Abstract
Objectives
The aims of this study were to describe the radiographic features of cardiogenic pulmonary oedema (CPE) in a large group of cats with left-sided cardiac disease, and to determine the association between the radiographic features of CPE and the underlying cardiac disease.
Methods
Thoracic radiographs of cats with CPE and echocardiographic evidence of left-sided cardiac disease and left atrial enlargement (LAE) were reviewed, and cardiac silhouette, pulmonary vessels and pulmonary parenchyma evaluation were performed. Interstitial and/or alveolar patterns were classified according to their distribution (ie, diffuse, multifocal or focal) and location (ie, craniodorsal, cranioventral, caudodorsal, caudoventral and perihilar). A Student’s t-test and Mann–Whitney U-test, or the two-proportion z-test, were used to compare continuous or categorical variables, respectively, between cats affected by the two most represented cardiac diseases, namely hypertrophic cardiomyopathy (HCM) and restrictive cardiomyopathy (RCM).
Results
Seventy-one cats were included; among them, 46 (64.7%) and 13 (18.3%) had presented for HCM and RCM, respectively. Subjective and objective cardiomegaly, and subjective and objective LAE were detected in 97.2% and 91.9% of cats and in 80.3% and 40.6% of cats, respectively. Pulmonary artery abnormalities, in particular caudal pulmonary artery dilation, were found in 77.5% of cats. Pulmonary artery to pulmonary vein ratio = 1 was found in 71.8% and 55% cats on right lateral and ventrodorsal or dorsoventral views, respectively. Interstitial (57.8%) and mixed interstitial–alveolar (38%) pattern, multifocal (84.5%) and symmetrical (75%) distribution with prevalent ventrocaudal (65.6% of cats) and ventrocranial (60.9% of cats) locations were most frequently observed. No difference was found for any of these radiographic features between cats with HCM and RCM.
Conclusions and relevance
Moderate-to-severe cardiomegaly and LAE, caudal pulmonary artery and vein dilation, as well as a ventral, multifocal and symmetrical interstitial pulmonary pattern, were the main radiographic features of CPE in evaluated cats. Underlying cardiac disease did not influence the aforementioned radiographic features.
Keywords
Introduction
Cardiogenic pulmonary oedema (CPE) is the most frequent cause of respiratory distress in cats, secondary to increased vascular hydrostatic pressure due to left-sided congestive heart failure (L-CHF).1–6 A presumptive diagnosis of CPE is based on history and physical examination findings; however, other respiratory disorders may cause similar clinical signs. Thoracic radiography is a useful diagnostic method to confirm and characterise CPE. Furthermore, diuretic treatment response confirms observed clinical and radiographic signs caused by CPE. In cats – differently from dogs – pleural effusion is often an additional presentation of L-CHF and it complicates radio-graphic assessment of pulmonary parenchyma alterations.3–12 The radiographic features of CPE in cats are well reported in veterinary textbooks.1,2,10 However, only a few original studies specifically focused on radiographic findings of CPE.4,13,14 Typically, CPE radiographic features can vary from peribronchial unstructured interstitial to mixed interstitial and alveolar lung patterns, depending on the severity of fluid accumulation.1,2 However, feline CPE is known by its variable appearance, especially regarding distribution and location. In one previous study conducted on 23 cats, the distribution of CPE was reported as predominantly diffuse/non-uniform on pulmonary fields with prevalent ventral location, 4 but no correlation was reported between CPE radiographic features and specific left-sided cardiac diseases. This study aimed to describe the most frequent CPE radiographic features in a large group of cats with left-sided cardiac diseases, and to determine an association between the radiographic aspects of CPE and the underlying cardiac cause.
Materials and methods
Animals
Cats included in this retrospective study were from referrals of the Veterinary Teaching Hospitals (VTHs) of Bologna and Padova University (July 2010–October 2020). Electronic databases from both VTHs were searched for cats with the following clinical characteristics and diagnostic imaging information: an unequivocal diagnosis of L-CHF with an acute onset based on historical and clinical findings of acute respiratory distress (ie, tachypnoea, laboured breathing or both); thoracic radiography with a report consistent with a diagnosis of CPE; echocardiographic evidence of left atrial enlargement (LAE) associated with left-sided cardiac disease; and the resolution of clinical and/or radiographic signs within 48 h of diuretic treatment. Cats were included only if two orthogonal thoracic radiographic views were available and radiographic and echocardiographic examinations were performed within 24 h of each other. Exclusion criteria were echocardiographic diagnosis not related to left-sided cardiac disease and diuretic administration 48 h prior to the radiographic examination. Cats with moderate-to-severe pleural effusion were also excluded.
