Abstract
Clinical importance:
Feline infectious peritonitis (FIP) is one of the most important infectious diseases and causes of death in cats; young cats less than 2 years of age are especially vulnerable. FIP is caused by a feline coronavirus (FCoV). It has been estimated that around 0.3% to 1.4% of feline deaths at veterinary institutions are caused by FIP.
Scope:
This document has been developed by a Task Force of experts in feline clinical medicine as the 2022 AAFP/EveryCat Feline Infectious Peritonitis Diagnosis Guidelines to provide veterinarians with essential information to aid their ability to recognize cats presenting with FIP.
Testing and interpretation:
Nearly every small animal veterinary practitioner will see cases. FIP can be challenging to diagnose owing to the lack of pathognomonic clinical signs or laboratory changes, especially when no effusion is present. A good understanding of each diagnostic test’s sensitivity, specificity, predictive value, likelihood ratio and diagnostic accuracy is important when building a case for FIP. Before proceeding with any diagnostic test or commercial laboratory profile, the clinician should be able to answer the questions of ‘why this test?’ and ‘what do the results mean?’ Ultimately, the approach to diagnosing FIP must be tailored to the specific presentation of the individual cat.
Relevance:
Given that the disease is fatal when untreated, the ability to obtain a correct diagnosis is critical. The clinician must consider the individual patient’s history, signalment and comprehensive physical examination findings when selecting diagnostic tests and sample types in order to build the index of suspicion ‘brick by brick’. Research has demonstrated efficacy of new antivirals in FIP treatment, but these products are not legally available in many countries at this time. The Task Force encourages veterinarians to review the literature and stay informed on clinical trials and new drug approvals.
Keywords
Introduction
Feline infectious peritonitis (FIP) was first described as a specific disease entity in 1963 by Dr Jean Holzworth and colleagues at the Angell Memorial Animal Hospital in Boston, USA. 1 FIP commonly occurs in cats originating from catteries, shelters and foster/rescue groups, as the prevalence of feline coronavirus (FCoV) infection is high in cats living in crowded conditions. 2 Cats that have recently undergone a stressful event are also more likely to develop FIP. In addition, young cats (less than 2 years of age) are especially vulnerable. 3
FIP is one of the most important infectious diseases of cats, and nearly every small animal veterinary practitioner will see cases, especially those working closely with shelters, rescue groups and/or breeders. More complex cases are often seen by specialists and at university hospitals. It has been estimated that around 0.3% to 1.4% of feline deaths at veterinary institutions are caused by FIP.3–5 FIP can be challenging to diagnose owing to the lack of pathognomonic clinical signs or laboratory changes, especially when no effusion is present. However, given that the disease is fatal when untreated, the ability to obtain a correct diagnosis is critical.
The 2022 AAFP/EveryCat Feline Infectious Peritonitis Diagnosis Guidelines have been developed by a Task Force of experts in feline clinical medicine to provide veterinarians with essential information to aid their ability to recognize cats presenting with FIP.


Helpful tips are provided throughout the document, together with multiple clinical images and tables; algorithms and other supplemental resources appear online (see list of supplementary material on page 928).
FIP was once considered a terminal diagnosis. Research has demonstrated efficacy of new antivirals in FIP treatment, but these products are not legally available in many countries at this time. Veterinarians are encouraged to review the literature and stay informed on clinical trials and new drug approvals.
Characteristics of feline coronaviruses
FIP is caused by an FCoV. FCoVs are enveloped, positive-stranded ribonucleic acid (RNA) viruses characterized by club-like spikes that project from their surface and have unusually large viral RNA genomes (see supplemental figure 1). 6 They belong to the order Nidovirales, family Coronaviridae and subfamily Coronavirinae.

Coronavirinae are subdivided into four genera based on genetic and antigenic characteristics: alpha (α)-, beta (β)-, gamma (y)- and delta (δ)-CoV. FCoV belongs to the α-corona-virus genus and is taxonomically distant from SARS-CoV-2, a member of the β -coronavirus genus and the causative agent of coronavirus disease 2019 (COVID-19).7,8 FCoV occurs in two serotypes (types I and II), which encompass numerous strains.9,10 FCoV serotypes differ in their neutralizing antibody reactions and have distinct spike (S) protein sequences.11–13 Type II is less prevalent in most cat populations compared with type I,14,15 and likely originated from recombination between FCoV type I and canine coronaviruses (see supplemental figure 2).12,16–18 While serotype II uses the feline aminopeptidase-N receptor present on the intestinal villi19,20 for cell entry, the cellular receptor for serotype I is still unknown.20,21 Serotype II FCoV is easily grown in cell culture, while serotype I FCoV is difficult to culture.
FCoV occurs as two different bio-/patho-types,9,22,23 which differ in virulence: a non-mutated harmless biotype (also called feline enteric coronavirus [FECV]), which is very common in multi-cat populations, and a mutated virulent biotype (also called feline infectious peritonitis virus [FIPV]), which causes FIP. Infection always involves the non-mutated biotype, but in a small proportion of cats infected with FCoV (7-14% in multi-cat environments), a spontaneous mutation will occur in individual cats leading to a change in cell tropism from enterocytes to an affinity for monocytes/macrophages.10,24–26 This biotype switch is a key event in the pathogenesis of FIP. Non-mutated and mutated FCoV bio-types from the same environment are >99% related, yet vary uniquely in their virulence. 24
Epidemiology of feline coronavirus infection
FCoV is found globally and is ubiquitous in most cat populations.27,28 It is highly contagious and spreads efficiently via fecal-oral transmission, allowing for high prevalence in multi-cat environments, such as breeding catteries, 2 shelter/rescue facilities and animal hoarding situations. 28 In particular, FCoV infection is common when living conditions are crowded, and litter boxes and feeding bowls are shared (see supplemental figure 3).29–34 In one study, real-time reverse tran-scriptase-quantitative polymerase chain reaction (RT-qPCR) determined the prevalence of FCoV shedding to be 77% in 179 cats from 37 German catteries. 2 None of the 37 tested catteries was free of FCoV. A few other studies on FCoV shedding in single catteries or shelters have revealed prevalences ranging from 74% to 100%.30,35 In an investigation of cats before and after being placed in shelters in California, USA, the overall prevalence of FCoV shedding upon admission was 33% in all cats, and 90% in kittens and young cats under 56 weeks of age; 36 the prevalence increased significantly, particularly among adult cats, when they were housed together in a shelter environment. 36
Initially, when a cat is infected with FCoV, the virus replicates within the mature apical columnar epithelial cells of the small intestinal villi from the distal duodenum to the cecum. 37 In some cats, persistent infection of colonic columnar epithelial cells occurs (see supplemental figure 4a).30,38,39 Cats start to shed virus within the first 7 days, usually as early as 2-3 days post-infection, and generally continue to shed virus for several weeks, with some cats shedding virus for up to 18 months or even lifelong.9,32,40 The amount of virus shed is especially high during the early phase of infection;9,38 in most cats the viral load then gradually decreases and shedding can stop entirely, but all remain susceptible to reinfection and will then shed again.9,30,32,36,41,42 Very few cats seem to be resistant and never shed the virus.9,17,30,32,36,40–42
Kittens in multi-cat environments where FCoV is endemic usually become infected within the first weeks of life;9,40 indeed, FCoV infection has been demonstrated in kittens as young as 2-4 weeks old. 43 These early infections have raised questions about the effectiveness of protection by maternal antibodies; nevertheless, early weaning of kittens is not recommended.28,44
Kittens less than 1 year of age are 2.5 times more likely to shed FCoV than adult cats, 2 and kittens typically shed significantly higher viral loads than older cats. 2 High amounts of viral shedding in kittens under 6 months of age can be explained by the immaturity of their immune system, allowing the virus to replicate efficiently.9,36 The higher viral loads of young cats imply higher levels of viral replication and, thus, increased risk of mutation of FCoV to the more virulent biotype. 9

Due to the high mutation rate of FCoV, infected cats shed a cluster of genetically related but distinct viral populations, known as quasispecies or mutant cloud. Infected cats can continuously be reinfected with the same or different FCoV strains.42,46 Several studies in catteries and other multi-cat environments have demonstrated at least one cat shedding FCoV at any one time.2,9,10,28,43,45 In the aforementioned study of cats before and after being placed in shelters in California, FCoV shedding increased 10- to 1 million-fold in some cats after only a week in the shelter. 36
Owing to the universal presence of FCoV in multi-cat environments, a priority is to prevent FCoV infection – and thus the potential for development of FIP – in vulnerable kittens and cats, where possible, by identifying persistent FCoV shedder cats. To detect FCoV shedders in these situations, each cat should undergo a series of at least three fecal RT-qPCR tests for FCoV at intervals of 1 week to 1 month.2,28,31,41,45,47,48
While most cats infected with less-virulent FCoV are asymptomatic and do not require treatment, some can develop gastrointestinal signs (eg, mild vomiting and/or diarrhea).46,49 If present, these signs are typically self-limiting and of short duration.9,37,49 However, in a small number of cats, diarrhea can last for weeks to many months.9,49,50 One study looking at feline enteric pathogens in 100 shelter cats found that FCoV was the only one out of 12 enteric pathogens identified that was significantly more prevalent in cats with diarrhea than in normal cats (58% and 36%, respectively). 51
Pathogenesis of FIP
Coronavirus genomes possess a high level of genetic variation owing to the high error rate of RNA polymerase and are, thus, prone to genetic mutations. Mutations in an individual cat leading to a switch in cellular tropism (gastrointestinal epithelium to monocytes/ macrophages) and infection of monocytes/ macrophages are crucial for systemic spread of FCoV (see supplemental figure 4b).26,52,53 Monocyte/macrophage entry alone is not sufficient for the development of FIP; likely, mutations leading to persistence and efficient replication within and activation of monocytes/macrophages are required (see ‘prerequisites’ box).54,55 FCoV strains from different cats with FIP in the same household show mostly unique genetic characteristics, demonstrating that these viruses develop independently in individual cats.56–58

To date, specific viral mutations leading to the biotype switch remain undetermined. Two single nucleotide polymorphisms in genes encoding the fusion peptide of the S protein, leading to amino acid changes (M1058L and S1060A), were initially shown to be linked to the conversion of the biotype;25,59 however, they were later suggested to only indicate systemic spread of FCoV irrespective of the development of FIP (ie, identified in healthy cats as well; see supplemental file 5a).60,61
Diagnosis of FIP
Diagnosing FIP can be straightforward if a cat with typical signalment (Table 1) presents with effusion, as tests using effusion generally have much higher positive predictive values (PPVs) than those using blood.78–80 However, if no effusion is present, diagnosis can become quite challenging due to the variety and non-specificity of possible clinical signs.28,81 FIP can present with systemic signs or signs specifically related to the organs involved, and with or without fever. One study documented fever in only 56% of cats with FIP, and fever was even less common in cats without effusion. 3 Some cats with FIP lack the serum biochemistry abnormalities (hyperproteinemia, hyperglobulinemia, hypo-albuminemia, hyperbilirubinemia) that are considered ‘typical’ for FIP. 78 FIP might not even be on the list of differential diagnoses in cases that do not fit the classic picture (eg, older cat from a single-cat household). Subtle indicators of FIP might also be missed when concurrent disease is present.
Risk factors influencing the development of FIP

The ability to definitively diagnose or have a high index of suspicion ante-mortem that a cat has FIP has been the focus of multiple studies.28,50,78,80,82–86 To arrive at a diagnosis of FIP, the veterinarian must consider the individual patient’s history, signalment and physical examination findings, and select diagnostic tests and sample types accordingly, in order to increase the index of suspicion ‘brick by brick’ (Figure 1).

