Abstract
Background:
The literature is full of papers reporting side effects of progestogens in cats; however, they are, in fact, safe drugs, as discussed in this article. Gonadotropin-releasing hormone (GnRH) agonists and melatonin are additionally a practical solution for controlling cyclicity in queens, but they also have some contraindications and need to be used carefully.
Clinical relevance:
Mastering the use of reproductive drugs allows feline practitioners to handle many more clinical situations than can be solved using surgery. It is not necessary to be a specialist in reproduction to be able to halt cyclicity in a valuable breeding queen using hormones.
Equipment and technical skills:
A sound knowledge of appropriate dosages and criteria for patient selection for all reproductive drugs currently used in feline reproduction is the best guarantee of owner satisfaction and of ensuring the queen’s health. Availability of a serum progesterone assay, either in-house or via an external laboratory, is also important, in order to confirm a queen is at an appropriate stage of the reproductive cycle for treatment.
Audience:
This article is aimed principally (but not exclusively) at veterinarians working with cat breeders and whose clientele is increasingly interested in alternative methods of controlling reproduction in pet queens. Surgery is no longer the only choice, and practitioners who manage to keep abreast of new developments will be able to address clients’ needs in a modern, professional way.
Evidence base:
This review draws on a vast body of scientific evidence-based literature. Previously, personal (and sometimes misinformed) opinions, such as the proposed dangerous side effects of progestogens, have been perpetuated in the scientific literature. The papers cited in this review have therefore been carefully scrutinised to distinguish reliable information based on controlled studies from non-evidence-based information.

Introduction
Cats are one of the most fertile domestic animal species, and one for which reproductive control measures have always been advocated in an effort to curb population growth. Controlling reproduction in queens has historically been achieved using surgery, but things started changing in the second half of the last century when purebred cat breeding began gaining momentum following the founding of the Federation Internationale Feline (FIFe) in 1949 (fifeweb.org). One of FIFe’s breeding and registration rules in order for pedigree certificates to be issued is a limit of three litters in 24 months. To fulfil this requirement, a breeding queen’s cycle therefore often needs to be blocked for a few months every year. This has gradually imposed a paradigm shift on veterinarians, as controlling reproduction in breeding queens could no longer entail an irreversible procedure.
For decades, reversible control of feline reproduction has meant the use of synthetic derivatives of progesterone, known as progestogens (or progestins). Unfortunately, progestogens have been misused for a long time based on an inadequate assessment of the evidence published within the veterinary literature. 1 This led to a widespread misconception about progestogens, which are, in fact, safe compounds provided that drug dosing and patient selection are based on scientific evidence. More recently, from the turn of the century, a few interesting alternative drugs for reversible control of reproduction in queens have also emerged, such as long-acting gonadotropin-releasing hormone (GnRH) agonists and melatonin. These have attracted a great deal of interest and are increasingly being considered the standard approach to controlling feline reproduction. Nevertheless, progestogens still maintain their niche in the reproductive armoury of feline practitioners, thanks both to their safety and reliability in terms of duration of action.
This review focuses on the clinical use of these three categories of commercially available drugs, describing how the queen’s reproductive cycle can be temporarily and reversibly blocked without any tangible consequence for her future health and fertility.
Progestogens
Synthetic analogues of progesterone, or progestogens, are reproductive steroids commonly used (principally but not exclusively) to control reproduction in many mammalian species, including humans. Their well-known negative feedback mechanism on the hypothalamus, which triggers a sequence of cascade-type events (inhibition of GnRH release, followed by arrest of pituitary luteinising hormone [LH] and follicle-stimulating hormone [FSH] release, and the consequent block of oestrogen and progesterone secretion) is harmless in itself. However, progestogens have a number of target organs within the reproductive and endocrine systems upon which they will inevitably produce an effect while exerting a (harmless) negative feedback on the hypothalamus.
Appropriate, pharmacological levels of progestogens will produce in any female a number of ‘physiological’ (ie, not harmful, and reversible) effects, as described in the box above. Such peripheral effects are normally as innocuous as the hypothalamic effect, given appropriate drug dosing and patient choice. Unfortunately, however, both drug dosing and patient choice have been made without adequate knowledge and/or consideration of the scientific evidence. As a result, major overdosing has occurred repeatedly. Many of these (unfortunate) queens have been the subject of case reports whose resonance in the scientific and clinical arena has earned progestogens a very negative reputation as dangerous drugs that induce mammary and uterine pathology. 1 Such a reputation is undoubtedly unfounded, as progestogens can be used safely in pet as well as breeding queens of almost all ages, provided that drug dosing is titrated correctly and queens at risk (see ‘patients to be avoided’ on page 867) are not treated.

In animals and humans, the administration of a progestogen allows the female’s cyclicity to be manipulated, essentially to suppress oestrous cycles by inducing a prolonged anoestrus regardless of the stage of the reproductive cycle at the time of treatment.2,3 In queens, long-acting drugs such as medroxyprogesterone acetate and proligestone are normally marketed as depot injections for oestrus postponement, while short-acting drugs such as megestrol acetate (which queens also respond well to when used long-term) are marketed as oral compounds for oestrus suppression.

Megestrol acetate
MA is the progestogen with the highest affinity for progesterone receptors (25 times greater than that of endogenous progesterone), as well as having a considerable affinity for androgen (75%) and glucocorticoid (37%) receptors. Because of its half-life of just a few hours, MA is, in fact, the only progestogen that can justifiably be labelled as ‘short-acting’. It is commercially available as a veterinary product in oral formulation (pills or syrup) throughout most countries of the world. Despite its short half-life, MA has proved helpful for controlling reproduction in queens even over prolonged periods of time. Clinical experience indicates that queens tolerate both oestrus suppression and prolonged treatments well when MA is given at (appropriately) low dosages.