Echocardiographic examination
Echocardiographic examination was performed on non-sedated cats by the same experienced operator in each centre (MBT and CG). Comprehensive transthoracic two-dimensional real-time mode, M-mode and Doppler were performed on all cats using two different echocardiographic machines (CX50 [Philips] and an iE33 ultrasound system [Philips Healthcare]) equipped with multifrequency phased-array transducers. Each echocardiographic examination was performed under electrocardiography monitoring using standard echocardiographic views.
LAE diagnosis was based on two different two-dimensional echocardiographic methods: left atrial (LA) and aortic root diameter (Ao) measurement in early diastole from right parasternal short-axis view at the heart base for LA:Ao ratio calculation, and LA diameter (LAD) measurement from a right parasternal long-axis four-chamber view at end-systole. LAE was considered for cats with an LA:Ao ratio and LAD >1.5 15 and >16 mm, 16 respectively.
Based on the echocardiographic findings, cats were divided into five groups of cardiac diseases. A hypertrophic cardiomyopathy (HCM) phenotype was considered when maximal end-diastolic left ventricular (LV) wall thickness in a short-axis view at papillary muscle level or in a long-axis four-chamber and LV outflow tract view was ⩾6 mm. Cats with an LV wall thickness of ⩽5 mm, increased LV internal diameter (ie, LV diameter at diastole >14 mm) and fractional shortening <28% were considered to be affected by a dilated cardiomyopathy (DCM) phenotype. Cats with LA or biatrial enlargement with or without mild thickening of the LV wall were considered to be affected by a restrictive cardiomyopathy (RCM) phenotype. All animals showing evidence of myocardial disease with echocardiographic findings not included in the above categories were diagnosed with a non-specific cardiomyopathy phenotype (NSCMP). 17 Cats with different types of congenital heart diseases (CHDs) associated with LAE were merged into a specific group. All echocardiographic diagnoses were made or reviewed by a board-certified veterinary cardiologist (MBT).
Radiographic examination
Right lateral (rLV) and ventrodorsal (VDV) or dorsoventral (DVV) thoracic radiographic views evaluation was performed by the same experienced radiologist (AD), who was blinded to the echocardiographic results.
First, qualitative and quantitative radiographic evaluation of the cardiac silhouette was performed. On rLV, the subjective absence or presence of cardiomegaly (if present, graded as mild, moderate or severe), concave aspect and/or presence of a notch at the level of the caudal border of the cardiac silhouette, and the absence or presence of ‘bulging’ at the area of the LA were considered. On VDV or DVV, the presence or absence of a ‘Valentine-shaped’ cardiac silhouette and a ‘double wall’ at the level of LA area were annotated. Quantitative evaluation of the cardiac silhouette was performed on rLV by measuring the cardiac long axis (CLA), cardiac short axis (CSA), vertebral heart score (VHS) 18 and LA-VHS 16 using previously described methods and reference values to identify cardiomegaly (ie, VHS >8.1) 19 and LA enlargement (ie, LA-VHS >1.30).14,16
The subjective evaluation of pulmonary vessels was performed by assessing abnormalities of the pulmonary veins (PVs) and pulmonary arteries (PAs), namely dilation, tortuosity and pruning. These abnormalities were then classified according to the involved lung lobe. Dilation of cranial PAs and PVs was then evaluated on rLV by comparing the vessel diameter with the thickness of the proximal third of the fourth rib; a ratio >1:1 was considered abnormal. Dilation of caudal PAs and PVs was considered when a superimposed image with the ninth rib on DVV or VDV was a rectangle. The PA to PV ratio (PA:PV) was evaluated and categorised as 1:1, PA >PV or PA <PV. 10
Evaluation of pulmonary parenchyma was performed when considering the presence of an interstitial or a mixed interstitial and alveolar pattern (ie, increase in lung opacity without or with the obliteration of air spaces, respectively). These radiographic patterns were then classified according to distribution as follows: diffuse (when all lung fields were involved); multifocal (when multiple areas of more than one lung lobe were involved); or focal (when a single area of one lung lobe was involved). An asymmetrical distribution of CPE was considered when only a single lung lobe or two ipsilateral lobes were involved on VDV or DVV, with all other distributions considered symmetrical. Multifocal and focal patterns were further classified according to their location as craniodorsal, cranioventral, caudodorsal, caudoventral or central (ie, perihilar) based on involved lung fields.
