Abstract
Objectives
The aim of this study was to establish the safety and efficacy of a novel multidrug lomustine-based chemotherapeutic protocol for cats with high-grade multicentric or mediastinal lymphoma, in an area endemic for feline leukemia virus (FeLV).
Methods
This prospective study included owned cats, diagnosed (cytologically) with multicentric or mediastinal lymphoma and treated with the LOPH (lomustine, vincristine [Oncovin; Antibióticos do Brasil], prednisolone and hydroxydaunorubicin [doxorubicin]) protocol. A complete blood count was performed before every chemotherapy session and any significant abnormalities recorded as possible related toxicities. Median survival time (MST) and disease-free interval were estimated by Kaplan–Meier curves.
Results
Twenty-one cats were included in this study. Nineteen (90.5%) tested positive for FeLV and were therefore considered to have persistent viremia. Complete response was reported in 81% (n = 17/21), while three had partial remission and one had no response. Seven cats finished the induction protocol within 20–31 weeks (23.1 ± 4.5; median 20) and all seven received a maintenance protocol. The MST (lymphoma-related survival) for the 21 cats was 214 days. The MST was 214 days for cats with mediastinal lymphoma (n = 13), but it was not reached for multicentric lymphoma (n = 8; P = 0.9). The MST of cats with persistent FeLV antigenemia was 171 days. Grade I anorexia and vomiting occurred in 19% of the cats (n = 4/21). Hematologic toxicity was found in 100% of the cats at some point during their treatment, but it was mostly grade I or II. Neutropenia, thrombocytopenia and anemia occurred in 16/21, 21/21 and 15/21 cats, respectively.
Conclusions and relevance
The LOPH protocol was well tolerated by cats with lymphoma and persistent FeLV viremia, and resulted in a better MST than similar studies with other protocols. Novel studies and controlled trials are necessary in order to evaluate the efficacy of different protocols according to the lymphoma subtype, anatomic form and FeLV status.
Introduction
Lymphomas are lymphoid neoplasias emerging from peripheral organs (lymphatic or extralymphatic) and are classified as nodal, when arising from the lymph nodes (eg, multicentric or mediastinal), or extranodal (eg, thymic, gastrointestinal, cutaneous, ocular, renal or spinal). 1 Lymphoma may represent up to 90% of hematopoietic neoplasms in cats and approximately one-third of all tumors in this species. 2 In areas where feline leukemia virus (FeLV) is not endemic, intestinal and mesenteric lymph nodes are the most affected sites, mainly in older cats (10–12 years).3–5
However, disease epidemiology is quite different in regions endemic for FeLV. Despite vaccine availability, FeLV remains highly prevalent in developing countries.6–8 The median survival time (MST) for cats with progressive FeLV infection (persistent viremia) is 2.4 years (median), and only 20% are alive after 3 years. 4 Most deaths are related to FeLV degenerative diseases or opportunistic infections, but up to 23% of these cats may develop hematopoietic neoplasms, mainly lymphoma. 9 FeLV plays a direct role in lymphomagenesis, through insertional mutagenesis, increasing the risk of lymphoma 62-fold. 10 A common integration domain is usually related to proto-oncogenes (MYC, FLVI-1, FLV1-2, FIT-1, PIM-1 and FLIT-1), and the U3 region of the long terminal repeat retrovirus is capable of promoting gene transcription and cell proliferation, which can increase genome instability and novel mutations.10,11 Diseased cats are mostly young (<3 years old), usually rescued from the street, with outdoor access or direct contact with feral cats. In these cats, lymphomas are usually high grade, and multicentric or mediastinal, although extranodal lymphomas may also occur. 12 Feline immunodeficiency virus (FIV) plays an indirect role in carcinogenesis and increases the risk of lymphoma five-fold. 10
A plethora of protocols are currently available for treatment of intermediate-to-high-grade feline lymphoma2,13–27 There is no standard care for feline lymphomas; however, high-dose cyclophosphamide, vincristine (ie, Oncovin; Antibióticos do Brasil) and prednisolone (COP) is widely used, and it is thought to provide the highest complete response (CR) rates and longest disease-free interval (DFI) and MST, with minimal treatment-related toxicity.2,13,14 While remission rates are similar among FeLV-infected or non-infected cats,2,23,25 the survival time is reduced in persistently viremic cats. 25 The MST for FeLV-related lymphoma ranges from 37 to 126.3 days,2,21,25 while for non-antigenemic cats it ranges from 105 to 388 days.2,25,27
The present study was conducted in an area where FeLV has a high prevalence and mortality associated with lymphoma, despite chemotherapy. In our experience, these cats have a very poor prognosis and treatment with most protocols results in survival times of only 30–60 days.21,25 Previous studies revealed a low efficacy of lomustine and doxorubicin as rescue agents in cats with intermediate-to-high-grade lymphoma.26,28 Nevertheless, lomustine appears to have potential as an induction agent, 22 as does doxorubicin in prolonging the duration of the first remission. 19 We have proposed a new intensive protocol for induction of remission, including lomustine, vincristine (ie, Oncovin), prednisolone and doxorubicin (hydroxydaunorubicin) (LOPH).
This study aimed to establish the safety and efficacy of a novel multidrug lomustine-based chemotherapeutic protocol for cats with high-grade multicentric or mediastinal lymphoma, in an area endemic for FeLV.
Material and methods
This research was approved by the Ethics Committee on Animal Use from the Universidade Vila Velha (Brazil), number 462/2018, and the cats’ owners consented to their participation in the study.
This prospective study included client-owned cats with a cytologically confirmed high-grade lymphoma (multicentric or mediastinal), presented between March 2018 and June 2019. No distinction was made between thymic and mediastinal nodal lymphoma. Cytologic criteria were based on a modified Kiel classification. 29 Cats that received no treatment or had received glucocorticoids for <10 days, or a single dose of vincristine within the last 10 days, were included.
