Abstract
Objectives
The aim of this study was to report the incidence of transfusion reactions in cats, including acute haemolysis (AH), occurring within 24 h of receiving a xenotransfusion. An additional aim was to determine whether cases with AH could be classified as having an acute haemolytic transfusion reaction (AHTR) as per the definition provided by the Association of Veterinary Haematology and Transfusion Medicine’s Transfusion Reaction Small Animal Consensus Statement.
Methods
Medical records of cats that received canine packed red blood cells (PRBCs) between July 2018 and September 2020 at a veterinary hospital were reviewed. The incidence of AH, AHTRs, febrile non-haemolytic transfusion reactions (FNHTRs), transfusion-associated circulatory overload and septic transfusion reactions were recorded.
Results
The medical records of 53 cats were retrospectively evaluated. Twenty-three (43%) cats had transfusion reactions. Thirteen (25%) cats had AH; however, only four (8%) met the definition of an AHTR. Ten (19%) cats were determined to have FNHTRs. Survival to discharge of cats affected by AH was 50% (25% for cases that met the definition of an AHTR). Survival to discharge of cats not suffering from AHTR was 40%.
Conclusions and relevance
This report indicates that a higher proportion of cats undergo AH (25%) when administered canine PRBCs than previously reported, although many could not be classed as having an AHTR due to an apparently adequate packed cell volume rise. Challenges with sourcing feline blood in emergency situations occasionally necessitates the use of xenotransfusion in transfusion medicine. Clinicians should be aware that haemolysis after xenotransfusion can occur within 24 h and that a repeat feline transfusion may be required sooner than anticipated in some cases.
Introduction
Xenotransfusion (the administration of blood products from one species to another) is occasionally used in the emergency setting in veterinary medicine and the administration of canine products to cats is well described in the literature.1–5 This practice is utilised owing to the challenges that exist in feline blood banking, including the sourcing of sufficient donors. 6 Several studies have demonstrated both the effectiveness and safety of xenotransfusions in cats with first-time use. However, it is well described that fatal transfusion reactions are likely if cats receive more than one xenotransfusion.2,3,7,8
Because the repeated exposure of cats to canine blood can result in a fatal outcome, the use of canine blood in feline transfusion medicine is largely avoided. However, in recent years several reports have described the relative safety of one-time administration of canine blood to cats.3,4,8 Delayed haemolytic transfusion reactions are expected to occur in the subsequent days as the feline recipient develops antibodies to the canine red blood cells. However, acute haemolytic transfusion reactions (AHTRs; ie, those occurring with 24 h) on the first administration of canine packed red blood cells (PRBCs) to cats have not been described. 3
The aims of this study were to describe the incidence of all acute transfusion reactions, including acute haemolysis (AH) in cats that received a first-time xenotransfusion, and to determine whether any AH noted was consistent with an AHTR, as defined by recent consensus guidelines. 9 A further aim was to determine whether previous feline blood transfusions increased the relative risk of developing an AHTR when cats received a xenotransfusion. Our hypothesis was that AHTRs occur more commonly in this population of cats than previously reported.
Materials and methods
This study was a retrospective review of the clinical records of cats that had received canine PRBCs between July 2018 and September 2020 at a veterinary teaching hospital. Cats were included if they were anaemic and had received a xenotransfusion between these dates. Electronic and paper records were searched and analysed, and where more than one xenotransfusion was performed, only the records of the first transfusion were reviewed. The recipients’ signalment, cause of anaemia, blood type, volume of canine PBRCs administered (ml/kg), pretransfusion packed cell volume (PCV) within 24 h of transfusion, post-transfusion PCV (including subsequent PCVs over a number of days, where available), total bilirubin (Tbil) pre- and post-transfusion (when available) and survival to discharge were recorded. The transfusion records were also reviewed for evidence of transfusion-related reactions and complications.