A dose of 2.5 mg MA given as one pill/week in adult cats (corresponding to 0.625 mg/week or 0.009 mg/kg q24h for a 4.0 kg cat) has been shown in two studies in the 1970s to be effective and safe for up to 30 weeks; the only side effects noted were increased appetite and increased body weight, occurring in 33% and 13% of treated cats, respectively.4,5 Unfortunately, there is little, if any, information on the clinical efficacy of this low-dosage treatment protocol as these two early studies have not been considered in subsequent investigations and scientific publications. Instead, overdosing of queens with MA appears to have been practised continuously over the subsequent decades, with numerous case reports and experimental studies detailing side effects resulting from MA protocols featuring much higher dosages than 0.009 mg/kg q24h. Adrenocortical suppression6,7 and temporary impairment of glucose metabolism leading to diabetes mellitus,8–12 have been reported in cats treated with 0.625 mg/kg q24h for 1 week or q48h for 2 weeks. Endocrine and diabeto-genic effects at such high dosages are probably due to the relative affinity of MA for the gluco-corticoid receptors.

Despite these endocrine side effects, such high treatment protocols have proved relatively safe in the past when used for no longer than 1-2 weeks for non-reproductive (derma-tological or behavioural) indications; diabetic and/or adrenocortical side effects are not observed in all cats and, when occurring, tend to be short-lived once treatment is discontin-ued.6,7,10 MA becomes dangerous when the above-mentioned 0.625 mg/kg dose is administered at a much higher frequency, such as daily or every other day for several weeks, or weekly for months. Unfortunately, such protocols have previously been repeatedly used long term for prolonged oestrus postponement, 1 as well as for reproductive reasons, and many cats (both intact and spayed queens, as well as tom cats) have developed mammary gland lesions (mammary hyperplasia, as well as benign and malignant tumours), skin lesions (cutaneous xanthomatosis), blindness (due to hyperlipidaemia causing opacity of the anterior chamber)13–20 and uterine lesions (cystic endometrial hyperplasia [CEH], pyometra, adenomyosis),21–30 as well as more severe and longer lasting endocrine side effects (diabetes and adrenocortical suppression).31–34
Contrary to the published misinformation, MA is a safe drug, assuming correct dosing (~0.009 mg/kg q24h) and careful patient choice (see later). 1 A formulation of MA is commercially available for cat-only use in
Italy (Estropill; MSD Animal Health), with a prescribed oral dosage of 11 - g/kg q24h and a maximum duration of treatment of 12 months. Preliminary reports from the University of Padova, Italy, on clinical use of this compound for periods of up to 2 months indicate that such a treatment protocol is efficacious and safe;35,36 longer durations of treatment are currently being tested.
Medroxyprogesterone acetate
MPA was the earliest available progestogen on the veterinary market. It is a longer-acting compound than MA when used both orally (half-life 12-17 h) or parenterally (half-life 40-50 days), but has a lower affinity for progesterone receptors (five times more potent than endogenous progesterone). MPA has historically been used for prolonged oestrus postponement as the need for only periodic (every 5-6 months) injections in order to induce anoestrus is a more practical approach than daily MA administration.
The induction of prolonged anoestrus was demonstrated in the very first experimental study on MPA in cats, conducted by Harris and Wolchuk around 60 years ago; these investigators used an oral dose of 0.05 mg/kg q24h for 12 months. 37 The protocol proved to be 100% efficacious for oestrus control and safe for general health as well as long-term fertility, as demonstrated by the birth of normal litters between 3 and 6 months after the end of the study. However, while the above study went almost unnoticed among clinicians and researchers, a paper published by Colton only 2 years later, in 1965, reporting on good clinical results of a simpler protocol (3-5-day oral treatment or a single subcutaneous [SC] injection), but using a much higher MPA dosage (achieving a dose range of 6.25-25 mg/kg for a 4 kg cat), 38 attracted much more attention, going on to be referenced widely in reviews and book chapters dealing with oestrus control in cats over the subsequent half a century (see Romagnoli 1 for a review).
Whatever the reason for this misinterpretation of the scientific literature, the emphasis given to Colton’s paper resulted in case reports on uterine and, in particular, mammary side effects in cats treated with high-dose MPA protocols appearing from as early as 1967 and continuing for several decades.39–43 Interestingly, even when used at dangerously high dosages, MPA has rarely, if ever, been reported to have diabetogenic effects in cats, compared with overdosing of MA; this is likely explained by MPA’s low affinity for the glucocorticoid receptor (1000 times less than that of MA). 44 In addition, neither glucocorti-coid nor mineralocorticoid derangements have been reported in cats treated with high MPA doses. 1 This feature of MPA might be useful when deciding upon a treatment.
