Abstract
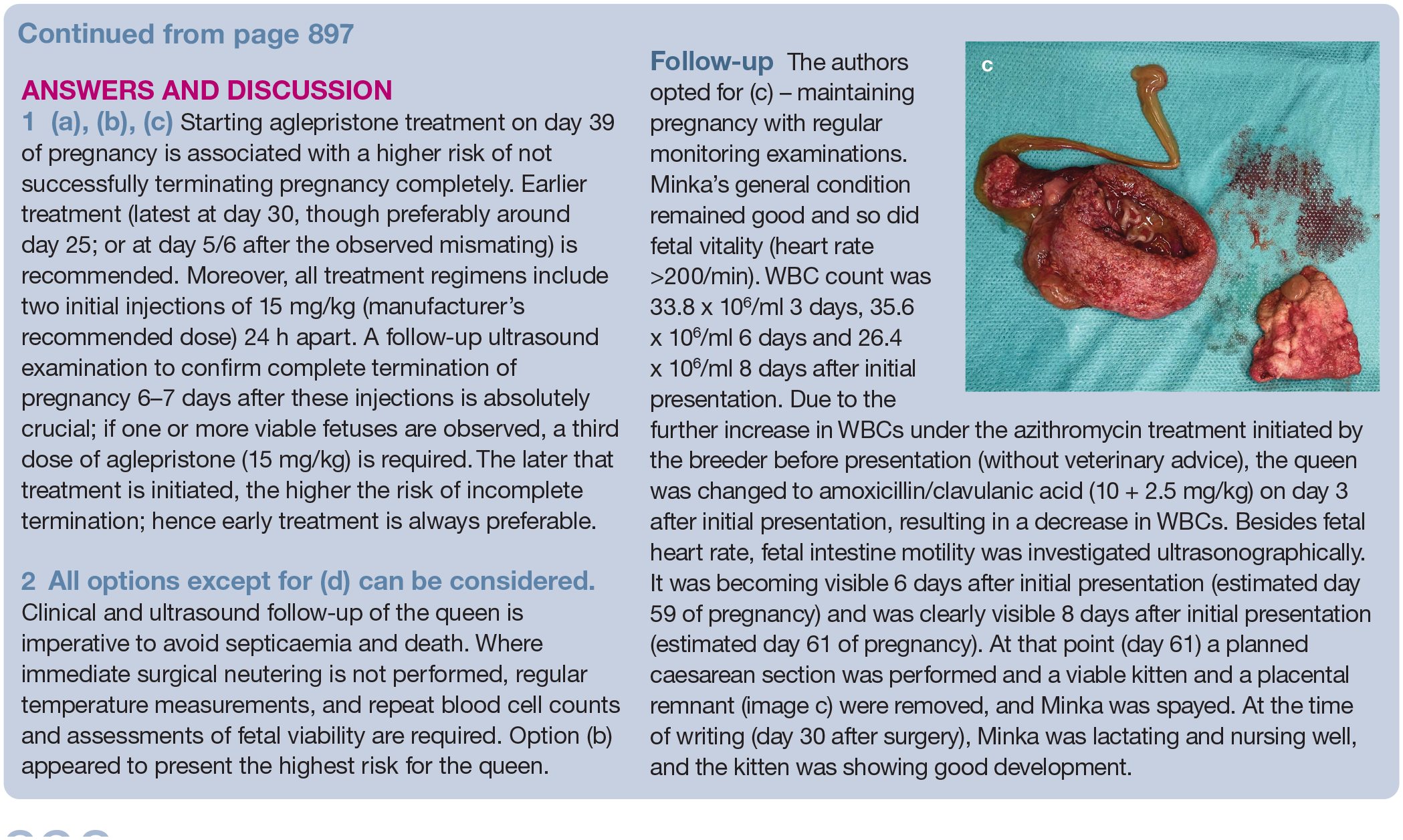
Practical relevance:
Many veterinarians – even those engaged in small animal reproduction – are uncomfortable with taking care of cat breeders or being in charge of catteries. Likely reasons for this are that feline reproduction is largely under-represented in undergraduate and graduate reproduction teaching, as well as in postgraduate education and science, whereas cat breeders themselves are often well informed and actively share knowledge with other cat breeders via the internet and social media.
Clinical challenges:
A variety of problems can exist within a cattery, and collaboration between veterinarian and breeder to solve these is ultimately beneficial for the breeder, for the veterinarian, and for the health and wellbeing of the individual cats and of the cattery as a whole.
Aim:
This review presents a comprehensive overview of aspects of cattery management that might negatively impact reproduction, including sanitation, hygiene and infectious disease control. It also discusses monitoring of reproductive performance, breeding recommendations, and the diagnostic and therapeutic approach to some common and specific problems.
Equipment and technical skills:
Reproductive management in the cattery requires no specialised equipment, as such. What it does need is an open mind, in terms of seeing how things are done, an open ear, for listening to the breeder, and the veterinarian’s clinical skills in palpation, auscultation and, where required, further examination of the animals. Keeping abreast of the latest information on infectious diseases, disinfection and genetics ensures proper advice is provided.
Evidence base:
Current knowledge of reproductive management in catteries is summarised in order to apply an evidence-based approach, whenever possible. Notwithstanding, much of the information remains empirical.

Introduction
Cat breeding has increased significantly in recent years. Teaching of small animal reproduction within veterinary curricula still, however, focuses mainly on canine reproduction. Similarly, research in the field of feline reproduction remains very limited and under-represented compared with canine reproduction. 1 As a consequence, knowledge about cat breeding and cat breeders’ practices is scarce among small animal practitioners, unless they are breeding cats themselves. A recent survey-based study reported that US dog and cat breeders ‘wanted veterinarians to learn more about reproductive health and disease’. 2
The perceived lack of knowledge and/or interest of veterinarians in cat breeding often results in cat breeders communicating between themselves, rather than with their vets. This does not necessarily result in the best solution for the animals. Crucial for good collaboration with cat breeder clients is improved veterinarian knowledge, and that is the aim of this article – not only knowledge specific to reproductive medicine, but also knowledge regarding general management of the cattery.
Cat reproduction as ‘herd health medicine’ – a new approach
Generally speaking, there are two veterinary approaches to the cat breeder:
✜ The traditional approach, whereby the cat breeder visits the veterinarian, usually with a single cat that has a problem;
✜ The veterinarian visits the cat breeder.
Currently, the latter is relatively uncommon, which is unfortunate as a direct visit to the cattery is a means of gaining deeper insights into management, housing and feeding practices, and so forth. In turn, this allows the veterinarian to more easily identify possible deficits, to provide specific feedback and to sample more animals to get a better picture of the infectious disease status of the cattery. The tasks (and opportunities) of these ‘visiting veterinarians’ are similar to what is well established in bovine and porcine herd health management. According to Whyte et al, 3 ‘the traditional role of herd health is to control or eliminate diseases and management inefficiencies that impact on animal welfare or limit productivity… ‘ In the present context, productivity can refer to reproductive performance, which in the authors’ opinion is about attaining realistic breeding goals. Rather than maximising the number of litters in a cattery or from one individual cat, the focus is on achieving pregnancies when attempted, with a satisfactory number of vital and healthy newborns that survive. Optimising general health and wellbeing of the cats is consequently an essential part and should be the primary goal of both veterinarians and cat breeders.

