Abstract
Objectives
This research aimed to evaluate the performance of a closed blood collection system and to compare it with an open system in terms of feasibility, tolerability by the donor, quality of blood collected and bacterial contamination.
Methods
Eight feline blood donors were prospectively and randomly subjected to both collection methods. Heart rate (HR), respiratory rate (RR) and blood pressure (BP) were evaluated before sedation, after sedation and after blood collection. The duration of the donation, the formation of a hematoma, and the degree of hemolysis and packed cell volume (PCV) of each blood unit were evaluated. Aliquot samples were aseptically collected from each unit and tested for bacterial contamination by culture and PCR on days 0, 14 and 28 of storage.
Results
There was no significant difference between collection methods for HR and RR at any time point. Before sedation, the mean systolic BP was significantly higher with the closed system (closed 169 mmHg, open 137 mmHg; P = 0.003). The average duration of collection was significantly shorter with the closed system (closed 3 mins 10 s, open 8 mins; P = 0.035); however, the prevalence of a successful blood collection with a single venipuncture and hematoma formation were not significantly different between systems. The mean unit PCV was significantly higher with the open system (closed 31%, open 34%; P = 0.026). On bacterial culture, 15/16 units were negative at all time points (closed 7; open 8). Using PCR, 5/16 units were positive for Ralstonia species for at least one time point (closed 3; open 2).
Conclusions and relevance
Our designed closed system appears to be well adapted to feline blood collection and was well tolerated by the donors, performing similarly to an open system, and could represent a valuable clinical device for the development of a feline blood bank, namely feline blood storage.
Introduction
The use of blood products has become essential in the supportive care of critically ill cats, in which the main indications of transfusion are blood loss anemia and erythropoiesis deficiency.1–3 Because of the small amount of blood collected and the lack of commercial closed collection systems, an open system composed of multistep manipulation of syringes is usually employed.2,4–7 Each point of attachment provides an opportunity for contamination. Therefore, the blood collection procedure in cats is considered to have a higher risk of contamination than in dogs or humans, precluding storage of feline blood products. Two reports have described the spontaneous contamination of feline whole blood units by Serratia and Pseudomonas species.8,9 This bacterial contamination could cause an immediate reaction and even fatal endotoxin-related shock in the recipient, although clinical cases in feline patients have not been described. 10
One previous report mentioned the use of a feline closed collection system that was not commercially available. 11 Another recent study evaluated the in vitro quality of feline blood stored for 35 days in a new commercially available closed collection system designed for cats and concluded that the system was appropriate for blood collection. 12 However, no published study has compared the feasibility and safety of an open system with a closed system.
The objectives of this study were to compare two whole blood collection systems in cats (closed system and open system) in terms of feasibility, tolerability by the donor, quality of the blood unit and bacterial contamination. We hypothesized that both systems would be identical for each criterion, except for the risk of bacterial contamination, which would be reduced with the closed system.
Materials and methods
Animals
Eight healthy adult domestic shorthair cats with blood type A (weight 4.5–5.9 kg, age 2–6 years) that belonged to the blood donor colony (Faculté de médecine vétérinaire, Université de Montréal) were enrolled. All cats had a normal physical examination and echocardiography before entering the colony. They tested negative for feline immunodeficiency virus, feline leukemia virus (SNAP FIV/FeLV Combo; IDEXX Laboratories), Mycoplasma haemofelis, Bartonella species, ‘Candidatus Mycoplasma haemominutum’, ‘Candidatus Mycoplasma turicensis’, Cytauxzoon felis, Anaplasma and Ehrlichia species (PCR testing, IDEXX Reference Laboratories, Canada) and were up to date on vaccinations and deworming, according to the current recommendations. 13 Each cat was subjected to both collection methods performed by a single phlebotomist, the order of which was attributed randomly. All blood collections were performed by the same phlebotomist, a health technician trained in good blood collection practices, in the presence of the same veterinarian, a small animal internal medicine resident.This study was approved by the Ethical Committee of the Faculté de médecine vétérinaire of the Université de Montréal (CEUA-Rech-927).