Exclusion criteria included recurrent/relapsed lymphoma and extranodal (ie, gastrointestinal, cutaneous, renal and spinal) and/or low-grade lymphomas. Cats in the so-called ‘leukemic phase’, diagnosed via bone marrow aspirate (cytology) or suspected on complete blood count (CBC; ie, bicytopenia associated with lymphocytosis higher than 20 × 109/l, or identifiable blasts) were also excluded. Further eligibility criteria were no other serious medical illness that would limit full compliance with the study.
All cats had a minimum database, including CBC and blood smear examination, serum biochemistry panel (urea, creatinine, alanine aminotransferase [ALT], aspartate aminotransferase [AST], gamma-glutamyltransferase [GGT], alkaline phosphatase [ALP], total protein and albumin), SNAP Feline Triple Test (IDEXX Laboratories), three-view chest radiographs (left lateral, right lateral, dorsoventral; although two cats had only the two lateral view radiographs) and abdominal ultrasound. Cats that were negative on the SNAP Feline Triple Test had whole-blood PCR analysis for FeLV provirus. Clinical staging was determined according to Terry et al, 30 as shown in Table 1. Unfortunately, complete staging was performed after initial treatment in cats that had previously received vincristine and/or prednisolone. Staging was based on echogenicity suggestive of spleen or liver involvement, but aspirates were not taken for confirmation. Bone marrow aspirates were not collected from the included cats. Immunophenotyping was also not performed.
Clinical staging system for feline lymphoma (adapted from Terry et al) 30

The LOPH protocol consisted of the weekly administration of cytotoxic agents, including reformulated lomustine, vincristine (ie, Oncovin), prednisolone and hydroxydaunorubicin (doxorubicin), as shown in Table 2. A CBC and blood smear examination was performed before every chemotherapy session, and a biochemistry panel (urea, creatinine, ALT, AST, GGT, ALP, total protein and albumin) was carried out at the start of every new cycle of chemotherapy (every 4 weeks).
LOPH protocol for feline high-grade lymphoma

Repeat five times (total 20 weeks)
Prednisolone was given continuously at 10 mg/cat/day in the first cycle, followed by a gradual decrease if in remission in the second cycle (25% every 2 weeks; ie, 7.5 mg/cat/day for 2 weeks, 5 mg/cat/days for >2 weeks and then discontinued)
Prior treatment with dexamethasone (0.5 mg/kg IV)
For cats in remission after finishing the 20-week protocol, a maintenance phase was initiated with chlorambucil 2 mg/cat Monday–Wednesday–Friday for 8 weeks, and vincristine 0.75 mg/m2 intraperitoneally as follows: two with a 2-week interval; three with a 3-week interval; four with a 4-week interval; and every 6 weeks until 2 years from diagnosis.
Response was assessed via physical evaluation and, for mediastinal presentation, with thoracic ultrasound, before every chemotherapy administration. Cats were classified as having a complete response (CR) (regression of all measurable disease and clinical signs), partial response (PR; decrease of >50% but <100% in the measurable disease) or no response (NR; decrease of <50% or an increase). 2 DFI and MST were estimated by Kaplan–Meier curves. DFI was calculated from clinical remission to disease recurrence, and overall survival (OS) from the first dose of chemotherapy to death related to the disease (lymphoma). Unrelated deaths (including FeLV-related anemia or bone marrow aplasia) were censored, along with those lost to follow-up. The Cox–Mantel log-rank test was used to compare DFI and MST according to anatomic classification, stage, sex, age and weight. Statistical analyses were performed in Graph Pad Prism version 6.02.
Reference intervals (RIs) for hematologic parameters were defined according to Klaassen, 31 as routinely used in the university’s laboratory, as it was not possible to determine our own RI based on the attending population, which was commonly affected by chronic diseases. Adverse events were graded at the time the cat attended, according to the Veterinary Cooperative Oncology Group – Common Terminology Criteria for Adverse Events following chemotherapy version 1.1, 32 as shown for hematologic toxicities (Table 3).
Severity of adverse events related to chemotherapy in cats according to the Veterinary Cooperative Oncology Group version 1.1 32
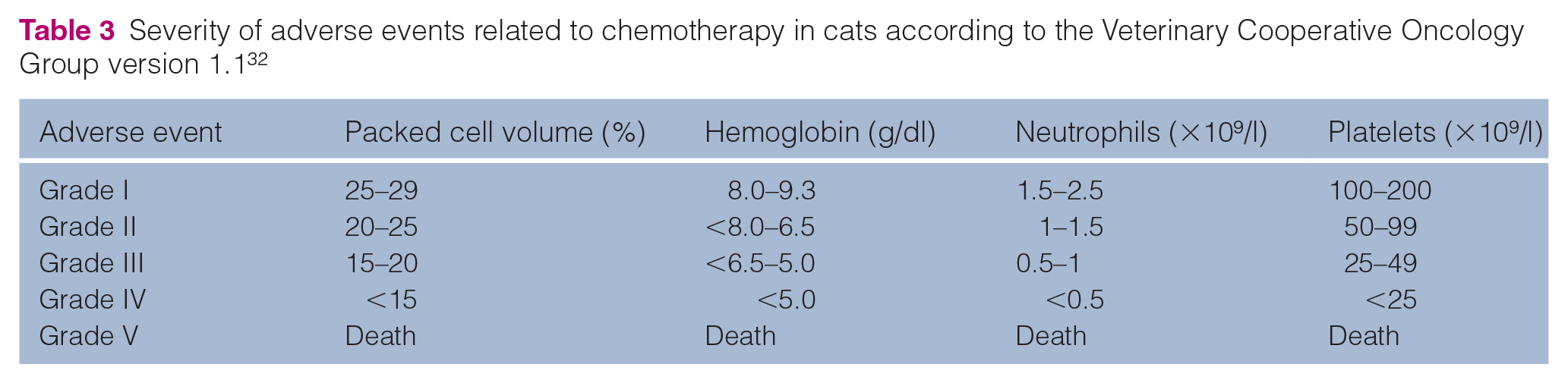
Chemotherapy was performed if neutrophil counts were >1.5 × 109/l and platelets were >50 × 109/l. Antibiotics – norfloxacin (22 mg/kg q12h) or marbofloxacin (5 mg/kg q12h) – were started if the neutrophil count was <1 × 109/l, but they were withdrawn when the neutrophil count was > 1 × 109/l. Granulocyte colony-stimulating factor (G-CSF; ie, filgrastim 5 µg/kg SC) was used to increase neutrophil counts and allow treatment continuation if there was severe neutropenia. A single dose was used for grade II neutropenia, and two doses with a 24 h interval were used for grade III or IV neutropenia. Refractory cases (neutrophil count <1.5 × 109/l after 72 h) were treated with two doses (within a 24 h interval) using twice the usual dose (10 µg/kg SC).