Finally, the presence of a bronchial pattern and other miscellaneous radiographic findings such as subjective mild pleural effusion, caudal vena cava dilation and aerophagia were recorded.
Data analysis
Data were collected via an online survey tool (Google Form). Survey answers were automatically transferred into a Microsoft Excel spreadsheet.
Numerical continuous data were assessed for normality using the Shapiro–Wilk test, and normally and non-normally distributed data are presented as mean ± SD and median (range), respectively. Categorical data were presented as number and percentage. Student’s t-test and Mann–Whitney U-test, or the two-proportion z-test were used to compare continuous or categorical variables, respectively, between cats affected by the two most represented cardiac diseases, namely HCM and RCM. All statistical analyses were performed using a commercially available software (MedCalc version 12.6.1.0). A P value <0.05 was considered to be statistically significant.
Results
Animals and echocardiographic diagnosis
Altogether, 103 cats with a diagnosis of acute L-CHF were initially included in the study. Of these, 17 were excluded because of moderate/severe pleural effusion, the availability of only one radiographic projection or the absence of LAE on echocardiographic examination. A further 15 cats were excluded owing to a lack of improvement in clinical and radiographic features after diuretic treatment. Therefore, 71 cats fulfilled inclusion criteria. Forty-four cats were male (62%; 30 castrated and 14 intact) and 27 were female (38%; 22 spayed and five intact). Age ranged from 2 months to 20 years (mean 9.6 ± 3.8), and body weight ranged from 0.6 to 11 kg (mean 4.1 ± 1.0). Fifty-four cats were domestic shorthairs (76.0%), eight were Maine Coons (11.3%), seven were Persian (9.9%), and there was one Siamese (1.4%) and one Chartreux (1.4%). Based on the echocardiographic phenotypes, 46 cats had HCM (64.8%), 13 had RCM (18.3%), six had NSCM (8.5%), four had DCM (5.6%) and two had CHD (2.8%), namely a ventricular septal defect and atrioventricular valves dysplasia, respectively.
Radiographic findings
Subjective cardiomegaly was detected in 69 cats (97.2%), which was classified as mild, moderate or severe in 12 (16.9%), 36 (50.7%) and 21 (29.6%) cats, respectively. In 57 cats (80.3%), LAE was subjectively diagnosed on rLV based on the concurrent presence of a cardiac silhouette with a concave caudal border and a bulging LA area, whereas a cardiac silhouette with a notch on the caudal border was only detected in 21 (29.6%) cats (Figure 1). A ‘Valentine-shaped’ cardiac silhouette was observed in 38 (53.5%) cats (Figure 2a), and a ‘double wall sign’ was detected in 19 (27%; Figure 2b). No statistically significant difference was found in the percentage of cardiac radiographic findings in cats with HCM vs RCM (P = 0.206, P = 0.858, P = 0.903, P = 0.985, P = 1.000, P = 0.667 and P = 0.656 for overall moderate cardiomegaly, severe cardiomegaly, concave caudal border, notch on caudal border, presence of a mass in the LA area, Valentine shape and double wall, respectively). VHS and LA-VHS measurement could not be performed in nine and seven cats, respectively, because of vertebral degenerative disease or a silhouette sign on the cardiac border due to mild pleural effusion. Overall median VHS and LA-VHS were 9.2 v (range 7.5–14) and 1.3 v (0.8–3.1 v), respectively, with 57/62 (91.9%) and 26/64 (40.6%) cats having a VHS and LA-VHS >8.1 v and >1.3 v, respectively. No statistically significant difference was found for median VHS and LA-VHS (P = 0.288 and P = 0.197, respectively) between cats with HCM and RCM, or for CLA and CSA (P = 0.666 and P = 0.174, respectively). Table 1 presents the results of cardiac silhouette evaluation and measurement.