Blood collection procedures
Prior to sedation and blood collection, packed cell volume (PCV) and total solids were evaluated on blood collected from a peripheral venipuncture. All donors were sedated with ketamine (4 mg/kg) and diazepam (0.25 mg/kg) administered in the cephalic vein. All cats were placed in lateral recumbency and a jugular vein was punctured. Duration of the donation was recorded, from the time of venipuncture for blood collection until completion of the collection. Serial physical examinations, including heart rate (HR), respiratory rate (RR) and blood pressure (BP) measured with an oscillometric monitor (PetMAP graphic, Ramsey Medical), were performed by the same veterinarian prior to sedation (presedation), immediately after sedation (post-sedation) and after blood collection (post-collection). The site of venipuncture was evaluated post-donation for the presence of a hematoma or of active bleeding.
Blood collection systems
For the open system, 56 ml of blood was collected via a 19 G butterfly needle connected by a three-way valve to two 30 ml syringes prefilled under aseptic conditions in a laminar flow hood with citrate phosphate dextrose adenine (CPDA) anticoagulant extracted from a 450 ml single unit for human blood collection (Terumo Medical) in a ratio of CPDA:blood of 1:7.2,6,7,14,15 Eighteen syringes (eight for the study and 10 for the functioning of our blood bank) were prepared at the same time using two different human blood collection bags.
To create a closed system, a 150 ml single pediatric bag (transfer pack; Fenwal) was aseptically welded (TSCD-II apparatus; Terumo Medical) to the same type of human bags used for the open system, in a laminar flow hood. Eight milliliters of CPDA were transferred to the pediatric bag in order to obtain the same ratio of CPDA:blood, after which the bag was welded to a single 19 G apheresis needle (Fistula Needle Set; JMS). The final product (Figure 1) was sealed into a bag for storage (Safe-Seal; Medicom) and stored in a cupboard in the blood bank room. A volume of 56 ml of blood (weighing 59 g for a blood specific gravity of 1.053) was collected by suction using a vacuum chamber (Animal Blood Resources International) placed on a digital scale. Negative pressure of the vacuum was set at –1 inHg.

(a) The final closed system before sealing into the bag for storage. (b) The closed system in its storage bag
Samples and analysis of whole blood
For the open system, 6 ml blood from the syringes were aseptically transferred to a collection line (Baxter extension set with two interlink injection sites). For the closed system, the blood in the bag was gently mixed and the collection line was filled with blood. Three aliquots of 2 ml and one aliquot of 0.3 ml were produced on collection lines for both systems using an electric thermal sealer (Hematron 3; Baxter). All blood units collected during the study were transfused to anemic feline patients, and only aliquots were analyzed at all time points.
PCV (measured on a microhematocrit tube after centrifugation) was assessed immediately postdonation on the 0.3 ml aliquot, and the degree of hemolysis was visually graded according to the following scale: absent (ie, plasma is nearly transparent [0]); mild (ie, plasma is light red [1+]); moderate (ie, plasma is red [2+]); and severe (ie, color of the plasma cannot be visually differentiated from red blood cells [3+]). Bacterial contamination was assessed by testing aliquots by culture and by conventional PCR on days 0 (T0 at room temperature), 14 (T14 at 5°C ± 3°C) and 28 (T28 at 5°C ± 3°C). These time points represent the beginning, the middle and the end of the shelf life of the feline whole blood bag. 16 Three aliquots of 2 ml from one unused specimen of each collection system were analyzed at T0, T14 and T28 (‘closed and open system negative controls’). Three aliquots from the collection line of a 450 ml human single bag containing CPDA were analyzed at T0, T14 and T28 (‘CPDA negative control’). The CPDA bag used to perform the CPDA negative control was obtained from the same lot as the bags used to produce the closed systems.
At the time of the analysis, the blood contained in the aliquot was aseptically transferred into an empty sterile tube in a laminar flow hood to facilitate manipulation. Aerobic and anaerobic cultures were performed by inoculation of 500 µl of blood on Columbia agar with 5% sheep blood (Difco) incubated at 35 ± 2°C with 5% carbon dioxide, and 500 µl on Tryptone soya agar (Oxoid) with 5% sheep blood and gentamycin sulfate (Enzo) incubated at 35 ± 2°C in anaerobic conditions. Also, 10 µl of blood was added to 5 ml of brain heart infusion broth (Bacto) and incubated at 35 ± 2°C. All plates were examined after 18–24 h and 48 h of incubation. The incubation time for the negative plates was prolonged for up to 5 days. Identification of growing bacteria was performed using matrix-assisted laser desorption/ionization–time of flight mass spectrometry.
