Abstract
Case series summary
Urinary bladder masses in four cats were treated with palliative radiation therapy (RT). Three cats were previously diagnosed with chronic kidney disease (CKD): International Renal Interest Society (IRIS) stage 2 in two cats and IRIS stage 3 in one cat. One cat had a diagnosis of hyperthyroidism and inflammatory bowel disease. Three cats had urinary tract infections diagnosed by urine culture and susceptibility testing prior to or during treatment. All patients had urine cytospin cytology performed; one case showed suspect urothelial carcinoma and three had no cytological evidence of neoplasia. All clients declined further sampling from the bladder masses. Therefore, cytologic/histologic diagnosis in all cases was not available. An abdominal ultrasound was performed in all cats, which revealed urinary bladder mass(es) prior to referral for RT. Three cats had pretreatment thoracic radiographs, which revealed no evidence of pulmonary metastasis. An abdominal CT was performed in all cases and one case had thoracic CT performed for staging. The thoracic CT showed a focal lesion of unknown etiology in the right caudal lung lobe. Palliative RT was performed with four weekly 6 Gy fractions (24 Gy in total). The urinary signs in all cats resolved over the course of RT: after the first RT treatment in two cats and after the second RT treatment in two cats. There were two Veterinary Radiation Therapy Oncology Group grade 1 gastrointestinal and one grade 2 genitourinary acute RT side effects.
Relevance and novel information
This is the first report in the literature of a standardized RT protocol as a treatment option for feline urinary bladder masses.
Introduction
Feline transitional cell carcinomas (TCCs) are uncommon, with a reported incidence of 0.18–0.38% in a large study. 1 The most common feline lower urinary tract tumors include TCCs, but others such as squamous cell carcinoma, adenocarcinoma, lymphoma, rhabdomyosarcoma, fibrosarcoma and cystadenoma have been reported.2–4 In a recent large study of urinary tract TCCs in cats, the locations of the bladder tumors were the trigone (27.1%), ventral bladder wall (23.7%), bladder apex (17%), dorsal bladder wall (13.6%) and multifocal/diffuse (8.5%); 11.9% had urethral involvement and 11.9% had ureteral obstruction. In this study, 7% of cats presented with urethral obstruction and 5.1% had evidence of both urethral and ureteral involvement. 5 Based on the aforementioned study, the trigone appears to be the most common location of feline TCC, which limits local treatment options. Clinical signs are often urinary signs affecting the quality of life (QoL) of patients.5,6
One of the most important components in the management of bladder tumors is clinical signs associated with the tumor, and therefore local therapy is important. 5 A previous study suggested surgical excision and non-steroidal anti-inflammatory drugs (NSAIDs) should be the primary treatment. In this study, radiation therapy (RT) was performed in three cats and 2/3 experienced improvement in clinical signs, but RT protocols differed and detailed information on these protocols was unavailable. 5 Two studies have evaluated RT for the treatment of canine bladder tumors and showed promising outcomes.7,8 However, intrapelvic irradiation may be associated with severe late RT side effects such as chronic colitis, cystitis, rectal perforations and strictures in 19–56% of cases.7,9,10 Previous studies of RT in dogs with genitourinary carcinoma showed relatively high rates of grade 3 Veterinary Radiation Therapy Oncology Group (VRTOG) late gastrointestinal and genitourinary side effects such as ureter obstruction and rectal stricture formation in 19% of cases.7,11 These papers suggested using a smaller radiation fraction size of <3 Gy, and smaller radiotherapy fields to minimize late RT side effects.RT-induced side effects following pelvic irradiation in cats have not been reported.
Therefore, the aim of this case series was to report the clinical outcomes and adverse events of a standardized RT protocol for feline bladder masses.