Right lateral views of the thorax of two cats with cardiogenic pulmonary oedema and different degrees of left atrial (LA) enlargement (LAE). Radiographic border of left atrium caudodorsal aspect is outlined (yellow dotted line). (a) Moderate LAE (maximum LA craniocaudal diameter as quantified by two-dimensional echocardiography = 23 mm) with a notch at cardiac caudal border (arrow). (b) Severe cardiomegaly and LAE (LA = 35 mm) characterised by an increased length and width of the cardiac silhouette, increased convexity of all cardiac borders and rounding of the cardiac apex. A marked dorsal displacement of thoracic trachea (arrowheads) and moderate distension of caudal vena cava are also evident
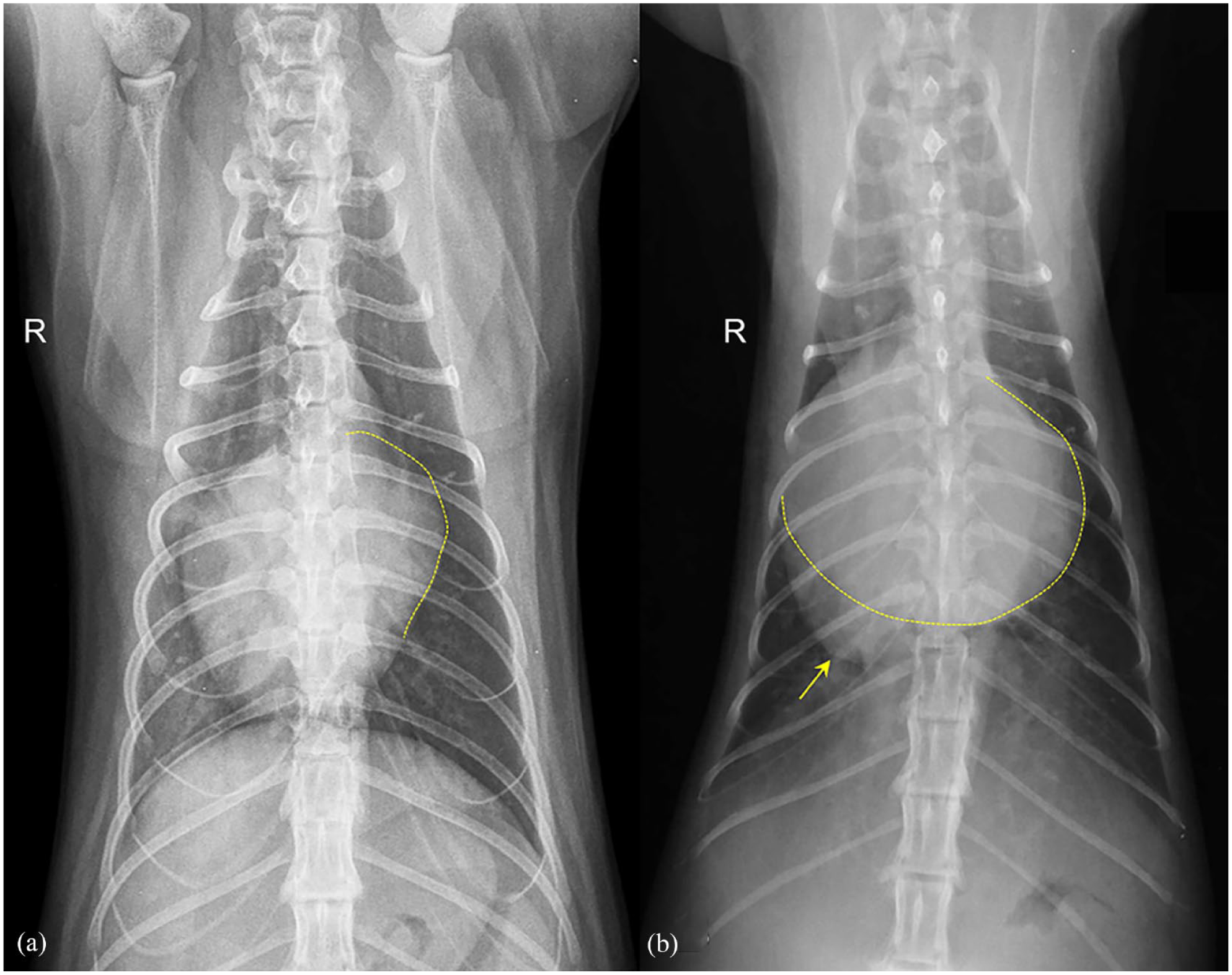
Dorsoventral views of the thorax of two cats with cardiogenic pulmonary oedema and different degrees of left atrial (LA) enlargement (LAE). (a) Moderate LAE (LA = 24 mm) is visible as a bulging in the left cardiac border (yellow dotted line). (b) Severe LAE with ‘double wall sign’ (yellow dotted line) at the level of the LA area. The cardiac apex (arrow) is displaced to the right
Subjective and objective radiographic findings regarding cardiac abnormalities in 71 cats with cardiogenic pulmonary oedema

Data are presented as n (%) unless otherwise indicated
HCM = hypertrophic cardiomyopathy; RCM = restrictive cardiomyopathy; DCM = dilated cardiomyopathy; NSCMP = non-specific cardiomyopathy phenotype; CHD = congenital heart disease; rLV = right lateral view; NE = not evaluable; LA = left atrial; VDV = ventrodorsal view; DVV = dorsoventral view; CLA = cardiac long axis; CSA = cardiac short axis; VHS = vertebral heart score
Fifty-five (77.5%) cats had PA abnormalities and dilation was detected in 52 (94.5%) of them, with similar percentages among the different cardiomyopathies. The left and right caudal pulmonary arteries were most affected (50 cats, 91% for both arteries), whereas the right and left cranial PAs were abnormal in only 22 (40%) and 24 (43.6%) cats, respectively (Figure 3). PA:PV was calculated in 57 (80.3%) and 56 (78.9%) cats on rLV and DVV or VDV, respectively, due to reduced visualisation secondary to lung opacity in other cats. PA:PV = 1 was found in 51 (71.8%) and 39 (55%) cats on rLV and DVV or VDV, respectively. PA:PV >1 was observed in three (4.2%) and 16 (22.5%) cats on rLV and DVV or VDV, respectively. The PA:PV <1 was observed in three (4.2%) cats and one (1.4%) cat on rLV and DVV or VDV, respectively. No significant difference was detected in the percentages of pulmonary vessel abnormalities between cats with HCM and RCM (P = 0.342, P = 1.000, P = 0.322, P = 0.398 and P = 0.773 for overall PA abnormalities, cranial vessel dilation, caudal vessel dilation, PA:PV = 1 on rLV and PA:PV = 1 on DVV or VDV, respectively). Table 2 presents the results of the evaluation of the pulmonary vessels.