DNA was extracted from a 500 µl aliquot of blood (Biosprint 96 One-For-All Vet Kit; Qiagen) according to the manufacturer’s instructions. PCR was performed with universal primers for 16S rRNA gene of bacteria. Amplification was performed in a T Professional Basic Biometra thermocycler (Biotech). Negative (sterile water) and positive (known 16S rDNA of Listeria species) controls were included in every set of PCR. PCR products were detected by 1.5% agarose gel electrophoresis, and bacteria were identified by sequencing the PCR product (Sanger method) and comparing it with a genetic sequence database (GenBank NCBI).
Analysis and statistics
A mixed model, in which collection method and time (presedation, post-sedation, post-collection) were included as fixed effects and the subject was included as a random effect, was used to compare HR, RR and BP between methods. Similarly, a mixed model, in which the collection method was included as a fixed effect and the subject was included as a random effect, was used to compare the duration of collection and donor PCV (presedation) and blood unit PCV. A McNemar test was used to compare the prevalence of success with a single venipuncture, the prevalence of hematoma formation and the degree of hemolysis between both collection methods. Statistical analyses were performed using SAS version 9.4 (SAS Institute). Results were considered to be statistically significant when P <0.05.
Results
Patients
All cats had been enrolled in the feline blood donor colony for a minimum of 6 months (median 13.5 months [range 6–24 months]) and had participated in an average of 5.7 donations (range 1–10 donations) before entering the study. The median duration between collections for the purpose of the study was 5 months (range 2–7 months) and cats had participated in 0–2 donations ‘outside’ of the study during the study period.
Assessing the impact of each method on the donor
No significant differences in mean HR and RR were found between collection methods for each given time point. Before sedation, mean systolic BP was significantly higher (169 mmHg) with the closed system (open system: 137 mmHg; P = 0.003), but no significant difference was noted at other time points (Table 1). The prevalence of hematoma was not statistically different between collection methods (closed: 12.5% [n = 1]; open: 25% [n = 2]; P = 0.13). Of the three cats that developed hematomas, two