Abstract
Objectives
This study aimed to evaluate renal measurements by B-mode ultrasound in kittens during their first 3 months and compare them with those of young adult cats.
Methods
Forty-eight healthy mixed-breed cats (24 males and 24 females) were enrolled in this study. The cats were divided into six age groups: groups 1–5 (kittens) and group 6 (young adult cats). Each kidney was examined using B-mode imaging and the following measurements taken: renal length (RL), renal height (RH), renal width (RW), cortical thickness (CoT), medullary thickness (MdT) and corticomedullary ratio (CMr). The ratio and correlation of RL to aortic diameter (AoD) and the length of the fifth/sixth lumbar vertebrae (LLV) were also determined.
Results
Among groups, there was a statistically significant difference between RW, renal volume (RV), CoT, MdT and CMr. The smallest renal dimensions were obtained in the youngest kittens (up to 10 days of age), including RL (1.84 ± 0.40 cm), RH (1.14 ± 0.17 cm), RW (1.25 ± 0.20 cm) and RV (1.47 ± 0.68 ml). A strong correlation was observed between RL:AoD and RL:LLV, allowing for linear regression equations and reference intervals for RL:AoD and RL:LLV to be created for each age group.
Conclusions and relevance
B-mode ultrasound showed that renal measurements in kittens differed from those in young adult cats, and that the renal dimensions, renal CoT and CMr increased with the growth of the cat. The RL-LLV ratio is a reliable method for measuring RL according to age group.
Introduction
Domestic cats have a higher predisposition to kidney diseases than dogs, and chronic kidney disease (CKD) is one of the most common conditions in this species.1–3 Although the aging process predisposes all cats to kidney injuries and CKD due to cellular senescence, several factors can predispose young or adult cats to renal parenchymal damage, such as the environment, nutrition, other diseases and use of anesthetics, sedatives, therapeutic agents or other drugs.2–4 Genetic factors and phenotypic variations must also be considered as they can be predisposing factors for congenital anomalies that result in morphological alterations such as renal dysplasia or hypoplasia.2,5
The kidneys are considered immature at birth and undergo a gradual process of growth and maturation during the first weeks of postnatal life.6–9 During this period in human infants and canine puppies, the glomeruli grow and distribute throughout the renal cortex and the tubular system develops, which causes changes in the corticomedullary ratio (CMr) and an increase in kidney dimensions.7,8,10 One study showed that the cell and tubular volumes per nephron increased by 249% and 303%, respectively, in Beagle dogs from 14 to 200 days of age. 6 In kittens, the glomerular filtration rate increases up to 9 weeks of age before reaching adult levels. 5
Conventional ultrasound is an important imaging tool for the assessment of the urinary tract in small animals, as it can provide relevant information on morphological features and parameters, and allow for the characterization of local or diffuse lesions.3,11–13 The ultrasonographic appearance of normal kidneys in adult domestic cats is well described, including reference intervals (RIs) for renal dimensions; however, information about the kidneys in kittens has not yet been described, considering growth and physiological functional changes in the postnatal period.5,8,10,12,13
To the best of our knowledge, an ultrasonographic study showing the influence of age on renal dimensions in cats has not been previously reported. Thus, this study aimed to evaluate renal measurements by B-mode ultrasound in kittens during the first 3 months of age and compare them with those of young adult cats.
Materials and methods
Cat selection
Forty-eight healthy mixed-breed cats (24 males and 24 females) were enrolled in this study. The cats from breeding catteries or client-owned cats were divided into six age groups of eight cats (four males and four females per group) as follows: group 1 (G1; up to 10 days old); group 2 (G2; from 11 to 20 days old); group 3 (G3; from 21 to 30 days old); group 4 (G4; between 1 and 2 months old); group 5 (G5; between 2 and 3 months old); and group 6 (G6, between 1 and 3 years old). Based on the Feline Life Stage Guidelines, G1–G5 were classified as kittens, and cats from G6 were classified as young adults. 14 Cats in G1 to G5 were considered healthy by physical examination, hydration status assessment and the Apgar scoring system. 4 Cats from G6 were considered healthy by physical examination, complete blood count, serum biochemistry, urine analysis and seronegativity for feline immunodeficiency virus antigen and feline leukemia virus antibody. All cats in G6 were neutered. The study design was approved by the Institutional Ethics Committee for the Use of Animals (CEUA no. 0152/2019).