The work from Harris and Wolchuk 37 demonstrated that, when used at low dosages, MPA can be effective and safe. Unfortunately, however, there is a lack of studies assessing the effect of low dosing of MPA in cats. Most of the MPA-based drugs that are commercially available for veterinary use around the world are parenteral formulations, which makes low-dose daily treatments difficult, if not impossible, to administer. Nevertheless, SC dosages of MPA, varying from 2.0 mg/kg every 3 months to 2.5 mg/kg every 5 months, are probably safe when administered to the correct patient and for the appropriate length of time (see later). Dosages lower than these are likely effective and should be further investigated; notably, a 1.5 mg/kg MPA dose administered intramuscularly every 13 weeks was capable of inhibiting ovulation in Beagle bitches, albeit hypersecretory lobules and some neoplastic mammary nodules were observed following prolonged administration over a total of 40 months. 45 Safety of an intramuscular (IM) 1.5 mg/kg dose every 13 weeks has never been tested in queens; however, such a low dose is likely to be effective and safe provided healthy adult queens are treated with no more than two consecutive administrations. Although caution should be exercised with any inter-species extrapolation, it is worth mentioning that in the study by Harris and Wolchuk the daily oral dose of 0.05 mg/kg MPA that was effective in queens was also capable of suppressing heat in bitches for 12 months. 37
Proligestone
PRG is a drug used solely in veterinary medicine that has been marketed and authorised since the early 1990s for induction of prolonged anoestrus in small animals. In cats it is administered at an SC dose of 100 mg/cat (or 25-30 mg/kg) repeated every 5 months. When compared with other progestogens, it is claimed that PRG is equally effective for the control of oestrus (ie, on the hypothalamic-pituitary-gonadal axis) but less active with respect to the queen’s reproductive (uterus and mammary glands) and endocrine systems or glucose metabolism. 46 A study comparing the efficacy of 30 mg/kg
PRG and 25 mg/cat MA, both SC, in two groups of 15 queens, reported a duration of anoestrus of approximately 8 months for PRG vs 3-5 months for MA. 47 No evidence of mammary or uterine side effects was observed in queens in the PRG group, while two cases of pyometra (but no mammary disease) were observed in the queens in the MA group. It is noteworthy that the treatment protocol for both drugs in this study involved the induction of ovulation with GnRH when queens were in heat. Ovulation induction should not be performed prior to administration of a progestogen, as this will lead to overdosing (through the development of corpora lutea and occurrence of dioestrus), which might have contributed to the development of pyometra. Furthermore, while 30 mg/kg of PRG is an appropriate dose for a cat, an SC dose of 25 mg MA is probably much higher than normal as there is no published information on parenteral use of MA in cats. A fairer comparison would have been between a 30 mg/kg dose of PRG and daily treatment with an appropriate oral dose of MA, permitting safety of treatments to be assessed based on equal duration of efficacy. Nevertheless, a 30 mg/kg dose of PRG allows for an anoestrus of 8 months’ duration without mammary or uterine side effects. 47
However, a case report involving a prepu-bertal 7-month-old Maine Coon queen weighing 4.2 kg that developed benign mammary hyperplasia 4 weeks after being treated with a fairly low dose of 0.75 ml (17.8 mg/kg) PRG cautions that perhaps the mammary gland remains an important target for the action of PRG. 48 In female rabbits, PRG causes a significantly lower degree of endometrial proliferation compared with both MA and exogenous progesterone, 49 although it is unknown whether a lower uterine effect likewise occurs in queens. Similar to other progestogens, PRG can also cause calcinosis circumscripta (local deposition of calcium salts presenting as nodules, often at an SC injection site) in cats. 50 Although the above studies and case reports point to a similarity in the range of side effects between PRG and other progestogens, the currently available information regarding uterine and endocrine side effects of PRG in cats seems to indicate a lower affinity of PRG for the uterus as well as for glucose and adrenal metabolism in cats.
Altrenogest
Also termed allyl-trenbolone, ALT is marketed for oral use in horses and farm animals for oestrus synchronisation. ALT has progesta-tional and antigonadotropic effects but limited oestrogenic and androgenic effects, while its glucocorticoid effects have not been inves-tigated. 51 In a study of this progestogen in the queen, oral dosages of 0.044, 0.088 and 0.352 mg/kg q24h were administered for 38 days to groups of five, five and six queens, respectively. 52 All dosages were effective in controlling heat and inducing anoestrus, but not in suppressing heat in queens with elevated oestrogen concentrations at the beginning of treatment (these queens completed their heat before entering a drug-induced anoestrus). 52 Post-treatment follicular activity started soon after the end of the study, and in a fairly synchronous way (within 10-16 days), for the 0.088 mg/kg treatment. There were no side effects in treated queens. 52 Similar dosages (0.088 or 0.176 mg/kg q24h) were used in six Pallas’s cats for 14-30 days prior to artificial insemination; ovarian function was suppressed equally in the six queens, which subsequently came into heat and ovu-lated, with no difference being seen in terms of numbers of follicles or corpora lutea following the two dosages. 53
Although the above data point to a clear efficacy for the control of reproduction in queens, little, if anything, is known about the safety of ALT for use in cats.
Levonorgestrel and etonogestrel
LEV and ETO are human drugs. The affinity of LEV for the progesterone, testosterone, aldosterone, cortisol and glucocorticoid receptors is 323%, 58%, 17%, 7.5% and 1%, respectively. 54 LEV is frequently combined with ethynilestradiol (EE) as a human oral contraceptive treatment. In one study, an oral formulation of 0.15 mg LEV combined with 0.03 mg EE was used in five cats daily for 90 days. 55 All treated queens displayed regular oestruses during treatment, with an average of 2.6 heats/cat. Failure to induce anoestrus in these queens may have been due to the oestrogen component of the LEV+EE formulation. As there is no information on potential side effects of EE in queens, it would probably be wise to avoid using human contraceptive treatments combining progestogen and oestrogen in queens.
