Abstract
Objectives
The aim of this study was to evaluate the change in packed cell volume (PCV) and total protein following intramuscular preanesthetic sedation with one of three drug combinations in cats.
Methods
Thirty client-owned cats were enrolled in this prospective, randomized, blinded, clinical study. A venous blood sample was obtained prior to administration of any sedation and PCV, total protein, electrolytes (Na+, K+, Cl–, iCa2+), glucose and lactate were measured. Cats were randomly assigned to receive one of three intramuscular sedation protocols (n = 10 cats/protocol): methadone 0.2 mg/kg + acepromazine 0.02 mg/kg (MA), methadone 0.2 mg/kg + dexmedetomidine 5 µg/kg (MD) or methadone 0.2 mg/kg + midazolam 0.2 mg/kg + alfaxalone 2 mg/kg (MMA). Twenty-five minutes later, cats were assessed for level of sedation followed by another venous blood sampling to evaluate the same variables as above.
Results
There were no significant differences in demographics (age, weight, sex) between groups. Level of sedation was significantly higher in MMA cats. Within groups, after premedication, PCV and hemoglobin significantly decreased in all groups, total protein significantly decreased in the MA and MMA groups and glucose significantly increased in the MD group. For electrolytes, statistical changes were not clinically relevant; Cl– mean difference was significantly different between MA and MD; in the MD group Na+ and Cl– significantly decreased and in the MMA group Cl– significantly increased.
Conclusions and relevance
All three sedation protocols caused significant decreases in PCV and hemoglobin in healthy cats.
Introduction
Cats are commonly administered premedication as part of a balanced anesthetic protocol to facilitate intra-venous catheter placement, provide pre-emptive analgesia, anxiolysis and decrease anesthetic induction drug and inhalant dose requirements. It has been previously documented in cats that certain anesthetic drugs (acepromazine and dexmedetomidine) and different combinations (midazolam + butorphanol, midazolam + butorphanol + ketamine, midazolam + butorphanol + dexmedetomidine, ketamine + dexmedetomidine) can result in a decrease in packed cell volume (PCV),1 –3 which can have a significant effect on diagnostic test interpretation and clinical treatment.
Acepromazine is a phenothiazine tranquilizer that causes sedation by blocking pre- and post-synaptic dopaminergic receptors, 4 and it is commonly used for sedation and anxiolysis in cats.5,6 Dexmedetomidine is an alpha (α)2 agonist used for sedation and analgesia, through spinal and supraspinal effects, in cats. 7
Methadone is a racemic mixture that provides analgesia as a mu (µ)-opioid receptor agonist (L-isomer) and as an N-methyl-D-aspartate antagonist, as well as inhibiting norepinephrine (noradrenaline) re-uptake (D-isomer). 8 Methadone has been found to provide adequate analgesia when administered as a premedication in combination with dexmedetomidine or acepromazine in cats undergoing ovariohysterectomy or castration.9,10 The effect of methadone, or any pure µ-opioid agonist, in combination with common sedative agents on PCV in cats has not been evaluated.
Midazolam is a benzodiazepine that causes central sedation and muscle relaxation via potentiation of the gamma-aminobutyric acid (GABA) receptor. Its water solubility allows its intramuscular (IM) administration. Midazolam is a poor sedative when administered alone in cats, paradoxically causing excitement sometimes and difficulty in handling. 11 However, when administered as a co-sedative with α2 agonists or induction agents (ketamine), midazolam can be effective in providing sedation.1,12
Alfaxalone is a synthetic, injectable agent that can be used to induce anesthesia. Alfaxalone can also be administered IM in veterinary species to provide heavy sedation. Clinically, it provides better quality sedation when coadministered with other agents,13,14 than when administered alone. 15
The use of combinations of sedatives is frequent in cats to obtain a higher and more reliable degree of sedation. The aim of this study was to evaluate the effects of three common sedative combinations used at this referral center – methadone with acepromazine, methadone with dexmedetomidine, and methadone with midazolam and alfaxalone – and their effects on PCV, hemoglobin (Hb), total protein (TP), electrolytes, glucose and lactate. Our hypothesis was that all sedative combinations would result in decreases in PCV, Hb and TP.