For the purposes of this study AH was defined as an increase in Tbil concentration of at least 2.5% from baseline, a level which was higher than that explicable by within-run imprecision with the institution’s in-house analyser. 10 The second Tbil level also needed to be above the reference interval (>5 µmol/l). If cases had new-onset haemoglobinaemia (detected by visual inspection of recipient plasma through centrifugation), haemoglobinuria, bilirubinuria (detected visually or by dipstick) or if new ghost cells were seen on blood film evaluation, they were included in the AH group. These signs of haemolysis needed to be seen within a 24 h period to be ‘acute’. Where increases in Tbil were used to determine if AH had occurred, the patient’s underlying disease was examined as a potential cause. If the diagnostic work-up (clinical signs, imaging and laboratory findings) suggested underlying hepatic, biliary or pancreatic disease, and provided an explanation for the acute increase in bilirubin, the case was not included as one with possible AH.
Following recent consensus Transfusion Reaction Small Animal Consensus Statement (TRACS) guidelines, the diagnostic criteria for an AHTR was AH that occurred within 24 h of the transfusion starting, where an inadequate PCV increase was also seen. 11 Any case that was suspected to have AH also had its expected PCV post-transfusion calculated, to assess for a possible AHTR. An expected PCV post-transfusion was calculated using the formula 2 ml/kg PRBC × body weight, which will result in a 1% PCV increase from baseline. 12 An inadequate PCV increase was defined as a post-transfusion PCV that did not reach the expected PCV as per this formula minus 0.35%, as this was the mean negative bias reported for this formula. 12 The expected post-transfusion PCV was calculated using PCVs taken up to 24 h after the end of the transfusion. This allowed enough time for cats with AH to reveal if their post-transfusion PCV would later fall after an initial expected increase in PCV, and therefore establish whether they fitted the diagnostic criteria for having an AHTR.
Physical examination parameters consistent with transfusion reactions were recorded, including a body temperature increase of ⩾1°C from baseline or an increase to ⩾39.2°C, tachycardia and hypotension, although these were not necessary to meet the definition of an AHTR. Other clinical signs were noted during the transfusion, such as cardiovascular or respiratory parameters and whether the transfusion was discontinued. Where a transfusion reaction or complication occurred but was not suspected to be haemolytic, the type of event was determined from the transfusion record and the clinician’s notes. Classifications of reactions or complications that were not haemolytic were febrile non-haemolytic transfusion reaction (FNHTR), septic transfusion complication and transfusion-associated circulatory overload (TACO). FNHTRs, TACO and septic transfusion reaction were defined as per recent guidelines. 11
The institution where the study took place follows the Association of Veterinary Haematology and Transfusion Medicine’s TRACS guidelines when a recipient’s body temperature increases by more than 1°C during a transfusion. 13 Cases were excluded if inadequate transfusion recording was available to determine if a reaction had occurred.
Statistics
Data were assessed for normality using the Shapiro–Wilk test. Normally distributed data were presented as a mean ± SD. Non-normally distributed data were presented as a median (range). Comparison of means was performed using a paired Student’s t-test. Comparison of medians was performed using a Mann–Whitney U-test. The relative risk for developing an AHTR was calculated for cases that had received previous feline transfusions.
Results
Population
Fifty-six xenotransfusions were administered between July 2018 and September 2020 to 55 cats. One cat was excluded due to inadequate information in the transfusion record. One cat had two xenotransfusions administered within 24 h and only information from the first transfusion was evaluated, and one cat had a normal pretransfusion PCV. The result was that 53 xenotransfusions were included in the study. Twenty-nine of the recipients were male castrated cats, 20 were female spayed, three were female entire and one was male entire. Thirty-three cases were domestic shorthairs, six were British Shorthairs, five were domestic longhairs, two were Siamese, two were Bengal, two were crossbreeds, one was a Ragdoll, one was a Persian and one was a Scottish Fold. The median age of the cats was 7.6 years (range 0.2–16.2).