Currently available human LEV preparations can induce anoestruses of variable durations in cats. Oral LEV formulations are characterised by a short duration of action (half-life of 9-14 h) in humans and, therefore, may require once or twice daily administration in cats. Anoestrus was successfully induced in queens with oral administration of 0.750 mg LEV pills divided into two, with half a pill given per day for 37 days. 56 This dosage was effective in keeping queens out of heat, although ovarian activity was not suppressed during treatment based on faecal oestrogen assay and ovarian laparoscopy; reproductive and general health side effects of this LEV oral treatment were not determined. 56
Slow-release SC LEV implants have been tested in queens in experimental studies. When administered twice at a 2-month interval, a dose of 40 mg/kg LEV mixed with poly(lactic-co-glycolic acid) and methylpyrro-lidine induced 36 weeks of anoestrus in cycling queens. 57 A 16 mg LEV silastic implant placed subcutaneously was able to maintain 3/4 treated queens in anoestrus until removal 12 months later. 58 Reproductive activity has been suppressed in queens without any relevant side effects on the mammary glands, body weight and glucose or adrenocortical metabolism,58,59 although elevations in baseline fecal estradiol concentrations have been observed during treatment. 60 Three of the four queens treated with the silastic implant exhibited oestrus and conceived within 2 months of implant removal, confirming normal fertility following cessation of treatment; endometrial hyperplasia and fluid accumulation occurred in 1/4 cases, suggesting the potential for development of pyometra following long-term use. 58
First generation LEV implants were taken off the market in 2002 due to difficulties with the insertion procedure and replaced by the parent compound ETO, which has a longer half-life (25-29 h) than LEV and less androgenic activity, but a slightly higher affinity for the glucocorticoid receptor (14%). Currently available ETO implants, which feature an improved disposable trocar for insertion, have been used in cats in an experimental study in Brazil in which a single 68 mg ETO implant induced anoestrus for the entire duration of treatment until removal 37 days later. 56 Ovarian activity was suppressed during treatment based on faecal oestrogen assay and ovarian laparoscopy; reproductive and general health side effects of the ETO implant were not assessed. 56
In women, ETO implants provide up to 3 years of continuous contraception. Therefore, ETO implants might present an interesting alternative in countries where veterinary products for the control of reproduction are not available to feline practitioners; however, general health and reproductive safety for queens need to be demonstrated. As the daily release rate of ETO (60-70 ug/day until month 5-6, gradually decreasing to 30-40 ug/day during the second year 61 ) is similar to the daily release of LEV, one might speculate that long-term use of ETO implants may produce the same uterine side effects in queens as LEV implants. Furthermore, the size of ETO implants (40 mm x 2 mm) may be a concern for use in queens.
Chlormadinone acetate and delmadinone acetate
CMA and DMA are progestogens that are also available in some countries as veterinary products. DMA is marketed in several Western countries as a veterinary-only product. It is licensed for treating reproductive problems in male dogs and, as such, has not been used extensively in cats. CMA is widely marketed all over the world as a human contraceptive compound; it is also licensed for cattle in Austria and has been used for oestrus control studies in queens. CMA’s affinity for the progesterone receptor is 33% higher than that of endogenous progesterone. 62 CMA has a low affinity for the glucocorticoid receptor, a negligible affinity for androgen or oestrogen receptors and no affinity for the mineralocorticoid receptor. 62 Despite its negligible affinity for the androgen receptor, when used at high concentrations CMA may compete effectively with andro-gens in target tissues to block their effects.63,64 The efficacy of CMA in cats has been extensively studied in Japan. Sawada and colleagues treated 15 immature queens for 24-43 months with weekly oral doses of 2.0 mg/cat, while 10 mature queens were treated with weekly oral doses of 2.0 mg/cat (n = 7) or 6.25 mg/cat (n = 3) for 12 months. Queens in oestrus at the start of the study were treated with higher oral doses (12.5 or 25 mg/cat) for the first few weeks. 65 None of the pre- or postpubertal queens treated with the above dosages of CMA showed heat during the entire duration of the study; a 15% increase in body weight was observed in these queens at the end of the study. 65 Despite a claim that no uterine or mammary side effects were observed throughout the study, treated queens were only assessed clinically, and no diagnostic imaging or histopathological examinations were carried out. 65 In another study, SC implants of CMA used in 13 queens at doses of 0 (n = 3), 2.5 (n = 3), 5 (n = 3) or 20 (n = 4) mg/kg suppressed oestrus signs for 12 months; for the 2.5 and 5.0 mg/kg doses, there were no uterine changes, mild mammary lobular development with proliferation and secretion, and no effects on general health; 66 the higher (20 mg/kg) dose was associated with endometrial hyperplasia with mild uterine fluid accumulation. 66 In a longitudinal study involving 24 queens, 19, 16 and four cats were treated for 6, 8 and 10 years, respec-tively. 67 Mammary nodules and vaginal discharge or pyometra were recorded in 11% of cats treated for 6 years, 38% of cats treated for 8 years and 25% of cats treated for 10 years. 67 The only information available for DMA in cats is that a single oral dose of 2.5 mg DMA (also 5.0 mg CMA) is reportedly efficacious in causing failure to conceive when given to queens 24 h after the onset of a heat during which the queen was mated. 68