Results
Thirty-five cats with cytologically diagnosed high-grade lymphoma were identified and 21 met the inclusion criteria. Cats were excluded from the study if they were diagnosed with gastrointestinal (n = 2), spinal (n = 2), hepatic (n = 1) or nasal (n = 1) lymphoma, or in the so-called ‘leukemic phase’ (n = 5) or the owner did not consent to treatment (n = 3). The individual features of the 21 cats included are provided in the table in the supplementary material. There were no pure-breed cats in this study. Age ranged from 7 to 42 months (mean ± SD 23.5 ± 12 months) and weight from 1.8 kg to 5.9 kg (mean ± SD 3.7 ± 1.1 kg). Fifteen cats were male (seven castrated) and six were female (three spayed). Seven cats (33.3%) had previously received vincristine and prednisolone (unknown dosages), and the remaining cats (67.7%) had no prior treatment. Of 21 cats, 19 were diagnosed with persistent FeLV viremia by positive results on the SNAP Feline Triple Test (90.5%). The two cats that tested FeLV negative were also negative on PCR for provirus (whole blood). The FIV test was negative for all 21 cats. Lymphoma was anatomically classified as mediastinal (n = 14; all in stage I), and multicentric (n = 7; n = 4, n = 2 and n = 1 in stages III, IV and V, respectively). Spleen and liver involvement were based only on ultrasonographic features and may have been over- or underestimated. 30 Cytologic classification beyond high grade was performed in 12 cases: six were centroblastic, three were immunoblastic and three were lymphoblastic.
At presentation, a single cat was anemic (macrocytic normochromic). Packed cell volume (PCV) ranged from 19.8% to 48.1% (mean ± SD 38.0 ± 7.6%; median 40%; RI: 29–48%). Platelet count ranged from 99 to 550 × 109 thrombocytes/l (mean ± SD 193.0 ± 148.9 × 109/l; median 150 × 109/l; RI: 200–600 × 109/l) and thrombocytopenia was found in 13 individuals. Neutrophil counts ranged from 1.3 to 17.7 × 109/l (mean ± SD 8.0 ± 4.6 × 109/l; median 7.5 ×109/l; RI: 2.5–12.5 × 109/l). Neutropenia was diagnosed in three cats (2.0, 1.3 and 2.1 × 109/l, respectively). Lymphocyte counts ranged from 0.4 to 10.0 × 109/l (mean ± SD 3.2 ± 3.0 × 109/l; median 1.9 × 109/l; RI: 1.5–7.0 × 109/l). A single cat was azotemic (urea 113.8 mg/dl [RI: 40–60 mg/dl]; creatinine 2.44 mg/dl [RI: 0.8–1.8 mg/dl]). Urinalysis was not performed in this cat, which presented with mediastinal lymphoma and had a poor response to therapy (PR and OS of 52 days). Two cats had increased serum levels of ALT (213 and 171 U/l, respectively; RI: 6–83 U/l) and AST (169.4 and 120 U/l, respectively; RI: 23–43 U/l). One cat had increased activity of ALP (278.5 U/l; RI: 25–93 U/l) and one cat had low albumin levels (1.4 g/dl; RI: 2.3–3.1 g/dl). All of these abnormalities were mild and did not compromise treatment.
Median follow-up time was 162 days for all cats (range 17–739 days), 136.5 days for censored cats (lymphoma-unrelated death; range 17–263 days) and 450 days for those still alive or lost to follow-up (range 162–739 days). Only one cat was lost to follow-up (after 162 days). CR was reported in 81% of patients included in this study (n = 17/21), while three had a PR and one had NR. There were 14 reported deaths, 10 related to lymphoma recurrence and progression (five of those were euthanized). Two cats died owing to FeLV-related anemia or bone marrow impairment (erythroid and megakaryocytic aplasia); a single cat died from undefined causes; and one cat died from sepsis after the second dose of chemotherapy; therefore, these cats were censored. Seven cats finished the induction protocol in a median of 20 weeks (range 20–31 weeks; mean ± SD 23.1 ± 4.5 days) and all seven received the maintenance protocol. To date, only one has finished the maintenance protocol, but six were still alive at the end of the study (one died of unrelated causes).
