Abstract
Objectives
This study aimed to determine the prevalence of clinically relevant bacteria on the surface of hospital-issued iPads and to assess the effectiveness and residual effect of a new cleaning regime using 70% alcohol and 2% chlorhexidine wipes.
Methods
Hospital-issued iPads were swabbed to determine the presence of clinically relevant organisms. The iPads were wiped using 70% alcohol and 2% chlorhexidine. Further samples were taken 5 mins, 6 h and 12 h after implementation of the cleaning regime. Cultured bacteria were tested for antimicrobial resistance.
Results
A total of 25 hospital-issued iPads were analysed. Seventeen iPads (68%) sampled in this study were contaminated. Bacillus species (21%) were the most predominant, followed by Pasteurella species (14%), Acinetobacter species (11%), Eikenella species (11%), beta-haemolytic streptococci (11%), coagulase-positive staphylococci (7%), Escherichia coli (7%), coagulase-negative staphylococci (7%), alpha-haemolytic streptococci (3%), Enterococcus species (4%) and Pseudomonas species (4%). Of the isolated bacteria, 89% were resistant to at least one of the tested antibiotics. Of our isolates, 24 (75%) were resistant to clindamycin. After the cleaning regime, there was no bacterial growth from any of the devices at 5 mins, 6 h and 12 h despite repetitive use within the hospital.
Conclusions and relevance
A variety of nosocomial pathogens, including antibiotic resistant pathogens, were isolated from the iPads. Cleaning with 70% alcohol and 2% chlorhexidine wipes is recommended every 12 h during use, between patient contacts and after witnessed contamination. A variety of nosocomial pathogens, including antibiotic-resistant pathogens with potential devastating effects on both human and animal health, were isolated from the iPads. Infection prevention strategies related to the devices should be employed in a hospital setting.
Keywords
Introduction
Hospital-acquired or nosocomial infections can cause considerable morbidity and mortality and pose a large financial burden upon human and veterinary healthcare. Bacterial contamination of personal electronic devices is well recognised in the human and veterinary setting.1,2 Mobile devices, such as mobile phones or personal digital assistants (PDAs), represent a rather novel ‘surface’ in the hospital setting that may also play an important role in the transmission of nosocomial pathogens. Many hospital-associated pathogens are capable of surviving on environmental surfaces and these surfaces, therefore, are a potential reservoir for transmission of infection.
In human healthcare settings, patient side electronic devices (PSEDs) are commonly used for point-of-care data capture and electronic health records, and such devices are increasingly used in veterinary healthcare provision. In contrast to personal devices, PSEDs are typically consulted and handled by ward staff, directly before and directly after patient interaction. These can include intravenous catheter management, urinary catheter management, administration of medications, drain management or the cleaning of soiled bedding. PSEDs are also used during anaesthetic monitoring, in patient preparation areas and in surgical theatres, and can be handled by multiple staff members, increasing the risk of cross-contamination.
The design of the iPad suggests convenient cleaning because of the simple, smooth oleophobic surface and few buttons. Touchscreen phones have been found to harbour fewer microbes than equivalent keypad devices. 3 Data are limited regarding effective disinfecting protocols in the human medical literature. 4 An electronic literature search was performed in June 2022 and in August 2022 using the PubMed database with the following keywords: iPads; disinfection; and cleaning protocols. To the best of the authors’ knowledge, there have been no studies conducted to evaluate the effectiveness of disinfection protocols in a veterinary clinical setting.
Howell et al 4 examined the efficacy of iPad disinfection in a non-clinical laboratory setting using different disinfectant wipes after contamination with a microbial broth (methicillin-resistant Staphylococcus aureus [MRSA], vancomycin-resistant Enterococcus [VRE] and Clostridium difficile). Sani-Cloth CHG 2% not only demonstrated significant efficacy in removing the majority of MRSA and VRE from the surfaces of the iPads, but a single disinfection prevented further contamination of the devices for up to 12 h, in spite of repeated exposure of the device. In a veterinary clinical environment, however, there may be ongoing repeated contamination of the device, which may reduce the duration of the residual effect. The aims of the present study were to determine the prevalence of bacterial contamination on PSEDs in a veterinary hospital and to test a new cleaning regime using a 70% alcohol and 2% chlorhexidine surface wipe.