Housing and husbandry of the cats
Usually cat breeders keep multiple animals, but the number of cats per breeder is extremely variable. Studies in the literature have reported (mean) numbers of cats per breeder of 3 – 4 intact adult queens and one tom cat, 4 4.9 ± 2.3 cats, 5 6.1 ± 2.4 female cats:0.8 male cats, 6 and (median) 12 cats (range: 5 – 29). 7 All these studies vary with respect to the number of participating breeders and breeds represented, as well as the origin of breeders.
Romagnoli et al 5 summarised data from six Bengal, five Maine Coon, eight Norwegian Forest and seven Persian breeders from Italy; Klein-Richers et al 7 selected 37 breeders from Germany for sample collection for non-reproductive purposes; Strom Holst and Frossling 6 included data from 264 Swedish breeders; and Hackfort and Goericke-Pesch 4 compiled data from 404 breeders from 24 countries, with various breeds represented.
Housing requirements and recommendations for cats used in research are determined by US and European government regulations and scientific guidance documents.8–11 Dependent on the country, national laws might exist providing an official, legal basis for large catteries, but ‘hobby’ catteries tend to be less regulated; and, especially in the latter, official veterinarians only become involved if complaints arise relating to animal welfare.
Whereas queens are often kept within the household as pets, it is not uncommon for intact toms to be kept individually, either within individual rooms of the accommodation or in separate ‘garden houses’. This is principally to prevent unwanted matings, but also the smell, urine marking and possible aggressive behaviours of toms. Careful observation during the visit is important, as a bathroom or a dark shelter in the garden is not the optimum location for a cat. As well as the cats’ behavioural welfare, hygiene and environmental conditions should also be assessed (Figure 1).

(a,b) Hygiene and environmental conditions in the cattery – including the kittening area – should be assessed, as well as the cats’ behavioural welfare. Courtesy of Ricarda Schaper and Embricabengal
Specific requirements pertain to the kittening area. It must be clean, dry and warm, allowing for privacy of the queen. Boxes are most often made from wood or cardboard lined with clean soft cloth, pads and/or carpet material. However, from a hygiene perspective, plastic and sterilisable materials are generally preferable. Straw, hay and sawdust should be avoided for queens with kittens, as some substrates can be dangerous when kittens start playing and eating them. Placing a clean, fresh and individual litter box in the kittening area can prevent potentially serious faecal and urinary retention, which is not uncommon during the first days after parturition. Sufficient numbers of litter boxes should be available.
Plentiful clean cool fresh water should be available to all cats at all times. Adequate water consumption plays an important role during growth and lactation. Frequent changing of water and cleaning of the water bowls can be helpful in encouraging drinking, as can the provision of water fountains for some cats. 12
Infectious disease status of the cattery
Multi-cat households, animal shelters and breeding catteries are high-risk environments for infectious diseases.6,13 These, in turn, can have a significant impact on reproductive performance; a specific aetiological agent may have a direct effect on oestrous cyclicity, mating behaviour, fertility/pregnancy and birth rates, or an indirect effect as a result of, for example, poor general health or fever. Strom Holst and Frossling’s 2009 study 6 highlighted a lack of knowledge about the relevance and prevalence of infectious diseases in breeding catteries. Unfortunately still very little is known today.
To the best of the authors’ knowledge, only two studies exist, both from Sweden: one based on a 2006 serosurvey comparing antibody titres against Chlamydophila felis and feline coronavirus (FCoV) in Swedish purebred vs mixed-breed cats; 14 and the other, the Strom Holst and Frossling questionnaire study regarding C felis, FCoV/feline infectious peritonitis (FIP), feline panleukopenia virus (FPV), feline immunodeficiency virus (FIV) and feline leukaemia virus (FeLV) in Swedish catteries, which received 264 completed responses from breeders. 6 As the latter study stated: ‘Information regarding management and prevalence of infectious diseases in breeding catteries is valuable when discussing prophylactic measures, treatment and other measures to undertake after diagnosing an infection,’ and thus there is a significant need for further diagnostics and research.
Infectious diseases that might negatively affect reproduction
While there are excellent reviews of viral, bacterial and protozoal causes of feline pregnancy loss,15,16 prevalence data is lacking. Table 1 summarises infectious and non-infectious causes affecting reproductive performance in cats.
Infectious and non-infectious causes affecting reproductive performance in cats, with an indication of time point(s) when pregnancy is disturbed