Materials and methods
The study was carried out in accordance with guidelines of the Canadian Council on Animal Care and was approved by the Institutional Animal Care Committee at the University of Guelph for ethics and scientific merit (number 4393).
Animals
Cats that presented for any surgical or diagnostic procedure requiring premedication or sedation over an 11-month period were considered for the study. Written owner consent was obtained prior to the study. All cats received a full physical examination by a veterinarian prior to any drug administration. Exclusion criteria included aggressive/fractious cats, pre-existing cardiac or respiratory disease, and pre-existing hematologic abnormalities.
Study design
This was a randomized, prospective, blinded clinical study. Cats were assigned to receive one of three sedation protocols.
Sedation and hematology
Cats were fasted for 8 h prior to sedation but had free access to water until IM sedatives were administered. Cats were allowed to acclimatize to a kennel in a quiet environment for 1 h prior to handling for blood sampling. A venous blood sample (1 ml) was obtained from the left or right medial saphenous vein prior to administration of any drugs. The PCV was measured manually following centrifugation (MX12; LW Scientific) at room temperature at a relative centrifugal force of 12,000 rpm (13,533 g) for 3 mins. TP was measured from the plasma fraction using a refractometer (Refractometer 726000; Eickemeyer). A bench analyzer (ABL90 Series; Radiometer Medical ApS) was used to measure Hb, electrolytes (Na+, K+, Cl–, iCa2+), glucose and lactate. Cats were randomly assigned to receive one of three sedation protocols: (1) methadone (Comfortan; Dechra Veterinary Products) 0.2 mg/kg + acepromazine (Atravet; Boehringer Ingelheim) 0.02 mg/kg (MA); (2) methadone 0.2 mg/kg + dexmedetomidine (Dexdomitor; Zoetis) 5 µg/kg (MD); or (3) methadone 0.2 mg/kg + midazolam (Midazolam Injection; Sandoz Canada) 0.2 mg/kg + alfaxalone (Alfaxan; Jurox) 2 mg/kg (MMA). All sedative combinations were administered as a single injection in the epaxial muscles. Cats were left undisturbed in a quiet kennel for 25 mins after sedation was administered. Cats were assessed for degree of sedation using a score 15 from 0 to 16 by a registered veterinary technician who was blinded to the treatment. Immediately after, a left or right cephalic catheter (BD Insyte; Becton Dickinson Infusion Therapy Systems) was placed and a second venous blood sample (1 ml) was obtained from the catheter and the same measurements as above were repeated.
Statistical analysis
Data were assessed for normality using the Anderson–Darling test, the D’Agostino-Pearson test and the Shapiro–Wilk test. Age, body weight and sedation score were compared with a one-way ANOVA, and multi-pairwise comparisons were carried out with a Tukey test. Sex was compared after transformation with a χ2 test. All remaining variables were analyzed with a two-way ANOVA for repeated measures, and multiple comparisons between groups corrected with a Tukey test and within groups with a Sidak test, to determine the effects of premedication treatment (MA, MD and MMA). Differences were considered significant at P <0.05. All statistical analysis were performed using GraphPad Prism (Prism 9 for macOS, version 9.1.2 for macOS; GraphPad Software).
The number of cats in the study was determined by assuming a difference of 20% in PCV (eg, 40% before sedation and 32% after sedation) with a SD of 5%, an α value of 0.05 and a power of 0.90, which required of a sample size of nine cats per group.
Results
Animals
A total of 30 cats presenting to Toronto Animal Health Partners were enrolled in the study after confirming eligibility. Breeds included domestic shorthair (n = 19); domestic longhair (n = 2); Bengal (n = 2); Persian (n = 1); Oriental Shorthair (n = 1); British Shorthair (n = 1); Ragdoll (n = 1); American Shorthair (n = 1); Maine Coon (n = 1); and Exotic Shorthair (n = 1). There was no significant difference in the demographics (age, weight and sex) of the three groups (Table 1).
Description of cats sedated with intramuscular methadone + acepromazine (MA), methadone + dexmedetomidine (MD) or methadone + midazolam + alfaxalone (MMA)