Abdominal ultrasound and body parameters
All ultrasonographic examinations were performed by the same experienced operator using portable ultrasound (Logiq E; GE Medical System) with a linear transducer (8–12 MHz). Only cats in G6 fasted for 12 h before the procedure. Ultrasonographic examinations were performed in a home environment using feline friendly handling techniques, including a quiet environment, synthetic feline facial pheromone spray and a silent clipper.
All cats were manually restrained and placed in the supine position with a V-shaped foam pad. Prior to ultrasound, fur was clipped from the ventral and lateral abdomen, and an acoustic coupling gel was used. Initially, both kidneys were located and evaluated in B-mode, ruling out any morphological or congenital abnormalities. Each kidney was then examined in the longitudinal and transverse scan planes using B-mode imaging, and measurements were taken as follows: renal length (RL) in the longitudinal plane, renal height (RH), renal width (RW), cortical thickness (CoT), medullary thickness (MdT) and CMr in the transverse plane (Figures 1 and 2). The CMr was calculated by dividing the CoT by the MdT. Renal volume (RV) was automatically calculated by the ultrasound device using the ellipsoid volume formula after the determination of RL, RH and RW. 15 In addition, to measure the aortic diameter (AoD) and determine the renal:aorta ratio (RL:AoD), cross-sectional images of the abdominal aorta (Ao) just caudal to the origin of the left renal artery were obtained as previously described (Figure 1).16,17 The last lumbar vertebra was located using the abdominal approach. The lengths of the fifth and sixth lumbar vertebrae (LLV) were measured in the longitudinal plane, and the RL:lumbar vertebra ratio (RL:LLV) was obtained (Figure 1). 16

(a) Longitudinal and (b,c) transverse ultrasonographic images of the left kidney of a young adult cat, demonstrating measurement of renal length (RL), renal width (RW), renal height (RH), cortical thickness (CoT) and medullary thickness (MdT). (d) Longitudinal ultrasonographic image of vertebral bodies of the fifth (L5)/sixth (L6) lumbar vertebrae, demonstrating measurement of the length of the lumbar vertebra (LLV). (e) Transverse ultrasonographic image of the aortic lumen just caudal to the origin of the left renal artery, demonstrating the aorta (Ao) and the measurement of the aortic diameter (AoD)
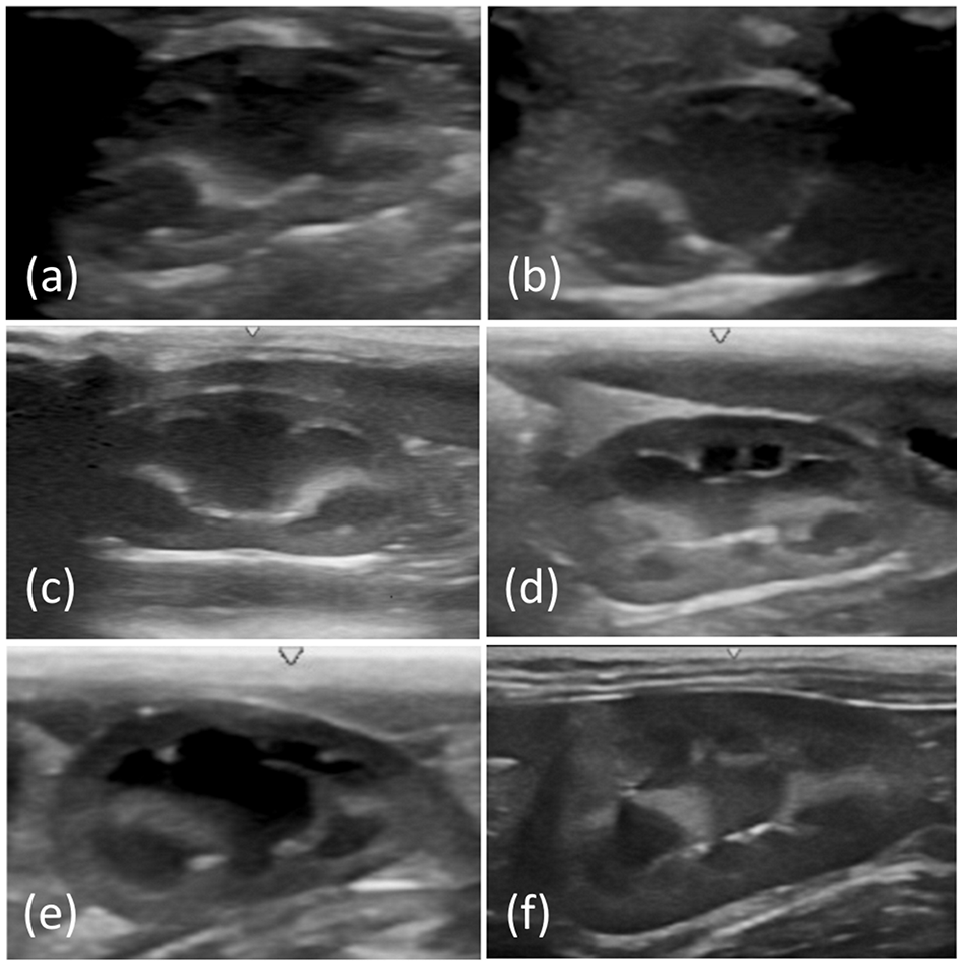
Longitudinal ultrasonographic images of the kidney obtained in (a) group 1 (up to 10 days old), (b) group 2 (11–20 days old), (c) group 3 (21–30 days old), (d) group 4 (1–2 months old), (e) group 5 (2–3 months old) and (f) group 6 (1–3 years old)