Causes in brackets are of minor relevance
Time point when pregnancy is disturbed: 1 = embryonic death; 2 = abortion/fetal death; 3 = stillbirth; 4 = fetal defects
† = cerebellar hypoplasia; ? = Effect postulated, but not generally confirmed
Infections with FPV, FIV, FeLV, FCoV
(resulting in FIP) and possibly feline her-pesvirus (FHV-1) can cause failure to conceive, but also embryonic/fetal death.15,17–32 In utero FPV infection causes fetal death, abortion, fetal resorption and/or cerebellar hypoplasia,15,17-20,33 but it is well known that regular vaccinations can be protective against FPV.34,35 Thus some infectious disease problems within catteries can be avoided with the implementation of proper vaccination schemes. Unfortunately, there is no clinical study published about the vaccination status of catteries and, to the best of the authors’ knowledge, descriptions of the impact of FIV and FeLV on reproductive problems, abortion, kitten death and postnatal infections are limited to the above-mentioned questionnaire-based study. 6 According to breeders participating in that study, neither FPV nor FIV infections had been diagnosed in the previous year in their catteries, and FeLV was diagnosed in one cattery only. A limitation of the data, however, is that the number of analyses was not reported by participating breeders. 6 A study from Brazil confirmed that FPV causes reproductive failure, with associated clinical signs, in queens. 33
Another well-recognised problem in catteries is FIP, which is a common cause of feline death; transplacental transmission, stillbirth and delivery of weak newborn kittens have been described. 36 In the Swedish questionnaire-based study, 3% of breeders had lost a cat due to FIP in the previous year, and 7% (n = 17) had sold a cat that went on to develop FIP within the first year of age (n = 11) or later (n = 6). It was reported that 15% of breeders answered that they tested cats for FCoV antibodies, and 34% of those (5.1% of all participating breeders) had FCoV-positive cats. Overall, 32% of breeders were aware of FCoV antibodies in their cattery. 6 Likewise, Strom Holst et al (2006) 14 had earlier identified a relatively high overall prevalence of antibodies against FCoV in purebred cats (n = 64, 65%). Moreover, 59% of breeders had never checked antibody titres in their cattery, 6 indicating a need for better awareness and more diagnostics in order to achieve a greater understanding of FCoV. There is also a need for research on the impact of this virus on reproduction as only very few proven cases have been published. 36
Among countries other than Sweden, there are recent data from a study in Germany confirming a high prevalence of FCoV: none of 37 catteries studied was FCoV (RNA) free (analysis by real-time PCR [RT-qPCR] of faecal samples of 179 cats). 7 These data support the present authors’ clinical experiences. A recent study confirmed that cat antibody titres correlate with the likelihood and frequency of FCoV shedding and with faecal viral load identified by RT-qPCR. 37 It is important to stress that for effective management of FCoV in the cattery and identification of shedders, at least three consecutive faecal samples for RT-qPCR analysis are recommended. 37 Interestingly, in a separate study by the same authors, the number of cats per cattery, breed, hygiene management, husbandry conditions and outdoor access were not significantly associated with FCoV shed-ding. 7 Readers are referred to recent articles reviewing the diagnosis of FIP and the evidence supporting diagnostic methods.38,39
A variety of facultative pathogenic bacteria such as Escherichia coli var haemolytica and Streptococcus lancefield group G (Streptococcus canis) can affect reproductive performance (Table 1). Samples for bacteriological testing can be obtained from the vagina (eg, vaginal discharge) and uterus (eg, during caesarean section in the case of dead fetuses), and also directly from aborted or stillborn fetuses, fetal membranes, and so on. Interpretation of results is often challenging as the external genitalia harbour opportunistic bacteria. Therefore, excessive growth of facultative pathogenic bacteria on bacterial culture (often a monoculture) with associated clinical signs is usually considered supportive of a causal role, and this is the approach to diagnosis in the authors’ clinic. However, the available literature about these causes in cats is limited. 16 Salmonella typhimuri-um infection, associated with a raw chicken diet, has been reported as a cause of abortion in queens. 40 Experimental inoculation of pregnant cats with Mycoplasma species resulted in abortion, with mycoplasmal bacteria recovered from the heart blood of aborted kittens.16,41
C felis, an important pathogen in catteries, has been described as a possible cause of embryonic/fetal death and neonatal/paedi-atric infections. Seroprevalence of antibodies against C felis was found to be as high as 18% (n = 62) in one study of purebred Swedish cats. 14 In the Strom Holst and Frossling study, an even higher proportion of breeders (33.3%) reported conjunctivitis in kittens and/or adult cats; the majority of cases were not aetiologi-cally investigated (71%), but for the remainder C felis was most commonly diagnosed (7%). 6
In terms of treatment, data regarding safety of antibiotics in pregnant animals are scarce. In general, betalactams, as penicillin or amoxicillin (also combined with clavulanic acid), are the first-choice agents in pregnant bitches and queens. 42 Macrolides (azithromycin) are also considered of low risk during pregnancy. In contrast, in the light of the limited published data about use of fluoroquinolones, and the known side effects described in puppies (eg, on cartilage formation), this class of antibiotics should only be administered in strictly selected cases. C felis infection is usually treated with doxycycline over 4 weeks. 43 Tetracyclines should not be used in the first half of pregnancy, and after day 40 only with significant precautions (if unavoidable). The precise risks to cats cannot be specified currently, but various malformations were reported in a litter of Golden Retriever puppies subsequent to prolonged administration of doxycycline, prednisone and tramadol to the dam during pregnancy. 44
Although reported as a cause of feline fetal death, 45 the protozoan Toxoplasma gondii seems to be relevant only in the case of severe systemic illness of the queen. 16 Tritrichomonas fetus, a protozoan pathogen associated with bovine infertility and abortion, is known to cause chronic diarrhoea in cats but may have -as with any other chronic problem or disease – a negative impact on reproduction. Fatal infections with neonatal/paediatric kitten death and/or fading kittens have also been described. However, no evidence of direct T fetus infection of the reproductive tract was found in a study investigating reproductive tract tissue obtained from elective ovariohys-terectomies and castrations of 61 purebred cats from 36 catteries, even though in 15/61 cats and 22/33 catteries the presence of T fetus was demonstrated. 46 Moreover, no effect on the total number of litters, kitten mortality rate or birth defects could be proven, suggesting that the presence of T fetus in the reproductive tract plays no important role in disease transmission. 46 To prevent and manage tritrichomoniasis in cats, guidelines from the European Advisory Board on Cat Diseases (ABCD) should be followed. 47 Further detailed advice on sampling techniques (via fecal loop) and treatment is available in the literature,48–50 and it is generally recommended not to treat pregnant cats with ronidazole due to possible teratogenicity. 50
Ectoparasites, such as fleas and mites (cheyletiellosis), and dermato-phytes are not an uncommon problem in catteries. Similar to viral infections, the genuine prevalence of these parasites is largely unknown. The only reported incidence data derive from the 2009 Swedish study by Strom Holst and Frossling: fleas 1.5%, cheyletiellosis 0.4%, dermato-phytes 1.1%. 6 Reproductive performance might be affected due to stress or related to treatment not suitable for cats used for breeding. Recommended treatment is well described in the ESCCAP (European Scientific Counsel Companion Animal Parasites) guide-lines. 51 Whereas fipronil and selamectin, as treatment options for fleas and Cheyletiella mites, are also suitable for pregnant and lac-tating queens, no antimycotic treatment is registered for pregnant cats. For treatment of dermatophytes, guidelines published by Moriello et al 52 should be followed. With any ectoparasiticide, appropriate treatment of both animals and the environment is required for animal wellbeing and good reproductive performance, and also because of the zoonotic potential of some diseases. 51
Risk factors for infections in the cattery
Various risk factors for infectious diseases exist in catteries, the principal ones being:
✜ Outdoor access of individual cats or all cats;
✜ Attendance at cat shows;
✜ Mating with toms from other catteries;
✜ Purchase of animals from other catteries. Data about the extent to which catteries
allow their cats outdoor access and/or mate their queens with toms from other catteries are very limited. Although the majority of pedigree breeding cats are kept indoors,4–6 25% of Swedish breeders in the Strom Holst and Frossling study answered that all or some cats have free outdoor access. 6 Other breeders have fenced enclosures on their properties,4,5 preventing cats from leaving the premises and limiting contact with other cats (Figure 2). The risks associated with outdoor access are very relevant, as even a single cat can have an important role in the transmission of infectious diseases such as FIV and FeLV, either through direct contact (eg, mating or fighting) or indirect contact (eg, via owners and equipment) with the indoor residents. Whereas the relative risk of transmission is high, the absolute risk depends on whether the cattery is located in an endemic area, the amount of direct contact that cats with outdoor access have with indoor cats, and the disinfection protocols and means of avoiding transmission via human contact that are in place.

A fenced enclosure on a breeder’s premises, providing outdoor access while limiting contact with non-resident cats. Courtesy of Ricarda Schaper and Embricabengal
Similar risks of disease transmission arise from exchanging cats between catteries or receiving queens/toms from other catteries as a stud service. However, no published information is available about whether breeders ask for specific infectious disease testing before accepting cats for breeding and, indeed, about how diseases can be spread in the cattery. Strom Holst and Frossling (2009) 6 reported that, in Sweden, 58% of breeders performed matings between cats within the owner’s cattery, but 56% reported also that they took their queens to toms in other catteries for mating. Interestingly, while most breeders (68%) did not take in queens from outside, 5% had received queens from other catteries on over five occasions in the previous year. In a large study in France, 93.3% of breeders reported using a male within their own cattery for breeding, 53 which is in good agreement with the authors’ observations. Various questions and important considerations in terms of disease control are listed in the box. Whatever the specific circumstances, quarantine and testing against FIV and FeLV are strongly recommended when introducing cats to the cattery (whether after purchase or for breeding).

Cat shows are another potential ‘spreader’ of infectious disease. Although there are no recent data available, the majority of breeders participate actively in cat shows. Strom Holst and Frossling reported that only 13% of breeders in their 2009 Swedish study did not attend cat shows in the preceding year, whereas the majority (61%) had attended between one and nine cat shows. 6 The remaining breeders had attended cat shows 10-26 times in the year. Conjunctivitis and other signs of upper respiratory tract disease were observed after shows by around 10% and 15%, respectively, of breeders. 6 It seems likely that viral infections, such as FHV-1 and feline calici-virus (FCV), and also C felis are important contributors in these conditions. Although the role of cat shows on transmission and/or reactivation of FHV-1, FCV and C felis infection has never been investigated, the incidence of these diseases is likely to increase following attendance at cat shows where cats are not pre-screened for such diseases.
Hygiene and sanitation within the cattery
Good hygiene and sanitation are crucial in any multi-cat environment, be it a household, cattery or shelter. However, among private ‘hobby’ catteries, in particular, knowledge of hygiene and sanitation principles is often poor; not uncommonly, hygiene management is a low priority, or even non-existent, until problems arise.
General principles and a practical approach to good hygiene are summarised in Table 2, and useful examples of hygiene plans are available in guidelines from the Cat Protection Society of New South Wales. 55
Ten practical principles for effective sanitation/disinfection