The most common reasons for a transfusion included blood loss (n = 17; 32%), immune-mediated haemolytic anaemia (n = 11; 21%), neoplasia (n = 8; 15%) and infectious causes (n = 7; 13%). Blood loss was either due to trauma (n = 5/17; 29%), gastrointestinal loss (n = 4/17; 24%), urogenital loss (n = 3/17; 18%), spontaneous haemabdomen (n = 4/17; 24%) or surgical loss (n = 1/17; 5.9%). Infections included feline infectious peritonitis (n = 4/7; 57%), feline immunodeficiency virus (n = 1/7; 14%) and feline leukaemia virus (n = 2/7; 29%). Blood dyscrasias accounted for 11% of cases of anaemia (n = 6/53): erythroid leukaemia (n = 2/6; 33%), erythroid hypoplasia (n = 1/6; 17%), toxicity (n = 1/6; 17%), red cell aplasia (n = 1/6; 17%) and myeloid leukaemia (n = 1/6; 17%). The remaining causes of anaemia were hepatobiliary disease (n = 2/53; 4%) and ectoparasites (n = 1/53; 2%), with one undetermined cause (n = 1/53; 2%). Some cats had concurrent neutropenia and/or thrombocytopenia with the anaemia. Three cats had concurrent neutropenia and thrombocytopenia, three cats had concurrent thrombocytopaenia only and one cat had concurrent neutropenia.
Blood tests
Fifty-two of 53 cats were blood-typed prior to transfusing. Thirty-four cats were of blood type A (n = 34; 65%), 19 cats were of type B (36%) and no cats were of blood type AB. One cat was not blood-typed prior to starting the xenotransfusion, owing to difficulty obtaining blood and patient instability. Canine donor blood type was available for 52/53 transfusions performed. Thirty-four (64%) xenotransfusions were performed using dog erythrocyte antigen 1 (DEA-1)-postive PRBCs and 18 (34%) were performed using DEA-1-negative PRBCs. The mean total volume of xenotransfusion administered was 51.9 ml and 14.4 ml/kg, respectively. This included cases that had their transfusion discontinued. Transfusions were stopped in four cases prior to completion, owing to concerns over AH (Table 1). In one case, a transfusion was stopped due to a suspected FNHTR (Figure 1).
Clinical signs during transfusion, how evidence of haemolysis was identified and outcomes of cats with suspected acute haemolysis (AH)

Met the criteria for an acute haemolytic transfusion reaction
PCV = packed cell volume; HR = heart rate; bpm = beats/min; Tbil = total bilirubin; TACO = transfusion-associated circulatory overload

Pretransfusion packed cell volume (PCV) and post-transfusion PCV in (a) cats that were suspected to have acute haemolysis (AH) vs (b) cats that did not have AH. Cats in (a) that fitted the criteria for acute haemolytic transfusion reactions (AHTRs) are marked by a triangle
Pretransfusion PCV was measured in 51 cases. The median pretransfusion PCV within 24 h prior to xenotransfusion was 11% (range 6–18). The median post-transfusion PCV performed within 24 h (available in 38 cases) was 21% (range 9–42). In the cats that did not suffer from AH, the median pretransfusion PCV (n = 38) was 11% (range 6–18) and the median post-transfusion PCV (n = 25) was 22% (range 11–42). In the 13 cats that had AH, the median pretransfusion PCV was 12% (range 7–17) and the median post-transfusion PCV (n = 12) was 21% (range 9–34). The mean post-transfusion PCV was not significantly different when comparing cats that had AH with cats that did not (P = 0.6033).
Tbil was measured within 24 h before the transfusion in 51 cases. The median pretransfusion Tbil was 6 µmol/l (range 0–258). Post-transfusion Tbil (within 24 h) was measured in 28 cases. The median post-transfusion Tbil was 26.9 µmol/l (range 0–341). In cats that were not suspected of having AH where Tbil was measured (n = 37), the median pretransfusion Tbil was 5.15 µmol/l (range 0–258) and the median post-transfusion Tbil was 12 µmol/l (range 0–341). Eight cats were placed in this group despite having acute increases in Tbil (which were >2.5% above baseline and above the reference interval) during their transfusion because they had underlying diseases that made it difficult to rule out an aetiology other than haemolysis (Figure 2). These eight cats were found to have large hepatic masses (n = 2), diffuse infiltrative neoplasia (n = 1), intrahepatic cholestasis (n = 1), extrahepatic cholestasis and pancreatitis (n = 1), infectious cholangiohepatitis (n = 1), vacuolar hepatopathy with biliary hyperplasia and cholestasis (n = 1) and neutrophilic hepatitis (n = 1; Figure 2). In cats that had AH, the median pretransfusion Tbil was 6.7 (range 0–42) and the median post-transfusion Tbil was 43.7 (range 7–182). The smallest percentage increase in bilirubin from baseline in AH was 40%. There was no difference in volume in ml/kg of transfusion administered between cats that had AH and cats that did not (P = 0.3241).