Diagnostic work-up for FIP. The veterinarian must consider the patient’s history, signalment and physical examination findings, and then select diagnostic tests and sample types based on these, in order to build the index of suspicion ‘brick by brick’. ADR = ‘ain’t doing right’. For explanation of other abbreviations, see box on page 906. This figure is available to download from the supplementary material (supplemental figure 8). Graphic designed by Vicki Thayer and Susan Gogolski, based on Melissa Kennedy’s diagnostic brick wall
When pursuing a diagnosis of FIP, the same or different test modalities can be performed on a variety of sample types, including blood (whole blood, serum, plasma, peripheral blood mononuclear cells [PBMCs]), effusions (thoracic, abdominal, pericardial), tissues, cerebrospinal fluid (CSF), aqueous humour, and/or tissue fine-needle aspirates (FNAs) and biopsies.
Signalment and history
The pathogenenesis of FIP is complex, and susceptibility to FIP in an individual cat involves the interplay between virus virulence factors, host factors such as genetic characteristics, age of the cat at the time of FCoV exposure, concurrent diseases and/or other stressors at a time when a young cat is fighting an FCoV infection.86–88 Host factors can affect the cat’s immune system status, including major histocompatibility complex (MHC) diversity, cytokine production and lymphocyte apoptosis. For example, aspects of MHC II can influence the quality of the immune response.73,89 Additionally, a link between cytokine- and receptor-associated single nucleotide polymorphisms and increased susceptibility to FIP has been suggested. 62
A number of risk factors for the development of FIP, related to a cat’s signalment and history, have been identified (as listed in Table 1 and outlined below).
Physical examination findings
Cats with FIP commonly have non-specific clinical signs such as anorexia, lethargy, a waxing/waning fever (often non-responsive to antibiotics), lymphadenopathy and weight loss or, for kittens, failure to gain weight (Table 2).3,86,88,94 These non-specific clinical signs can occur irrespective of the presence of effusion, and may either be reported by owners or, particularly in the early stages of the disease, noted during examination.3,88,94
Signs associated with FIP
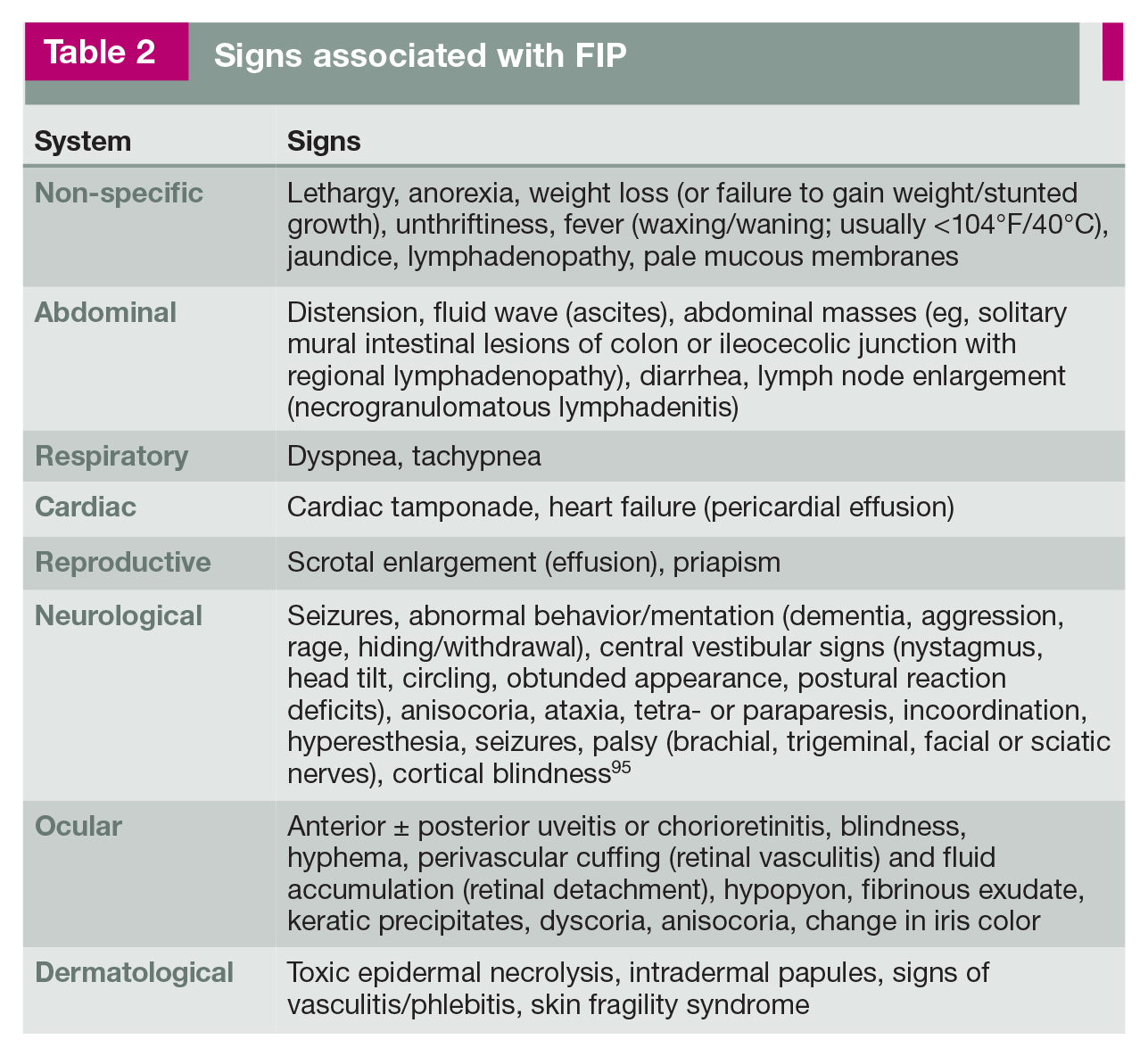
Differentiation between ‘wet’ and ‘dry’ FIP is not useful since these are not two different disease entities. Although a distinction between ‘effusive’ (non-parenchymatous) and ‘non-effusive’ (parenchymatous) forms of FIP (terms that have been used in earlier times) is important for diagnostic purposes, cats initially presenting with non-effusive FIP will commonly develop effusions at a later stage.28,46,48 Furthermore, pyogranulomatous lesions are found at necropsy in cats presenting with effusive FIP. 28 Since FIP is easier to diagnose when effusion is present, repeated ultrasound examinations should be performed to look for small pockets of abdominal or thoracic effusion that can be sampled for cytological analysis (see later).
Given that the clinical presentation of FIP is so highly variable (see Figures 2-13 in ‘the many faces of FIP’ box on pages 911-912), a comprehensive physical examination is essential (Figure 14). Mild fever in an otherwise healthy kitten can be the first clue of illness. Therefore, kittens should have their temperature taken in a cat friendly manner (eg, ear thermometer; Figure 14a) during kitten visits.


Kitten from a rescue group showing lethargy (listlessness), a common early sign of FIP. LIkewise, failure to thrive or unthriftiness is a frequent complaint in a kitten or young cat with FIP. This kitten eventually developed seizures from neurological FIP. Courtesy of Julie Jacobs

Male Ragdoll with a history of anorexia, fever and weight loss of 2 lb (0.9 kg) over 2 weeks, as well as effusion, caused by FIP. Courtesy of Stacey DeVaney

Female Oriental Shorthair from a cattery, showing significant weight loss. Jaundice and effusion were also present. Courtesy of Lisa Callaway

(a) Female spayed Persian showing depression, weakness and tremors (see supplemental video 10, which shows this patient’s neurological signs). (b) Same cat with progression of neurological signs to decreased consciousness, weakness and focal seizures. Images courtesy of Tammy Evans

Cortical blindness in a male Bengal manifesting as bilateral dilated pupils. Courtesy of Glenn Olah
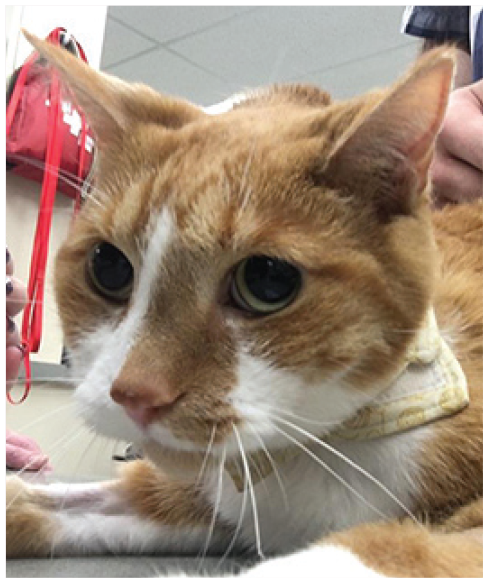
Five-year-old male castrated domestic shorthair cat with non-effusive FIP presenting as progressive weight loss, anemia and development of blindness. The cat was PCR positive on FNAs of kidney, liver, spleen and MLNs. Courtesy of Matthew Kornya

Oriental Shorthair with anterior uveitis, and corneal edema due to development of glaucoma. Courtesy of Lisa Callaway

Weight loss and progression of neurological signs accompanied the presence of anisocoria in this 12-year-old male neutered domestic shorthair cat. Courtesy of Glenn Olah
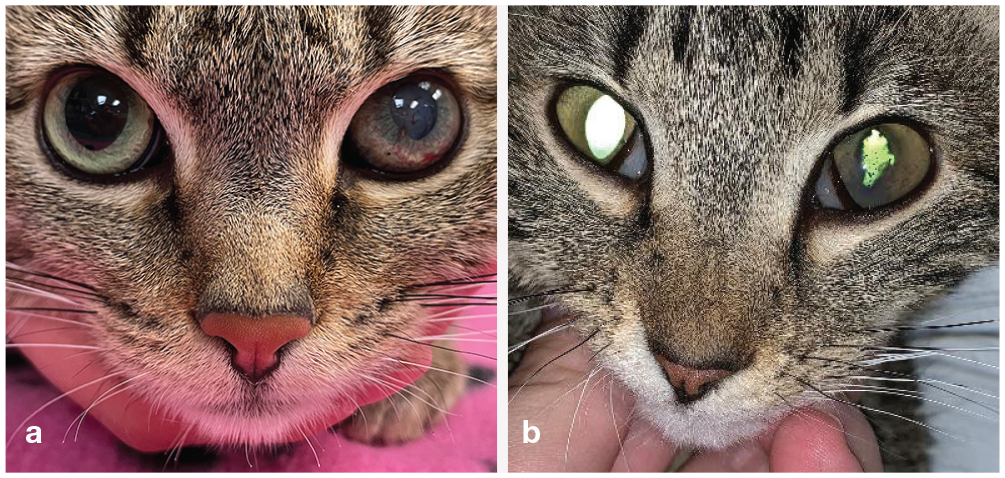
(a,b) Two cats demonstrating subtle differences in the clinical presentation of anterior uveitis caused by FIP: iris color changes, haziness in the anterior chamber (hypopyon or hyphema) and lesion(s) (keratic precipitates) in the cornea. The young cat in (b) has additional evidence of irregular pupils, which can occur with anterior uveitis caused by FIP. Repeated ophthalmoscopic examinations are indicated in difficult-to-diagnose FIP cases. Images courtesy of Marybeth Rymer (a) and Haley Batemen (b)