Immediately after ultrasound examination, the following body parameters were obtained: body weight (BW), body length (BL; distance from point of the shoulder to tuber ischium) and the distance from the tibiotarsal joint to the stifle joint (TSL) as previously described, 18 to be correlated to RL.
Statistical analysis
The variables (RL, RH, RW, RV, CoT, MdT, CMr, AoD, LLV, RL:AoD ratio and RL:LLV ratio) were subjected to the Shapiro–Wilk normality test and quantitative descriptive analysis. Based on the normality of the data distributions, the variables were subjected to non-parametric (Kruskal–Wallis) or parametric (ANOVA) tests to compare the kidney side (left vs right), sex (male vs female) and age groups (G1, G2, G3, G4, G5 and G6). Spearman’s correlation test was used to assess the correlations between RL and LLV, AoD and body parameters (BW, BL and TSL). All statistical analyses were performed using GraphPad Prism 7.0. Statistical significance was set at P <0.05.
Results
Most variables (RL, RH, RW, RV, AoD, LLV, RL:AoD ratio and RL:LLV ratio) had a parametric distribution, whereas CoT, MdT and CMr had a non-parametric distribution. Within age groups, no statistically significant differences were found in the variables according to kidney side or sex (P >0.05). Thus, the mean value of each variable for each age group was calculated (Tables 1–3).
Renal length (RL), height (RH), width (RW) and volume (RV) in 48 healthy cats in different age groups*

No statistically significant difference between kidney side (right vs left) and sex (female vs male) (P <0.05)
Cortical thickness (CoT), medullary thickness (MdT) and corticomedullary ratio (CMr) of the kidneys in 48 healthy cats in different age groups*

No statistically significant difference between kidney side (right vs left) and sex (female vs male) (P <0.05)
Diameter (cm) of the abdominal aorta, obtained by cross-section, caudal to the origin of the left renal artery
Length (cm) of the fifth/sixth lumbar vertebrae (no statistically significant difference was observed between the vertebrae [P >0.05])
Renal length (cm) obtained by the sagittal section (see Table 1)
No statistically significant difference between sex (female vs male; P <0.05)
ANOVA showed a statistical difference in most variables (RW, RV, CoT, MdT, CMr, AoD, LLV, RL:AoD ratio and RL:LLV ratio) among the groups (P <0.05). However, statistical differences were only observed in RH between G1 and G2 and between G5 and G6 (P <0.05), and in RL among G1 and G2, G3 and G4, and G5 and G6 (P <0.05).
Owing to the differences observed by the ANOVA test, RIs (mean ± 2 SD) of the RL:LLV ratio 15 were determined for each age group (Table 3) as follows: 3.4–5.1 (G1); 3.5–4.5 (G2); 3.2–4.1 (G3); 2.9–3.7 (G4); 2.5–3.3 (G5); and 1.6–2.3 (G6).