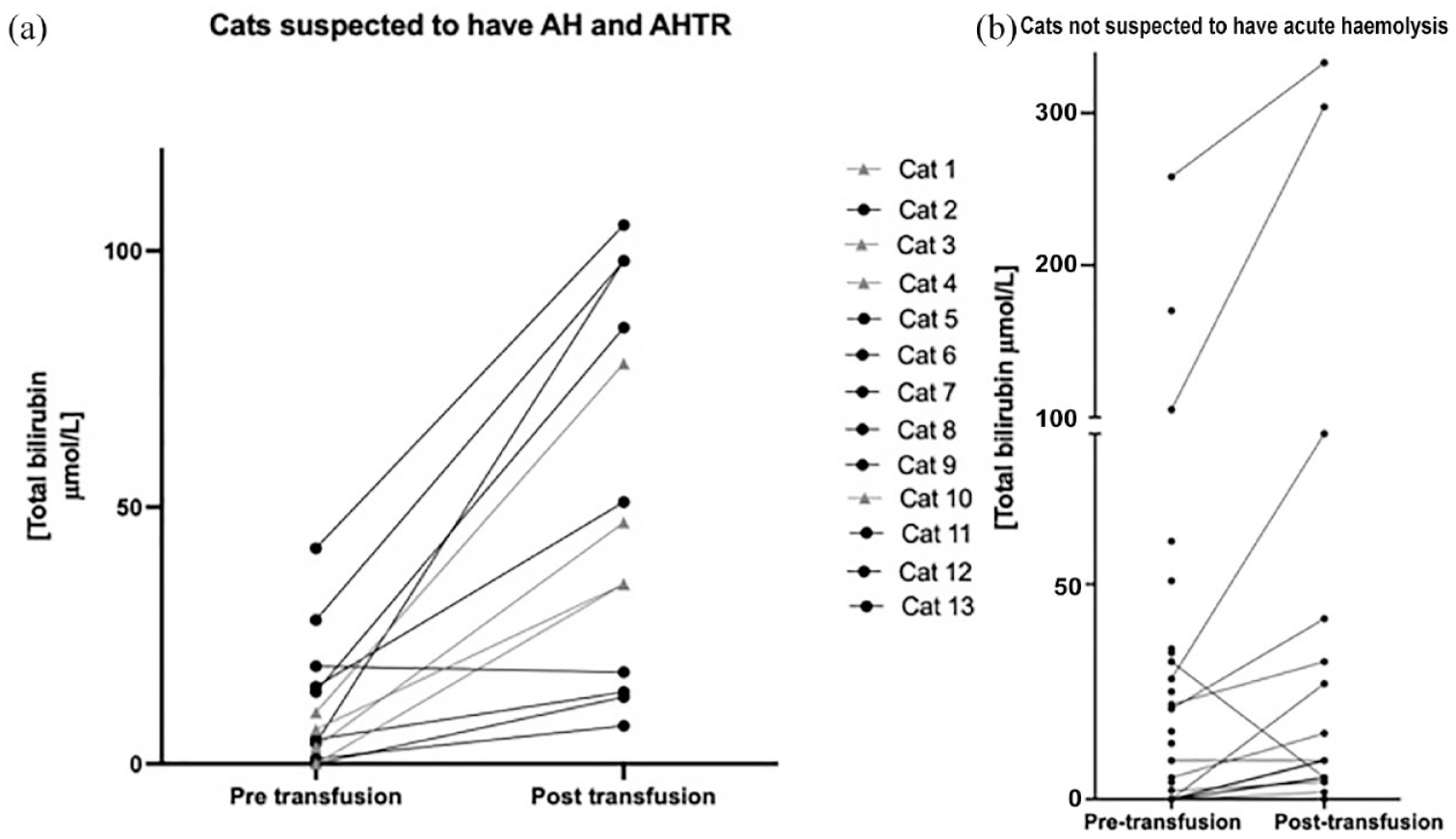
Pretransfusion total bilirubin (Tbil) and post-transfusion Tbil in (a) cats that were suspected to have acute haemolysis (AH) and (b) cats that did not have AH. Cats in (a) that fitted the criteria for acute haemolytic transfusion reactions (AHTRs) are marked by a triangle. Note that cats in (b) that had peritransfusion increases in Tbil were not included with the AH cases because of the presence of underlying disease precluding this (eg, hepatic/biliary disease)
Nine cats had received feline blood transfusions prior to receiving a xenotransfusion. Of these nine cats, five had AH post-xenotransfusion. These cats are included in Table 1 as cats 1, 4, 8, 9 and 12. Four of five cats that developed AH and had received a feline blood transfusion prior to receiving the xenotransfusion had received their feline transfusion 5–18 days prior to the xenotransfusion. One cat had the feline transfusion only 24 h before the xenotransfusion, which was due to it suffering from a bleeding diathesis and haemorrhage. The relative risk of developing AH when having a xenotransfusion following a feline transfusion was higher than if a xenotransfusion was given with no prior feline blood exposure (3.13; 95% confidence interval [CI] 1.32–7.37).
Seven of the 40 (18%) cats not suffering from AH needed feline blood post-xenotransfusion. The mean number of days to feline transfusion post-xenotransfusion in the non-AH group was 2.7 days (range 1–4). Three of 13 (23%) cats that suffered from AH required repeat transfusion with feline blood, and the mean number of days until repeat transfusion was 1.3 days (range 1–2).
Twenty-two (42%) cats had transfusion-related reactions or complications within 24 h of administering the xenotransfusion. Thirteen (25%) cats suffered from suspected AH, four of which were diagnosed with an AHTR (8%). Of the 13 cats that had AH, nine were blood type A and four were blood type B. Ten (19%) cats were determined to have FNHTRs. Two cats were suspected to have TACO (new-onset respiratory signs, increase in left atrium size and B-lines on lung ultrasound noted). These cats also each had an AHTR and an FNHTR, and were administered 19.91 ml/kg and 7.54 ml/kg canine PRBCs over 18 and 4 h, respectively. No septic complications or transfusion-associated lung injury or other acute transfusion reactions were seen in this patient population.
Outcome
Twenty-three of the 53 (43%) cats survived to discharge and 30 did not survive (57%). Of the cats that did not survive, 27 were euthanased (51%) and three died in hospital (6%). Of the 13 cats that had AH, six survived to discharge (46%) and seven did not survive (54%). Of the non-survivors, five were euthanased (71%) and two died of natural causes (29%). Seventeen of 40 (42%) cats that did not have AH survived to discharge. AH conferred a relative risk of death (non-survival to discharge) of 0.91 (95% CI 0.26–3.23) compared with those not suffering an AH. Three cats that underwent AH went on to have a feline unit within 2 days (Table 1).