underwent repeated phlebotomy. The average duration of collection for these three cats was 12 mins 40 s. The average duration of collection with the closed system (3 mins 10 s) was significantly shorter than with the open system (8 mins; P = 0.035). The prevalence of success with a single venipuncture was not significantly different between collection systems (closed: 87.5%; open: 75% [P = 1]) (Table 2).
Clinical parameters measured in eight feline donors using closed and open collection systems before sedation, after sedation and after collection
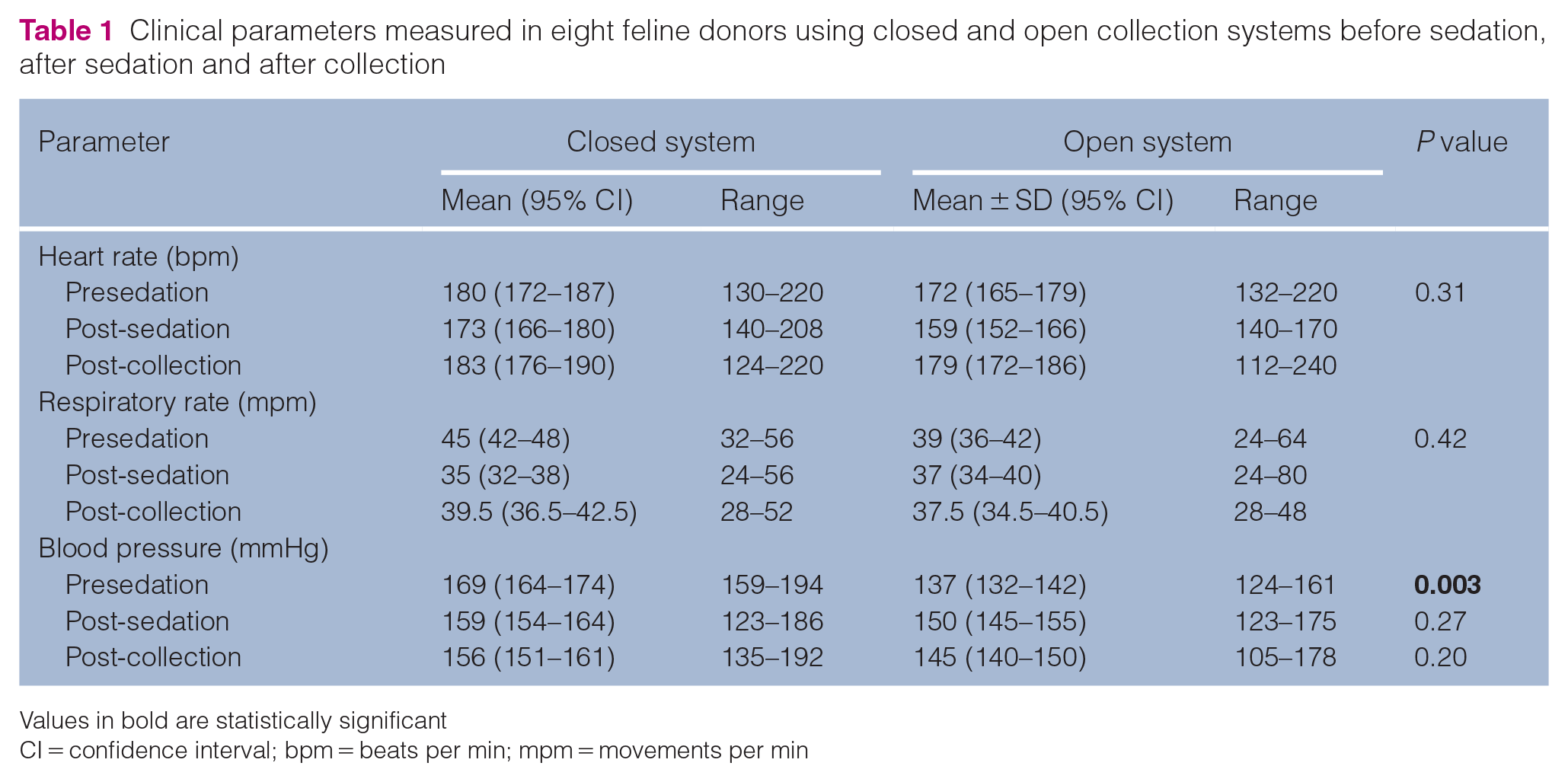
Values in bold are statistically significant
CI = confidence interval; bpm = beats per min; mpm = movements per min
Collection parameters in eight feline donors using closed and open collection systems
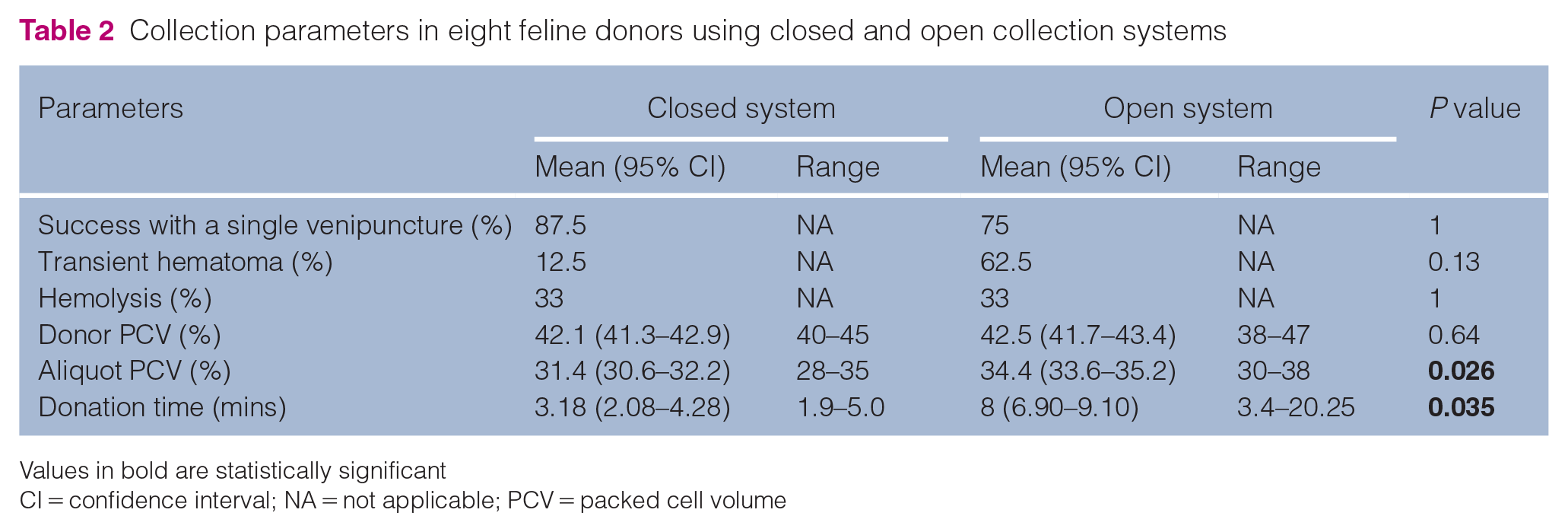
Values in bold are statistically significant
CI = confidence interval; NA = not applicable; PCV = packed cell volume
Quality of the blood product
The degree of hemolysis of all samples was either 0 or 1+. The prevalence of mild hemolysis (1+) was not significantly different between collection methods (P = 1). The mean donor PCV prior to donation was identical for both collection methods (42%; range 38–47%), which was significantly higher than the mean PCV of the aliquots for the two systems combined (33%; range 28–38% [P <0.0001]). The mean unit PCV was significantly higher (34%) with the open system compared with the closed system (31%; P = 0.026).
Bacterial culture and PCR
Fifteen of the 16 (94%) blood units and all the ‘open and closed system negative controls’ and ‘CPDA negative control’ aliquots were negative for bacterial culture at all the time points. One unit collected with the closed system was positive for Staphylococcus species after enrichment at T0 but negative at all other time points. Five units were only positive with PCR (n = 5/16; 31%), including 4/5 units positive at T0 but negative at all other time points. Two ‘closed system negative controls’ aliquots (T0 and T28) and one ‘CPDA negative control’ aliquot (T0) were also positive. All of the detected bacteria by PCR were Ralstonia species (Table 3). Blood collected by the open system was transfused the day of donation. Blood collected by the closed system was transfused after 0–19 days of storage (mean 8 days). All blood transfusions were cross-matched compatible and closely monitored, and were uneventful.