Spearman’s test indicated a strong positive linear correlation among RL and BL, BW, LLV, TSL and AoD (Table 4). Linear regression analysis confirmed this finding. However, RL:BW and RL:AoD presented low r² values, indicating a low influence of BW and AoD on RL (Table 4). Using the values obtained by linear regression analysis, it was possible to create the following regression equations, demonstrating the relationship between RL and BL, LLV and TSL:
Spearman’s correlation between renal length and body parameters obtained in 48 healthy cats
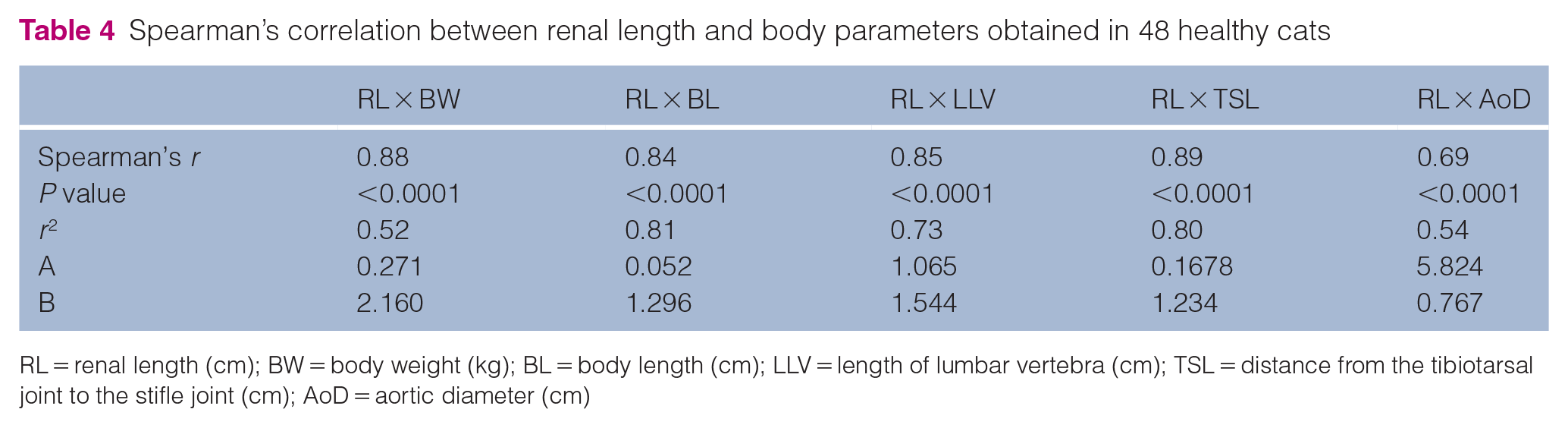
RL = renal length (cm); BW = body weight (kg); BL = body length (cm); LLV = length of lumbar vertebra (cm); TSL = distance from the tibiotarsal joint to the stifle joint (cm); AoD = aortic diameter (cm)



Discussion
This study showed the influence of age on ultrasonographic renal measurements in cats, with smaller renal dimensions and ratios in kittens than in young adults. A gradual increase in kidney dimensions, renal CoT and alterations in CMr were also observed in this study according to age group. Changes in the ultrasonographic appearance of the kidney according to normal growth have also been described in humans, such as differences in size, corticomedullary differentiation and CMr in neonates, children and adults.8,19–21
The kidneys of kittens after birth (G1) were approximately half the size as those of young adults (G6), with CoT half the thickness as that of young adults and a smaller CMr. These findings differ from those described for adults.12,13 Measurement of CoT and of CMr can be used to assess the stage or progression of CKD in adult cats. 4 Thus, kittens younger than 3 months of age with a renal CoT similar to that of adult cats should not be considered normal.
In humans, renal dimensional differences may persist until adult BW is obtained when the physiological growth period ends.19,20,22,23 In small animals, body growth is divided into rapid and slow growth periods. 5 In domestic kittens, the rapid growth period lasts until 5–6 months of age, when 75% of adult weight is reached.5,24 After 6 months of age, growth begins to slow and stops at 1 year in most domestic cats, except for large breeds, which continue to grow until the cat is 18 months of age.5,24 The age at which kittens had renal measurements similar to adult cats could not be determined in this study. However, the values differed between the kittens in G5 and young adults (G6).