The MST for the 21 cats was 214 days (lymphoma-related survival), but if the two FeLV-related (not lymphoma) deaths were not censored, the median was 171 days. The MST (lymphoma-related survival) was 214 days for mediastinal lymphoma (n = 14), but it was not reached for multicentric lymphoma (n = 7; P = 0.9) (Figure 1a). Three cats with multicentric lymphoma died of the disease at 50, 70 and 124 days, respectively, while three were still alive at 162, 335 and 739 days, respectively; the remaining cat died of unrelated causes (bone marrow aplasia, probably FeLV related) at 100 days. If FeLV-related deaths were not censored, the MST was 172 days for mediastinal and 169 days for multicentric lymphoma. FeLV-positive cats had an MST of 171 days (159.5 days if FeLV-related deaths were censored), and both FeLV-negative cats were still alive and well at the end of the study. The DFI for the 21 cats was 165 days and the same result was obtained for mediastinal lymphoma or considering only the FeLV-positive cats, while the median for multicentric lymphoma was not reached (P = 0.8) (Figure 1b). On univariate Cox regression analysis there was no difference in DFI or MST between anatomic forms of lymphoma, stage, sex, age and weight. Rescue treatment was attempted in five cases (one with only a PR after 3 weeks of LOPH) using doxorubicin, cytosine arabinoside, melphalan and dexamethasone. Only one cat had complete remission with an additional DFI of 62 days (mediastinal lymphoma).

Kaplan–Meier curves. (a) Survival (%) of cats with mediastinal (n = 14; median 214 days) or multicentric (n = 7; median not reached) lymphoma. (b) Disease-free-interval (%) of cats with mediastinal (n = 14; median 165 days) or multicentric (n = 7; median not reached) lymphoma. Cox–Mantel log-rank did not differ (P >0.05)
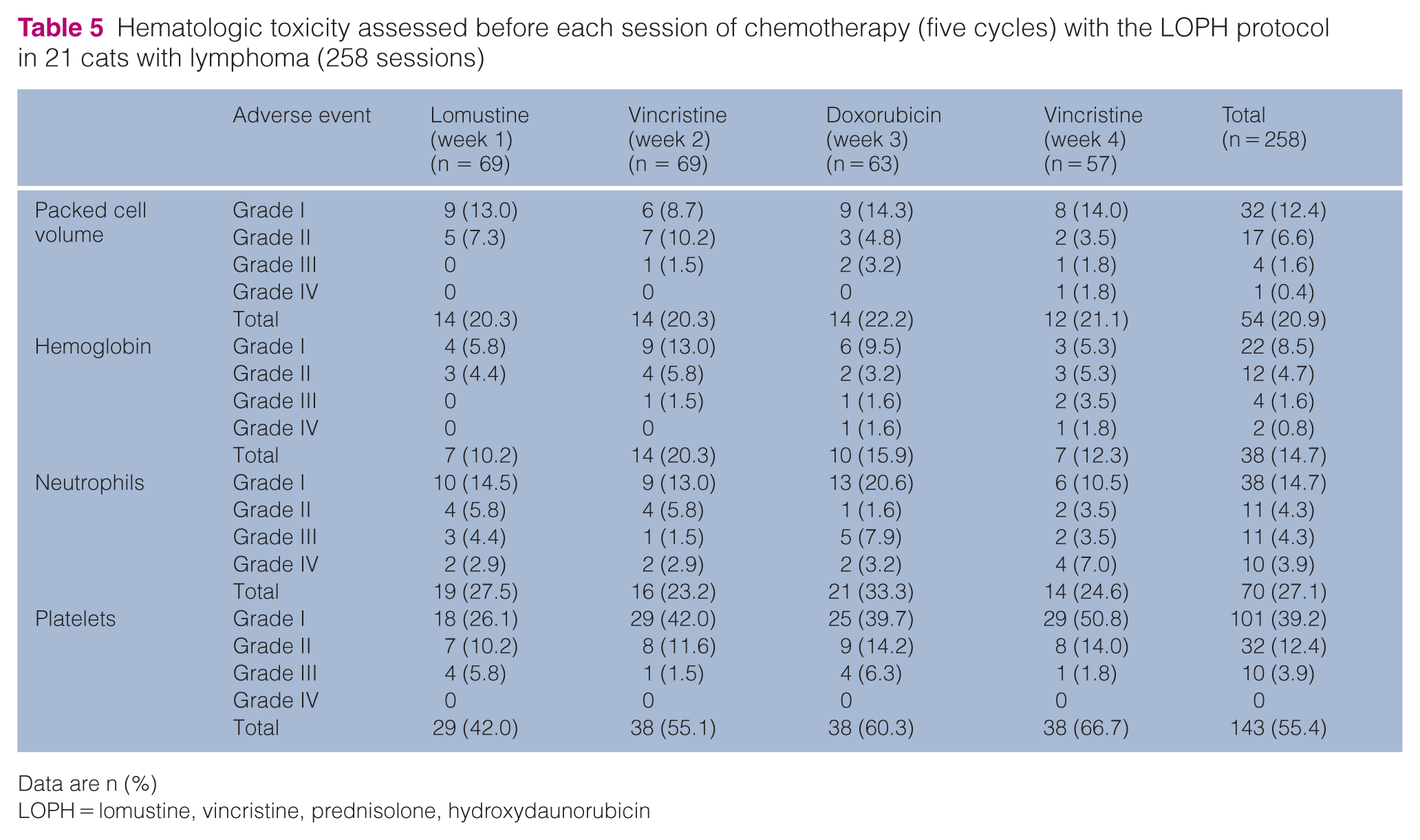
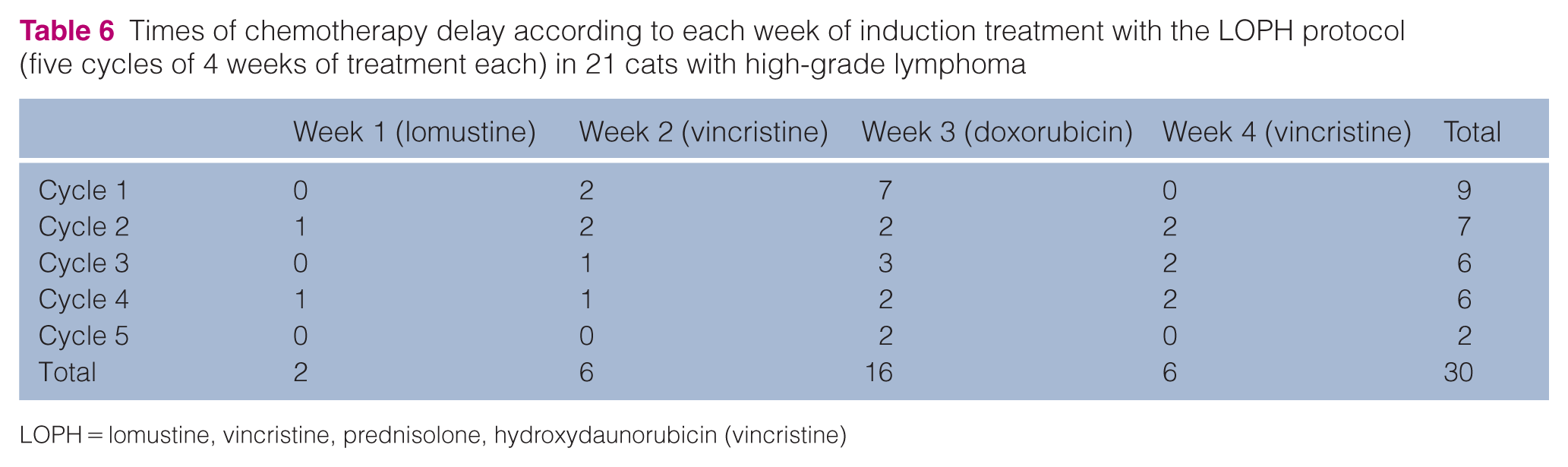
Grade I anorexia and vomiting occurred in 19% of cats (n = 4/21). Hematologic toxicity was found in 100% of the cats at some point during treatment according to the chemotherapy cycle (cycle 1–5), and according to the values obtained immediately before each session (or week) of chemotherapy within one cycle (Tables 4 and 5). During this study, a total of 260 doses of chemotherapy were administered: 84 in the first cycle (21 cats); 64 in the second (16 cats); 51 in the third (15 cats); and 32 in the fourth (10 cats). Chemotherapy was postponed for 2–14 days (median 7 days) on 30 occasions (Table 6). Most delays (n = 16/23) occurred on the third week of chemotherapy and were attributed to the lomustine 14-day nadir (combined with the vincristine 7-day nadir). There was a single record of sepsis and death related to chemotherapy (grade V toxicity). Neutropenia, thrombocytopenia