(a) Uveitis and leakage of fibrin into the anterior chamber of the eye. (b) Rubeosis iridis and a clump of fibrin. Images courtesy of Jessica Meekins

(a,b) Cat with abdominal effusion (ascites) presenting with abdominal distension, weight loss and decreased muscle mass. This patient is shown in supplemental video 11. Images courtesy of Stephanie Newton

Ventral inguinal area of an 8-year-old male neutered domestic shorthair cat with significant dermal vasculitis and neutrophilic splenitis that later developed effusion and was diagnosed with FIP. Cats with skin changes can be difficult-to-diagnose FIP cases. These images show the initial (a) and later (b) presentation of the vasculitis. Courtesy of Matthew Kornya Courtesy of Matthew Kornya
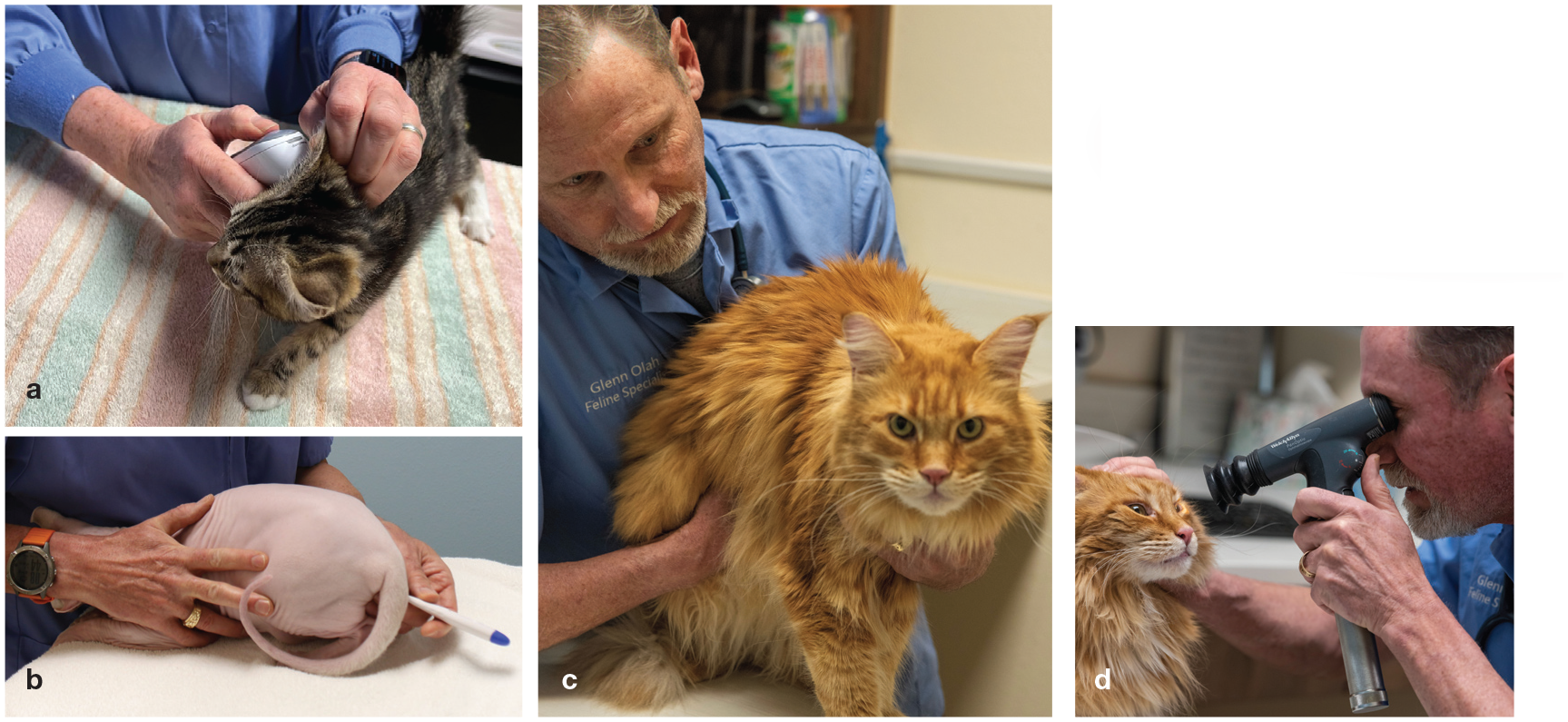
Essential components of a comprehensive physical examination. (a,b) Fever is one of the earliest non-specific signs of FIP. A temperature should be measured during routine visits in a cat friendly manner (eg, using an ear thermometer); if elevated, a rectal thermometer is used to confirm the existence of fever. (c) Palpation of the abdomen is an essential part of the physical examination for detecting enlarged MLNs, or nodular irregularities on the surface of kidneys or liver due to pyogranulomatous lesions. In addition, abdominal effusion, which is common in cats with FIP, can be noted by the presence of a fluid wave during this procedure. (d) Ocular involvement can be subtle yet creates a number of different changes including keratic precipitates, anterior and/or posterior uveitis, anisocoria and retinal detachment. A full opththalmic examination should be incorporated into the physical examination to obtain a diagnosis of FIP. Image (a) courtesy of Dianne Brown; images (b), (c), and (d) courtesy of Glenn Olah
FIP commonly leads to increased vascular permeability, vessel fluid leakage and development of protein-rich effusions.3,67,84 Effusion is the most typical change in cats with FIP.3,88,92 In one study of 224 cats with confirmed FIP, 78% of the cats had effusions. 3 In another study of 127 cats suspected to have FIP, 86% had effusions, with 92/109 having ascites and 11/109 pleural effusion. 92 By the same token FIP is one of the most common causes in cats presenting with effusion. In one study involving 197 cats with effusion, 41% of cases had FIP, 96 while in a more recent retrospective study involving 306 cats diagnosed with pleural effusion of established etiology at a university hospital, only 9% of cats had FIP. 97
Effusion can occur in various forms – ascites (most common, often with abdominal distension; Figure 12), pleural effusion (dyspnea, tachypnea and muffled heart sounds may be observed), pericardial effusion or, rarely, scro-tal effusion.3,28 In a study of pleural effusions, FIP and trauma were found to be the two most common causes in kittens and young cats. 98
FIP can cause pyogranulomatous lesions and masses. Clinical signs depend on which organ(s) are affected, with kidneys, liver, intestines (including local lymph nodes), brain and eyes most commonly involved.46,66,88,99
Pyogranulomatous lesions can be detected upon abdominal palpation as masses (eg, enlarged mesenteric lymph nodes [MLNs]) or palpable nodular irregularities on the surface of normal-sized or enlarged kidneys or liver (Figure 14c).46,66
When gastrointestinal signs occur in cats with FIP, this can pose a particular diagnostic challenge. These cats can present with a history of vomiting, diarrhea or constipation, and commonly do not have a fever. MLN enlargement occurs due to necrogranulomatous lymphadenitis,100,101 or solitary mural intestinal lesions of the ileocecocolic junction or the colon with associated regional lymphadenopathy. 102 It can be difficult to differentiate these lesions from neoplasia, 100 mycobacterio-sis 103 or toxoplasmosis. 104 Moreover, these intestinal tract manifestations can cause protein-losing enteropathy and result in low total protein and globulin values – the opposite of what would be expected in a cat with FIP.
The nervous system (eg, brain, spinal cord or meninges) is commonly affected, with neurological signs reported in up to 30% of cats with FIP in some studies.105,106 Neurological sequelae are reflective of the neuroanatomic localization and can be focal, multifocal or diffuse.28,107 In one study, 99 48% of cats had FIP-induced meningoencephalitis; in another study, 108 8% of cats had myelitis caused by FIP and, in 56% of cats with neurological FIP, the lesions were multifocal. Clinical signs can include seizures, abnormal mentation or behavior, abnormal postural reflex reactions, ataxia, hyperesthesia, nystagmus, anisocoria/dyscoria, cranial nerve deficits or cortical blindness (Table 2).
Ocular FIP manifests as unilateral or bilateral uveitis and/or chorioretinitis.88,109,110 In two studies of uveitis, FIP was a common cause, diagnosed in 16% of cats.111,112 Aqueous flare, hyphema, hypopyon, fibrinous exudate and keratic precipitates within the anterior chamber may be detected during ophthalmic examination (Figures 10 and 11).75,85 Chorio-retinitis can present as retinal perivascular cuffing due to inflammatory cell infiltrates or as focal to diffuse subretinal detach-ment.110,111 Since clinical signs of FIP can change over time, new ocular changes can arise, and therefore repeated ophthalmoscop-ic examination is indicated in difficult-to-diagnose cases. 28
Other presentations of FIP include dermato-logical changes. These can manifest as multiple non-pruritic or pruritic nodules or papules113–116 caused by pyogranulomatous necrotizing dermal phlebitis or vasculitis (Figure 13). Skin fragility syndrome has also been reported. 117 Priapism has been described as a result of granulomatous changes in tissues surrounding the penis. 118
Another example of a difficult-to-diagnose case is a cat that presents with glomerulo-nephritis caused by FIP-associated immune complexes, without any other signs of FIP. 119
Differential diagnoses
Due to the range of clinical signs and pathological manifestations of FIP, exclusion of differential diagnoses is an essential part of the diagnostic process (Table 3). An example of a case of mycobacteriosis – a potentially challenging differential diagnosis – is provided as supplemental file 12; the veterinarian was concerned about FIP, until cytology confirmed Mycobacteria species.
Diagnostic testing
A good understanding of each diagnostic test’s sensitivity, specificity, predictive value, likelihood ratio (LR) and diagnostic accuracy is important when diagnosing FIP; these are common statistical terms used to describe how accurately a diagnostic test can determine whether a cat has FIP (see box). Sensitivity, specificity and LRs are not influenced by the prevalence of disease in the studied population, while positive (PPVs) and negative predictive values (NPVs) are influenced by prevalence. 121 PPVs will be higher when the prevalence of disease is high and lower when prevalence is low, 123 and predictive values should not be applied to different populations of cats unless it is known that the prevalence of the disease is similar between these populations. If the prevalence of disease is known, a high PPV signals a good chance a cat with a positive test result has FIP. In a highly fatal disease such as FIP, the specificity of the diagnostic test is more important than the test’s sensitivity since it will help prevent euthanasia of cats misdiag-nosed with FIP. 122