Based on Lawler and Bebiak 54
Regular cleaning and disinfection of litter boxes, water/food bowls and other equipment, as well as bedding hygiene and proper food management (storage, etc), are as important as good personal hygiene (hand washing/ sanitation) of people taking care of the cats. Use of hot water for washing (>60°C) and dryers for laundry is a beneficial means of sanitisa-tion. Even nearly 40 years on, the words of Lawler and Bebiak 54 are still pertinent: ‘The sanitation program is an integral part of the activities of a feline colony and is a valuable adjunct to other health and general management procedures if correctly employed.’
The choice of disinfectant and its application are other important considerations. The disinfectant has to be effective against the types of microorganisms relevant in the individual cattery, suitable for the specific environment and material, and finally non-toxic for cats. Selecting the right disinfectant requires good knowledge of the potential pathogens in the cattery, and this can only be obtained by regular health checks, laboratory analysis and post-mortem examinations (incorporating bacterial, viral or other sampling/analysis), especially in the case of increased mortality in a specific age group. General and disease-specific guidelines, including recommendations for safe and effective disinfectants, are available from the ABCD.56,57 It is also recommended that veterinarians involved in cattery management consult relevant national literature and resources (eg, kennel and cattery guidelines, German DVG database).58,59
Handling of cats in age order, with the youngest handled first, and prioritising pregnant cats over other adult cats, as well as isolation and separation of ill cats and new arrivals (quarantine), are appropriate means of infection prophylaxis, as overcrowding and inability to physically isolate sick cats are leading contributors to disease transmission. 55 Moreover, health programmes incorporating at least an annual physical examination and laboratory profile (eg, at the time of annual vaccination) facilitate early detection of possible diseases. This, in turn, promotes the health of the cattery and prevents infections from becoming endemic.
Common problems, especially in larger catteries, are respiratory tract infections, ecto- and endoparasites and dermatomycosis, with the last being a potential zoonosis and thus having public health implications. In well-managed catteries, respiratory tract infections should only affect individual cats or small groups of cats, with mild signs and rapid recovery. 55 Periodic parasitological examinations of faeces are recommended in adult animals, especially those fed raw food diets or having access to outdoor areas and/or contact with animals with outdoor access. 60 Regular deworming of kittens is also recommended, or at least faecal examinations before weaning. In the event of disease, a proper diagnostic work-up is important to identify the cause, where possible, and to inform the treatment approach.
Vaccination programmes for infection prophylaxis
According to World Small Animal Veterinary Association (WSAVA) guidelines on the vaccination of (dogs and) cats, 61 core vaccines for cats are those that protect against FPV, FHV-1 and FCV. Regular vaccination using modified-live vaccines is recommended to reduce the risk of negative effects of these viruses in the cattery in general, but also on reproduction and the offspring (see box). However, pregnant animals should not be vaccinated with modified-live vaccines, emphasising the need to check the vaccination status of animals before mating. If vaccinations are not up to date in already pregnant animals, only inactivated (killed) core vaccines can be used. 61
Additionally, modified-live vaccines are not recommended for animals infected with FIV and FeLV according to widely held expert opinion 61 – once again, indicating the need for regular examinations in the cattery, especially in those at higher risk (see above). In catteries with sustained risk of exposure, non-core vaccines against FeLV (killed or recombinant sub-unit adjuvant vaccines, in FeLV-negative cats only) and/or FIV (killed adjuvant vaccine, FIV-negative cats only) should be considered.

Nutrition
Nutrition is important for the health and well-being of the animals in the cattery, but also for successful reproduction. A queen in optimum body condition fed a balanced diet will have the best reproductive performance. Thus breeding queens should be weighed on a regular basis, as well as at various stages in the reproductive cycle – before mating, regularly (eg, once weekly) during pregnancy, before and after parturition, and at weaning.
Not only will a body condition that is too lean or obese have a potentially detrimental effect on reproductive performance, an unbalanced diet can similarly have a negative impact. The importance of dietary taurine for cats is well known, with deficiency causing dilated cardiomyopathy, blindness and death.62,63 Sufficient taurine is, however, also required for good reproductive performance. Taurine-depleted females suffer from severe retinal degeneration, and their surviving offspring exhibit neurological abnormalities.64,65 Low copper concentrations are associated with an increased risk of fetal malformations and neonatal ataxia in sheep. 66 In one study, even though no clinical signs compatible with copper deficiency, such as angular limb deformities, were observed in kittens born to queens consuming low copper diets, copper concentrations were negatively correlated with time to conception. 67 The observation that cats receiving 4.0 mg copper per kg diet required significantly longer to conceive than those receiving 10.8 mg copper per kg diet resulted in the conclusion that the current National Research Council (NRC) recommendation of 5 mg cu/kg diet ‘is probably marginal for optimal reproduction in the queen’. 67 This is especially valid if a copper source with a lower availability than the studied copper sulfate is used. 67
Breeding queens have increased nutrient requirements. 68 After mating until parturition, energy requirement has been found to increase by about 30-70% (average 50%) depending on the litter size. During lactation the energy requirement increases by 100-300% depending on the time point of lactation (Figure 3). 68 Clearly, a complete, balanced adult diet formulated for maintenance cannot provide the required energy for these stages of increased demand. Instead highly palatable diets with a label claim for the specific reproductive period should be fed. Usually this means that cats are on a diet for pregnant and/or lactating queens, or a diet for performance or growth, like a kitten diet. 68 Food should always be available ad libitum during pregnancy.

A queen in optimum body condition fed a balanced diet will have the best reproductive performance. Depending on the stage of lactation, the queen’s energy requirement increases by 100–300%. Courtesy of Ricarda Schaper and Embricabengal
For breeders considering feeding raw food diets, requirements and contents should be determined by a qualified veterinary nutritionist, as unbalanced diets might not only negatively affect reproductive performance but can have a direct negative impact on the offspring. Other concerns include the risk of microbiological contamination of raw food (Salmonella species, 40 Campylobacter species, E coli, etc) or contamination with endopara-sites, such as Toxoplasma, Sarcocystis, Cystoisospora, Neospora and Hammondia species, the nematodes Toxocara and Trichinella species, and the tapeworms Taenia species, Echinococcus granulosus and Echinococcus mul-tilocularis. 60 Mycotoxins 69 – as a contaminant of industrial food – as well as environmental pollutants such as bisphenol A 70 can also exert negative effects on reproductive performance and/or general health.
During pregnancy, the queen’s body weight should increase continuously, aiming for 150% of initial body weight at the time of parturition. Energy and protein needs of cats during maintenance, gestation and lactation are described elsewhere.68,71 Sufficient water intake is also of the utmost importance, not only for development in general, but specifically for milk production; adding water to a highly digestible and palatable diet is a strategy for increasing the water intake of the lactat-ing queen. Queens that lose excessive weight during lactation have to be closely monitored and restored to good body condition before the next mating. 55
Appropriate nutrition is crucial for toms, too, and unbalanced diets have been shown to result in oligo- and even azoospermia in felids. 72 Only male cats that are heavily used as sires require special feeding, namely high nutrient density rations. However, particular attention should also be paid to male cats that are exposed to cycling queens over prolonged periods, as they may lose weight due to reduced food intake and increased stress. Reduced grooming by some actively breeding toms has also been described, leading to the recommendation that animals should be carefully checked for skin or haircoat associated conditions. 55
Reproductive history and performance
General considerations
Cat breeding is regulated by various specialised breeding organisations, each with their own rules or guidelines for their members, which can vary quite substantially. One example is the Federation Internationale Feline (FIFe), which has member groups in around 40 countries; 73 another is the Governing Council of the Cat Fancy (GCCF) in the UK. Many general rules represent good common sense, but they are not always written down or based on published evidence.
Age of breeding animals and frequency of breeding
Cats should only be bred when fully grown. Depending on the breed, the age of a queen at first litter should ideally be 18-24 months. Queens younger than 1 year can rarely be considered fully grown and should consequently be prevented from breeding, as this could have a significant, and possibly lifelong, impact on the queen’s health. For large-breed cats, and depending on the season, the age at first litter might even be 24 months or more. Only healthy, fully vaccinated animals in good body condition should be used for breeding. A full health check-up should be performed and genetic disorders, as well as general and breed-specific diseases, should be excluded by specific tests.
Ideally, queens older than 5-6 years, or for large breeds possibly 7 years, should no longer be bred.