and anemia occurred in n = 16/21, n = 21/21 and n = 15/21 cats, respectively. Anemia, according to PCV values, occurred on 62 occasions (out of 300 complete blood counts; 20.7%) and they were classified as normocytic normochromic (79.0%; n = 49/62), normocytic hypochromic (12.9%; n = 8/62) and macrocytic normochromic (8.1%; n = 5/62). Grade I adverse events were more common and mild decreases in PCV, hemoglobin, neutrophil and platelet count were observed in 11.7% (n = 35/300), 8.0% (n = 24/300), 15.0% (n = 45/300) and 27.0% (n = 81/300) of the CBCs performed.
Hematologic toxicity within each chemotherapy cycle with the LOPH protocol in 21 cats with lymphoma (293 complete blood counts)

Data are n (%)
LOPH = lomustine, vincristine, prednisolone, hydroxydaunorubicin
Hematologic toxicity assessed before each session of chemotherapy (five cycles) with the LOPH protocol in 21 cats with lymphoma (258 sessions)
Data are n ()
LOPH = lomustine, vincristine, prednisolone, hydroxydaunorubicin
Times of chemotherapy delay according to each week of induction treatment with the LOPH protocol (five cycles of 4 weeks of treatment each) in 21 cats with high-grade lymphoma
LOPH = lomustine, vincristine, prednisolone, hydroxydaunorubicin (vincristine)
A total of 21 doses of G-CSF (ie, filgrastim) was used to increase neutrophil counts and allow treatment continuation. Of these, 42.9% (n = 9/21) were performed in the third week of chemotherapy, 38% (n = 8/21) in the fourth and 14.3% (n = 3/21) in the second week. According to the chemotherapy cycle, 9.5%, 14.8%, 14.8%, 33.3% and 28.5% of G-CSF administrations occurred in the first, second, third, fourth and fifth cycle, respectively.
The individual characteristics of the patients in this study can be found in the table in the supplementary material.
Discussion
A large number of protocols are currently available for the treatment of feline intermediate-to-high-grade lymphoma, and the outcome data of different studies are presented in Table 7. Lymphomas are highly chemosensitive neoplasms, irrespective of FeLV status.2,23,25 Nevertheless, FeLV antigenemia is considered a poor prognostic factor for feline lymphoma with an MST of 37–126.3 days.21,25 LOPH was designed considering the poor outcome of cats with lymphoma attending a referral practice in a region endemic for FeLV, where prevalence ranges from 11.7% to 47.5%.6–8 Persistent viremia was detected in 90% of cats included in this study and only two cats were negative for FeLV based on the SNAP Feline Triple Test (also negative on whole blood PCR). These patients were rescued from the streets and they were young at the time of diagnosis of high-grade lymphoma, which makes FeLV lymhpomagenesis likely but unexplained.