When FIP is a differential diagnosis, typical baseline laboratory work includes a complete blood count, serum biochemistry, urinalysis and FeLV/FIV testing. While not patho-gnomonic, many cats with FIP will have a mild non-regenerative anemia, lymphopenia, hyperbilirubinemia, hyperglobulinemia3,83 and an albumin to globulin ratio (A:G) <0.4,3,67,123 and increased serum amyloid A and alpha-1-acid glycoprotein (AGP) concen-trations.79,124 Other clinicopathological abnormalities can be present depending on the organ system affected (see Tables 4 and 5 for a summary of diagnostic testing).
Routine diagnostic testing for FIP – an overview*
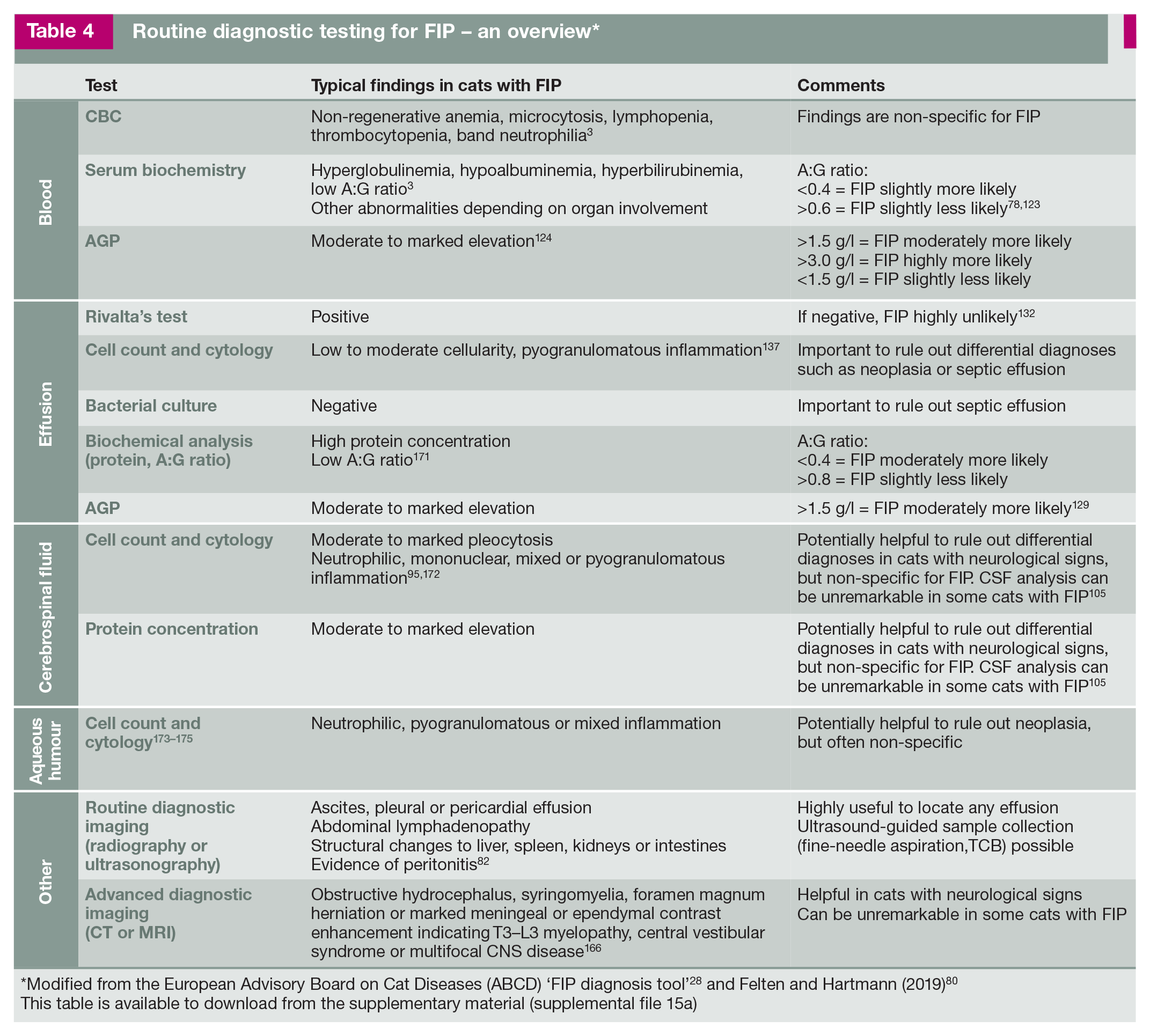
‘Modified from the European Advisory Board on Cat Diseases (ABCD) ‘FIP diagnosis tool’ 28 and Felten and Hartmann (2019) 80 This table is available to download from the supplementary material (supplemental file 15a)
Methods for direct and indirect detection of FCoV for diagnosis of FIP – an overview*

Modified from Felten and Hartmann (2019) 80
This table is available to download from the supplementary material (supplemental file 15b)
Effusion analysis
Multiple studies using different testing modalities have found that diagnostic accuracy is highest with effusion analysis, so fluid should be analyzed whenever available (see box of tips below). If there is no evidence of effusion, either overtly or via imaging, then it becomes much more difficult to diagnose FIP ante-mortem.78,80
Some FIP cases, however, can be challenging to diagnose even when effusion is present. While effusion caused by FIP is typically yellow and sticky in consistency (Figure 15), some cats have effusions that are more pink-tinged or watery/non-sticky.

Abdominal effusion due to FIP collected for sample analysis. (b) Initial assessment of an effusion sample due to FIP. The yellow color and sticky consistency (reflecting the high protein concentration in the fluid) is typical. Images courtesy of Emi Barker (a) and Matthew Kornya (b)
Moreover, the location of effusions can be unexpected. For example, intact male cats might only present with scrotal effusion and enlargement due to a serositis involving the tunica vaginalis of the testes. 127 Pericardial effusions occasionally occur without effusions in other body cavities.70,128
Typically, FIP effusions contain high protein concentrations and low white blood cell counts and, thus, vary in classification between a modified transudate or exudate, depending on the total protein concentration and total cell count. 28 However, some cats with FIP can have very high cell counts in the effusion (eg, secondary bacterial peritonitis). Even chylous effusions have occasionally been reported in cats with FIP. 71 Effusions sometimes clot upon exposure to air. Gentle warming in a water bath can aid dissolution of a clot to obtain fluid for further analysis.

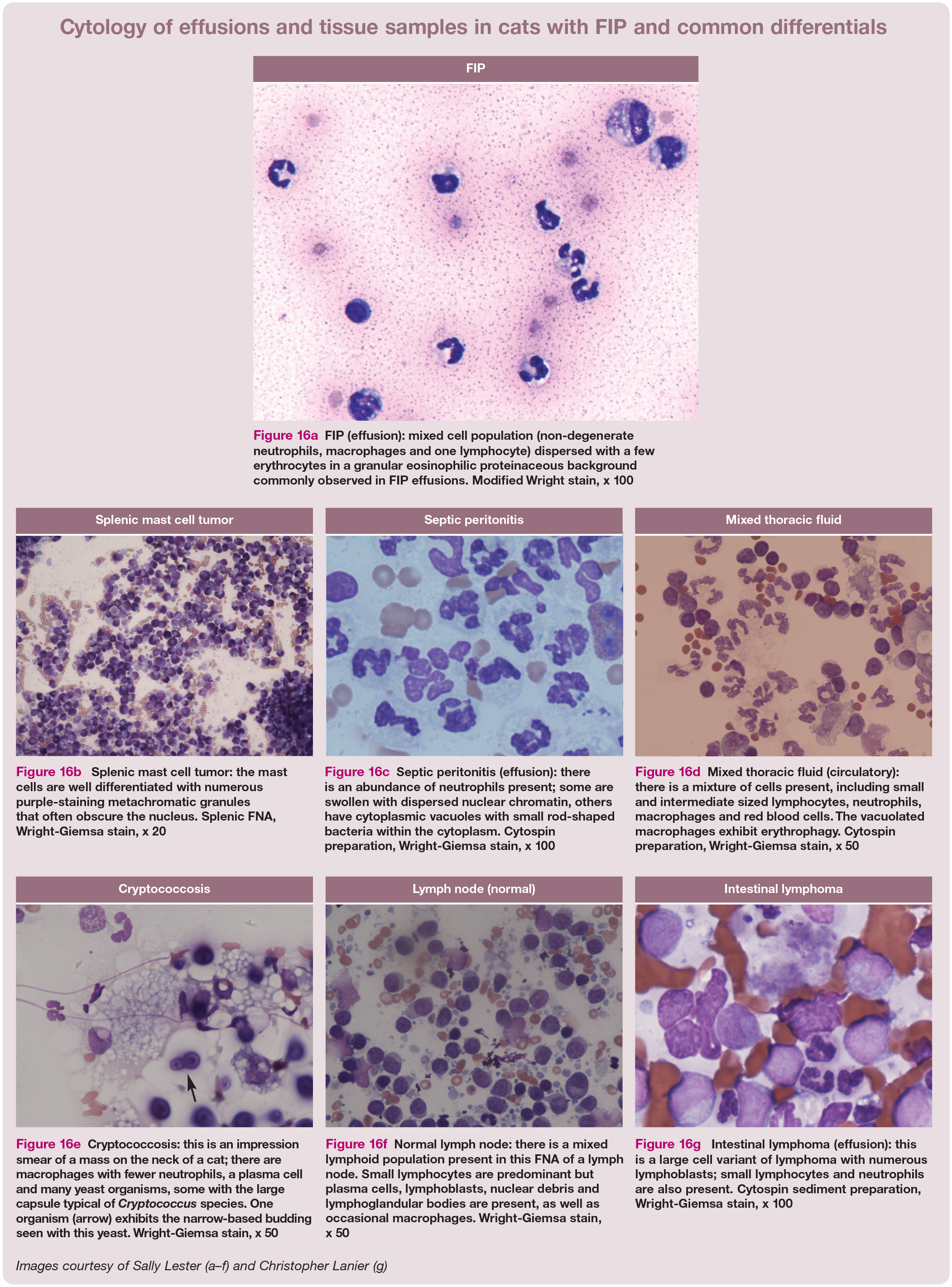
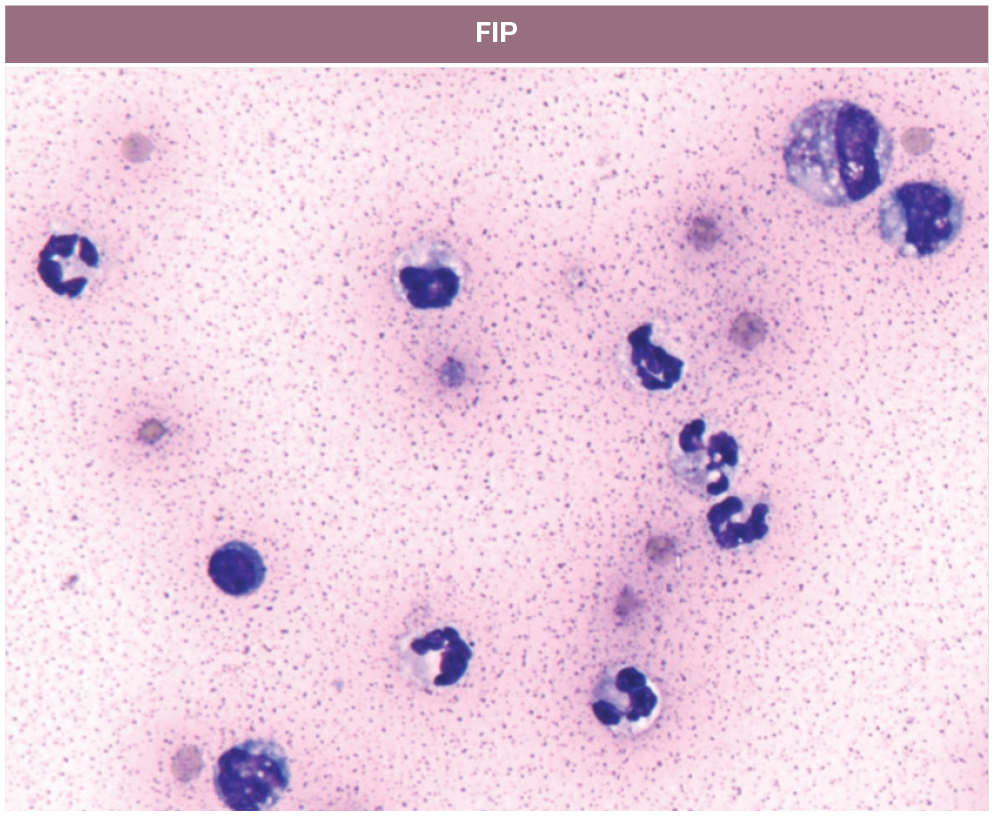
FIP (effusion): mixed cell population (non-degenerate neutrophils, macrophages and one lymphocyte) dispersed with a few erythrocytes in a granular eosinophilic proteinaceous background commonly observed in FIP effusions. Modified Wright stain, x 100