Bacterial culture (all time points combined) and PCR results (at days 0 [T0], 14 [T14] and 28 [T28] of storage) for the samples with at least one positive result

Discussion
Considering the small blood volume collected from cats, the use of a closed collection system for donation is rare, and the collection of feline blood has been performed mainly via open systems.2,4–7,12,17 For this study, we decided to design a closed system not composed of syringes, but rather one that looks like human and canine standard closed donation systems, in order to avoid points of attachment that can be disconnected, which could compromise the sterility of the blood product. Our goal was not to commercialize this system; as it is easy to produce, its availability is only dependent on the ability to invest in welder apparatus.
Overall, blood collection using both systems was well tolerated by the donors, as demonstrated by the lack of significant variation of the studied clinical parameters (HR, RR, BP) immediately after sedation and donation for both methods. Most importantly, no episode of hypotension was observed with all BPs maintained at >100 mmHg. These results are in accordance with those of other reports showing that, despite a variety of sedation protocols, blood donation is well tolerated by cats.5,18,19
Despite the same environment before sedation for both methods, the mean systolic BP was unexpectedly higher with the closed system. It is possible that the phlebotomist was more stressed when manipulating the newly designed closed system, which was perceived by the donor. Stress behavior parameters were not evaluated to reinforce this hypothesis.
The mean duration of blood collection with the open system (8 mins) was comparable to what has been previously reported.17,18 No study has published the duration required to collect feline blood with a closed system using a vacuum chamber. A study conducted in dogs reported a time of collection by suction of 4 mins 14 s, which is comparable to the time measured in our study. 20 The average duration of collection by the closed system was significantly shorter than by the open system, and it remained well tolerated by our donors. A shorter duration is a real advantage of the closed system because it ensures that cats are manipulated when sedation is still active, which is ideal to reduce stress and discomfort.