Discussion
Haemolytic transfusion reactions can be either acute or delayed. Delayed haemolytic transfusion reactions are caused by the formation of antibodies to red blood cell antigens after a transfusion has occurred, and occur between 24 h and 28 days post-transfusion. 9 Delayed haemolytic reactions are a known consequence of transfusing canine blood to feline recipients and have been previously reported to occur at a median of 2 days post-transfusion and, at the earliest, 1 day. 3 AHTRs occur within 24 h as a result of a type II hypersensitivity (where preformed antibodies target erythrocyte antigens). 14 These reactions can result in intravascular or extravascular haemolysis, can manifest in a range of clinical signs and have not previously been reported in cats receiving dog blood. 14 Early experimental studies in cats indicated that AHTRs do not occur in first-time administration of canine PRBCs, 7 suggesting that preformed antibodies to canine RBC antigens do not exist in cats. However, recent studies have shown incompatible cross-match testing between canine and feline blood when there was no known previous exposure of the cats to canine blood.3,15,16 Major cross-match testing of canine RBCs and feline plasma in a benchtop study found that agglutination with or without haemolysis occurred 57.6% of the time, indicating a high risk of severe AHTRs when transfusing canine blood to cats. 16 Furthermore, another study showed major cross-match incompatibilities between canine donor and feline recipients in 69% of cases. 3 Preformed antibodies to feline erythrocytes may also exist in the canine donor plasma, which is plausible given that minor incompatibilities were reported in 30% of cross-matches in one study. 3 Despite this, clinical studies have not described AH when canine blood is administered to cats.3,15 The reason for clinical studies not documenting AH with xenotransfusions could be because this blood product is normally reserved for severe cases when alternatives are not available. It is possible that more critically ill patients lack the robust immunological response to reveal the reactions clinically. With the retrospective nature of this study and the heterogeneous population of cats, it would be difficult to determine whether the AH seen was related to the nature or severity of the underlying disease. A further prospective study would be needed to stratify patients into groups by severity of illness and look at the incidence of AH to determine this.