Close up of (a) right lateral and (b) dorsoventral views of two cats with cardiogenic pulmonary oedema and pulmonary lobar vessels abnormalities. (a) Mild dilation of both cranial pulmonary arteries (arrows) and veins (arrowheads) is visible. (b) Severe dilation of the caudal lobar pulmonary arteries (arrows) and veins (arrowheads) with mild tortuosity of the right caudal artery are seen
Radiographic findings regarding pulmonary vessel abnormalities in 71 cats with cardiogenic pulmonary oedema

Data are n (%)
HCM = hypertrophic cardiomyopathy; RCM = restrictive cardiomyopathy; DCM = dilated cardiomyopathy; NSCMP = non-specific cardiomyopathy phenotype; CHD = congenital heart disease; PA = pulmonary artery; NE = not evaluable; RCau = right caudal; LCau = left caudal; RCr = right cranial; LCr = left cranial; PV = pulmonary vein; rLV = right lateral view; A >V = artery > vein; V >A = vein > artery; DVV = dorsoventral view; VDV = ventrodorsal view
CPE presented radiographically as an interstitial and mixed interstitial–alveolar pattern in 41 (57.8%) and 27 (38%) cats, respectively. In only one (1.4%) cat, CPE presented as an alveolar pattern; the pulmonary pattern was not evaluable in two (2.8%) cats. A multifocal (n = 60; 84.5%) distribution of CPE, mainly localised in the ventrocaudal (n = 42; 65.6%) and ventrocranial (n = 39; 60.9%) positions, was most commonly observed (Figure 4). If multifocal, the distribution was symmetrical and asymmetrical in 45 (75%) and 15 (25%) cats, respectively, with prevalent right lung involvement in 13 cats (21.7%; Figure 5). No statistically significant difference was found between cats with HCM and those with RCM regarding pulmonary pattern, including type (P = 0.883 and P = 1.000 for interstitial and mixed patterns, respectively), distribution (P = 0.271, P = 0.377 and P = 0.304 for multifocal, symmetrical and right asymmetrical, respectively) and location (P = 0.867, P = 0.180, P = 0.331 and P = 0.304 for dorsocaudal, ventrocranial, ventrocaudal and perihilar, respectively). A bronchial pattern was present in 40 (56.3%) cats but was not statistically significantly different between cats with HCM and those with RCM (58.7% and 38.5%, respectively; P = 0.318). Table 3 presents the results of the lung parenchyma evaluation.