Data from different chemotherapeutic protocols for the treatment of intermediate-to-high-grade lymphoma in cats

Progression-free interval and not disease-free interval (DFI)
COP = cyclophosphamide, vincristine (Oncovin), prednisolone; CHOP = includes doxorubicin (hydroxydaunorubicin); LOPH = lomustine, vincristine, prednisolone, hydroxydaunorubicin; FeLV = feline leukemia virus; NA = not available
The results of this study are more likely to be comparable with research conducted in the 1980s, before the widespread use of FeLV vaccines, when 60–70% of cats with lymphoma were persistently viremic.13,16,21,25 Vail et al 25 performed a retrospective study of 145 cats with lymphoma. FeLV association was reported in 25% of cases (73% for mediastinal lymphoma and 31% for multicentric lymphoma), and most cats were treated with the COP protocol, although some received additional doxorubicin, idarubicin or mitoxantrone. CR was obtained in 60% of cats, despite FeLV status and anatomic site; nevertheless, multivariate analysis revealed that FeLV status significantly influenced the DFI (27 days for FeLV-positive cats vs 146 days for FeLV-negative cats; P = 0.026) and OS (37 days for FeLV-positive cats vs 170 days for FeLV-negative cats; P = 0.021). Similarly, a retrospective study by Mooney et al 21 reported a variant of COP with L-asparaginase: 62% achieved CR with an MST of 4.21 months for FeLV-positive and 9.08 months for FeLV-negative cats (P <0.001). MST did not differ according to FeLV antigenemia in the studies by Fabrizio et al 14 and Collete et al; 2 however, both studies were underpowered to evaluate the impact of FeLV, with only 9% (n = 5/55 and n = 9/102, respectively) being persistently viremic cats.
The COP protocol is the most popular protocol for feline intermediate-to-high-grade lymphoma. It was first described by Cotter 13 with a CR of 79%. The MST was not reached, but the DFI was 150 days. Although it was not possible to statistically compare these results with those obtained with LOPH, the remission rates were similar, but an increased DFI was obtained in the present study (especially for multicentric lymphoma), even though the FeLV infection rate was 90%. It is possible that older studies may have included low-grade lymphomas, particularly those located in the gastrointestinal tract, resulting in better disease control. 2 The VCM protocol is an intensified protocol, similar to CHOP but with methotrexate instead of doxorubicin and the inclusion of L-asparaginase. 16 It resulted in a CR rate of 52–62%,16,21 and an MST of only 60 days. 16 Further studies, with reduced FeLV infection rate, showed a different scenario. In the Netherlands, a study of 61 cats with lymphoma had only 7.4% (4/54) with FeLV infection and COP treatment provided a CR rate of 75.4% with a median DFI and MST of 251 and 266 days, respectively. 24 After a decade, the same group published a second study, 27 including 26 cats (0% with FeLV infection), with intraperitoneal delivery of vincristine and cyclophosphamide resulting in a CR rate of 76.9% and an MST of 388 days, although 38.5% (n = 10/26) of patients had nasal lymphoma, which is known to carry a better prognosis. 2
Hydroxydaunorubicin (doxorubicin) is one of the main drugs used to treat lymphoma in humans and dogs; however, its use is extremely controversial in cats. As a single first-line agent (25 mg/m2 or 1 mg/kg every 21 days), it resulted in a CR in only 26.3% of cats (n = 5/19), with an MST of 92 days. 17 As a rescue treatment, CR was noted in only 9% of cats (n = 2/23) with DFIs of 1.5 and 47 months, respectively. 26 As a maintenance agent, doxorubicin significantly increased the MST of cats with lymphoma (approximately 18% were infected with FeLV), after the COP induction phase; nevertheless, the study was underpowered, with only seven cats treated with this protocol, with an MST of 281 days (vs 83 days with conventional COP; P = 0.006). 19
Multiagent protocols including doxorubicin (CHOP) provided CR in 38–74%.2,14,15,18,20,23 The Wisconsin–Madison protocol was described in 1996, but the first study in cats was published by Milner et al, 20 with 38 cats (47% with CR and an MST of 210 days). Originally, this protocol included a maintenance therapy (with vincristine, chlorambucil and methotrexate), 20 but it underwent several modifications. The 24-week VELCAP-C protocol (combining vincristine, cyclophosphamide, doxorubicin, prednisolone and L-asparaginase) resulted in CR in 43% of cases, but the MST was only 62 days. Only 6/61 cats (10%) completed the protocol, achieving an MST of 1189 days, which may support the efficacy of doxorubicin in prolonging the duration of first remission. 15 Simon et al 23 evaluated a prolonged combination protocol, including L-asparaginase, vincristine, cyclophosphamide, doxorubicin, methotrexate and prednisolone, for treating feline intermediate-to-high-grade lymphoma (only one multicentric and four mediastinal lymphomas). The MST was 232 days, there were no FeLV-infected cats and 30% had FIV infection (n = 7/23). With a similar but shorter CHOP-based, maintenance-free 12-week protocol, Limmer et al 18 had 23% of cats with progressive FeLV infection (n = 6/26) and an MST of only 58 and 62 days, respectively, for mediastinal (n = 3; two FeLV infected) or multicentric (n = 4; two FeLV infected) lymphoma. The modified CHOP protocol of Collete et al, 2 lasted 25 weeks, and allowed CR in 38% of cats with intermediate-to-high-grade lymphoma, and an MST of 97 days. All cats that did not receive doxorubicin had progression of the disease and 21/87 cats (24.1%) that received doxorubicin completed their CHOP protocol. This study excluded low-grade lymphomas but included 63% of gastrointestinal lymphomas (intermediate-to-high-grade), which were less likely to achieve CR than other anatomic forms. 2 Fabrizio et al 14 did not find a statistical difference between the COP and Wisconsin–Madison protocols, either considering the response rate (61.5% vs 66.7%, respectively; P = 0.26) or MST (484 days vs 211 days, respectively; P = 0.89).