Recommended dosages of progestogens in the queen and reported side effects
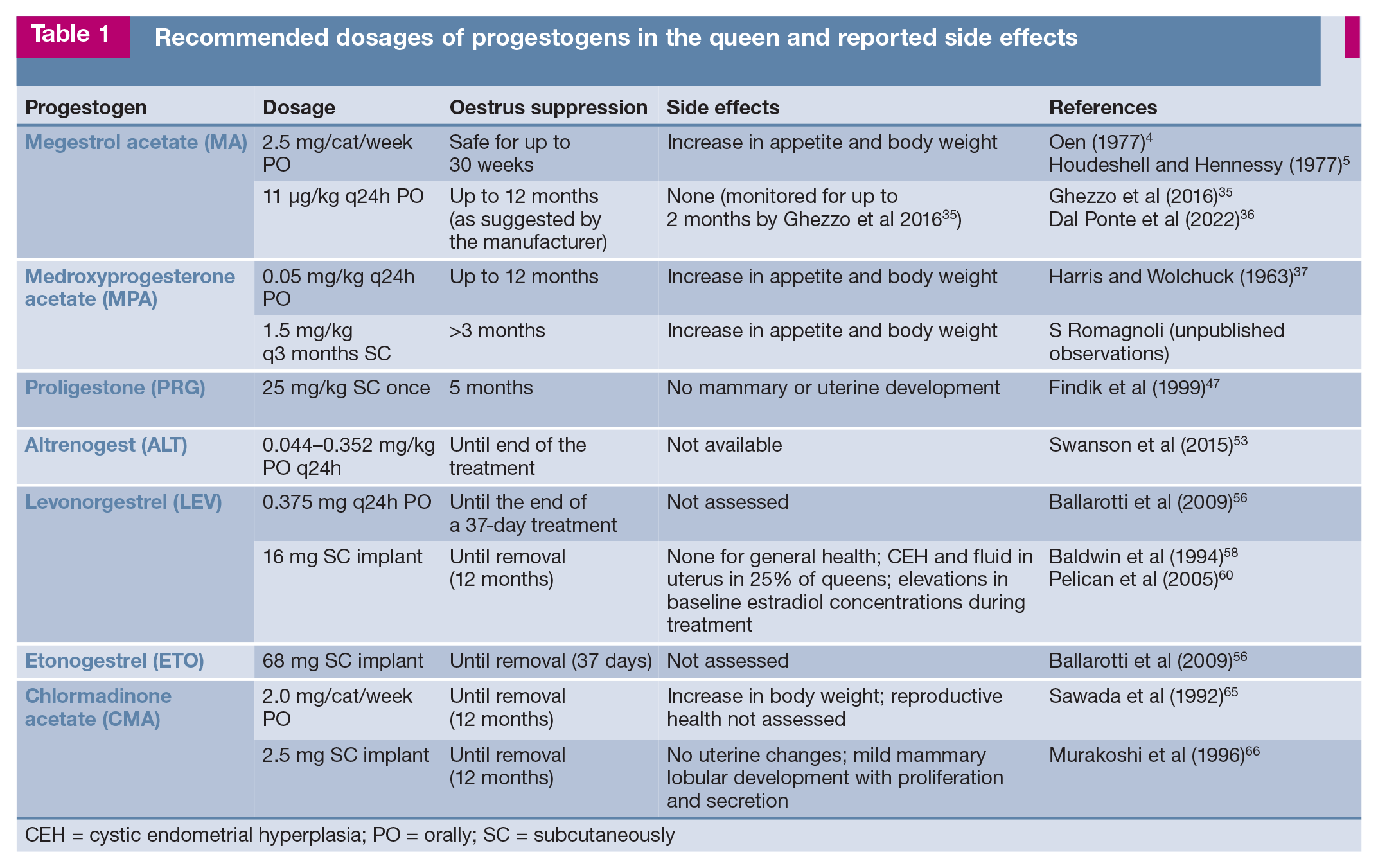
CEH = cystic endometrial hyperplasia; PO = orally; SC = subcutaneously
Long-acting GnRH agonists
Long-acting GnRH agonists block reproduction through a different type of feedback mechanism from the one used by progestogens. Whereas progestogens simply inhibit the production of hypothalamic GnRH, and consequently the pituitary gonadotropins FSH and LH (through negative feedback), GnRH agonists actually initially provide powerful stimulation (positive feedback) to the pituitary causing the release of FSH and LH for up to 2 weeks. 69 Two types of GnRH agonists have been used in queens, namely deslorelin and azagly-nafarelin.
Deslorelin had traditionally been (and continues to be) marketed as an SC implant for reversible pharmacological castration of male dogs, with a duration of action of at least 6 (4.7 mg) or 12 (9.4 mg) months. The use of both formulations in cats has been attracting the interest of researchers and clinicians, and is becoming increasingly popular among breeders, particularly since the 4.7 mg deslorelin implant has recently (June 2022) been approved for use in male cats in Europe, and hence no longer constitutes extra-label use. A 20 mg implant of azagly-nafarelin has been used in cats with good success, 70 but the product has never been launched (despite meeting European registration requirements) and, therefore, there is a lack of clinical data on its use in queens. Unless otherwise stated, the following discussion focuses on the use of deslorelin in cats.
Pharmacokinetic data available for adult male dogs treated with the 4.7 mg deslorelin implant indicate that serum deslorelin concentration peaks during the first week post-treatment and then decreases gradually, reaching undetectable levels around 80 days post-treatment. 71 Although similar data on deslorelin do not exist for adult cats, clinical evidence suggests that an initial peak during the first week post-treatment most likely occurs in this species as well; the span of the gradual decrease may differ, however, as the clinical efficacy of deslorelin implants in cats extends for much longer than in dogs.
The endocrine features of the 2-week post-implantation period, commonly referred to as the ‘flare-up phase’, have been poorly characterised in queens. 72 The immediate result of the strong gonadotropin secretion caused by deslorelin may or may not be the induction of heat, depending on the stage of the reproductive cycle (see later). Once the flare-up phase is over, downregulation of pituitary GnRH receptors will occur, causing temporary pituitary desensitisation with suppression of the function of the hypothalamic-pituitary-gonadal axis. In the absence of gonadotropin stimulation, the queen’s ovaries will stop functioning and she will enter anoestrus.
In intact queens, deslorelin may be used to delay puberty, induce oestrus or induce anoestrus. Regardless of the rationale for treatment and the phase of the cycle when the queen is treated, anoestrus is the endpoint of any deslorelin treatment of adult queens, as even when oestrus is induced, a prolonged phase of reproductive quiescence will inevitably follow (unless the implant is removed). Although this reproductive quiescence is often referred to as ‘medical (or chemical) sterilisation’, in terms of the reproductive cycle this is simply either an extension of normal anoestrus (if treatment is administered during anoestrus) or an anoestrus occurring during the breeding season. Oestrus induction is not a common requirement in feline practice and can be a nuisance in a queen presented for oestrus control; however, inducing a heat may be useful in queens presented for prolonged anoestrus or undergoing artificial insemination (as discussed in an accompanying review in this series 73 ). 74
Delaying the onset of puberty
Prepubertal treatment of queens with a 4.7 mg deslorelin implant causes a delay in the onset of puberty, with reports of puberty occurring between 9 and 36 months of age in treated cats.75,76 The mechanism by which this delay occurs has not yet been closely investigated. Prior to puberty, pituitary GnRH receptors are likely to be insensitive to GnRH. Therefore, rather than a desensitisation of pituitary receptors, a GnRH agonist is likely to cause a premature block of the hypothalamic-pituitary axis just before the onset of pubertal sensitivity.