Case series description
Signalment, clinical signs and staging examination findings
Four cats with bladder masses were treated with palliative RT at our institution from March 2021 to August 2021. The mean age was 15 years (range 13–16). Three cats were spayed females and one was a castrated male. Three cats were domestic shorthairs and one was a Siamese. All cats presented to their primary care veterinarian for urinary signs, including: hematuria in all cases; stranguria in two cases; pollakiuria in one case; and inappropriate urination in one case. All cases had chronic comorbidities: International Renal Interest Society (IRIS) stage 2 chronic kidney disease (CKD) in two cases; stage 3 in one case; and hyperthyroidism and inflammatory bowel disease (IBD) in one case. The cat with hyperthyroidism and IBD had been receiving oral methimazole (0.71 mg/kg PO q12h) and prednisolone (0.7 mg/kg PO q24h) chronically prior to referral. All cats had an abdominal ultrasound performed by their primary care veterinarian, which revealed mass(es) in the urinary bladder or thickening of the bladder wall. The imaging report for the bladder mass(es) was available in 3/4 cats. The differential diagnoses from the reports were neoplasia such as TCC, and a benign disease process, including granuloma, polypoid cystitis, inflammatory cystitis and hematoma. Two cases had a description of the bladder mass(es) as isoechoic, and one was described as a lobular mass. There was no evidence of ureteral obstruction or bladder stones in any of the cases. Physical examination findings were not clinically significant upon presentation for all cases. Bloodwork was performed in all patients prior to and throughout RT. Two patients with IRIS stage 2 and one patient with stage 3 CKD had creatinine values ranging from 2.5 to 3.7 mg/dl (reference interval [RI] 0.8–2.4) before RT. The other cat did not have CKD (creatinine 1.2 mg/dl). Urinalysis was performed in all patients, which identified a urine specific gravity ranging from 1.011 to 1.029. No other clinically significant findings were noted in the complete blood count (CBC), chemistry panel and urinalysis in all cases. A urine culture and sensitivity (UCS) was performed in all cats at various time points: prior to referral in three cats and 3 weeks after RT in one cat. Three cats had evidence of a urinary tract infection (UTI) and received a course of oral antibiotics based on the UCS. An unknown type and dosage of oral antibiotic was prescribed by the primary care veterinarian 3.5 months before referral in one cat, but no improvement in lower urinary signs was observed. One cat was prescribed pradofloxacin (7.6 mg/kg q24h PO for 7 days) by the primary care veterinarian at the time of referral to our facility. The last cat with a UTI noted 3 weeks after RT is described in detail later.
All patients had urine cytospin cytology performed by clinical pathologists; one patient showed suspected urothelial carcinoma and three had no cytological evidence of neoplasia. The urine cytology of the cats where no neoplasia was evident was normal in one cat, inflammatory in one and consistent with hematuria in one. All clients declined further diagnostics for cytological or histological sampling of the bladder masses after discussion of additional diagnostics. Therefore, definitive diagnoses in all cases were not available.
Three cats had thoracic radiographs reviewed by a radiologist, which showed no detectable pulmonary metastasis. One cat had a thoracic CT reviewed by a radiologist, which revealed a 3 mm focal lesion of unknown etiology in the right caudal lung lobe. Differentials included early formation of a cavitary mass, focal fibrosis in an area of a bulla or emphysematous lesion. But follow-up evaluation of the lung lesion was not performed. For three other cats, additional thoracic CT was not elected by the clients.
All clients elected for palliative RT without further diagnostic tests or alternative treatments after discussion of additional diagnostic and treatment options, which included surgery and cystoscopy with biopsy.