Splenic mast cell tumor: the mast cells are well differentiated with numerous purple-staining metachromatic granules that often obscure the nucleus. Splenic FNA, Wright-Giemsa stain, x 20

Septic peritonitis (effusion): there is an abundance of neutrophils present; some are swollen with dispersed nuclear chromatin, others have cytoplasmic vacuoles with small rod-shaped bacteria within the cytoplasm. Cytospin preparation, Wright-Giemsa stain, x 100

Mixed thoracic fluid (circulatory): there is a mixture of cells present, including small and intermediate sized lymphocytes, neutrophils, macrophages and red blood cells. The vacuolated macrophages exhibit erythrophagy. Cytospin preparation, Wright-Giemsa stain, x 50

Cryptococcosis: this is an impression smear of a mass on the neck of a cat; there are macrophages with fewer neutrophils, a plasma cell and many yeast organisms, some with the large capsule typical of Cryptococcus species. One organism (arrow) exhibits the narrow-based budding seen with this yeast. Wright-Giemsa stain, x 50

Normal lymph node: there is a mixed lymphoid population present in this FNA of a lymph node. Small lymphocytes are predominant but plasma cells, lymphoblasts, nuclear debris and lymphoglandular bodies are present, as well as occasional macrophages. Wright-Giemsa stain, x 50

Intestinal lymphoma (effusion): this is a large cell variant of lymphoma with numerous lymphoblasts; small lymphocytes and neutrophils are also present. Cytospin sediment preparation, Wright-Giemsa stain, x 100 Images courtesy of Sally Lester (a-f) and Christopher Lanier (g)
Laboratory analysis will typically reveal a protein content >35 g/1,
9
an A:G ratio <0.4 and a cell count <5
Cytology and bacterial culture can play an important role in differentiating FIP from other causes of effusion. Examples of cytology samples obtained from effusions and tissues are illustrated in Figure 16, highlighting the importance of this diagnostic technique. Generally, cytology of FIP effusions shows pyogranulomatous inflammation with macro-phages, non-degenerate neutrophils and very few lymphocytes. 80 This contrasts, for example, with neutrophilia, toxic neutrophils and intracellular bacteria present on a cytological specimen, which would point toward a bacterial peritonitis/pleuritis; or neoplastic cells, such as large lymphoblasts with prominent nucleoli, which would indicate lymphoma. The box on page 915 contains helpful tips for preparing diagnostic cytology slides in-house.
The Rivalta’s test is a simple, quick and inexpensive diagnostic tool for examining effusions.78,131 It is a valuable method to rule out FIP, especially when there are cost concerns, such as in shelter situations. The test is performed by adding a drop of effusion to a slightly acetic solution (see box of helpful tips). High protein content and the presence of inflammatory mediators – characteristics of effusions caused by FIP – will cause the drop to precipitate, indicating a positive test result (Figure 17). If the drop dissipates, this is considered a negative test result. Effusion storage for up to 3 weeks does not influence test results. However, test result interpretation can be challenging due to subjectivity and is dependent on the experience of the clinician. 131


Positive Rivalta’s test. Courtesy of Katrin Hartmann, LMU Munich
In earlier studies, the Rivalta’s test demonstrated a high sensitivity and specificity for the diagnosis of FIP.78,96 However, in a large study of 851 cats, sensitivity and specificity were lower (91% and 66%, respectively), although the NPV was still high and the PPV reached almost 90% in cats less than 2 years of age. 132 This means that a negative Rivalta’s test result makes FIP very unlikely. A positive Rivalta’s test result increases the suspicion of FIP, especially in a young cat; however, this must be confirmed with other tests, as positive results can also occur in cats with bacterial peritonitis/pleuritis or lymphoma. 132 Fluid cytology and bacterial culture can help discriminate between these causes. 133
Diagnostic imaging
Abdominal focused assessment with sonography for trauma, triage and tracking (AFAST) is a first-line screening test that allows the attending clinician to quickly identify whether free fluid and soft tissue abnormalities of the relevant target organ are present. This test is widely used in the emergency setting for the quick detection of free fluid but it also can be used as a point-of-care ultrasound (POCUS) screening in cats suspected of having free fluid associated with FIP. The technique, which involves placement of the ultrasound probe in four different locations on the abdomen, is relatively easy to learn. Moreover, AFAST is a rapid (<3 mins), realtime imaging modality that has low patient impact. 134 Since many cats do not initially present with a noticeable abdominal fluid wave, or their disease transitions from being primarily non-effusive to effusive, serial AFAST examinations are an excellent diagnostic tool for identifying these cases.
Figures 18–21 are positive AFAST images showing anechoic free fluid (alongside each image [a] is an identical, labeled version [b]). Such findings in a cat suspected of having FIP are an indication for immediate fluid sampling and analysis.
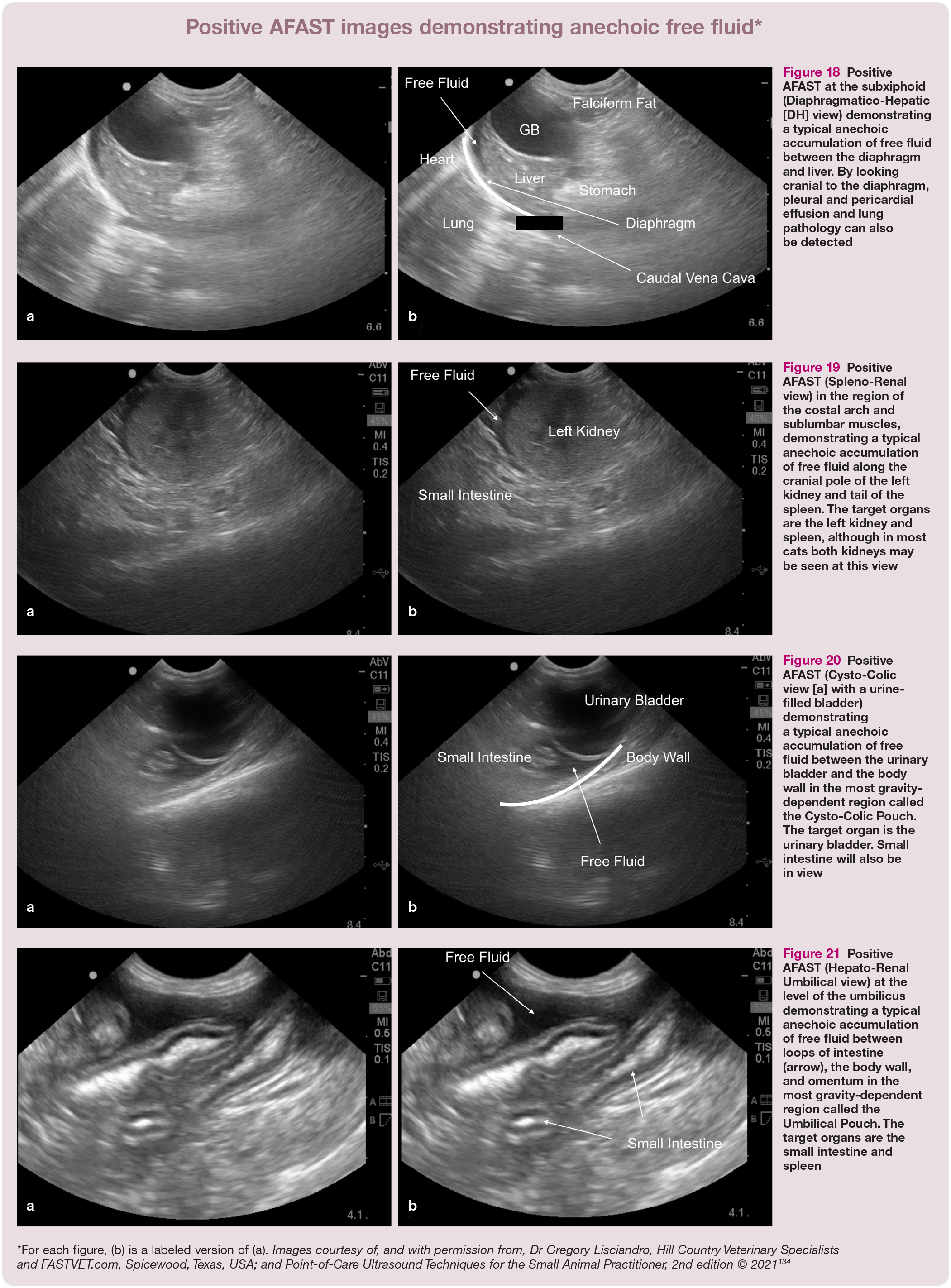
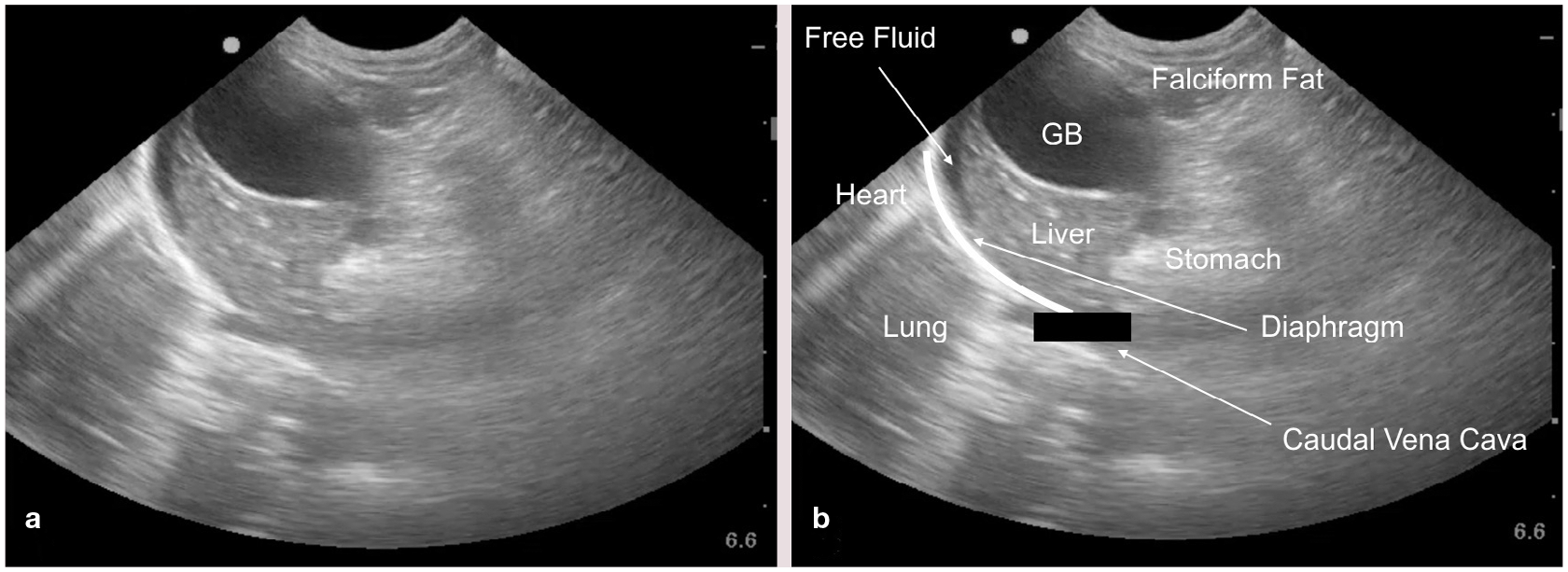
Positive AFAST at the subxiphoid (Diaphragmatico-Hepatic [DH] view) demonstrating a typical anechoic accumulation of free fluid between the diaphragm and liver. By looking cranial to the diaphragm, pleural and pericardial effusion and lung pathology can also be detected