In an experimental study, Risso and colleagues treated 100-day-old queens (maintained in a laboratory environment in Argentina) with a 4.7 mg deslorelin implant, observing a significant delay in the onset of puberty in all 13 treated cats, which reached puberty at 9 months, compared with 6 months in the 15 control cats. 75 A longer delay in the onset of puberty was reported by Cecchetto et al who, in Italy, treated nine prepubertal, client-owned queens, aged between 5 and 9 months, with a 4.7 mg deslorelin implant. Four out of nine queens completing the study showed onset of puberty at between 21 and 36 months, and a fifth queen was spayed at 24 months while still prepubertal. 76 The difference between the two studies in the extent to which puberty was delayed may be due to latitude (45.24°N vs 34.5°S), breed, age of the queens entering the study, season at the point when the implant’s activity ceased (unknown in both studies) and/or the living conditions of the cats (laboratory vs family home).
On average, the onset of puberty in domestic cats is reported to occur from as early as 3-6 months to as late as 12-16 months, depending on the breed.77–79 Deslorelin treatments used to delay feline puberty should therefore probably be started 3-6 months before the expected age of pubertal heat. Queens treated close to their natural onset of puberty may show either a full heat response (as reported for one queen in each of the two above-mentioned studies75,76), or some degree of vaginal epithelial keratinisation. 76 While a full heat means the attainment of puberty, a keratinised vaginal smear followed by a silent heat in a young queen implanted with deslorelin likely indicates that the pituitary is gradually maturing and is partially capable of responding to GnRH, producing minor LH pulses. In prepubertal bitches, or those in late oestrus, such minor LH pulses are typically seen when the pituitary is gradually becoming (but is not yet) responsive to GnRH. 80 Therefore, a queen that shows only vaginal epithelial keratinisation following treatment with deslorelin is considered to remain pre-pubertal unless a normal heat develops immediately afterwards.
Induction of oestrus
In adult queens, administration of a deslorelin implant will stimulate follicular growth and oestradiol secretion, which is frequently followed by the display of a normal oestrus. Almost all studies on the use of deslorelin in queens have documented the occurrence of oestrus post-treatment in varying proportions of the cats.81–87 Such oestrus is fertile and the queen will conceive if mismated 87 or artificially inseminated. 74 The occurrence of oestrus depends on the stage of the reproductive cycle at the time of treatment. Among queens in anoestrus or interoestrus (with a low [<1.5 ng/ml] serum progesterone concentration), 1/1 84 and 9/9 87 showed a normal oestrus within 2-5 days of treatment. In contrast, oestrus was not observed in 9/9 queens implanted during dioestrus 84 or in 7/7 queens implanted following treatment with MA while in proestrus or interoestrus. 82 However, even if oestrus is not displayed, oestradiol production (based on faecal assay) increases in all queens treated with deslorelin, irrespective of the stage of the cycle or treatment with a progestogen. 82 When 10 queens were implanted during the follicular phase (2-4 days after the beginning of oestrus), oestrus manifestations stopped 4 days later, but resumed in one queen on day 6. 84 Indeed, the occasional occurrence of vaginal epithelial keratinisation and/or resumption of oestrus manifestations has been reported during the first few months following implanta-tion;81,82,84,85,87,88 such heats, which are difficult to explain, tend to be isolated and self-limiting.
Ovulation in queens implanted during anoestrus or interoestrus, as demonstrated by an increase in serum progesterone concentration, has been reported both after deslorelin 84 and azagly-nafarelin treatment. 71 Ovulation in these queens is probably the result of the continuous secretion of LH during the flare-up phase. Persistent heat following implantation with deslorelin has been reported in two queens failing to ovulate. 87 In the bitch, the development of follicular cysts leading to persistent heat has been confirmed post-deslorelin treatment.89–91 However, the reason for persistent heat in the two queens of the above-mentioned study was not determined, as there was neither macroscopic nor histolog-ical evidence of pathological ovarian cystic structures following ovariohysterectomy. 87
Induction of anoestrus
Regardless of whether the queen is prepubertal or adult, and irrespective of the stage of the reproductive cycle, treatment with deslorelin will, as noted earlier, inevitably cause a prolonged phase of reproductive quiescence. Shortly after treatment, serum progesterone will increase due to ovulation caused by high levels of LH during the flare-up phase. 84 Subsequently, in non-pregnant queens, serum progesterone will peak at around 2 weeks post-implantation, then plateau during the third and fourth weeks post-treatment, before reaching a nadir at 2 months. 84 Queens implanted in dioestrus may see their luteal phase prolonged to 60 days or more, 83 presumably as a result of increased luteal production subsequent to increased LH secretion during the flare-up phase. The fact that the duration of the luteal phase in queens treated with deslorelin is roughly similar to the duration of pregnan-cy83,84 leads to the speculation that prematureluteal failure (reported for bitches mated at an oestrus induced with deslorelin) does not occur in queens treated with deslorelin. Indeed, this hypothesis seems to be confirmed by the normal duration of the luteal phase in deslorelin-treated queens 83 and by a case report of a queen carrying a pregnancy to term after being treated with deslorelin following an (unplanned) mating 8 and 9 days prior to implantation. 92 The queen produced four live normal kittens, indicating normal progesterone secretion throughout gestation; however, she failed to lactate normally and did not show appropriate maternal behaviour. 92 Interestingly, a queen treated with MA and then implanted with deslorelin showed a fully fledged pseudo-pregnancy. 82 Additionally, galactorrhoea was observed in a queen 2 months following implantation with deslorelin; the queen was not pregnant and early abortion was ruled out. 85 As mammary signs did not resolve following two courses of treatment with meter-goline, the implant was removed and 20 days later milk secretion ceased. 85

Although the above observations may seem rather conflicting (lack of milk secretion vs excessive milk secretion), taken together they confirm that, as in the bitch, feline mammary function relies on a fine interplay between progesterone and prolactin that can be easily disrupted. Prolonged secretion of progesterone might inhibit the prolactin peak, leading to agalactia, while a sudden fall in progesterone (or a regular fall but from a higher than normal concentration) might trigger prolonged milk production.