Characteristics of bladder masses and other findings on abdominal CT
Abdominal CT was performed for RT planning and staging purposes using the same equipment in all cases concurrently (Somatom Confidence RT 64 Slice, 120 kVp, 0.5 mm slice thickness; Siemens Healthineers) (Figures 1–4). A contrast CT study was only performed in two cats; the other two cats were not given the intravenous (IV) contrast owing to their CKD. Iohexol (OMNIPAQUE 300) at a dose of 662 mgI/kg was used for contrast study in these cats. Locations of the bladder masses in each patient were multifocal lesions in the apex and dorsal wall (cat 1 [Figure 1a]), trigone (cat 2 [Figure 2a]), dorsal wall of the bladder neck (cat 3 [Figure 3a]) and the ventral wall (cat 4 [Figure 4]). No cases showed CT evidence of urinary tract obstruction by masses in the ureter or urethra. The median bladder mass size on CT in the cranial caudal, dorsoventral and mediolateral dimensions were 1.4 cm (range 1.0–1.9), 0.8 cm (range 0.5–2.8) and 1.3 cm (range 0.8–2.4), respectively. Other abdominal CT findings included a mildly enlarged left sublumbar lymph node in one cat, mineral foci in renal pelvis bilaterally in three cats and mild irregularity in the right kidney in one cat. There was no overt concern for the etiology of the enlarged sublumbar lymph node and fine needle aspiration was declined by the client.

(a) Non-contrast CT transverse view before radiotherapy (RT) in cat 1, showing the irregular shaped, hyperattenuating, space-occupying urinary bladder mass (arrows) with multiple foci of mineralization in the apex bladder wall. (b) Abdominal ultrasound sagittal view 21 weeks post-RT in cat 1, showing the multiple irregular-shaped, heterogeneously echogenic, space-occupying urinary bladder masses (arrows) newly extending to the trigone, which was defined as progressive disease

(a) Non-contrast CT transverse view before radiotherapy (RT) in cat 2, showing the dome-shaped, hyperattenuating urinary bladder mass (arrows) on the left lateral wall and extends to the trigone. (b) Abdominal ultrasound transverse view 8 weeks post-RT in cat 2, showing the irregular-shaped, heterogeneously echogenic urinary bladder mass (arrows) extending to the trigone

(a) Contrast CT transverse view before radiotherapy (RT) in cat 3, showing the irregular-shaped, markedly contrast enhanced urinary bladder mass (arrows) in the right dorsal wall. (b) Abdominal ultrasound transverse view 22 weeks post-RT in cat 3, showing no evidence of grossly visible urinary bladder mass
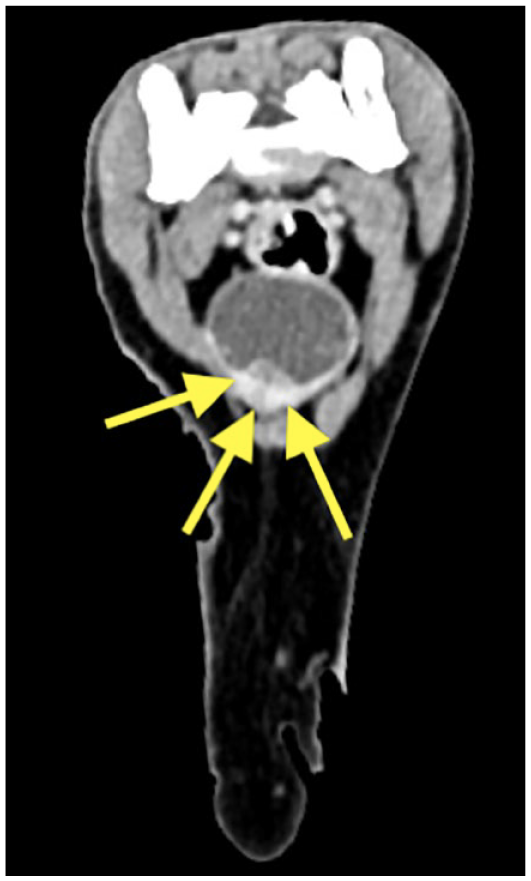
Contrast CT transverse view before radiotherapy in a cat 4, showing the irregular-shaped, heterogeneously contrast-enhanced urinary bladder mass (arrows) in the ventral wall. No follow-up abdominal ultrasound image was available
RT prescription, planning and delivery
Palliative RT was performed using 4 or 6 MV photons from a linear accelerator (Varian Edge). The radiation prescription consisted of four fractions of 6 Gy administered once weekly. During treatments, all cats were anesthetized and placed in lateral recumbency using a vac lock bag (Civco) as a positioning device. The gross tumor volume (GTV) was defined as a visible mass in the urinary bladder on CT and clinical target volume (CTV) was defined as GTV plus the whole urinary bladder and pelvic urethra on CT. The pelvic flexure and penile urethra were not included in the CTV in the male cat. The planning target volume (PTV) was defined as CTV plus 5 mm to account for positioning uncertainty. All treatment plans and contouring were performed on the Varian Eclipse Treatment Planning System (version 15.6). Plans were designed as parallel opposed pairs in all cases, with the goal of delivering 95% of the dose to 95% of the GTV. All plans were reviewed by a medical physicist prior to delivery and the dosimetry was verified by an independent calculation software (RadCalc; LifeLineSoftware). Verification of patient positioning was performed by either orthogonal kV images or cone beam CT prior to delivery of each fraction to position the patient. No management of urinary bladder size was attempted during simulation or treatment other than consistent early-morning treatment scheduling. The urinary bladder was matched with the PTV consistently in all cases using on board image guidance with minimal shifting required at each fraction.
All patients had an IV catheter placed, and induction of general anesthesia for each treatment was achieved using either propofol or alfaxalone. Maintenance of general anesthesia was performed using sevoflurane. Patients were monitored for blood pressure, capnography, cardiogram and pulse oximetry, and given IV fluids throughout treatments. There were no significant anesthetic events, including hypotension, during treatment in all cases.
Follow-up information during and after RT
Follow-up information was available in all cases with a mean follow-up time of 23 weeks (range 4–46) after completion of RT.
Prior to each RT fraction, chemistry panels were performed, which showed stable azotemia throughout RT in the three patients with CKD. There were no other significant findings in the chemistry panels in all cases throughout the course of treatment.
The urinary signs in all cats resolved over the course of RT as reported by the owner: after the first RT treatment in two cats and after the second RT treatment in two cats. This resolution of their lower urinary signs was characterized as back-to-normal urination and defecation habits in three cats, and normal frequency of urination in another cat. All cats also showed subjectively increased energy and appetite throughout the course of RT, based on conversations with clients at follow-up visits.
Cat 1 was prescribed a course of transmucosal buprenorphine (0.013 mg/kg q8–12h PO) at the beginning of RT until a week after its completion, as needed (as the clinician’s preference for prophylaxis), to alleviate the potential for pain from urethral muscle spasms secondary to acute RT toxicity. This cat was re-evaluated 6, 14 and 21 weeks after RT with abdominal ultrasound. However, a new, small lesion was found in the trigone that was categorized as progressive disease 14 weeks after RT. Further progressive disease was noted 21 weeks after RT owing to the increased size of multifocal bladder masses along with the trigone lesion in this cat (Figure 1b). There was no radiographic evidence of pulmonary metastasis 21 weeks after RT. This cat was doing well at home without urinary signs during follow-up. This cat developed mild constipation, which was suspected to be a grade 1 VRTOG gastrointestinal acute side effect 6 weeks after completion of RT; it resolved without further treatment. This cat was still alive and doing well at home 46 weeks after completion of RT, despite documented progression of the mass based on imaging.
Cat 2 was re-evaluated with abdominal ultrasound 8 weeks after RT, and showed stable disease without urinary signs with a normal appetite and energy level (Figure 2b). This cat died approximately 16 weeks after completion of RT. No further information, such as the cause of death, was available, as additional follow-up visits with our clinic were not elected by the owner after completion of RT.