In this study, young adult cats (G6) had mean RL and CoT values within the RI established for adult cats.4,12,13,25–27 MdT and CMr were measured in the transverse plane, and the mean values were similar to those reported in a study in adult cats using the same methodology. 4 In contrast, the values differed from those of other studies in cats whose measurements were performed in the dorsal plane.27,28
In this study, no statistical differences were observed between sexes or in the measurements between the right and left kidneys. However, differences between right and left RLs have been reported in cats, even though they were not statistically significant.4,28–30 Sex-related differences and reproductive status have been described, with the RL being smaller in females than in males, and smaller in neutered cats than in intact cats.25–27,30 Sex hormones have been thought to contribute to these differences as studies in mice and rats have shown that androgens positively influence kidney dimensions. Estrogens may have a negative influence, resulting in sexual dimorphism.31,32 In addition, one study showed that kidney weight decreased when male mice were castrated in adulthood. 31 As the cats were prepubertal (<5 months old) or young adults (neutered going through puberty at 5–6 months of age), this may be related to the absence of sex differences in this study. Considering the role of gonadal hormones,30–32 these levels were probably insufficient to influence renal dimensions in the actual study.
Except for the very small (Singapura or Munchkin) or large (Highlander or Maine Coon) cat breeds, body size variation was less critical in cats than in dogs, as observed in this study. The RL for adult cats in ultrasound examination has been described to be 3.0–4.5 cm but can measure up to 5.3 cm.12,13,25 To help reduce subjectivity in renal size determination, quantitative methods have been developed to assess the correlation between RL and body parameters in dogs, such as RL:AoD and RL:LLV ratios.16,17 One study showed a positive linear association between RL and AoD in dogs, suggesting that RL reduced or increased when RL:Ao ratios were <5.5 or >9.1, respectively. 17 Owing to this wide range of normality, another study evaluated a correlation between RL and L5 or L6 length, establishing a ratio between 1.3 and 2.7, considered more sensitive for determining changes in RL. 16
In this study, a moderate positive linear correlation was obtained between RL:AoD and a strong positive correlation between RL:LLV, regardless of age group. In addition, the normal ranges for the RL:AoD ratio (7.9–9.4) and RL:LLV ratio (1.6–2.3) in young adult cats (G6) were narrower than those described for adult dogs,16,17 suggesting that changes in RL can be detected more clearly in cats. However, one study obtained a higher RL:AoD ratio (12.15 ± 1.48) in neutered adult cats 27 than in young adult cats in this study (8.6 ± 1.4). As the RL:AoD correlation showed a low r² (0.54) in this study, other variables were likely to influence the data. Based on these findings, the RL:AoD ratio can be assumed to be less accurate than the RL:LLV ratio (r² = 0.73) in domestic cats. Thus, the RL:LLV ratio may be considered the most useful parameter to determine RL in cats, as the renal size in neutered young cats should be considered decreased with an RL:LLV ratio <1.6 and increased with an RL:LLV ratio >2.3.
Owing to the similar body sizes of different cat breeds, other parameters could be used for the ultrasound assessment of renal dimensions. One study showed a strong linear correlation between BW and renal measurements in dogs, but linear regression analysis (r² = 0.60–0.73) showed that other factors influenced renal measurements in addition to BW. 33 Likewise, a high Spearman’s r (0.88) but moderate r² (0.52) was verified in this study. In contrast, BL and TSL have been considered consistent morphometric measurements in cats. 18 Both measurements showed a strong correlation with RL in this study. However, TSL was easier to obtain than BL in awake cats, in addition to taking less time to measure and was less stressful for the animals.
Our study had some limitations. First, our findings were based on kittens in the first 3 months of life only, and not in kittens aged 3–12 months; thus, further studies including this age group are needed. Second, laboratory tests were not performed for the kittens to support their health status because they were not authorized by the owners.
Conclusions
B-mode ultrasound showed that renal measurements in kittens differed from those in young adult cats, and that the renal dimensions, renal CoT and CMr increased with the growth of the cat. The RL:LLV ratio is a reliable method for measuring RL according to age group.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors wish to thank FINEP (Financiadora de Estudos e Projetos grant 01.12.0530.00) and Capes (Finance Code 001).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.