Our hypothesis was that PSEDs would be found to be colonised with typical human skin bacteria, and also with bacteria of clinical significance, which could increase the risk of hospital-acquired infections.
Our second hypothesis was that after implementation of the cleaning regime using a 70% alcohol and 2% chlorhexidine product (Sani-Cloth CHG 2% Professional Disposables International), we would see an immediate and residual decreased level of contamination of the PSEDs.
Materials and methods
This study was conducted between January and September 2022 at the authors’ hospital. Random samples for culture were obtained from iPads used by the healthcare workers and nursing and veterinary team in both the hospital and clinic settings. Every iPad was systematically swabbed three times on all sides using a grid pattern to include the screen and the buttons with sterile culture swabs (transport swabs with Amies charcoal medium). The samples were refrigerated and then cultured within 24 h of collection.
With a gloved hand, the iPads were then wiped three times over the entire screen surface with the same wipe 5 using 70% alcohol and 2% chlorhexidine wipes (Sani-Cloth CHG 2%) and left to dry for 5 mins at room temperature. 4 A second sample collection was carried out after decontamination as described previously. Further samples were obtained at 6 h and 12 h after implementation of the cleaning regime. During that time, the iPads were used by the veterinary healthcare workers in the hospitalisation area as routine.
The swabs were inoculated onto Columbia agar with sheep’s blood, MacConkey without salt, Columbia Naladixic Acid Agar (CNA) for Staphylococcus and Streptococcus species, Sabouraud Dextrose agar with chloramphenicol plates and incubated at 37°C for up to 48 h. Organisms were identified with a combination of colonial morphology, Gram stain and analytical profile index (API), and supplementary biochemical phenotypical tests (catalase, coagulase and oxidase) were carried out.
Cultures were tested for resistance to amoxicillin, amoxicillin–clavulanic acid, cephalexin, sulfamethoxazole/trimethoprim, marbofloxacin, erythromycin and clindamycin using a Stokes comparative disc diffusion test and then by using a broth microdilution test to EUCAST guidelines and breakpoints. 6
Results
A total of 25 hospital-issued iPads were analysed. Of the iPads sampled in this study, 17 (68%) were contaminated with either single or mixed bacterial agents. The results of antibiotic resistance are listed in Table 1. Bacillus species (21%) was the most common, followed by Pasteurella species (14%), Acinetobacter species (11%), Eikenella species (11%), beta-haemolytic streptococci (11%), coagulase-positive staphylococci (7%), Escherichia coli (7%) and coagulase-negative staphylococci (7%). Those with the lowest prevalence were alpha-haemolytic streptococci (3%), Enterococcus species (4%) and Pseudomonas species (4%) (Figure 1).
Specific organisms and antibiotic resistance pattern identified on each iPad
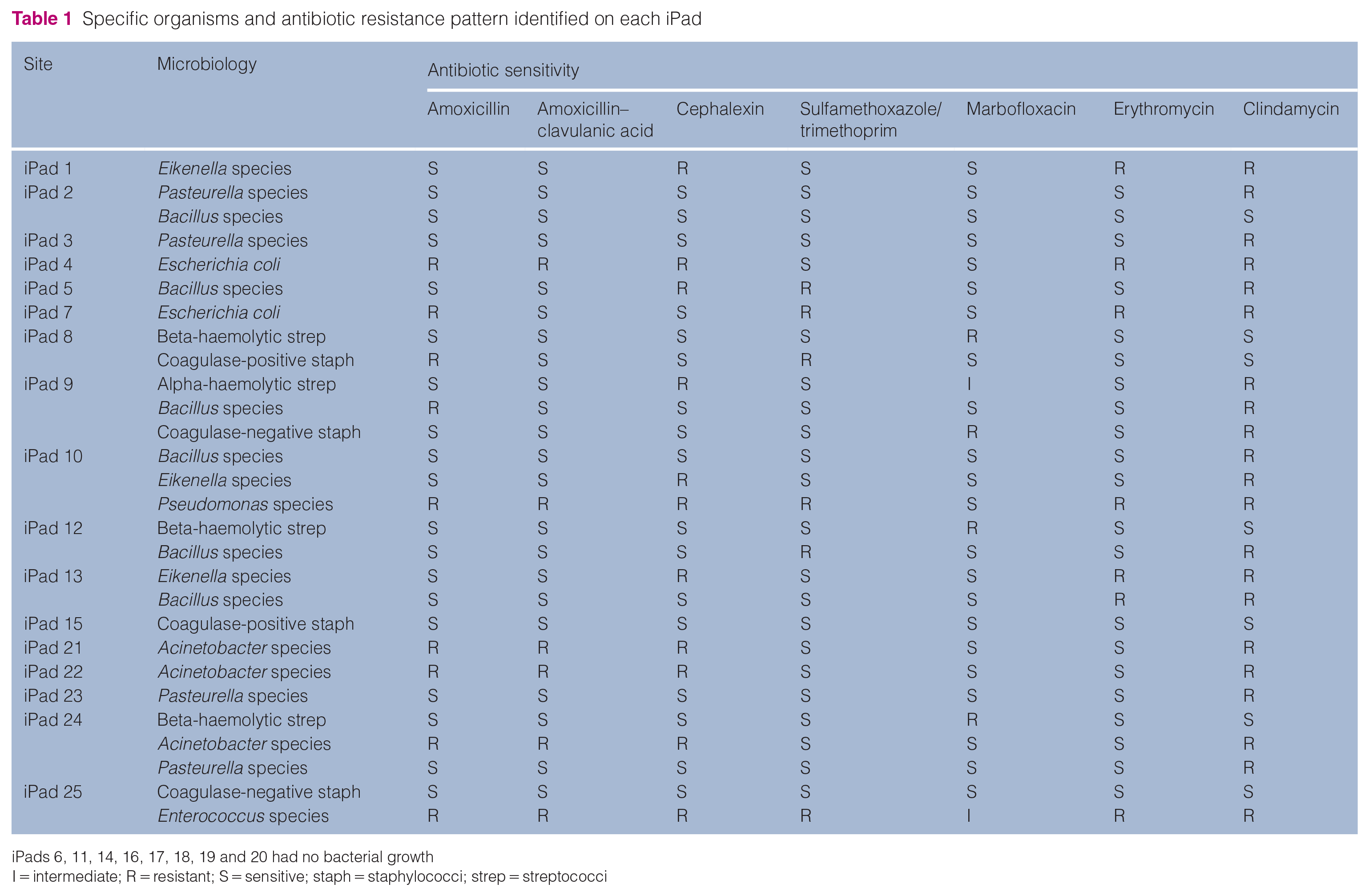
iPads 6, 11, 14, 16, 17, 18, 19 and 20 had no bacterial growth
I = intermediate; R = resistant; S = sensitive; staph = staphylococci; strep = streptococci
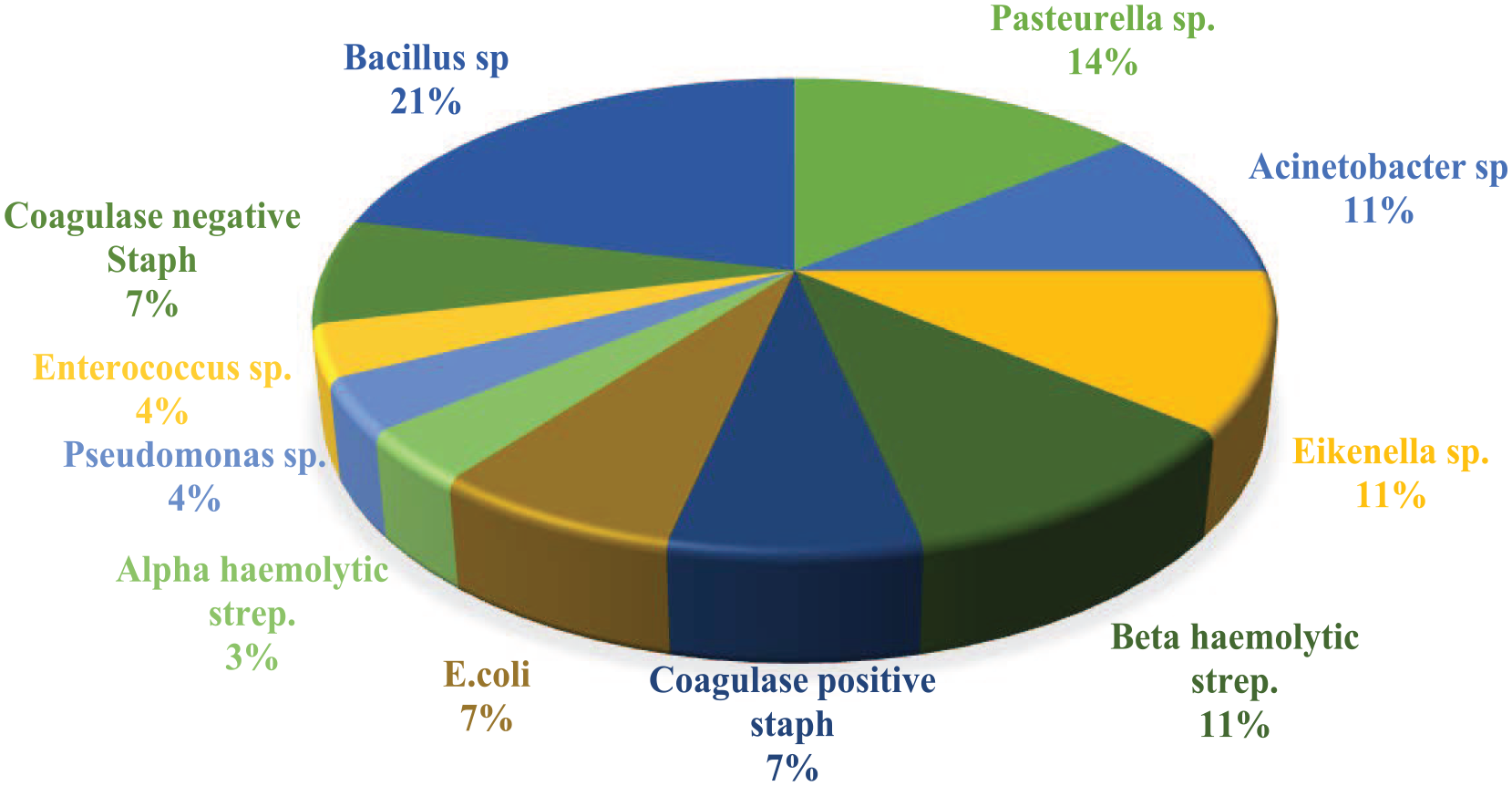