Cat 3 developed straining to defecate, which was suspected to be a grade 1 VRTOG gastrointestinal acute side effect, and increased straining to urinate, which was suspected to be a grade 2 genitourinary acute side effect or UTI after the first RT treatment. This cat was prescribed amoxicillin–clavulanic acid (17.8 mg/kg PO q12h for 5 days) along with transmucosal buprenorphine (0.01 mg/kg q8–12h PO) for this clinical sign. Clinical signs of straining to urinate/defecate had resolved by the next RT fraction. Three weeks after completion of RT, this cat was hospitalized by a local emergency clinic owing to the acute onset of vomiting. Physical examination findings were an elevated rectal temperature of 105.2°F with 5% dehydration; CBC revealed neutrophilia (30,738/µl; RI 2500–8500) with toxic changes and the presence of bands (981/µl). A chemistry profile revealed mildly elevated albumin (4.0 g/dl) and blood urea nitrogen (42 mg/dl) with a creatinine of 2.2 mg/dl. Urinalysis revealed gross bacteria and UCS was positive for Escherichia coli and sensitive to amoxicillin–clavulanic acid. This cat received IV ampicillin–sulbactam (22 mg/kg q8h for 48 h), followed by a course of amoxicillin–clavulanic acid (13 mg/kg q12h PO for 14 days) along with IV dexamethasone sodium phosphate (0.1 mg/kg q24h) and a continuous rate infusion of metoclopramide at 2 mg/kg/day for 48 h. The cat responded to treatments and was discharged after 2 nights of hospitalization at the local emergency clinic. No UCS was repeated after this event. This cat was re-evaluated by abdominal ultrasound 7 and 22 weeks after RT, both of which showed no detectable bladder mass (Figure 3b). There was no radiographic evidence of pulmonary metastasis by a board-certified radiologist at this recheck. This cat was alive and had resolution of urinary signs 27 weeks after completion of RT, as determined by a telephone conversation with the owner.
Cat 4 was euthanized for unknown reasons by the primary care veterinarian 1 month after the completion of RT, but no further imaging or follow-up was performed by our clinic. However, this patient maintained stable azotemia throughout RT, with no lower urinary signs or changes in defecation noted by the client.
Discussion
The use of palliative RT for the treatment of imaging-diagnosed feline urinary bladder masses was well tolerated in the short term at our institution. Although the sample population was small, palliative RT showed tolerable focal/localized RT-induced acute side effects and all patients’ urinary signs were alleviated by palliative RT and other medical management such as oral antibiotic and oral transmucosal buprenorphine.
The largest study of 118 cases of feline lower urinary tract carcinoma suggested that treatment with partial cystectomy and NSAIDs resulted in the longest survival. Palliative RT was only performed in three cases; one cat was treated with RT and NSAIDs. 5 Stenting of obstructive lower urinary masses has also been described in cats.5,12 In our case series, after discussion of the treatment options, including partial cystectomy, definitive RT, chemotherapy, NSAIDs or combinations of these treatments, palliative RT was elected by clients as the primary treatment in an effort to alleviate clinical signs and to improve QoL while minimizing complications associated with treatments.
Canine TCC has been found to have a low-to-moderate mean alpha:beta ratio of 3.27 (range 2.67–4.0), which suggests that TCC may respond to RT, similar to a late-responding normal tissue. Therefore, a hypofractionated RT protocol with larger fraction size may be beneficial. 13 However, based on a review of canine intrapelvic irradiation of >45 Gy, complications associated with RT occurred at a high rate. 10 Acute side effects included epithelial desquamation and mucositis, and late effects occurred in 19–56% of patients, which included chronic colitis/cystitis, strictures (of the urethra, vagina and rectum) and rectal perforation.7,9,10 Therefore, intrapelvic irradiation, especially with a large fraction size, should be performed carefully. Each patient in our series was treated using image guidance, and small-margin PTVs were used to minimize the potential for acute and late side effects.
In a prior study of palliative RT in 13 dogs with TCC, an overall response rate of 61.5% was obtained. 8 Another benefit of RT was that the patients’ urinary signs, at least transiently, improved during treatment in 10/13 cases, even with urethral or ureteral obstruction in four patients.