Positive AFAST (Spleno-Renal view) in the region of the costal arch and sublumbar muscles, demonstrating a typical anechoic accumulation of free fluid along the cranial pole of the left kidney and tail of the spleen. The target organs are the left kidney and spleen, although in most cats both kidneys may be seen at this view

Positive AFAST (Cysto-Colic view [a] with a urine-filled bladder) demonstrating a typical anechoic accumulation of free fluid between the urinary bladder and the body wall in the most gravity-dependent region called the Cysto-Colic Pouch. The target organ is the urinary bladder. Small intestine will also be in view

Positive AFAST (Hepato-Renal Umbilical view) at the level of the umbilicus demonstrating a typical anechoic accumulation of free fluid between loops of intestine (arrow), the body wall, and omentum in the most gravity-dependent region called the Umbilical Pouch. The target organs are the small intestine and spleen

Mesenteric lymphadenopathy. The hypoechoic structure in the mid-abdomen may be screened for during AFAST and its target organ approach and detected during complete abdominal ultrasound studies. In (a) the image is unlabeled and in (b) labeled with the mesenteric lymph node outlined with an oval and small intestine (SI). Enlargement has been defined by measurements of thickness of being no more than 5 mm; however, some may be considered normal up to 7-8 mm. The ratio of short axis to long axis is another parameter that may be used, with normal <0.5. 135 Images courtesy of, and with permission from, Dr Stephanie Lisciandro, Hill Country Veterinary Specialists and FASTVET.com, Spicewood, Texas, USA © 2021
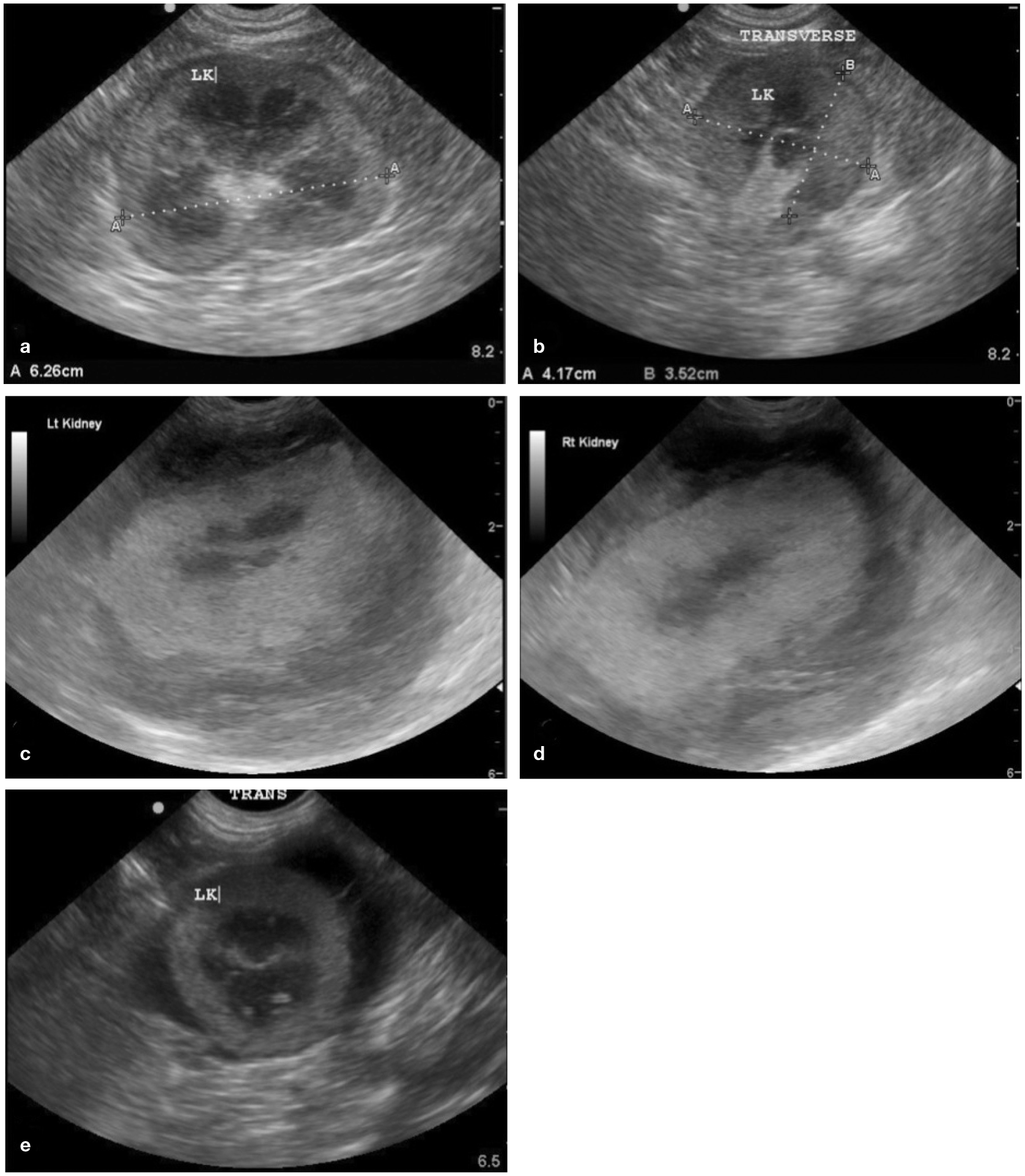
Renomegaly, Poor Architecture, Perirenal Fluid. In (a) and (b) there is renomegaly and a small rim of subcapsular fluid that may be suspected during AFAST. The length should never exceed 4.5 cm in a cat and this kidney clearly measures over 6 cm. Measurements are also increased for width and height in its transverse plane. In (c) and (d) there is obvious loss of expected architecture in both kidneys; again, a finding that could be suspected during AFAST using its target organ approach. There is perirenal fluid associated with these kidneys as well. In (e) is another example of perirenal fluid. By detecting these abnormalities during AFAST as part of the initial diagnostics of the cat, the information is captured quickly and the diagnostic plan is potentially more streamlined. LK = left kidney. Images courtesy of, and with permission from, Drs Gregory and Stephanie Lisciandro, Hill Country Veterinary Specialists and FASTVET.com, Spicewood, Texas, USA; and Point-of-Care Ultrasound Techniques for the Small Animal Practitioner, 2nd edition © 2021 134