Bacterial frequency in samples collected from iPads. Sp = species; staph = staphylococci; strep = streptococci
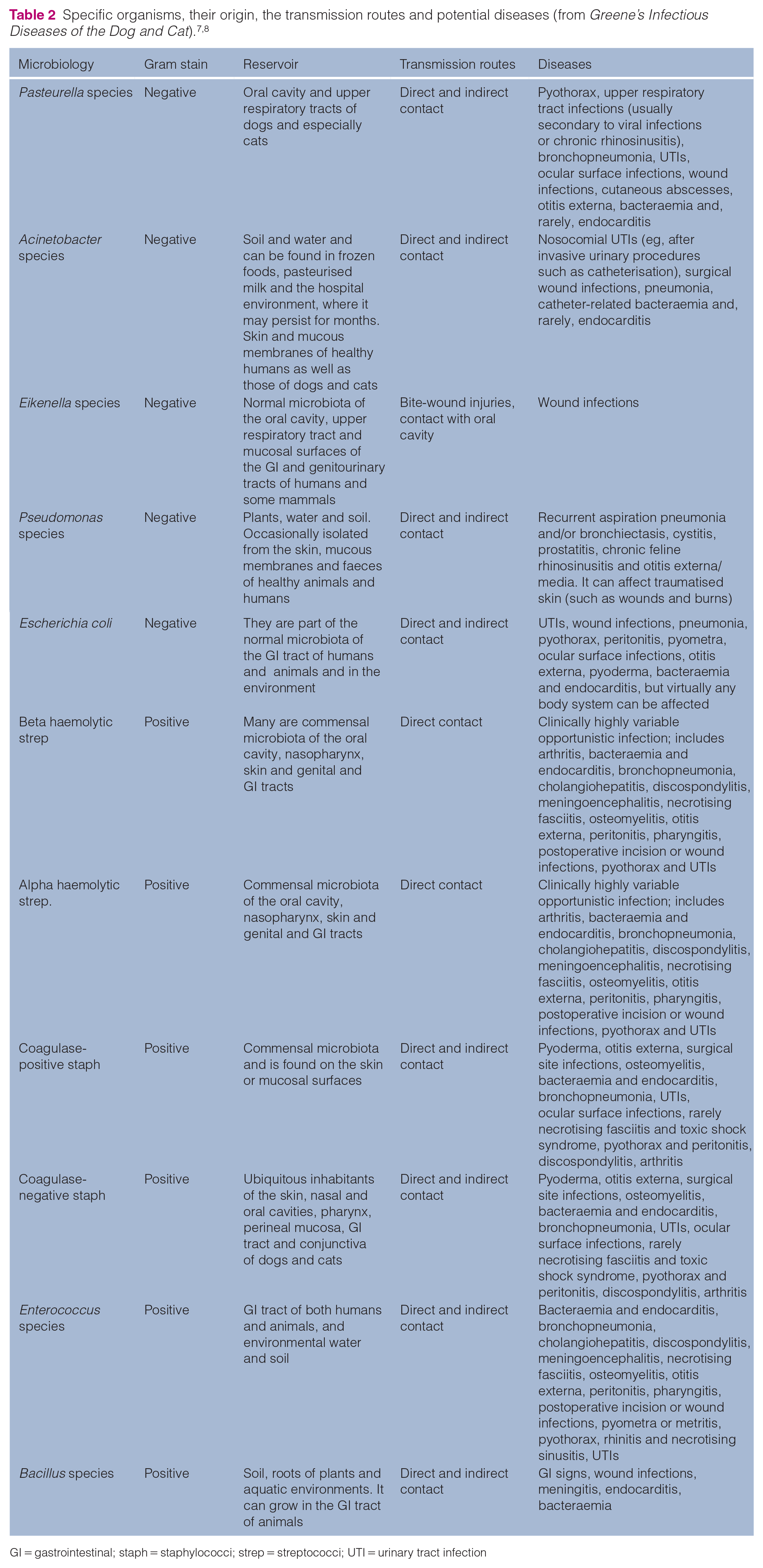
Of the 11 isolates, five were Gram-negative bacteria and six were Gram-positive with the potential to cause disease. A summary of the organisms isolated, their origin, the transmission routes and potential diseases are listed in Table 2.
GI = gastrointestinal; staph = staphylococci; strep = streptococci; UTI = urinary tract infection
In this study, 89% of the isolated bacteria were resistant to at least one of the tested antibiotics. Pseudomonas and Enterococcus species were resistant to all the tested antibiotics, with the exception of marbofloxacin. Of our isolates, 21 (75%) were resistant to clindamycin (Figure 2).

Frequency of antimicrobial-resistant bacteria isolated from iPads
After disinfection with the disinfectant wipes, elimination of microbial contamination was observed. Disinfection using 70% alcohol and 2% chlorhexidine wipes was completely effective at removing microbes from the iPads. There was no growth from any of the devices at 5 mins, 6 h and 12 h after cleaning, despite normal use within the hospital.
Discussion
Veterinary health professionals are increasingly switching to paperless recording systems to detail patient monitoring and treatment records. PSEDs are a common replacement for the countless charts, files and forms in every hospital, many of which then have to be scanned into a digital format. Routinely, PSEDs are in contact with the fingertips, and intermittent handling of those devices during and between procedures is a