diagnosis during the evaluation. In addition, normal ovaries that are not in the luteal phase or with cystic follicles are found in approximately 15–23% of queens with pyometra. 5
The reduction in catalase immunostaining observed in the uterus of cats with pyometra may also be involved in the pathogenesis of this disease, since reduced SOD and catalase activity has also been demonstrated in the uterus and plasma of dogs with pyometra 32 and catalase is one of the main enzymatic antioxidants present in most aerobic organisms. 33 Interestingly, the reduction in uterine expression of catalase in cats with pyometra was accompanied by an increase in gene and protein expression of ERα and PR, contrary to what occurs physiologically in the uterus of cats in diestrus. 34 In addition, previous studies in rats demonstrated that the administration of E2 increased catalase expression in the uterus, while ovariectomy reduced its expression.35,36 This differs from the present study, in which cats with pyometra showed reduced uterine expression of catalase, even with high plasma levels of E2 and P4 and higher endometrial expression of ERα. Studies in rats suggested that the increase in catalase in the uterus after treatment with E2 occurs to control the oxidative stress caused by this hormone.17,37 Thus, we suggest that the reduction of catalase observed in the uterus of cats with pyometra in the present study may favor the uterine oxidative stress observed in these animals by 8-OHdG immunostaining and the occurrence of this disease, since the increase in reactive oxygen species that occurs in oxidative stress causes damage to the uterine luminal epithelium, favoring the entry of pathogenic bacteria. 29 However, more studies are needed to establish whether this oxidative stress is the end product or perhaps one of the initial steps of the pathogenesis of pyometra in cats.
However, unlike the results of catalase immunostaining, cats with pyometra showed increased protein and mRNA expression of GPX1 in the uterus. This is different from what has been observed in dogs with pyometra, which demonstrated a reduction in the uterine concentration of this enzyme. 12 It is plausible that this increase in GPX1 in the uterus of cats with pyometra may be associated with an increase in endometrial ERα and PR, since we observed a physiological reduction of Gpx1 in the endometrium of cats in diestrus, concomitant with the reduction of ERα and PR. 34 Furthermore, a positive correlation was observed between plasma E2 and uterine levels of GPX in women, 38 like that observed in uterine GPX expression in rats treated with E2, 39 in agreement with the results of the present study. The increase in GPX1 in the uterus of cats with pyometra may also reflect increased uterine oxidative stress observed in these animals, as the body increases the expression of antioxidant enzymes in order to control the increase in reactive oxygen species released by neutrophils and macrophages in the endometrium.40,41
Conclusion
The findings of this study showed that pyometra in domestic cats is associated with high plasma P4 levels and oxidative stress in the uterus, in addition to alterations in the uterine expression of sex steroid receptors, mainly ERα and PR, and of antioxidant enzymes, such as catalase and GPX1, suggesting that changes in their profiles may be associated with the development of this disease in cats.
Footnotes
Acknowledgements
The authors thank Ivo Arouca (State University of Santa Cruz, Ilhéus, Brazil) for the technical support provided.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Universidade Estadual de Santa Cruz (UESC) and Coordenaçao de Aperfeiçoamento de Pessoal de Nivel Superior (Capes).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (“best practice”) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