Right lateral (a) and ventrodorsal (b) thoracic radiographs of a cat with pleural effusion. Images courtesy of, and with permission from, Dr Gregory Lisciandro, Hill Country Veterinary Specialists and FASTVET.com, Spicewood, Texas, USA; and Point-of-Care Ultrasound Techniques for the Small Animal Practitioner, 2nd edition © 2021134
As a screening test for free fluid and soft tissue abnormalities, AFAST can be utilized as both a monitoring tool and an extension of the physical examination. 134 (See supplemental file 14 for specific instructions.) Further, if free fluid is not detected and the history, signalment and physical examination findings are highly suggestive of FIP, then a complete abdominal ultrasound examination is indicated. There have been limited studies investigating the diagnostic utility of abdominal ultrasound in the ante-mortem diagnosis of FIP. A retrospective study that reviewed abdominal ultrasonographic results associated with FIP in 16 cats demonstrated that, when FIP is under consideration, certain findings such as abdominal lymphadenopathy (mesenteric [Figure 22], splenic, etc), renal architectural changes, presence of effusion (peritoneal or retroperitoneal [Figure 23]) and intestinal changes should elevate FIP to the top of the differential diagnosis list. 82
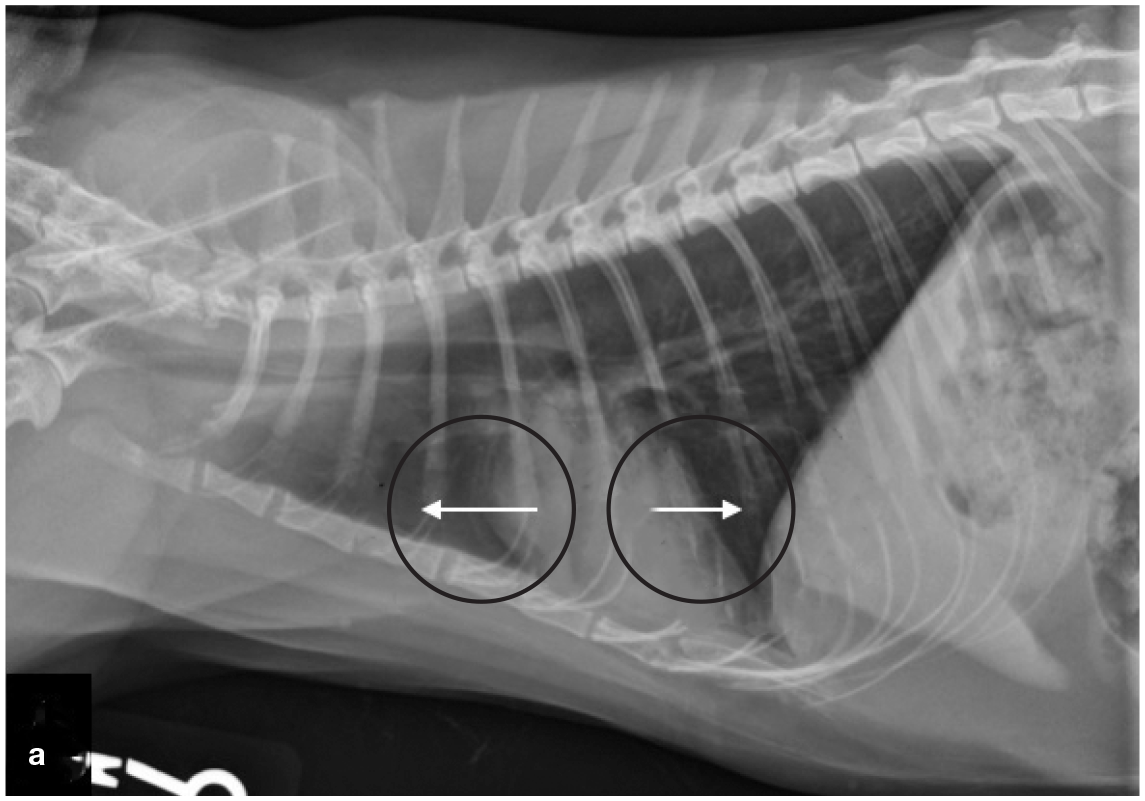
Depending on the clinical presentation, physical examination findings, the imaging equipment available and the veterinarian’s assessment of the patient’s clinical status, thoracic radiographs may be indicated if pleural effusion is suspected (Figure 24). If the cat is extremely dyspneic, a standing lateral or dorsoventral radiographic view is preferable or thoracic POCUS since these cats are at an increased risk for respiratory arrest.
In cases where there is no obvious respiratory distress but fluid is still suspected, thoracic focused assessment with sonography for trauma, triage and tracking (TFAST) should be performed since, as with abdominal effusion, ultrasound is more likely to detect small amounts of fluid (Figures 25 and 26). 85 (See supplemental file 14 for specific instructions.) The AFAST DH view can be combined with the right pericardial site (PCS) TFAST view to identify whether pleural effusion is present. 134 Pericardial effusion has been associated with FIP, but is uncommon. 70 However, while radiography of the thorax is usually diagnostic for pleural effusion, the gold standard for pericardial effusion is cardiac ultrasonogra-phy. The entire heart is imaged so that the hyperechoic pericardium is visible. If fluid has accumulated between the heart and the pericardium, the apex of the heart will appear rounded (Figure 27). 134

Pleural effusion located in the cardiac diaphragmatic pouch. During the ‘TFAST slide’, the probe is moved caudally from the heart to the diaphragm where the most gravity-dependent area is located, the cardiac diaphragmatic pouch. Images courtesy of, and with permission from, Dr Gregory Lisciandro, Hill Country Veterinary Specialists and FASTVET.com, Spicewood, Texas, USA; and Point-of-Care Ultrasound Techniques for the Small Animal Practitioner, 2nd edition © 2021 134
(a,b,c) Pleural effusion located in the cardiac cervical pouch. During the ‘TFAST slide’, the probe is moved cranially across the heart to the cardiac cervical pouch, which is the most gravity-dependent area in the standing or sternal patient positions and is located cranial to the heart, which is identified by the arrows. Images courtesy of, and with permission from, Dr Gregory Lisciandro, Hill Country Veterinary Specialists and FASTVET.com, Spicewood, Texas, USA; and Point-of-Care Ultrasound Techniques for the Small Animal Practitioner, 2nd edition © 2021 134

Pleural and pericardial effusion. The entire heart is visible. Images courtesy of, and with permission from, Dr Gregory Lisciandro, Hill Country Veterinary Specialists and FASTVET.com, Spicewood, Texas, USA; and Point-of-Care Ultrasound Techniques for the Small Animal Practitioner, 2nd edition © 2021 134
Histopathology
Histopathology of lesions is quite specific for a diagnosis of FIP, but sampling is invasive or can only be performed post mortem. Generally, sampling of multiple tissues is preferred to improve sensitivity 58 and can include MLNs, small and large intestines, omentum, spleen, kidney and liver. 136 Pathology will typically show vasculitis with perivascular necrosis. Pyogranulomatous lesions can be identified around the vasculature, especially on serosal surfaces.137,138 Immunohistochemistry (IHC; see later discussion on ‘Detection of viral antigen’) will reveal FCoV antigen associated with macrophages within the lesions; thus IHC is more specific than histopathology alone 136 and is considered the gold standard for diagnosis. 80
A cat with suspected FIP is often too sick or debilitated to undergo surgical sample collection for histopathology and IHC. Recent investigations of ante-mortem diagnosis of FIP have used minimally invasive techniques such as ultrasound-guided fine-needle aspiration58,139,140 or Tru-cut biopsy (TCB) of kidney and liver. 141 One study investigated whether certain combinations of diagnostic tests (histology, IHC and/or nested RT-PCR [RT-nPCR]) performed on TCB samples obtained post mortem could be utilized in making an ante-mortem diagnosis of FIP. 136 Histological lesions previously associated with FIP 88 were found to be present in some organs, but not all; and some cats with organ lesions consistent with FIP had diseases other than FIP. Based on these findings, it was recommended that biopsies be obtained from more than one organ in order to increase the odds of diagnosing FIP. 136 Tips for obtaining a diagnostic FNA are provided in the box.


Detection of viral nucleic acid
Detection of viral RNA is one way of confirming FCoV as the underlying etiological agent (see box below). Real-time RT-PCR (also referred to as RT-qPCR) for FCoV nucleic acids can be performed on blood, other fluids (eg, effusion, CSF or aqueous humour), and tissue samples or FNAs. However, since less-virulent FCoV can spread systemically in cats without FIP, and PCR methods are very sensitive, the detection of FCoV RNA by RT-qPCR in blood, fluid or tissue is not necessarily specific for a diagnosis of FIP (see box on page 923).38,122,146,147 One study looked into the diagnostic utility of RT-qPCR of MLN FNAs in cats without effusion and found an overall sensitivity of 90% and specificity of 96.1%. 140 Additionally, it has been shown that the sensitivity of RT-qPCR did not differ significantly between FNAs and incisional biopsies; thus, the more invasive biopsy technique is not mandatory for obtaining sample material for RT-qPCR. 58
RT-qPCR can indicate the amount of virus in the sample. Cats with FIP generally have higher viral loads than healthy FCoV-infected cats,60,61,148 meaning that a positive RT-qPCR result with high viral load moves FIP up the list of differential diagnoses. Fluids and fresh tissue are best suited for RT-qPCR, but fixed tissue is acceptable. 149

Mutations within the S protein gene can also be detected by special S gene real-time RT-PCR using specific primers58,122,150–152 or by sequencing after real-time RT-PCR.60,79,153 For the mutation-specific RT-qPCRs, these typically will be combined into one assay, referred to as multiplexed, for interpretation of results. The assay will involve one PCR amplicon set targeting a conserved region that will detect all FCoV species, and a second set where one of the primers (forward or reverse) is specific at the 3’ end for the mutation being looked for, and the mutation-specific primer will only bind if the mutation is present. It will not bind to sequences other than the mutation being looked for, so there will be several possible outcomes for the diagnostic:
✜ FCoV RT-qPCR−/spike mutant RT-qPCR−(eg, no FCoV infection);
✜ FCoV RT-qPCR+/spike mutant RT-qPCR−(eg, FCoV infection, non-spike mutant);
✜ FCoV RT-qPCR+/spike mutant RT-qPCR+ (eg, FCoV infection, spike mutant); and
✜ FCoV RT-qPCR-/spike mutant RT-qPCR+ (eg, something is wrong with the assay).
In a recent study, FCoV containing S gene mutations was found in at least one body fluid or tissue in all cats with FIP, but the distribution varied from cat to cat, hence a recommendation that multiple samples be analyzed to increase sensitivity. 58 The specificity of detection of S gene mutations for FIP is highly controversial. While specificity of the S gene real-time RT-PCR was good in effusion and tissues in some studies, false-positive results have been frequently demonstrated.122,152,154 The usefulness of the special S gene RT-PCR using specific primers has been questioned and detection of S gene mutations by sequencing might be more specific for diagnosing FIP. However, even the sequencing approach was not able to improve specificity significantly in a few studies.60,79
Detection of viral antigen
FCoV antigen can be detected by staining the antigen within its target cells, fluid or tissue macrophages, using different immuno-staining methods. The binding of antibodies to host cell-associated FCoV antigens is subsequently visualized (by IHC, ICC or immunofluorescence [IF]), producing a color change or fluorescence.
FCoV antigen detection in macrophages of affected tissues via immunostaining is highly specific and reliable,136,155,156 as long as it is performed with appropriate controls and reagents that prevent non-specific binding of anti-FCoV antibodies to the tissues. Hence, it is considered the gold standard method for diagnosing FIP.28,80,88,136,157 Samples of affected tissues can be taken during exploratory laparotomy, by laparoscopy or ultrasound-guided biopsy, or at necropsy. Although immunostaining cannot differentiate between non-mutated and mutated viruses, the presence of a high amount of antigen, which is necessary for positive staining, indicates high virus replication rates within macrophages -the key event in the pathogenesis of FIP. However, negative IHC results do not exclude FIP because FCoV antigens can be variably distributed within lesions.58,88,136,141 Ideally, samples should be taken from areas with histopathological changes.
FCoV immunostaining can also be performed on cytology samples from tissues, effusions, CSF or aqueous humour using ICC or IF.
ICC on MLN FNAs has been suggested as a non-invasive method to diagnose FIP, but one study showed only moderate sensitivity of 53% and a false-positive result in a cat with lymphoma. 139 A study comparing the efficacy of FCoV immunostaining in hepatic and renal TCBs and FNAs found that sensitivity in both tissues was poor (24% and 17-31% for hepatic TCBs and FNAs, respectively; 17% and 11-20% for renal TCBs and FNAs, respectively), although combining the two sampling techniques slightly increased sensitivity (to 38% for liver and 14% for kidney samples). 141
FCoV immunostaining of effusion has had variable sensitivity in different studies, ranging from 57% to 100%.78,96,130,158,159 False-negative results are possible if the effusion does not contain enough macrophages or if anti-FCoV antibodies mask the FCoV anti-gen.78,160 Further, while immunostaining has long been thought to be very specific, false-positive results have occurred in more recent studies. In one study, 2/7 cats with effusion due to other diseases (heart failure and cholan-giocarcinoma) had false-positive results by IF. 159 In another study, 8/29 cats with effusions due to other diseases (heart failure in three cats and neoplasia in five cats) tested false positive by ICC. 130 ICC with dual staining for macrophages as well as FCoV antigen might potentially increase specificity. 161
FCoV immunostaining using ICC was successful in detecting FCoV antigen in the CSF of a cat with neurological FIP. 162 However, one larger study evaluating CSF ICC in cats with and without FIP (with or without neurological signs) found positive results in 17/20 cats with FIP but also in 3/18 cats without FIP (one cat each with mediastinal lymphoma, menin-goencephalitis, and hypertensive angiopathy with brain hemorrhage), indicating low specificity. 163 FCoV antigen has also been detected by ICC in aqueous humour samples, with a sensitivity of 64% and specificity of 82%; false-positive results occurred in one control cat with lymphoma and one control cat with pulmonary adenocarcinoma.130,151
Detection of anti-FCoV antibodies
Detection of antibodies against an infectious agent is an indirect method of identifying an underlying etiological agent. Anti-FCoV antibodies can be detected by enzyme-linked immunosorbent assay (ELISA), or indirect immunofluorescence antibody (IFA) or rapid immunomigration tests. 164 Due to a lack of standardization among antibody targeting methods, results of different tests and from different laboratories can vary; hence they are difficult to compare, and some can be unreliable. Additional considerations for laboratory test submissions are summarized in the box.
Measurement of anti-FCoV antibodies can be used to detect FCoV infection; however, the presence of antibodies is not useful for diagnosing FIP, since antibodies are not only present in cats with FIP, but also in healthy FCoV-infected cats or FCoV-infected cats with other diseases. 78 Thus antibodies against FCoV in feline serum or plasma indicate only that the cat has had contact with FCoV through natural infection or vaccination, with antibodies usually developing on days 10-28 post-infection.39,165 Although cats with FIP tend to have higher anti-FCoV antibody titers than cats without FIP, there is often no difference in median anti-FCoV antibody titers between healthy cats and cats with FIP. 78 Moreover, negative antibody test results do not rule out FIP, since up to 10% of cats with FIP do not have anti-FCoV antibodies. 78 This percentage was even higher in cats with neurological FIP without effusion. 166