source of recontamination and does indirectly reduce the effectiveness of hand washing. Humans constitute the largest reservoir of the Staphylococcus species as part of the normal human flora of the skin and mucous membrane. These microorganisms can spread through direct contact with inanimate surfaces and may cause infections in patients with critical clinical conditions. 9
The main agents of nosocomial infections are Gram-positive cocci (staphylococci and enterococci), members of the Enterobacteriaceae, and non-fermentative Gram-negative bacilli (Acinetobacter species and Pseudomonas species). 10 Gram-negative bacteria are ubiquitous agents of opportunistic infections in dogs and cats and primarily cause disease when host defences are impaired. 8 The mode of transmission is by direct contact or contact with contaminated fomites. Like Gram-positive cocci, some Gram-negative bacteria have the ability to form biofilms, which form on living or non-living surfaces. These protect the bacteria, and organisms in biofilms can resist the effect of antimicrobial drugs and disinfectants. 8
Recommendations for iPad cleaning are limited to cleaning with a lint-free cloth (recommended by Apple); however, this was proven insufficient after contamination with common pathologic bacteria in a previous study performed by Howell et al. 4
Increasing rates of multi-drug-resistant surgical site infections in our institution led to the question of whether the iPads could be colonised with pathogenic organisms. These infections can cause significant morbidity and may pose a large financial burden on the clients and the institution.