Right lateral view of the thorax of four cats with different distributions of cardiogenic pulmonary oedema. (a) An interstitial pulmonary pattern is mainly visible in the ventrocranial part of the lungs. Generalised cardiomegaly and rounding of the cardiac apex are also visible. (b) A moderate interstitial–alveolar pulmonary pattern is visible in the cranioventral and caudoventral parts of the lungs, partially obscuring the cranial cardiac border and caudal vena cava. (c) An interstitial–alveolar pulmonary pattern is mainly visible in the caudodorsal lung lobes. This patient also has cardiomegaly. (d) Severe and diffuse interstitial–alveolar pulmonary pattern involving all lung fields and obscuring the cardiac silhouette and caudal vena cava

(a) Ventrodorsal and (b) dorsoventral views of the thorax of two cats with cardiogenic pulmonary oedema. (a) An interstitial–alveolar pulmonary pattern with prevalent asymmetrical distribution in the right caudal lung lobe obscuring the right caudal pulmonary vessels is seen. Severe cardiomegaly with a rounded cardiac silhouette shape is also evident. (b) Severe, diffuse and symmetrical interstitial–alveolar pulmonary pattern. Marked biatrial enlargement with prominent caudal lobar pulmonary vessels is seen
Radiographic findings of lung parenchymal abnormalities in 71 cats with cardiogenic pulmonary oedema

Data are n (%)
HCM = hypertrophic cardiomyopathy; RCM = restrictive cardiomyopathy; DCM = dilated cardiomyopathy; NSCMP = non-specific cardiomyopathy phenotype; CHD = congenital heart disease; NE = not evaluable; RA = right asymmetrical; LA = left asymmetrical
Mild pleural effusion was detected in 45 (46.4%) cats, with no statistically significant difference between animals affected by HCM (n = 26/46; 56.5%) and RCM (n = 9/13 [69.2%]; P = 0.598). Caudal vena cava dilation and aerophagia were detected in 16 (22.5%) and 19 (26.8%) cats, respectively, without a statistically significant difference between animals affected by HCM (17.4% and 28.3%, respectively) and RCM (30.8% and 23.1%, respectively; P = 0.545 and P = 0.985, respectively). Table 4 presents the miscellaneous radiographic findings.
Miscellaneous radiographic findings in 71 cats with cardiogenic pulmonary oedema

Data are n (%)
HCM = hypertrophic cardiomyopathy; RCM = restrictive cardiomyopathy; DCM = dilated cardiomyopathy; NSCMP = non-specific cardiomyopathy phenotype; CHD = congenital heart disease; CVC = caudal vena cava; NE = not evaluable
Discussion
The prevalence of cardiac diseases associated with L-CHF in this study was similar to findings from previous reports,5,14,19 with congenital cardiac disease being less frequent than acquired cardiac disease and, among the latter, HCM being the most represented. The main results of this study were that pulmonary interstitial patterns with a multifocal and symmetrical distribution located in a ventral position are the most common radiographic features of CPE in cats, regardless of the underlying cardiac disease. Cardiomegaly and LAE are radiographically discernible in most affected cats, as are PA and PV dilation. Although a bilateral, symmetrical and multifocal lung pattern is also characteristic of non-cardiogenic pulmonary oedema in dogs and cats, 20 a prevalent ventral pulmonary pattern distribution, cardiomegaly and dilated pulmonary vessels were typical of CPE in this study and are useful to discriminate both forms of oedema.