There is no current consensus surrounding treatment of feline lymphoma, which results in a great variability of therapeutic approaches between different institutions. 2 Despite the large number of studies, there is a paucity of information for populations with a high prevalence of FeLV.18,23 This is the first study to show reasonable survival results, with a population highly exposed to FeLV and 90% with persistent viremia. Both FeLV-negative cats (one with multicentric and the other with mediastinal lymphoma) were still alive at the end of the study, with no reported relapse, but the number of cases was too small for statistical inference. Although mediastinal lymphoma is commonly associated with a worse prognosis,2,13,16 a high MST was obtained in this study. In the current study, the DFI and MST were not influenced by clinical stage; nevertheless, it was not accurately determined since liver, spleen and bone marrow aspirates were not performed and one-third of patients were staged after receiving vincristine and prednisolone. In general, LOPH results were superior to most previous COP protocols (considering only FeLV-positive cats in the studies of Mooney et al 21 and Vail et al 25 ) or CHOP reports for feline intermediate-to-high-grade lymphoma (also for feline intermediate-to-high-grade gastrointestinal lymphoma), except for the studies conducted by Teske et al,24,27 with COP; Milner et al 20 , with CHOP; and Fabrizio et al 14 , using COP or CHOP, but those studies had up to 10% FeLV antigenaemia.
LOPH was created owing to the need for a more intensive protocol for FeLV-infected cats, based on the evidence that lomustine is more likely to be of value in the induction phase than for rescue,22,28 and that doxorubicin may contribute to a prolonged DFI in cats that achieved CR.2,15,19 Lomustine is an alkylating agent, widely used in dogs as a first or rescue agent for mast cell tumors and cutaneous and/or T-cell lymphomas.33–35 Although it has been used in cats for several neoplasias, including T-cell cutaneous lymphoma,36,37 there is still a lack of information surrounding its use. Cats with naive intermediate-to-high-grade gastrointestinal lymphoma were treated with lomustine (30–60 mg/m2 every 4–6 weeks) resulting in CR in 21.9% of cats (n = 7/32) and partial remission in 28.1% (n = 9/32), with an MST of 108 days. 22 Lomustine does not appear to be as useful in rescue protocols, 28 which partially justified its inclusion in an induction phase. In a study performed by Dutelle et al, 28 a DFI of only 39 days was obtained in cats with recurrent lymphoma. Traditionally recommended at 50–60 mg/m2 every 4–6 weeks, or 10 mg/cat, a lower dose was applied in the present study. We chose the lower range used in larger studies22,37 so it could be included in an intensive induction protocol (30–40 mg/m2 with a mean dose of 35.7 ± 8.9 mg/m2 and median of 36 mg/m2). The dose was also defined according to the oncologist’s experience and available reformulated capsule size: 6 mg (<3 kg), 9 mg (3–4.5 kg), 12 mg (4.5–6 kg) and 15 mg (>6 kg).
Voorhorst et al 38 showed that vincristine had 100% bioavailability when administered intraperitoneally to cats (systemic exposure similar to the IV administration), with no specific adverse events related to its route of administration. Intraperitoneal delivery was considered easier, faster and safer, and should be considered as an acceptable alternative to intravenous (IV) administration of vincristine in cats, although, as for the IV route, it is highly recommended to use a closed system drug transfer device, in order to prevent environmental contamination and reduce the exposure of veterinarians, students and nurses. 27
The evidence for the inclusion of doxorubicin was based on its efficacy as a maintenance drug for prolonging DFIs in cats with lymphoma previously treated with an induction phase of COP, although the number of cases was small (n = 7) and the study underpowered. 19 Nevertheless, prolonged DFIs were also seen in other studies that included doxorubicin in cats that achieved CR.2,15 This might be partially justified by the anthracycline resistance mechanism, mainly related to the expression of efflux pumps on tumor cells, such as P-glycoprotein and the breast cancer resistance protein (BCRP), transcribed by the ABCB1 and ABCG2 genes, respectively. Ramirez et al 39 identified four specific modifications, in highly conserved regions, of feline BCRP, with an inefficient efflux of its substrates, partially explained by the abnormal intracellular expression of such protein, detected by immunofluorescence. It is possible that BCRP substrates, such as mitoxantrone, doxorubicin, tyrosine kinase inhibitors and fluoroquinolones, have their efficacy, but also their toxicity increased in cats, owing to the ABCG2 polymorphism and hypofunctional BCRP, both in normal or tumor cells, also delaying the development of tumor resistance. In spite of this, doxorubicin was well tolerated by the cats in this study. Neutropenia usually occurs after 7–10 days and there is a risk of nephrotoxicity related to the release of reactive oxygen species. 40 Mitoxantrone (5 mg/m2 IV) might be used to substitute doxorubicin in the LOPH protocol in cats with stage II–IV chronic kidney disease; nevertheless, this was deemed unnecessary in this study. 40
Treatment is mainly based on two phases: induction of remission and rescue (re-induction). It is also possible to propose intensification and maintenance protocols; 1 however, there is a current tendency to implement aggressive and multi-agent protocols during the induction phase of remission, suppressing those phases.2,15 LOPH is an intensive protocol that might exclude the maintenance phase. Nevertheless, maintenance was encouraged in the present study considering the poor prognosis and usually fatal outcome of feline high-grade lymphoma associated with FeLV.12,25 The maintenance protocol is a modified version of that proposed by Vonderhaar and Morrison, 41 which uses vincristine and chlorambucil. This was adapted for cats, as a simple measure, with the intention of improving the duration of the first remission. Vincristine was chosen despite the high number of doses already administered in the induction phase, as it is commonly included in maintenance therapy in other protocols and significant cumulative toxicity has not been reported.13,14,20,21,23,24,27 Only a small number of patients received the maintenance protocol, and its efficacy remains unclear, although it may have affected the DFI and OS.