The threat of the spreading of isolated microorganisms is valid, since 17 (68%) devices evaluated in this study showed bacterial contamination. In this study, common skin flora such as coagulase-negative staphylococci were collected from two (8%) iPads; nevertheless, it is reasonable to consider coagulase-negative staphylococci as potentially pathogenic and able to cause disease of various body sites, particularly in hospitalised and otherwise compromised animals. 7 Worryingly, problematic nosocomial pathogens such as Pasteurella species, Acinetobacter species, Eikenella species, beta-haemolytic streptococci, coagulase-positive staphylococci, E coli, alpha-haemolytic streptococci, Enterococcus species and Pseudomonas species were also isolated.
Health-workers’ hands are the main pathway of germ transmission; therefore, hand hygiene is the most important measure for avoiding cross-contamination. The goal of effective hand hygiene is to reduce the number of microorganisms on the hands, but more specifically the number of microorganisms that are part of the transient microflora of the skin, as these include the majority of opportunistic pathogens on the hands. 11 The World Health Organization (WHO) hand hygiene guidelines state that hand hygiene should be performed at five key moments: before touching a patient; before a clean/aseptic procedure; after body fluid exposure risk; after touching a patient; and after touching patient surroundings. 11 At the authors’ institution (MZ/TR), washing facilities are readily available as an addition to alcohol-based handrub dispensers in all wards and departments. There are also WHO posters on ‘How to Handrub’ and ‘How to Handwash’ at washing stations as a reminder. However, sharing portable equipment such as PSEDs between patients carried into the patient zone presents additional risks, with constant movement across different areas of the hospital; therefore, regular and effective disinfection of electronic devices should be stressed to help prevent the spread of nosocomial pathogens.
Restricting the use of the PSEDs is not realistic; in fact, the benefits of those electronic devices are to improve quality healthcare services – being able to store patient information digitally on a mobile device helps veterinary staff to work more efficiently, as the information is easily accessible. 12 However, veterinary health professionals should be aware that such devices can contain and convey harmful microorganisms, which can be disseminated both in and outside the hospital environment.
Chlorhexidine gluconate is a cationic bisbiguanide compound that is known to be an effective antimicrobial agent, with a residual effect on health workers’ hands, 13 which also helps to prevent bacterial colonisation and biofilm formation. 14 In this study, we aimed to investigate the residual effect of Sani-Cloth CHG 2% wipes. A previous study evaluated the residual effect of Sani-Cloth CHG 2% wipes on laryngoscope handles. 15 It concluded that this product conferred additional advantages over routine autoclaving or the use of disposable handles owing to a powerful residual antimicrobial effect. Moreover, the product also appears to have antiviral properties and is cheap and convenient. 15
The authors’ hospital guidelines for the cleaning of iPads changed after the study. It is now recommended to clean all iPads every 12 h on a scheduled basis (at the beginning of the day and night team shift), between users and after witnessed contamination. Each iPad should be thoroughly wiped for approximately 10 s with the Sani-Cloth CHG 2% wipe and allowed to air dry. If the patient is known, or suspected, to be infectious (eg, nurse barrier patient), the iPad (as well as other necessary equipment) should be kept within the isolation area and only be used on the affected patient. This is a cautious approach to minimise the risk of infection control breakdowns if contamination between uses has occurred.
The present study has some limitations. Our sample size is modest, and we focused only on the iPad screen and buttons, although the whole unit, including charging accessories, has the potential for contamination. Potential physical effects of the cleaning agent on the screen of the device were also not considered.
Conclusions
A variety of nosocomial pathogens, including antibiotic-resistant pathogens were isolated from the iPads. Infection prevention strategies should be employed in a hospital setting; however, this should not replace good hand hygiene techniques to help prevent the spread of nosocomial pathogens.
Footnotes
Acknowledgements
The authors gratefully acknowledge the contribution of SYNLAB, the Veterinary Pathology Group (VPG) for assistance with the laboratory work. The authors would also like to express their deepest appreciation to Dr M Lopez Martin, Biophysics Unit, School of Medicine, Autonomous University of Barcelona, for his generous advice regarding this paper.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research is sponsored by Research Fund, IVC Evidensia.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.