Subjective and objective cardiomegaly was detected in >90% of cats and was subjectively defined as moderate or severe in around 80% of animals. Furthermore, the observed median VHS of 9.2 v was similar to the 9.3 v threshold previously reported as highly associated with heart disease in cats with acute respiratory distress, 21 and well above the threshold of 7.9 v that is useful in distinguishing healthy cats from those with left-sided cardiac disease. 22 Cardiomegaly was still recognisable, even though mild pleural effusion was present in nearly two-thirds of the cats included in the study. These findings suggest that cardiomegaly is either subjectively or objectively recognisable in nearly all cats with CPE, including those affected by prevalent LV concentric cardiac hypertrophy (eg, HCM). These findings are similar to those observed in another feline study, in which the cardiac silhouette was subjectively and objectively enlarged in most cats with CPE. 4
Enlargement of the cardiac chambers is closely related to CHF pathophysiology and volume overload. In particular, LAE results from a chronic adaptive compensatory mechanism to increased LA pressure in cats with diastolic dysfunction and associated left-sided cardiac disease, and it closely reflects LV filling pressure and volume overload.14,16,23,24 The caudal border concave aspect of the cardiac silhouette and mass effect in the LA area were radiographic evidence of LAE in 80.3% of studied cats. However, a caudal border notch of the cardiac silhouette in rLV, and a ‘Valentine shape’ or the presence of a double wall on DVV or VDV were less frequently observed (29.6%, 53.5% and 26.8% of cats, respectively). These findings suggest that rLV is the best suited radiographic view to subjectively diagnose LAE in cats. Conversely, a Valentine-shaped heart, reported as a specific radiographic marker of LAE, 25 was found in half of the cats with CPE in this study.
Measurement of LA-VHS was not more sensitive than subjective evaluation of LAE in cats with CPE as fewer than half of cats in this study had a LA-VHS higher than upper normal limit of 1.3 v. 16 Similarly, LAE was not detected after subjective and objective radiographic evaluation in approximately one-third and more than one-half of cases, respectively, in a previous study on 100 cats with acute L-CHF. 14 Radiographic recognition of LAE remains challenging in cats because of LA and auricle superimposition on the cardiac silhouette. 7 Different studies have tried to determine the accuracy of subjective and objective radiographic assessment of LAE in cats with cardiac disease at various disease stages, with conflicting results.14,16,22
Over 70% of cats included in this study presented with abnormalities of the pulmonary vessels (both PAs and PVs), particularly dilation. This result agrees with that of another study on feline CPE in which pulmonary vessel dilation was observed in 71% of cats. 4 Interestingly, caudal pulmonary vessel dilation was seen in 70.4% of cats, whereas cranial pulmonary vessel dilation was detected in only 14% of cats. These findings are likely due to both the inherently greater dimensions of feline caudal pulmonary vessels vs cranial ones, 26 and better visualisation of the former on DVV secondary to the superimposition of minor thoracic structures. In healthy cats, PVs have a smaller diameter than the corresponding arteries, as recently observed on CT and echocardiography,26,27 and this would lead to a radiographic PA:PV of >1:1. Thus, the observed PA:PV of 1:1 in most cats in the present study likely suggest prevalent PV dilation, as would be expected owing to the histologically more distensible structure of PVs vs PAs. 28 Conversely, in a previous study on 100 cats with L-CHF, approximately one-half and two-thirds of the cats had PV dilation and PA enlargement, respectively, as assessed subjectively or semi-quantitatively. 14 This discrepancy could be due to different venous constriction effects because of the variable sympathetic stress in cats of the present study vs cats evaluated by Schober et al. 14 However, the overlap between the range in vein size and also in the PA:PV ratio of the cranial pulmonary vessels between normal cats and those with HCM on thoracic radiographs suggests that these measurements are unlikely to be clinically useful in deciding whether a cat has CHF or not. 29
Regarding lung opacity, an interstitial pattern and mixed interstitial–alveolar pattern were the most represented, corresponding to >95% of cases in our study. Prevalent interstitial patterns of CPE were also previously reported in both cats and dogs because they represent the most frequent pulmonary pattern in the first phase of L-CHF.4,6,30 Conversely, a prevalent multifocal and symmetrical distribution of CPE associated with ventrocranial and ventrocaudal location of pulmonary infiltrates observed in cats in the present study contrasts with the results of a previous study. 4 Both studies included cats with echocardiographic evidence of LAE and without previous diuretic treatment, but the sample size differed, with more than triple the number of cats included in the present study. Furthermore, a precise echocardiographic diagnosis of cardiac diseases associated with CPE was not included in the study by Benigni et al. 4 However, the current study did not present any significant difference in percentage of different type, distribution and location of pulmonary patterns between the two most frequently diagnosed feline myocardial cardiac diseases, namely HCM and RCM.