The box above contains general guidance concerning the frequency of breeding in any individual queen. In practice, little is known about the average number of litters from breeding queens in both small and large catteries. However, recent registration rules of The Cat Fanciers’ Association in the USA, including no limit on the total number of litters per cat, no limit on the number of litters per year (but at least 65 days apart – which is the duration of pregnancy, so biologically impossible otherwise), no rules pertaining to the maximum age at breeding and ‘no rules against closely related breedings of any kind nor restrictions on the pedigree relationship of the prospective sire and dam’ (including mother/son, sire/daughter, siblings, etc) are ethically highly questionable in the authors’ opinion.
Whereas mating too early is detrimental for the queen, mating at a young age (around puberty) is not harmful for the tom. However, using a peripubertal or a young, inexperienced male is a risk for the breeder as semen quality might not be optimum, there may be insufficient semen to impregnate the queen (and, moreover, due to induction of ovulation an increased risk of pyometra for the queen in pseudopregnancy) or the male might even refuse or not succeed in mating, resulting in anovulatory cycles in the queen. Ideally the tom should also be 18-24 months of age before being used for breeding, assuming he has reached sexual maturity. Especially in long-haired breeds, such as Persians, some breeders report successful matings from an age of 24 months, indicating relatively late sexual maturity. As for queens, regular health checks and vaccinations, as well as testing for genetic disorders, are recommended for toms used for breeding.
Inbreeding
Inbreeding, often also called line breeding, is a practice commonly used by breeders of domestic and companion animals to transfer desirable traits to the offspring in a predictable way. As discussed further in an accompanying review in this series, inbreeding increases homozygosity; 75 however, this not only preserves desirable traits but also the risk of hereditary diseases by combining deleterious genes. Pedigree cats are characterised by a relatively high individual inbreeding level (coefficient of inbreeding) as a result of close inbreeding practices. 76 This can increase the risk of genetic disorders and so-called ‘inbreeding depres-sion’,77–79 negatively impacting the individual animal and also the population at large. 80
Increasing genetic diversity will result in increased reproductive fitness, and represents an important aim of breeding programmes. Developing better mating schemes, including mating distant relatives within a breed, can minimise the risks of genetic disorders and inbreeding depression, and also contribute to a reduction in genome-wide inbreeding lev-els. 81 The benefits are especially relevant where a particular gene is associated with a disease through Mendelian or complex inheritance. 81 Indeed, currently more than 90 pathogenic gene variants or mutations are known to be associated with inherited diseases in cats.82,83
Inbreeding is also recognised to have a significant negative impact on reproduction itself. Loss of genetic diversity is related to reduced semen quality in several wild felids, 84 and this might be true for pedigree cats as well. To date, this negative correlation has been confirmed with respect to teratozoospermia. 85
Evaluation and recording of reproductive performance
Careful records should be kept of reproductive performance of all cats in the cattery (see Table 3 for recommended parameters for recording in the queen and tom). The environment is an additional key factor, and needs considering in its widest sense in order to optimise hygiene, and the health and well-being of the cats.
Parameters to be considered and evaluated for reproductive performance

Fertility and good reproductive performance have been shown to be heritable in the dog, 86 and it seems likely that this is also true in the cat
In cats, natural delivery is common. However, depending on the underlying reason, previous caesarean section does not necessitate exclusion of queens from breeding, nor does it likely have a negative impact on subsequent fertility, as recently shown in dogs 87
? = difficult to diagnose
Prevention of unwanted pregnancy
Prevention of unwanted pregnancy is a crucial aspect of cattery management. The aim is not only to avoid undesirable mating and inbreeding (eg, between blood relatives), but also to avoid unwanted litters. Consequently, short-term reversible suppression of fertility is of the utmost importance for all responsible breeders, not only those co-housing male and female cats and/or allowing their animals free outdoor access.
Use of contraceptive medication in queens
Due to the cat’s unique reproductive physiology, repeated oestrous cycles during the breeding season are normal and, especially in Orientals and Siamese, overlapping follicular waves appear as ‘prolonged’ or ‘persistent’ heats (authors’ personal observations). The seasonally polyestrous nature of cats can be stressful for both the animals and their owners. It can have a substantial impact on body condition score, with unmated queens that experience prolonged and multiple oestrous cycles during the season becoming too lean. Aside from the impact on the cat’s health and wellbeing, the loss of condition, which may also include the development of a rough coat, can compromise the chances of success for cats attending shows. 88 A further consideration is that, although an induced ovulator, spontaneous ovulations have been identified in 35–87% of cats,89–91 and progesterone exposure is known to predispose to pyometra. 92
Consequently, suppression of the oestrous cycle (reversible contraception) is relevant (and common) in queens that are intended for future breeding, particularly in the light of the fact that, as discussed, many cat breeding associations permit a maximum of three litters over a period of 2 years for a queen. 73 In a recent questionnaire-based study by one of the current authors (Goericke-Pesch) only 27% of breeders reported that they do not use contraceptives in their cats. 4 While surgical neutering is the method of choice for non-breeding animals, progestogens (progestins), gonadotropin-releasing hormone (GnRH) agonist slow-release implants (SRIs) and melatonin are used for contraceptive purposes and reversible suppression of oestrus in queens (Table 4). Progestogens are the only drugs registered for contraceptive use in queens to date; administration of melatonin and GnRH agonist SRIs represents off-label use.
Practical options for contraception in adult queens