Our study identified that 13/53 (25%) xenotransfusions resulted in AH. The incidence of AH in this study was high, but far fewer cases (n = 4/13) could be classified as having an AHTR, according to consensus guidelines. An explanation for this could be that AHTRs manifest with a range of severities, with milder reactions being harder to identify. The retrospective nature of this study meant that the pre- and post-transfusion PCV sampling was not performed at set time points. This could have affected our expectations for the post-transfusion PCV, meaning that an ‘inadequate rise in PCV’ could have been missed if the PCV was lower than recorded at the time point when the xenotransfusion was started. In addition, the formula selected for calculation of expected post-transfusion PCV in cats was found to be the most accurate in a study assessing the administration of allogenic feline blood transfusions and it is unknown which formula would be most suitable for a xenotransfusion. Interestingly, the expected PCVs of several cats using this formula were lower than their actual post-transfusion PCVs. This indicates that the formula for allogenic blood use could underestimate the actual PCV rise, meaning the true expected post-transfusion PCV value could be higher with a xenotransfusion than when calculated for feline transfusions. While this is speculation on the accuracy of the formula for this setting, if the formula does underestimate expected post-transfusion PCVs for xenotransfusions, then more cases in this study would fit the definition of an AHTR. The Centre for Disease Control (CDC) has various clinical criteria to make a definitive, probable and possible diagnosis of an AHTR in humans. 17 A definitive diagnosis can be made when a combination of any new-onset signs, including chills, backpain, gross visual haemolysis (eg, pigmenturia), hypotension, epistaxis, renal failure and two or more of the following are noted: decreased fibrinogen, decreased haptoglobin, elevated bilirubin, elevated lactate dehydrogenase, haemoglobinaemia, haemoglobinuria, plasma discolouration with haemolysis and spherocytes on blood film. Based on this definition, more cases of AH could have been classified as AHTR as the requirement for an inadequate rise in PCV is not present. Evidence of cross-match incompatibility can also be used to aid diagnosis of an AHTR. 13 Compatibility testing was not performed after the haemolysis was noted – a potential limitation of our study. Routine cross-matching of feline and canine blood for xenotransfusion was not performed as the administration of major incompatible blood has been previously reported to not result in AHTR. 3
An increase in body temperature of >1°C was noted in over half of the cats with AH (n = 8/13; 62%). However, cats 2, 6, 7, 10 and 11 exhibited AH without being coupled with the body temperature change (Table 1). Nevertheless, a temperature increase may have been masked in these cases, with cats 2 and 10 already being pyrexic at the start of their transfusion, cat 6 (which became icteric during the transfusion) was under general anaesthesia, and cats 7 and 11 were cardiovascularly unstable. In addition, reluctance to handle and cause stress to a critically unstable patient and patient temperament reduced the overall number of rectal temperatures measured.3,13
A possible explanation for the increased rate of haemolysis noted in this study vs previous studies could be that AHTRs and FNHTRs have a wide spectrum of presentations. The CDC describes different severities for both AHTRs and FNHTRs as non-severe, severe, life-threatening and death. 17 It is possible that mild AHTRs could be misclassified as FNHTRs if thorough investigations are not performed or are not possible due to patient factors. Measurement of bilirubin levels as performed in the current study could be a more sensitive method of detecting AH than visual inspection of spun serum in PCV tubes, observation of clinical icterus or pigmenturia, and might allow us to capture more cases with early haemolysis.