If mating and conception do not occur, the queen will transition from dioestrus into anoestrus. This may last - when using the 4.7 mg deslorelin implant - from as little as 4-14 months86,88 to 18-26 months81,82 and, in a few cases, up to 37-38 months.84,86 Similar to the postponement of puberty, the duration of induced anoestrus in adult queens also shows a puzzling variability. Potentially, this might be explained by differences in the breeds of cats treated, their living conditions (most of the above studies were performed on experimental cats in laboratory conditions, other than the studies from Pisu and Romagnoli 81 and Furthner et al, 86 which involved privately owned queens), other environmental factors (season, temperature, outdoor access) and/or the photoperiod. Feline anoestrus in privately owned queens following use of the 9.4 mg deslorelin implant is reported to last 26 ± 4 months. 93
Fertility following deslorelin-induced anoestrus has been confirmed in all studies in which this has been investigated, based on the capacity to ovulate, 83 the return to normal seasonal cyclicity, 84 the production of fertilis-able oocytes 85 and queens conceiving and giving birth to normal litters following the use of the 4.7 mg, 84 as well as the 9.4 mg, 93 implant. The scope to induce such a prolonged phase of reproductive quiescence that then allows for full reversibility to normal fertility is a great advantage for feline practitioners. However, there are two main disadvantages in using deslorelin to induce anoestrus in breeding queens: first, unpredictability in terms of onset and duration of action and, second, excessive duration of action.
Unpredictability is a concern as currently there are no means to predict when an implant’s activity will cease and the treated queen will come back into heat. Conceivably, episodes of vaginal epithelial keratinisation might be used to predict that an implant’s activity is waning, although such episodes may occur in the middle of a prolonged anoestrus period unconnected with the end of action of the implant.81,82,84,85,87,88 Excessive duration of action is likewise a concern, particularly for breeders, as cyclicity of breeding queens typically needs to be blocked for periods of time varying from a few months to 6-12 months, after which they need to be returned to normal fertility quickly. This problem can be circumvented by implant removal at a convenient time. In a study that involved removal of a deslorelin implant 3, 6 or 9 months after implantation, ovarian function resumed after 3 weeks for queens in an increasing photoperiod and up to 7 weeks for those in a decreasing photoperiod; moreover, queens that were mated conceived and produced lit-ters. 87 When removal is planned, the implant is best placed subcutaneously in the periumbili-cal area, as the subcutis is thinner, allowing implants to be located and removed more easily. Implants placed in between the shoulder blades are virtually impossible to remove.

Melatonin
Melatonin is a neurohormone produced by the pineal gland, a neuroendocrine organ that modulates many physiological functions (including reproduction) in a circadian rhythm. Melatonin is released into the general circulation in a continuous fashion, but with greater intensity and reaching higher concentrations during periods of darkness. 95 Therefore, the longer the night, the higher the concentration of melatonin. Serum melatonin concentrations reach maximum levels during the period between late autumn/fall and early winter as nights steadily increase in duration - this corresponds to the anoestrus season for cats. 96 As soon as nights become shorter in late winter and early spring, melatonin concentrations start to decrease and quickly attain basal values, which are maintained during the rest of the year - which is when cats have their breeding season. 96 Melatonin inhibits feline ovarian function and, similar to other seasonal species, its secretion is inversely proportional to the activity of the feline reproductive system. By binding to receptors located in the suprachiasmatic nucleus of the hypothalamus (the ‘central clock’ in mammals, generating circadian rythms), melatonin blocks tonic as well as surge secretion of GnRH.97,98
The effect of light-mediated secretion of melatonin on feline ovarian function is so well defined that, under laboratory conditions, queens will stop cycling shortly after the day length is reduced to 8 h/day, and will then resume cycling approximately 2 weeks after the photoperiod is brought back to 14 h/day.99,100 Furthermore, intravenous administration of melatonin can block ovarian function and cyclicity, 101 decrease LH secretion following mating and extend the duration of the refractory period that follows each coitus. 102 Following these findings from early studies on the control of feline reproduction with melatonin, various attempts have been made in the past two decades to find a more practical means of using melatonin for this purpose. Attention focused first on oral administration and, more recently, a melatonin implant marketed for use in small ruminants has been examined experimentally in queens.