For further information, see the accompanying review on reversible control of reproduction in queens 94
SRI = slow-release implant; PO = oral; SC = subcutaneous
It should be emphasised that even if less than 8 h of light induces anoestrus in queens, this manipulation of the photoperiod is not ethically acceptable for a cattery for the purpose of prolonged oestrus suppression.
Progestogens
Progestogens are the most frequently applied hormones for feline contraceptive purposes, 4 and are available as contraceptive ‘pills’ or ‘syrup’ or as injectables, with pills being most commonly administered. Medroxy-progesterone acetate (MPA) and megestrol acetate (MA) have been used to control reproduction in cats for over four decades, and, despite various reports about side effects, can be considered relatively safe if given on a short term basis at an appropriate dosage to carefully selected patients. 88 A review of the potential benefits and risks of progestogens – mainly MPA and MA – was published by Romagnoli in 2015, 88 and further detailed discussion is available in an accompanying review on the reversible control of reproduction in queens. 94 The major benefit of MA is its short half-life (a few hours),95,96 compared with the longer half-life of other progestogens (eg, 12-17 h for MPA after oral administration), making MA most likely the ‘safest’ progestogen. 88 Weekly administration of 2.5 mg/cat MA (the licensed dose for commercial MA-based formulations) for a maximum of 30 weeks can be considered relatively safe.88,95 Intermediate dosages (>0.625 mg/kg q24h for 1 week or q48h for up to 2 weeks) cause only a temporary and reversible adrenocortical suppression, but they may cause more significant, and potentially irreversible, damage when used for prolonged periods of time (eg, q24h or q48h for several weeks, or weekly for months or years). As reviewed by Romagnoli, 88 these include adrenocortical sup-pression,97–99 impaired glucose metabolism leading to diabetes,100–105 mammary gland lesions (eg, fibroadenomatosis, mammary tumours),106–112 uterine disorders (eg, cystic endometrial hyperplasia, pyometra),113,114 cutaneous xanthomatosis, 115 and blindness due to hyperlipidaemia 116 or secondary to diabetes mellitus (retinopathy, retinal detachment 103 ).
An injectable progestogen that is licensed and used in cats is proligestone.
GnRH agonist SRIs
Although just recently licensed for use in adult male cats (4.7 mg deslorelin), GnRH agonist SRIs were already in regular use by breeders in both male and female cats. According to the literature, they can be safely administered to significantly postpone puberty in postnatal and prepubertal animals.117,118 Reversibility in terms of suppression of oestrus has been proven in different studies, and data about subsequent fertility in breeding queens are also available, albeit in a limited number of cases.119–122
In the aforementioned questionnaire-based study, GnRH agonist SRI treatment for contraceptive purposes was reported for approximately 12% of adult breeding queens. 4 When used in adult queens in seasonal anoestrus or during postoestrus (with basal progesterone concentrations), oestrus induction is possible or even likely.122,123 Given that the induced oestrus might be fertile, contact with intact toms must be avoided to prevent unwanted matings and pregnancies. A risk of luteal failure has also been postulated, as described in dogs.124–126
GnRH agonist SRI treatment of recently mated cats needs to be avoided as queens can go into labour and deliver naturally at term, but might lack lactation and maternal instincts. 127 Further reported side effects include lack of efficacy, persistent oestrus and cystic endometrial hyperplasia; also lactation, pseudopregnancy and fibroadenomatosis, 120 likely related to induction of ovulation and progesterone increase as a consequence of treatment. In general, side effects seem to be more common in females than in males, as reported for bitches.120,126
The duration of efficacy of the 4.7 mg deslorelin SRI varies between studies – 16.0 ± 5.7 months (n = 26; range: 8–38 months) 120 versus 22.7 ± 2.1 months (n = 19; range: 16–37 months) 122 – and high individual variation is also seen. Nevertheless, reversibility has been proven in terms of the oestrous cycle and return of fertility, with reports of normal-sized healthy litters after the end of treatment.120,122 Due to the delayed onset of action, the duration of efficacy and high individual variability, the deslorelin SRI is more suitable for long-term suppression of fertility and ovarian function. Administration of divided implants (ie, cut in half) is not recommended, as sterility of the product cannot be maintained. Moreover, this does not necessarily result in a shortened duration of efficacy; rather, in more variability and less predictability. Consequently, on the grounds of both sterility and pharmacological concerns, this misuse of the GnRH agonist SRI should not be supported by veterinarians.
Data about the 9.4 mg deslorelin SRI are limited, but the fact that it postpones oestrus by more than 18.5 months 123 does not make it a better option for those breeders (the majority) requesting short-term contraception in the cattery.
Surgical removal of both deslorelin implants might be an option to shorten the duration of efficacy,128,129 but the time point for return to oestrus seems to be extremely variable 120 and might be influenced by season (authors’ personal observations). A recent study described resumption of ovarian function after an interval of 3–7 weeks, depending on the photoperiod. 130
Melatonin
Another option for contraception is the so-called ‘sleep hormone’ melatonin (N-acetyl-5-methoxytryptamine), which plays a critical role in feline reproduction and has been the subject of a dedicated review. 131 Melatonin concentrations and secretion by the pineal gland are proportional to the number of hours of night-time darkness. Increasing hours of light in the springtime are associated with suppression of melatonin secretion and increased gonadotropin concentrations, triggering ovarian activity and oestrus in cats (described as ‘long-day breeders’). In reverse, high concentrations of melatonin suppress GnRH and subsequently gonadotropin secretion.132,133
Melatonin, whether administered orally as a pill, as a long-term subcutaneous implant or even in injectable form, mimics longer dark phases associated with non-cyclicity through its ability to suppress ovarian function to an extent.129,131 More specifically, melatonin administration delays or prevents the post-coital surge in luteinising hormone (LH) needed for ovulation induction and increases the post-coital refractory period. However, even though melatonin can reduce LH release, 67% of cats showed oestrus and ovulated in one study. 133 As frequent injections are not practical, the following discussion focuses solely on oral and implant use.
No melatonin formulation or product is registered for use in cats. Off-label use of 18 mg melatonin implants licensed to induce oestrus in sheep (a short-day breeder) has a major limitation in that the product is available in large packages (2 x 25 implants) only, with just one cannula and syringe for repeated use. This is a critical consideration in terms of sterility, product safety and disease transmission between animals and catteries. On the other hand, oral treatment needs to be given on a daily basis and might not be easy for multiple cats. Notwithstanding, melatonin supplementation (30 mg/cat PO q24h, 3 h before lights-off) for 35 days was effective in suppressing ovarian activity; 96 cats returned to normal ovarian activity 21–40 days after the end of treatment, with some individuals (3/6) showing increased oestrogen concentrations within the first 25 days of supplementation. A lower oral dose (4 mg/cat PO q24h, 2 h prior to lights-off) in a later study produced similar results, 134 with oestrus suppression averaging 50 days from the beginning of treatment. In contrast to the earlier study, the authors did not report oestrus in any of the queens after the onset of treatment, but ovulation and pseudopregnancy were observed in 7/19 cycles (n = 12 cats), which could be a consequence of the oestrus prevention.
Low-dose melatonin (0.9 mg/cat) has been found not to be effective. 135 The same investigation also reported that oestrus signs during treatment (5 or 10 mg/cat PO q24h, 2–3 h before lights-off) were more frequent in breeds with short interoestrus intervals (Oriental, Sphynx, Devon Rex); 135 this indicates possible lack of or reduced efficacy of melatonin in these breeds. 131
Melatonin treatment from the end of oestrus until early interoestrus seems advisable. This is despite the fact that the prolonged duration of efficacy of the 18 mg melatonin implant in interoestrus (113.3 ± 6.1 days) compared with application in oestrus (61.1 ± 6.8 days) decribed in one study 136 was not reported in another study involving administration after oestrus until early interoestrus (63.8 ± 5.4 days). 134 The shorter duration of efficacy associated with application in oestrus might be explained by downregulation of melatonin receptors due to high oestrogen concentrations, as reported in rat and human ovaries, 137 though not to date characterised in cats. Schafer-Somi 131 described better efficacy in animals with more pronounced seasonality and a possible influence of the particular season when treatment is started. Successful prolongation of natural anoestrus might also be possible. 138 Further studies with larger populations of cats of different breeds are, however, necessary to confirm these hypotheses.
Regardless, strict separation of melatonin-treated queens from intact toms is crucial, as suppression of ovarian function is delayed (about 11 days) 136 and the initial (induced) oestrus can be fertile. 131 Reports suggest limited or no side effects, 131 and the majority of queens that were bred after treatment conceived (6/8, Gimenez et al 136 ; 12/17, Furthner et al 120 ). The melatonin implant is not suitable for postponement of puberty. 134
Use of contraceptive medication in toms
According to the recent questionnaire-based study by one of the authors (Goericke-Pesch), GnRH agonist SRIs are the most frequently used contraceptives in toms, 4 despite their use having been off-label until June 2022. This is not surprising given that GnRH agonist SRIs are the only drug that can induce temporary suppression of reproductive function in toms. Progestogens – also registered for male cats – have marginal effects on spermatogene-sis and, in turn, fertility. They are, however, able to modulate male behaviour, and reduce libido, as well as urine marking and aggres-sion.129,139 Little is known about the efficacy, mode of action and so forth of progestogens in male cats. No dose-side effect relationship of progestogens has been published to date, but they might induce side effects as described in queens, such as mammary hypertrophy (fibroadenomatosis). 140 Manipulation of the photoperiod and administration of melatonin is not suitable for either delaying puberty or suppressing reproductive function in toms.
In contrast, numerous studies deal with the use of GnRH agonist SRIs in toms. Following administration, SRIs induce short-term stimulation of reproductive function, similar to what has been described in dogs, followed by a long-term (but also individual) suppression of testicular endocrine function as indicated by basal (or at least significantly reduced) testosterone levels. Basal testosterone concentrations were reached in 9/10 cats between 3 and 11 weeks after treatment with a 4.7 mg deslorelin implant. 141 In the initial period after treatment, side effects include increased urine marking, libido and aggression; later on, reduced urine spraying and libido, and calmer behaviour have been reported. 141 As a consequence of testosterone withdrawal, testicular volume decreases significantly (around 60%),142,143 and penile spines regress and disappear over the course of a number of weeks (5.8 ± 1.1143; 9.4 ± 1.0141). The described duration of efficacy varies from 13.4 ± 3.2 months (n = 37, range: 8-21 months), 120 through 19.7 ± 3 months (n = 7, range 15–25 months), 144 to 22.7 ± 6 months (n = 9, range 6–30 months). 145 The package insert and summary of product characteristics for the 4.7 mg deslorelin implant (Suprelorin; Virbac) states: ‘clinical effects on urine odour, urine marking, testicular volume, penile spine size, and sexual behaviours begin to wane after approximately 12 months post implantation.’
It is noteworthy that some breeders have reported fertile matings at various time points during treatment with GnRH agonist SRIs, indicating that infertility might not be achieved, or may be of very short and unpredictable duration, in some toms. However, even though reversibility of effects on fertility has been demonstrated courtesy of a limited number of animals siring successfully post-treatment, 143 the veterinarian using the SRI in toms should never guarantee a return to fertility, even in the case of a proven breeder; there may be unforeseen influences on efficacy and/or strong individual variation. Reversibility of testicular endocrine function has been shown to occur quickly once efficacy wanes, 143 as well as after implant removal. 146 Testosterone concentrations were >0.1 ng/ml 23±6 days (following 3[n = 6]or6 [n = 5] months of treatment) and 22 ± 7 days (9 months of treatment, n = 4) after removal of a 4.7 mg deslorelin SRI in a recent study. 146
As stated for queens, dividing GnRH agonist SRI implants is not recommended due to compromised sterility and unpredictable duration of effects.
Management of unwanted pregnancy
While detection of unwanted pregnancy in free-roaming cats and pet cats with outdoor access is often relatively late, in breeding catteries unwanted matings might be observed immediately, in which case veterinary help may be sought for the prevention of pregnancy. However, especially in well-managed catteries that keep intact queens and one or more toms, mismatings are rare. Should they occur more frequently, or even on a regular basis, management of the cattery should be discussed with the breeder and contraceptive options (see above) considered.
No drug is licensed for termination of feline pregnancy and neutering of pregnant (stray or owned) animals is frequently performed in many countries, sometimes without knowledge of the pregnancy if at an early gestational age. While surgical neutering is not appropriate for queens intended for further breeding, if animals are at the end of their breeding career, it is without doubt the best option right after mating as it allows for permanent contraception and avoids the risk of repeated mismatings and reproductive disease. Collecting a history from the breeder (ie, mating and/or the queen’s post-coital reaction witnessed) supports the need for further procedures. Assaying serum progesterone helps to avoid unnecessary treatment as queens may fail to ovulate (indicated by low progesterone concentrations) even following normal breeding and post-coital reaction. In terms of medical options, antiprogestogens, prostaglandin F2 alpha (PGF2a) and cabergoline have been described in the literature for termination of feline pregnancy.
If available, antiprogestogens are the drug of choice, with high efficacy rates and low or no risk of side effects. Aglepristone (Alizin; Virbac) has been proven to be safe and effective for early and mid-term pregnancy termination in cats. The recommended treatment protocol includes administration on day 0 (first day of treatment) and day 1 at 10-15 mg/kg. If the aim is termination in mid-pregnancy, an ultrasound examination is required on day 6/7 to determine whether an intact pregnancy or fetal remnants can be visualised, in which case another aglepri-stone injection is given on the day and a subsequent examination is performed 1 week later.
Whereas success rates are 100% in the case of early termination (day 5/6), 129 efficacy has been reported to range between 87% and 88.5% if aglepristone is administered on days 25/26147 or 33.3 ± 4.2. 148 A study describing late termination (day 45/46 of pregnancy) in a very limited number of animals (n = 6), 149 reported effective abortion in only 4/6 cases (67%).
The lower termination rates in mid-term pregnancy clearly indicate the need for the above-mentioned subsequent ultrasound examination to confirm efficacy. Administration in late pregnancy is unethical in the authors’ opinion, due to low efficacy. Without doubt, earlier treatment is preferable. Ideally treatment with aglepris-tone should be started latest at day 30. Consequently, it is advisable to estimate/ determine the gestational age by ultrasound (see box) before the start of treatment, to help gauge the risk of incomplete abortion.