Chemotherapy is usually well tolerated by cats, but studies of adverse events in populations with high FeLV antigenemia are lacking.18,23 Limmer et al 18 evaluated the toxicosis of a 12-week CHOP-based protocol in 26 cats (23% with progressive FeLV infection). Neutropenia was detected in 46% of patients (44% with grade I neutropenia, 41% grade II, 11% grade III and 4% grade IV). In the present LOPH protocol, 76.2% of patients presented with neutropenia (n = 16/21). Thrombocytopenia (mostly grade I or II) occurred in 49% of the CBCs performed for the cats in this study. Nevertheless, it may represent a common artifact in cats (pseudothrombocytopenia), occurring in up to 50% of feline samples, as cats are highly susceptible to stress due to physical restraint and venipuncture, resulting in increased microaggregates. 42 Lomustine’s safety was demonstrated in cats in a phase 1 study by Rassnick et al. 43 It has been shown to have a high risk of hepatic toxicity in dogs; this has not been reported in cats, which are susceptible to an increased risk of neutropenia (7–28 days), thromobocytopenia (14–21 days) and sepsis.22,37 Neutropenia is the main dose-limiting toxicity. Grade I–IV neutropenia was reported in 52% of cats with lower values (nadir) mostly seen after 3 weeks. 22 Nevertheless, in the present study, most neutropenic episodes resulting in chemotherapy delay happened after 14 days, which overlapped with vincristine’s toxicity. Vincristine is a cell cycle-specific drug, which interferes with microtubule formation and mitosis, with the lowest neutrophil counts occurring in 7–9 days. 40 Luckily, most of the time, the low neutrophil count did not compromise the treatment after 14 days, as doxorubicin, in the third week of each cycle, was apparently the most safe and well tolerated drug in the LOPH protocol.
The level of evidence for using antibiotics or G-CSF is very low in veterinary patients and recommendations are made based on human guidelines. 44 There is no current specific recommendation for cats. In dogs, prophylactic antibiotics are indicated for neutrophil counts lower than 0.75 × 109/l, but they are not recommended for a neutrophil count higher than 1 × 109/l. As performed in this study, prophylactic antibiotic use is also considered in dogs presenting with a neutrophil count of 0.75–1 × 109/l if one or more of the following risk factors are presented: hematologic malignancies, concomitant disease, animals with reduced weight or body size (as in cats) and mutations in the ABCB1 gene (P-glycoprotein).44,45 Both lymphoma (as a hematopoietic malignancy) and persistent FeLV viremia (as a concomitant disease) may significantly compromise the patient’s immunity.1,2,4 Antibiotic choice should be based on the sensitivity of the resident pathogenic bacteria.44,46 Nevertheless, specific veterinary studies assessing resistance in chemotherapy patients are not available. The antibiotic must have a broad-spectrum efficacy, but anaerobe sparing and additional Gram-positive cover is not recommended. 44 Ideally, sulfonamides might be used, but they are not well tolerated by cats, and non-halogenated fluoroquinolones (such as norfloxacin or marbofloxacin) appear to represent a safer option, even though we recognize the risk of using tier 2 antibiotics in a prophylactic setting, which might lead to bacterial resistance.44,47 Treatment duration should be maintained if infection is confirmed or until the neutrophil count is higher than 1 × 109/l, 42 as performed in the present study.
There is very limited evidence on the use of G-CSF (ie, filgrastim) in cats and its use remains controversial in veterinary patients. 44 In humans, it reduces the duration of grade III and IV neutropenia, and reduces hospitalizations and the need for IV antibiotics. 47 In dogs, it accelerated the recovery and decreased the severity of neutropenias induced by cyclophosphamide. 48 There is a risk of cross-species antibody production leading not only to the neutralization of human G-CSF, but also endogenous feline G-CSF, which could lead to severe neutropenia, as reported in dogs. 49 Their use is not commonly advised in veterinary patients, except in cases of very severe neutropenia or in cases of a known chemotherapy overdose.49,50 Nevertheless, it is currently used in humans submitted to intensive chemotherapeutic protocols and it might be useful in cats, particularly those with persistent FeLV viremia, if necessary, allowing continuation of the chemotherapy protocol, as our experience demonstrated.
As an intensive protocol, LOPH might be harder to conduct in cats with lymphoma, if compared with the COP protocol, and complications might be more severe, especially if performed by feline practitioners without experience in clinical oncology, and also for oncologists with less experience of cats. The diagnosis of feline lymphoma may be acquired through cytologic analysis and the modified Kiel classification system was used for this purpose in the present study. There is a small chance of misdiagnosis based on cytology alone, especially for multicentric lymphoma, although the clinical responses noted make this less likely. Furthermore, it is well known that a more precise diagnosis might be obtained through histopathology and immunohistochemistry, using the classification system defined as REAL (Revised European-American Lymphoma Classification) and incorporated by the World Health Organization. 51 This was shown to have an impact on prognosis, 52 and it is reasonable to assume that FeLV-associated lymphoma might have a different profile compared with lymphoma not associated with FeLV. There are still no specific therapeutic recommendations for each type of lymphoma and a biopsy for such classification was not possible for this study, since many of the cats were presented in poor clinical condition and most of the owners had limited resources. Controlled multicentric studies are needed to investigate the clinical outcomes obtained with this treatment or other protocols, and to correlate them with FeLV status and the REAL.
Conclusions
The LOPH protocol was well tolerated for cats with lymphoma and persistent FeLV viremia and resulted in a better MST, if compared with similar studies with different protocols. There was no difference in the DFI or MST between anatomic forms of lymphoma and clinical stage. Novel studies and controlled trials are necessary in order to evaluate the efficacy of different protocols according to the lymphoma subtype, anatomic form and FeLV status.
Supplemental Material
Supplementary Table
Individual features of 21 cats with high-grade nodal lymphoma treated with the LOPH protocol.
Footnotes
Supplementary material
The following file is available online:
Table: Individual features of 21 cats with high-grade nodal lymphoma treated with the LOPH protocol.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals (owned or unowned) and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