Oral administration of a 4 mg melatonin pill 2 h prior to darkness, once daily until the following oestrus, induced an oestrous-free period of approximately 2 months in queens in one study. 103 All queens were kept in a laboratory environment and treated in interoestrus (at least 2 days after the end of oestrus). In 7/19 treated cycles the queen ovulated, demonstrating that ovulation induction following oral melatonin administration is not an unusual phenomenon when treating queens in interoestrus. The interval between treatment and the subsequent oestrus for these 19 cycles was 50 days, while the inter-oestrous interval (oestrus to treatment plus treatment to subsequent oestrus) was 63 days. A study using a higher oral melatonin dose of 30 mg/cat, given 3 h prior to darkness to six laboratory queens for 35 days, found that ovarian activity was inhibited from day 25. 104 During the first 25 days of melatonin treatment, 3/6 treated queens went through two heat periods (based on faecal oestrogen concentrations); there was no faecal evidence of oestrus in the other three queens. All six cats showed oestrus suppression from day 25 onwards that extended for an additional period of 33 days following treatment withdrawal. The stage of the cycle at the time of treatment was not identified. Furthermore, the three queens showing heat might have ovulated following their second heat, based on an interval to the subsequent heat of approximately 40-50 days. However, serum progesterone was not assayed in this study, making the incidence of melatonin-induced ovulation impossible to assess. 105
The availability of SC implants of melatonin produced for small ruminants prompted researchers and clinicians to test this new formulation in cats. In an early study on laboratory cats, eight queens were implanted with either one (n = 4) or five (n = 4) 12 mg mela-tonin implants (Wildlife Pharmaceuticals). 105 Similar to the study using 30 mg melatonin given orally, 104 these eight queens took, on average, 20 days to stop cycling. The mean period of ensuing oestrus suppression was 75 days, although this result was obtained only in 2/4 queens administered one implant and 3/4 queens administered five implants. 105 Unfortunately, the stage of the cycle at the time of treatment was not reported in this study either and serum progesterone was also not assayed, again making it impossible to assess the incidence of melatonin-induced ovulation. Interestingly, histopathol-ogy revealed uterine wall thickening and evidence of CEH. However, the presence of endometrial lesions prior to study onset was not ruled out. Furthermore, CEH may be due to and/or exacerbated by prolonged endogenous progesterone stimulation, pointing to an indirect role of melatonin in causing endometrial lesions (ie, an already compromised endometrium may develop a complicated CEH due to progesterone secretion rather than as a direct consequence of melatonin).
All subsequent studies on melatonin implants in cats were performed using an 18 mg implant marketed for use in sheep (Melovine; Ceva). The availability of this implant on the market as a veterinary product in many countries, together with the relative ease with which it can be administered to queens, has attracted the interest of feline practitioners and breeders alike for its potential anoestrus-inducing effect. However, the occurrence of oestrus after implantation and the short duration of induced anoestrus appear to be limiting factors in queens.

Post-implantation oestrus (based on oestrus behaviour and/or vaginal epithelial keratinisation within the first 7-10 days after implantation) was not identified in any of six queens implanted during seasonal anoestrus 106 or in 17 cycles of queens treated in very early interoestrus (within 2 days following the end of the previous heat). 103 However, when implantation was performed later on in interoestrus, oestrus was observed in 33% (3/9), 107 31% (6/19) 108 and 39% (12/31) 109 of treated queens. The incidence of post-implantation oestrus reached 78% (7/9) when queens were implanted in oestrus, while it was 0% (0/6) when queens were implanted in dioestrus. 109
The duration of effect (duration of induced anoestrus, or interval between treatment and the subsequent oestrus disregarding any post-implantation oestrus) in queens implanted with melatonin has been reported as approximately 100 days (in 5/6 queens), 106 64 ±5 days, 103 80 ± 25 days, 110 86 ± 50 days, 86 104 days 109 and 113 ± 6 days 107 in queens treated during anoestrus, while it was 61 ± 7 days for queens treated during oestrus. 107 Therefore, a number of conclusions can be drawn on use of the 18 mg melatonin implant in queens (see box above).
As for the GnRH agonists, the duration of melatonin-induced anoestrus in queens treated during the breeding season varies quite remarkably - ranging from <1 month to >7 months -and seems to be unpredictable. Although the longer intervals may be of interest from a breeder’s perspective, the high frequency of induction of ovulation in queens treated with melatonin is a concern owing to the potential negative impact of recurrent luteal phases on uterine health (ie, stimulating the development of CEH and, in turn, lowering fertility). Possibly the use of melatonin implants could be restricted to those queens that respond with a long interoestrus without ovulation at the first treatment - although the efficacy of sequential applications of this implant needs to be verified. There is certainly a need for more research on the effect of season at the time of implantation on the duration of melatonin-induced anoestrus, as well as on the role of the environment. The inhibitory effect of melatonin on the hypothalamic-pituitary axis is not as strong as that of deslorelin. As a consequence, resumption of cyclicity in melatonin-treated queens might be triggered by even minor stimuli, such as the presence of a tom or other queens in heat, or exposure to a long photo-period (eg, when cattery lights are kept on until late in the evening). Care should be taken to minimise any external stimuli when trying to induce anoestrus in queens with melatonin.


Key Points
✜ Progestogens, GnRH agonists and melatonin are commercially available drugs that can temporarily and reversibly block the reproductive cycle of the queen without any tangible consequence for future health and fertility.
✜ Contrary to previously published misinformation, progestogens are safe, provided that dosing and patient selection are based on scientific evidence. There is a large body of clinical and safety data available in cats for MA, MPA, PRG, LEV and CMA; only very basic information is available for ALT, DMA and ETO.
✜ Two GnRH agonists have been used to control the reproductive cycle of queens, namely deslorelin and azagly-nafarelin; both are regarded as safe. There is little clinical data for the latter in queens, while the extra-label use of deslorelin in queens has attracted the interest of researchers and clinicians, and it is becoming increasingly popular among breeders. In intact queens, deslorelin may be used to delay puberty, induce oestrus or induce anoestrus.
✜ Melatonin is also relatively safe to use in cats, is available as a veterinary product in many countries and can be administered 1 with relative ease to queens. The occurrence, in many instances, of oestrus after implantation and the short duration of induced anoestrus appear to be limiting factors, however.

Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Stefano Romagnoli receives funding from Virbac for research on deslorelin.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not specifically required for publication in JFMS.
Informed consent
This work did not involve the use of animals (including cadavers) and therefore informed consent was not required. No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