Abortion in the second half of gestation can also be induced by repeated injections of PGF2a or oral application of cabergoline;159–162 however, success rates are variable and there are higher risks of side effects subsequent to PGF2a treatment, such as salivation, vomiting and diarrhoea. Combined use of PGF2a and cabergoline is more successful, and PGF2a side effects are significantly reduced.162,163 Nevertheless, success rates of combinations are still not ideal, supporting the use of aglepristone.
Clinical approach to specific problems in the cattery
Queen fails to cycle
As discussed, queens are long-day breed-ers,164,165 and different climates and light patterns, as well as the latitude, affect season-ality. For example, cats cycle throughout the year close to the equator,166,167 whereas the majority of oestrous cycles occur from January to June in Italy 5 and February to August in Germany. However, due to exposure to artificial light, as experienced in the modern home setting, queens can also cycle throughout the year, as confirmed by a questionnaire study. 5 Furthermore, breed seems to have an impact on seasonality.168–171
Before considering oestrus induction, the underlying reason for the lack of oestrous cyclicity should be identified and investigated. Disease and malnutrition have to be ruled out as a first step.
Manipulation of the photoperiod is a simple, efficient and low-cost method of oestrus induction, with no known side effects, as artificial light has been shown to influence seasonality.168–171 Exposure to light (natural or artificial) for >12 h successfully induces reproductive cyclicity in the queen.133,167,172 Ideal stimulation of the hypothalamic-pituitary-gonadal axis appears to be reached with 14 h of light. 173 Excessive exposure is, however, not beneficial, as the frequency of cycles can decrease with 24 h of light. Keeping cats at <8 h/day of light to induce a temporary anoestrus seems to optimise the oestrus response after 12 h of light/day. 133 Similarly, 1 h light during the dark period has been shown to result in a faster oestrus response to light treatment. 174 The presence of other queens or intact toms (either direct [contact] or indirect [smell]) seems to stimulate oestrus induction 174 and can shorten the time to oestrus when combined with photoperiodic manipulation (see box). 174

Medical induction of oestrus can provide faster results, but might also be associated with side effects such as exaggerated and/or prolonged ovarian stimulation resulting in anovulatory follicles and ovarian follicular cysts. 171 Moreover, absence of progesterone-producing corpora lutea is crucial for efficacy of medical induction protocols, indicating the need for progesterone analysis before treatment. To improve the oestrus response, anoestrus can initially be induced by administration of progestogens (eg, MA or altrenogest).
Various protocols have been published for oestrus induction,175,176 the most frequently applied being administration of follicle stimulating hormone (FSH; porcine or human origin) or equine chorionic gonadotropin (eCG) (Table 5). Time to oestrus induction from the start of treatment usually varies between 3 and 7 days with FSH, 177 likewise with eCG. Natural mating or injection of LH-like acting human chorionic gonadotropin (hCG; 250 IU/cat, 177 50–100 IU/cat 179 ) can be used for induction of ovulation. Efficacy of eCG and hCG treatment can be affected by antibody formation observed after repeated use. 180 A 4-month break between treatments seems to be sufficient to avoid negatively affecting ovarian stimulation. 181
Selected protocols for medical oestrus induction in the queen

See text for discussion of protocols involving photoperiodic manipulation
Current recommendation (see comments in the text)
FSH = follicle stimulating hormone; eCG = equine chorionic gonadotropin; hCG = human chorionic gonadotropin; SC = subcutaneous; IM = intramuscular
Furthermore, use of GnRH agonist SRIs containing 4.7 mg deslorelin has been described by Zambelli and colleagues 178 for oestrus induction in queens, as reported also in the bitch.125,126,182 The 4.7 mg deslorelin implant induced oestrus in 13/13 cats after 4–11 days (mean 5.0 ± 2.2 days), with 7/13 showing behavioral oestrus and none having side effects. These authors successfully induced ovulation with 100 IU hCG at the peak of oestrus, and performed transcervical insemination 24–48 h after hCG injection; 3/3 cats conceived. Although GnRH agonist SRIs seem to be promising for oestrus induction, major limitations are the costs and the need for surgical removal of the implant (potentially requiring sedation or general anaesthesia). Furthermore, clinical experience of using deslorelin implants for oestrus induction is limited, as is information regarding subsequent pregnancy rates, indicating the need for further research before general recommendations can be made.
In the authors’ opinion, oestrus induction should only be considered in the case of prolonged anoestrus in otherwise healthy animals and should be used on a one-off basis rather than as a repeated treatment. If multiple animals in a cattery are affected, genetic causes, suboptimal housing or unbalanced diets should be considered, identified by diagnostics and addressed where possible.
Queen fails to conceive or to maintain pregnancy
Infectious causes for failure to conceive or to maintain pregnancy were discussed earlier and described in Table 1, which also provides insights into non-infectious causes. If pregnancy rates in a cattery are low or if abortion/ fetal death rates are high, diagnostics are crucial. A detailed history should be gathered including time to next oestrus (was ovulation induced?), clinical signs of illness or disturbed pregnancy, and whether any pregnancy was confirmed or just assumed in the case of resorption. This must be followed by a general physical examination, a gynaecological examination, possibly including a bacteriological examination of a vaginal swab, and ultrasound of the reproductive tract; based on the findings, blood work including virus (antigen/antibody) testing may be performed. Where dead fetuses (or placentas) are available, they should be submitted for further examination (necropsy, bacteriology/virology, etc) to identify the cause and inform appropriate treatment.
The queen does not have (sufficient) milk
Hypo-/agalactia is a serious problem in some queens after parturition. Early diagnosis is imperative, which is why regular weighing of kittens after delivery is strongly recommended (Figure 4). Ideally, kittens should be weighed after parturition, then at least once (preferably twice) daily until about 4 weeks of age and thereafter twice per week until weaning. Insufficient or lack of weight gain, or even loss of weight, are critical signs of an underlying problem and an indication for careful examination of the dam and the kittens (as discussed in accompanying reviews on feline breeding and pregnancy management 183 and fading kitten syndrome 184 ). An unsatiated, ‘crying’ kitten can also signify insufficient milk production by the dam. Whereas it is common that no milk can be expressed from the teats before the end of parturition in queens, it should be possible to obtain milk afterwards.