The prevalence of hematoma following blood donation was previously reported to be 4.2–7.9% in dogs and 11% in cats.5,20,21 The overall prevalence of transient hematoma was higher in this study (18.7%), but the prevalence of hematomas with the closed system (12.5%) was very close to the previously reported value in cats. A correlation with the duration of collection and the number of venipunctures was suspected, as 2/3 cats with hematomas underwent repeated phlebotomy, although this was not statistically significant owing to the sample size. Collection duration, repeated phlebotomy, size of the needle and pressure of the blood aspiration could be studied as risk factors for the development of venipuncture-related adverse events.
The mean unit PCV was significantly lower than the presedation PCV of the blood donors, which is in accordance with other reports showing that the PCV of feline whole blood units varies between 26.1% and 27.8%, while those of feline donors are >30%.6,7,12 First, this difference can be partly explained by a dilution effect of CPDA in the collection bag/syringe. 7 Second, sequestration of erythrocytes in the spleen and shifting of fluid from the extravascular to the intravascular compartment due to the anesthetic protocol could be considered.22,23 To support an effect of our sedation protocol on our donors’ PCV, it would have been interesting to measure their PCV after sedation and before collection. The mean unit PCV from the open system was also significantly higher (34%) than that of the closed system (31%), which was nonetheless adequate.6,7,12 As the sedation protocol was the same and the amount of hemolysis was not significantly different between methods, the difference is more likely related to the volume of collected blood. Indeed, when using the closed system, the blood collection was stopped when approximately 59 g of blood was collected, whereas with the open system the collection stopped when 56 ml were precisely measured. This point is a slight disadvantage of the closed system and could be improved by adjusting our CPDA:blood ratio and our assessment of the volume collected with the scale.
For collection with the closed system, we decided to use a vacuum chamber to mimic the protocol of blood collection commonly used in dogs. Negative pressure was set to the lowest value; therefore, it is likely that our closed system could work without using a vacuum chamber. A previous study conducted in dogs showed no significant difference between gravity and suction collections for blood donors’ clinical parameters, the prevalence of hematoma formation, the hemolysis index and the hematocrit of units. However, suction collection was faster than gravity collection. 20 A similar study could be conducted in cats to evaluate if collection by gravity is feasible, and how it compares with suction collection.
In our study, the percentage of negative bacteriological units was high (94%), which is similar to the 96–100% observed in previous studies on feline and canine blood products.12,24–26 Staphylococcus species were the only isolated bacteria. We attribute this growth to a contamination during the laboratory procedures, that is, the manipulations for microbiological evaluation described in the materials and methods section, because the sample was negative by PCR at all time points.
The percentage of units and aliquots that were positive by PCR was moderate (31% and 25%, respectively), and comparable between both systems. The detection of bacterial DNA in blood with PCR has been described in only a few veterinary studies.11,26 In one study, a PCR assay was performed on days 0, 21 and 35 on stored canine whole blood units and 53% of blood units were positive at one time point. 26 Interestingly, in the current study, four units were positive at T0 and negative thereafter. The failure to identify the bacterium at subsequent timepoints may be attributable to the residual phagocytic activity of blood leukocytes. 27 Furthermore, the level of contamination at the time of collection is estimated to be <1–10 colony forming units/ml in human blood products.28,29 Initially, the low bacterial load of the units was perhaps not uniformly shared between aliquots with some aliquots containing too few bacteria to be positive. This hypothesis could explain why one unit was positive at T0 and T28 but negative at T14. These results and explanations are only possible if the microorganisms identified were dead or degraded, so that the bacteria were already dead at T0 or that the refrigeration temperature killed the bacteria after T0 or inhibited their growth. It would have been interesting to use a real-time PCR to quantify the bacterial load of the aliquots.