These findings suggest that the distribution of pulmonary infiltration in cats with L-CHF is independent of underlying cardiac disease, unlike dogs in which an asymmetrical distribution of CPE was observed in animals with mitral valve disease (MVD) but not in those with DCM. 30 This difference can be explained by dissimilar pathophysiological mechanisms leading to increased LA pressure and, eventually, L-CHF in feline HCM and RCM vs canine MVD and DCM. Indeed, in both feline myocardial diseases, increased LA pressure is the consequence of LV diastolic dysfunction with preserved systolic function, 24 whereas canine cardiac diseases are characterised by predominant volume overload with preserved systolic function and normal to only mildly affected diastolic function (MVD) and LV systolic dysfunction associated with increased LV stiffness (DCM). 31 Furthermore, the presence and direction of mitral regurgitant jet differ between feline HCM and RCM and canine MVD and DCM. Although evaluation of mitral regurgitation (MR) has not been thoroughly carried out in small animals, feline HCM and RCM are associated with the variable presence of MR with prevalent posterior eccentric direction in HCM, 32 whereas canine MVD and DCM are characterised by high-velocity MR with an often eccentric jet direction and lower velocity MR jet with a central direction, respectively. 30 Nevertheless, an asymmetrical distribution of feline CPE involved almost exclusively the right lung lobes, as previously found in dogs with MVD and also in humans.30,33
An explanation for these findings is challenging and could be due to anatomical variability in the pulmonary vasculature (eg, pulmonary vascular branches) and pulmonary venous drainage into the LA (eg, number and location of PV ostia). 26 A mild amount of pleural effusion observed in nearly two-thirds of the cats in this study did not prevent evaluation of the above and other less-represented radiographic findings (ie, bronchial pattern, caudal vena cava dilation and aerophagia secondary to respiratory distress).
This study presented some limitations because of its retrospective design. First, LAE was considered among the inclusion criteria, even though some cats can develop acute L-CHF without LAE. 14 Furthermore, only linear, and not area or volumetric, echocardiographic measurements were used to identify LAE. However, LA diameter and LA:Ao measurement are two of the most commonly employed and easy to perform echocardiographic methods to identify LAE in cats. Moreover, the results of subjective radiographic identification of LAE could have been biased by the knowledge that all cats actually had echocardiographic evidence of LAE as per the study design. Second, the inclusion of cats without previous loop diuretic treatment and the exclusion of those without clinical signs and radiographic improvement after diuretic treatment may have resulted in the exclusion of some of the most severely affected animals (eg, cats that died of CPE shortly after admission). Finally, the small sample size of some groups of cats affected by less common cardiac diseases hampered further evaluations of their association with the radiographic features of CPE.
Conclusions
The main radiographic features of CPE in cats include moderate-to-severe cardiomegaly and LAE, recognisable by the caudal border concave aspect of the cardiac silhouette and the mass effect on LA area on rLV. PA and PV abnormalities are often present, recognisable as caudal pulmonary vessel dilation. An interstitial pulmonary pattern is the most frequently observed, with a prevalent multifocal and symmetrical distribution located in caudoventral and cranioventral positions. Underlying left-sided cardiac disease, namely HCM or RCM, does not influence the abovementioned radiographic features, although information on less common feline cardiac diseases deserves further study.
Footnotes
Author note
This paper was presented, in part, as an abstract at the 2021 EVDI Congress.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (either prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