The radiation protocol in our case series was a weekly protocol with a palliative intent. All cats in this case series had significant comorbidities such as CKD, IBD and hyperthyroidism. In addition, the first case had multifocal bladder masses, which precluded surgical resection. Palliative RT requires fewer anesthetic events rather than multiple consecutive treatments as in full-course fractionated definitive RT. 14 All patients in our series completed RT protocols, despite comorbidities, and achieved palliation of their clinical signs.
Although the survival benefit of RT is unclear, RT appeared to be well tolerated in all cats in this case series. Only two grade 1 gastrointestinal and one grade 2 genitourinary RT acute side effects occurred after the first RT treatment and 6 weeks after the completion of RT. One cat’s gastrointestinal side effect was self-limiting; the other cat’s gastrointestinal and genitourinary side effects resolved with medical management with oral antibiotic and oral transmucosal buprenorphine.
Two cats were prescribed oral transmucosal buprenorphine, including one case mentioned above, and the other cat was given buprenorphine prophylactically as needed at the beginning of RT for the possible development of urethral muscle spasms secondary to acute RT toxicity (clinician’s preference).
In this case series, 3/4 cats had UTIs based on UCS at various time points in the course of their disease. The response to oral antibiotics was not easily determined as no repeat UCS test was performed.
Two cats were euthanized or died during the follow-up period. One cat was euthanized for unknown reasons 1 month after the completion of RT. The client reported that there were no urinary signs at home at the time of euthanasia. The other cat passed away at home 16 weeks for unknown reasons after the completion of RT. The cause of deaths in these cases, such as progressive renal disease, progressive bladder masses or other, could not be determined owing to a lack of follow-up information.
Therefore, only mild RT acute side effects were noted in two cases in this case series. This could be a result of the cats being more radioresistant than dogs.15–17 Furthermore, clinical signs resolved after the first RT treatment in two cats. These two cases did not show relapse of lower urinary signs from 4 to 46 weeks after completion of RT.
Options for the treatment of bladder neoplasia in cats have been explored in the literature. The main factors that affected the clinical outcome in these patients was treatment of local disease with surgery and systemic treatment with NSAIDs. 5 After discussion with the owners of the cats in our case series about the benefits, risks and feasibility of systemic NSAIDs and surgery, these treatment options were declined in all cases. Therefore, RT was used as the first-line therapy in these patients. Although definitive diagnoses were not obtained in all patients, palliative RT was elected to improve QoL rather than maximally extend survival interval as in more definitive intent treatments such as surgery.
Limitations of this case series include small case numbers, an inconsistent follow-up schedule and variable diagnostics. In this case series, one case was diagnosed with suspect urothelial carcinoma via cytology of the urine sediment. Three cases were diagnosed with suspected urothelial tumors based on ultrasound and CT imaging characteristics of the bladder masses. Also, a contrast CT study was only performed in two cases in our study, and this could underestimate the extent of bladder masses. Lack of cytological/histological diagnosis limits the applicability of our results to the wider population of cats with bladder masses.
Although all clients reported that urinary signs resolved after RT, this may be subjective and biased. Clinical signs may have been alleviated by other concurrent medications in addition to palliative RT. Given the retrospective nature of this case series, acute side effects may also have occurred that were not documented. Longer follow-up is also required to evaluate the long-term survival of patients and to assess possible late RT side effects.
Lastly, we could not assess the local disease response to RT objectively by size with a single diagnostic imaging modality before and after RT in this study.
Based on our findings, palliative RT for urinary bladder masses in cats appears to be safe in the short term and may be associated with the alleviation of lower urinary signs.
Conclusions
In the short term, this palliative RT protocol was well tolerated in this population of cats with urinary bladder masses. No clinically significant acute RT side effects occurred in this case population. However, we cannot determine the RT efficacy, long-term outcomes and probability for late side effects with these retrospective data. Continued investigation with a large prospective study, standardized long-term follow-up and cytological/histological diagnosis will be required to assess the survival outcome and both acute and late RT toxicity profiles of this treatment modality for feline bladder masses.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