Ten cats in our study had FNHTRs (19%). This is slightly higher than in a previous study, where 10% of cats were affected by FNHTR. 3 The proportion of true FNHTRs can be overestimated in retrospective studies as it is harder to ascertain if an increase in body temperature is due to improved cardiovascular stability because of the transfusion or other factors, such as active warming. This report does not suggest that there is an increase in incidence of TACO in cats receiving xenotransfusions as only two cats showed signs consistent with this condition (4% of cases), and the incidence of TACO in cats receiving feline PRBC transfusions is reported to be 3%. 9
It is difficult to interpret survival numbers in this study because of the influence of euthanasia and the underlying morbidities resulting in the need for a transfusion. Clinicians may also be more likely to administer a xenotransfusion when the prognosis is already considered to be guarded. The survival to discharge rate was 43% in this patient population, similar to that recorded in a previous study that showed an overall survival rate in cases receiving a xenotransfusion of 37%. 3 Interestingly, the survival rate for cases suspected of having an AH (46%) was similar to those that did not have haemolysis documented (42%). In cases defined as AHTR, where an inadequate increase in PCV was seen, there was a lower survival rate (25%). With small numbers in this group, it is difficult to know whether the need for further transfusion affected this as 50% of the AHTR cats were euthanased. A lower percentage of cats in the AH group received repeat transfusions with feline blood. It is counterintuitive that fewer cats in the AH group went on to receive repeat feline transfusions, as we would expect them to have a higher requirement for this. It is possible that the need for repeat transfusion within a shorter period of time (1.3 days vs 2.7 days for the AH group) promotes a perception of poorer prognosis, resulting in discontinuation of treatment, although the survival rate was not lower.
The limitations of this study include the retrospective design, meaning recognition of transfusion reactions and AH relied on good clinical record-keeping, and consistent and regular monitoring. Cats did not have standardised times for checks for haemolysis, and we relied on transfusion monitoring sheets to evaluate clinical signs during the transfusions. Changes in the clinical picture of these patients could therefore have been missed. We suspected AH in many cases based on an acute increase in Tbil (or a new-onset icterus). Owing to the criteria for inclusion of haemolysis not being more stringent and including other additional markers of haemolysis in conjunction with this (such as new ghost cells on blood smear), it is possible that the acute rises in bilirubin were a result of another aetiology. In addition, not all cats had post-transfusion PCVs performed within 24 h, and although we used several modalities to assess for AH (ie, an increase in bilirubin, new-onset evidence of haemoglobinuria, bilirubinuria or new ghost cells on blood film evaluation), it is possible that some cases with AH were missed owing to bloodwork not being repeated. However, it is unlikely that the clinician would not have repeated such bloodwork if they had been suspicious of the transfusion being haemolysed. The definition of AHTRs used was any evidence of haemolysis noted within 24 h, along with an inadequate increase in PCV. Because of the retrospective design, although attempts to rule out other causes of inadequate PCV rise were made, other causes of a poor response such as blood loss or non-immunological causes of haemolysis could not be completely ruled out. With the knowledge of AH potentially occurring more frequently than previously estimated, an understanding of whether this has any consequences on renal parameters would be an avenue for further study. Previous studies have documented that azotaemia is a frequent change in cats receiving xenotransfusion. 3
Conclusions
This study revealed that AH should be a clinical concern for cats receiving xenotransfusion, as the incidence is higher than previously reported. The knowledge that haemolysis after a xenotransfusion can occur sooner than previously recognised allows clinicians to plan more effectively for repeat transfusions. This report raises concerns over the administration of canine blood to feline recipients as AH may not be only seen upon a second exposure. However, the relative risk of not surviving to discharge was not higher in patients undergoing AH. To this end, xenotransfusions can still be considered a life-saving method of stabilisation when feline blood products are not available.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental animals only (including owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional consent for publication was not performed.