Regular (daily or twice-daily) weighing of kittens after delivery is strongly recommended. Provision of an adequate environmental temperature is also important to facilitate maintenance of normothermia in newborns. Courtesy of Ricarda Schaper and Embricabengal
Hypo-/agalactia seems to be more common in some lines, indicating that further breeding of those cats should be carefully considered. Other common causes are caesarean section and insufficient postsurgical pain medication, or general or reproductive disease, such as retained fetal membranes and/or mastitis. Treatment of the primary cause is crucial for optimum success. Additionally, or where no obvious reason is present, drugs increasing prolactin production should be considered (see Table 6). Formerly, mild sedation using acepromazine was suggested, but the authors believe that nowadays better options are available and, combined with good owner care, are more beneficial and efficient.
Treatment approaches for hypogalactia/agalactia*

To maximise success, the primary cause must first be identified and addressed
Not helpful if ‘true’ hypo-/agalactia is present SC = subcutaneous; PO = oral
Increased neonatal mortality
Within the first 3-4 weeks of life, the queen and offspring represent a unit, and in the case of trouble with one, both parties must be examined carefully. Close monitoring of the kittens, including daily (or ideally twice daily) weight measurement, preferably in the presence of the queen, and regular assessment of the kittens’ muscle (flexor and extensor) tone, are essential for early detection of disease. Inadequate weight gain and growth are indicative of a problem involving the dam, kitten or potentially both.
In the case of suspected illness, a veterinary consultation including body temperature measurement is recommended. Even if treatment approaches are often initially supportive, early detection and instigation of treatment can be life-saving, and veterinarians should raise awareness of this among their breeder clients. Prevention and/or treatment of hypothermia and hypoglycaemia is always indicated, and can buy some time. Consequently, initial treatment includes provision of energy and fluids (prewarmed to body temperature), either as milk replacer (given orally via bottle or tube) or as glucose (given orally or, in severe cases, intravenously/ intra osseously). In mild to moderate cases, additional subcutaneous (prewarmed) fluid administration (eg, Ringer’s solution or Sterofundin [Braun]), plus milk or milk replacer, is beneficial. Provision of an adequate environmental temperature is important to facilitate maintenance of normothermia in newborns and kittens, and prevents excessive loss of energy (Figure 4). Further details on the veterinary approach in the case of suspected illness are given in an accompanying review on feline neonatology. 185

The risk of neonatal isoerythrolysis, a potentially fatal condition in kittens associated with blood group incompatibility between a type B queen bred with a type A tom, is discussed in accompanying reviews on feline breeding and pregnancy management, and fading kitten syndrome.183,184

Reproductive problems in toms
Cryptorchidism
Cryptorchidism, the most common abnormality of the feline male reproductice tract, is considered to be heritable; however, unlike for certain dog breeds,186–188 the genes involved have not been described in toms. An excellent overview of current knowledge of so-called disorders of sexual development (DSDs) in the cat is provided in a recent review 189 and also an accompanying review on DSDs in this series. 190 Environmental pollutants, such as diethylhexyl phthalate (DEHP) and polychlorinated bisphe-nol 153 (PCB153), have been associated with increased frequencies of cryptorchidism in puppies and babies. 191 The reported incidence of cryptorchidism is 1.5%.192,193 In the case of an increased frequency of cryptorchidism, pedigree analysis should be performed, and exposure to environmental toxins investigated. Cryptorchid toms should never be used for breeding.
Failure to mate or to achieve pregnancy
Failure to mate (ie, problems associated with libido/copulation or sperm delivery) can be associated with general (systemic) diseases, orthopaedic problems, hair rings, or pain due, for example, to gingivitis/stomatitis, abscessa-tion or orchitis. A prior bad experience with an aggressive or dominant queen can also result in an ‘unwillingness’ to mate. (Pituitary) hypogonadism, resulting in reduced testosterone concentrations and, in turn, poor libido, or phimosis, resulting in an inability to achieve intromission, are rare. Sexual inexperience or immaturity can also contribute to a failure to mate successfully (and consequently to induce ovulation in the queen). Late puberty has been described in Persians, Maine Coons and Norwegian Forest Cats.
Comparison of the affected tom with other intact toms in the cattery can be helpful for distinguishing physiological and pathological conditions. Aside from a detailed history, a thorough general physical and reproductive examination, possibly including blood work, is required to identify the underlying cause. In some cases, treatment might include pain medication. If no reason can be identified, ‘training’ with a placid and submissive queen should be considered.
Repeated failure to achieve pregnancy in different queens despite successful mating and induction of ovulation (confirmed with serum progesterone assay 3–5 days after breeding) is most likely related to poor semen quality. However, although teratozoospermia is common in cats,194,195 often this does not affect fertility in a natural mating situation, making it difficult to estimate the impact on fertility of an individual tom. Dietary supplements can be considered to improve semen parameters; however, scientific evidence about the benefits is lacking. As in other species, persistent azoospermia has a poor prognosis. Again, a good history, and thorough physical and reproductive examination, including semen collection by transurethral catheterisation (or electroejaculation) and possibly karyotyping, should be performed.
For further discussion, see the accompanying review on infertility in toms. 196
Key Points
✜ Only healthy, regularly vaccinated, sexually mature cats up to 6 years of age should be used for breeding. The general recommendation is a maximum of three litters from one cat within any 2-year period. The performance of regular health checks, including gene testing for breed-specific diseases, characterises the responsible breeder.
✜ Close adherence to hygiene and sanitation guidelines, including quarantine of new arrivals, as well as regular vaccination of all cats, is important for the prevention of infection and associated impacts on reproductive performance in the cattery.
✜ Appropriate contraception measures are crucial in catteries to prevent unwanted weight loss in animals not currently being bred, and also to prevent unwanted litters and unnecessary termination of pregnancy. The method of choice depends on the timeframe for when breeding is next planned.
✜ Further research is needed to provide detailed insight into catteries and management protocols across the world. The ultimate aim is to improve collaboration between veterinarians and cat breeders – and thereby J9 improve feline welfare.
Supplemental Material
Supplementary Table
Breeder questionnaire
Supplemental Material
Supplementary Figures
WSAVA vaccination recommendations for kittens and cats
Footnotes
Supplementary material
The following files are available online:
✜ Breeder questionnaire.
✜ WSAVA vaccination recommendations.
Conflict of interest
Sandra Goericke-Pesch has scientific and continuing education teaching sponsored by Virbac, and has also undertaken Virbac-funded research in relation to Alizin and Suprelorin. However, this did not directly influence this article or any other publication by the author.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