In addition to blood (serum, PBMCs or plasma), anti-FCoV antibody testing has also been performed on a variety of other sample types including effusion, CSF and aqueous humour. However, the diagnostic value in using these samples is also limited since antibodies can easily leak from the blood into high-protein effusions or cross a disturbed blood-brain barrier. 167 Additionally, negative results can occur in cats with FIP due to binding of antibodies by FCoV, as has been shown for effusion.164,168 Overall, antibody testing in serum, plasma or any other fluid is not recommended for the diagnosis of FIP. 80
Summary of diagnostic testing for FIP
The difficulty when trying to diagnose FIP is that both indirect and direct detection methods only indicate the presence of FCoV and not specifically the mutated virus that leads to FIP. FCoV is present in many cats and even detection outside of the intestinal tract in effusions, tissues, CNS or aqueous humour is not diagnostic for FIP, as harmless non-mutated FCoV can be found everywhere in the body.148,169,170 Detection of mutated FCoV (viruses containing certain S gene mutations) does not entirely solve the problem as viruses containing these mutations have been found in the tissues or fluids of cats without FIP.60,61,79,80,154
Probably the best approach for a relatively confident dagnosis is to find high quantities of FCoV RNA in the body, as it is well known that cats with FIP exhibit much higher viral loads than healthy FCoV-infected cats. Thus, a positive RT-qPCR result with a high viral load, if accompanied by consistent clinical signs and clinical pathology results, is at least very suggestive of FIP.60,61,154
Definitively diagnosing FIP remains a challenge. The only way to truly confirm FIP is to detect the viral antigen associated with macrophages within lesions using IHC.80,136 However, if the cat’s history and presenting clinical signs are consistent with FIP and the FCoV RNA is detected in fluid or tissue using real-time RT-PCR, then there is a significant chance that the cat has FIP. 80 Ultimately, the diagnostic approach to diagnosing FIP must be tailored to the specific presentation of the individual cat. Tables 4 and 5 (provided as supplemental files 15a and 15b) contain pertinent diagnostic test findings that may significantly increase the index of suspicion that FIP is the underlying cause of a cat’s clinical signs. Further, algorithms adapted for the different presentations of FIP have been provided by expert groups (see supplemental file 16) to help guide practitioners when working up a case.
Key Points
✜ FCoV infection, usually asymptomatic, is common in most cat populations because the virus is highly contagious and spreads efficiently via fecal-oral transmission, leading to high prevalence in multi-cat environments, such as breeding catteries, shelters, rescue facilities and animal hoarding situations.
✜ A diagnosis of FIP can be challenging due to a lack of pathognomonic clinical signs and laboratory changes, especially when no effusion is present. There is no one non-invasive confirmatory test available for cats ante-mortem without effusion. Likewise, no diagnostic procedure is available to predict which FCoV-infected cats will eventually develop FIP.
✜ Cats with FIP typically have non-specific clinical signs such as anorexia, lethargy, a waxing/waning fever (often non-responsive to antibiotics), jaundice, lymphadenopathy and weight loss or, in kittens, failure to gain weight. More specific clinical signs include presence of effusion, anterior uveitis or neurological signs.
✜ Several risk factors (host, viral and environmental) have been identified for the development of FIP, including a recent stressful event noted as part of the cat’s history.
✜ The clinician must consider the individual patient’s history, signalment and comprehensive physical examination findings when selecting diagnostic tests and sample types in order to build the index of suspicion ‘brick by brick’.
✜ Repeat examinations, including looking for pockets of effusions for cytological analysis, are often necessary in a difficult-to-diagnose case. If the cat presents with effusion, diagnosing FIP can be straightforward as tests using effusion generally have much higher predictive values than those using blood. The Rivalta’s test is one such example of an easy, rapid and inexpensive test on an effusion sample that has good sensitivity for excluding FIP (91-100%). If the Rivalta’s test is negative, other potential causes for the effusion are much more likely than FIP.
✜ If no effusion is present, diagnosis can become quite challenging due to the variety and non-specificity of possible clinical signs. A good understanding of each diagnostic test’s sensitivity, specificity, predictive value, likelihood ratio and diagnostic accuracy is important when building a case for FIP. Before proceeding with any diagnostic test or commercial laboratory profile, the clinician should be able to answer the questions of ‘why this test?’ and ‘what do the results mean?’
✜ When considering PCR testing, it is important to bear in mind that one RT-PCR may not be identical to another (eg, in terms of FCoV target sequence). Prior to submitting a sample, it is recommended the clinician contacts the laboratory for information regarding what that particular assay is targeting, to help guide sample selection for submission.
✜ Unlike many other feline diseases, blood samples are generally not helpful in the diagnosis of FIP.
✜ The only way to truly confirm FIP is to detect the virus (either by antigen staining or RT-PCR) within tissue and this requires taking histopathology samples. FCoV antigen detection in macrophages of affected tissues via immunostaining is considered the gold standard for diagnosing FIP.
✜ Ultimately, the approach to diagnosing FIP must be tailored to the specific presentation of the individual cat.

Supplemental Material
Supplemental figure 1
FCoV genome
Supplemental Material
Supplemental figure 2
origin of FCoV serotype II
Supplemental Material
Supplemental figure 3
Fecal-oral transmission of FCoV
Supplemental Material
Supplemental figure 4
Pathogenesis pathway from less-virulent FCoV infection to FIP-associated FCoV
Supplemental Material
Supplemental figure 5a
Pathogenesis of FIP
Supplemental Material
Supplemental figure 5b
Macrophage activation, complement activation and antibody-mediated viral infection of macrophages
Supplemental Material
Supplemental figure 6
Host immune response to FCoV infection
Supplemental Material
Supplemental figure 7
Purebred cat breeds with FIP representation in two studies
Supplemental Material
Supplemental figure 8
Diagnostic work-up for FIP ‘brick by brick’
Supplemental Material
Supplemental figure 9
Health-related history questionnaire
Supplemental Material
Supplemental figure 12
A complicated differential diagnostic case in a cat suspected of having FIP
Supplemental Material
Supplemental figure 14
Instructions for TFAST and AFAST
Supplemental Material
Supplemental figure 15a
Routine diagnostic testing for FIP – an overview
Supplemental Material
Supplemental figure 15b
Methods for direct and indirect detection of FCoV for diagnosis of FIP – an overview
Supplemental Material
Supplemental figure 16
Diagnostic work-up of FIP: diagnostic approach
Footnotes
Acknowledgements
The Task Force gratefully acknowledges the contribution of Dr Gregory Lisciandro, Dr Sally Lester, Dr Brian Geiss, Daniel Dominguez and Heather O’Steen in the preparation of the guidelines manuscript. ![]() was produced by graphic artist Rachael Weiss. Supplemental file 16 is included with kind permission of the European Advisory Board on Cat Diseases (ABCD).
was produced by graphic artist Rachael Weiss. Supplemental file 16 is included with kind permission of the European Advisory Board on Cat Diseases (ABCD).
Supplementary material
The following files are available online at jfms.com and can be accessed at catvets.com/fip and everycat.org/aafp-fip-guidelines:
✜ Supplemental figure 1: FCoV genome.
✜ Supplemental figure 2: Origin of FCoV serotype II.
✜ Supplemental figure 3: Fecal-oral transmission of FCoV.
✜ Supplemental figure 4: (a-d) Pathogenesis pathway from less-virulent FCoV infection to FIP-associated FCoV.
✜ Supplemental file 5a: Pathogenesis of FIP.
✜ Supplemental file 5b: Macrophage activation, complement activation and antibody-mediated viral infection of macrophages.
✜ Supplemental figure 6: Host immune response to FCoV infection.
✜ Supplemental file 7: Purebred cat breeds with FIP representation in two studies.
✜ Supplemental figure 8: Diagnostic work-up for FIP ‘brick by brick’.
✜ Supplemental file 9: Health-related history questionnaire.
✜ Supplemental video 10: Progressive neurological FIP in the cat shown in Figure 5.
✜ Supplemental video 11: Abdominal effusion due to FIP in the cat shown in Figure 12.
✜ Supplemental file 12: A complicated differential diagnostic case in a cat suspected of having FIP.
✜ Supplemental video 13: Rivalta’s test being performed.
✜ Supplemental file 14: Instructions for TFAST and AFAST.
✜ Supplemental file 15a: Routine diagnostic testing for FIP – an overview.
✜ Supplemental file 15b: Methods for direct and indirect detection of FCoV for diagnosis of FIP – an overview.
✜ Supplemental file 16: Diagnostic work-up of FIP: diagnostic approach.
Conflict of interest
The Task Force members have no conflicts of interest to declare.
Funding
These Guidelines were supported by an educational grant from Boehringer Ingelheim Animal Health USA Inc. The members of the Task Force received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