All bacteria identified by PCR were, unexpectedly, Ralstonia species. In previous studies, contamination by Serratia and Pseudomona species has been described in feline whole blood and packed red blood cell units.9,11 Ralstonia species are aerobic, non-fermenting, Gram-negative rods that can be found in water and soil. 30 In human patients with impaired immunocompetence, Ralstonia species have been shown to be the causative agent of invasive infections, including osteomyelitis, septic arthritis and meningitis. One veterinary case report described a septicemia due to Ralstonia pickettii in a dog with immune-mediated thrombocytopenia. 31 Ralstonia species have never been described in transfusion-related morbidities in human medicine or in veterinary medicine.It was not possible to determine the actual contamination source of these Ralstonia species, but we hypothesize that the contaminants were introduced during the processing of the units because they were already present at T0 for all positive units and the PCR assays were positive in 2/3 ‘closed system negative control’ aliquots. We do not believe that the contamination was related to inadequate asepsis during the production of our closed system for the following reasons: first, 1/3 ‘CPDA negative control’ aliquots were positive at T0; second, three aliquots from units collected with the open system were also Ralstonia species-positive. The hypothesis of contaminated CPDA units is considered likely because many cases of human infection were due to contaminated medical solutions, with the contamination supposedly occurring at the manufacturing stage.32–35 ‘Open system negative control’ aliquots were negative at all the time points, but it was not possible to compare the lot number of the CPDA units used to produce every system because the syringes of the open system were prefilled with anticoagulant months before the beginning of the study, and the lot number was not recorded at this time.
Alternatively, bacterial DNA contamination during PCR analysis at the laboratory is possible but seems less likely because negative and positive controls were undertaken at every step of the analysis. Furthermore, of 95 conventional PCR assays performed during the study period, Ralstonia species were detected in 9/57 samples from our study and 1/38 sample outside of our study (P = 0.045).
The clinical relevance of these PCR positive results is likely not significant because most of the positive units were negative after T0. Ralstonia species grow easily on conventional media, which led us to suspect that the bacteria were dead/degraded. Finally, all units were transfused to feline recipients at our veterinary hospital without detection of any transfusion reactions despite close monitoring.
One limitation of our study is that all blood parameters were evaluated on segments from the collection line. This method was previously described for human and canine blood units,26,36,37 but further studies are needed to assess the reliability of using segments as a proxy for evaluating quality parameters and bacterial contamination of feline blood units. For ethical reasons, the blood was used for actual anemic feline patients, which limited the volume of our aliquots to <10% of the total volume collected. If aliquots of a larger volume had been used, semi-quantitative measurement of the degree of hemolysis could have been assessed, as well as other quality tests.We also chose a duration of storage of 28 days because it represented the average of the two extremes of feline whole blood unit shelf life found in the literature (21–35 days), but the optimal shelf life of whole feline blood stored in CPDA is unknown.2,9,12,38 Recent studies have evaluated in vitro indicators of red blood cell survival after storage, which cannot accurately predict how a given sample of stored red blood cells will survive in a recipient.7,12,25,39 An in vivo evaluation where the stored cells are labeled and followed in the circulation is essential to meet the current US Food and Drug Administration requirements for stored human whole blood, which states that at least 75% of transfused red blood cells remain in circulation 24 h after transfusion.40 The objectives of our study were not to assess the storage lesions and shelf life of stored feline whole blood unit, but with the development of closed collection systems for cats, further studies are required to standardize the expiry of feline blood products.Contrary to one of the hypotheses, our study did not observe any difference in bacterial contamination between the two collection systems. However, it is important to note that collection conditions were optimized with, among others, an animal health technician trained in good blood collection practices. These conditions do not necessarily reflect the reality of all feline blood collection. Besides, the contamination was studied on a small number of blood units and further studies on a more significant number of cases is necessary. In addition, further research on the actual clinical relevance of positive PCR results on blood units are needed to assess the utility of using this test for evaluating bacterial contamination of blood products.
Conclusions
Our designed closed system appears to be as appropriate as an open system for blood collection of feline donors and should be considered as an alternative to open collection systems to limit the multistep manipulation of syringes. This closed system permits blood collection by suction using a vacuum chamber, which accelerated the process without being detrimental to the blood donor, therefore optimizing collection. Finally, this system could represent a valuable clinical device for the development of a feline blood bank.
Footnotes
Acknowledgements
The authors would like to thank Dr Guy Beauchamp for his assistance with statistical evaluation of the data.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was supported by the Fonds du Centenaire research grant (in-house grant, FC2018), as well as by the Fonds en Santé des Animaux de Compagnie research grant, supported by Zoetis (FSAC2018).
Ethical approval
This work involved the use of experimental animals; or involved the use of non-experimental animal(s) (owned or unowned) outside of established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