Data presented as mean ± SD
MC = male castrated; FS = female spayed; MI = male intact; FI = female intact
Sedation
The level of sedation was significantly higher in cats that received MMA (Table 2; Figure 1).
Mean difference (95% confidence interval of difference) sedation scores (range 0–16) in cats administered intramuscular methadone + acepromazine (MA), methadone + dexmedetomidine (MD), or methadone + midazolam + alfaxalone (MMA)

Indicates significant difference in the mean difference within treatments (P <0.05)

Sedation score in cats sedated intramuscularly with methadone + acepromazine (MA), methadone + dexmedetomidine (MD) or methadone + midazolam + alfaxalone (MMA). Ten cats per group
Hematology
Hematology data are presented in Figures 2–4. Within groups, PCV and Hb significantly decreased in all three groups, and TP significantly decreased in the MA and MMA groups (Table 3). In the MD group, glucose was significantly increased, and Na+ and Cl– significantly decreased after premedication (Table 4). In the MMA group Cl– was significantly increased after premedication (Table 4). The change in Cl– after premedication was significantly different between MA and MD.

Change in packed cell volume, hemoglobin and total protein in cats sedated intramuscularly with methadone + acepromazine (MA), methadone + dexmedetomidine (MD) or methadone + midazolam + alfaxalone (MMA). Ten cats per group
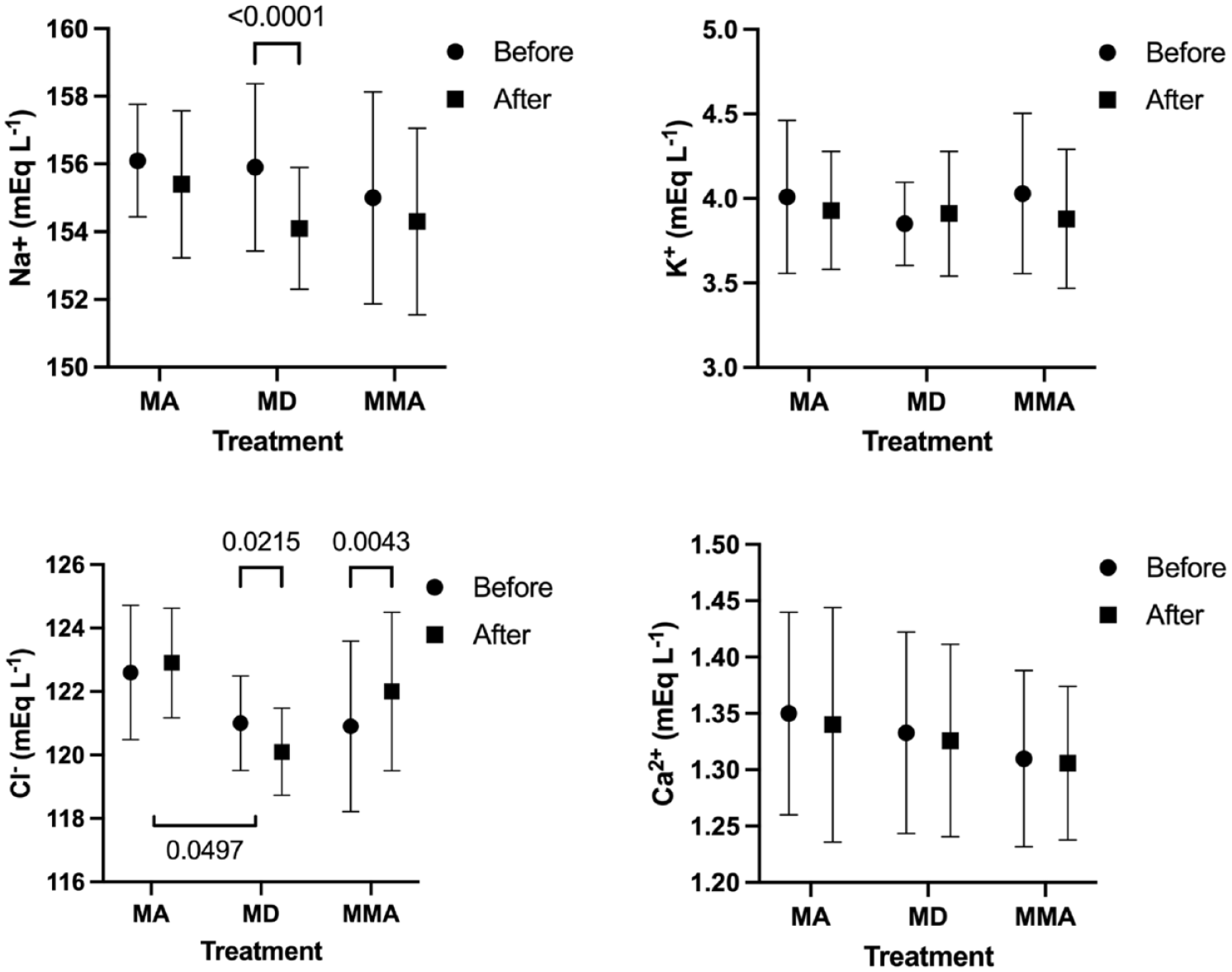
Change in sodium (Na+), potassium (K+), chloride (Cl–) and ionized calcium (iCa2+) in cats sedated intramuscularly with methadone + acepromazine (MA), methadone + dexmedetomidine (MD) or methadone + midazolam + alfaxalone (MMA). Ten cats per group

Change in glucose and lactate in cats sedated intramuscularly with methadone + acepromazine (MA), methadone + dexmedetomidine (MD) or methadone + midazolam + alfaxalone (MMA). Ten cats per group
Mean difference (95% confidence interval of difference) in packed cell volume (PCV), hemoglobin (Hb) and total protein (TP) in cats sedated intramuscularly with methadone + acepromazine (MA), methadone + dexmedetomidine (MD) or methadone + midazolam + alfaxalone (MMA)

Significant difference in the mean value before and after within treatments (P <0.05)
Mean difference (95% confidence interval of difference) in sodium (Na+), potassium (K+), chloride (Cl–), ionized calcium (iCa2+), glucose and lactate in cats sedated intramuscularly with methadone + acepromazine (MA), methadone + dexmedetomidine (MD), or methadone + midazolam + alfaxalone (MMA)

When superscript letters are present, values are significantly different (P <0.05) between treatments unless they have the same superscript
Significant difference in the mean value before and after within treatments (P <0.05)
Discussion
The results of this study have important clinical applications. Cats often require sedation for handling and diagnostic tests. It is important for clinicians to be aware of how sedation protocols may affect diagnostic test results and ongoing treatment. To our knowledge, this is the first study to evaluate the effect of a pure µ-agonist opioid (methadone) in combination with other sedative agents on PCV and TP.
The level of sedation was significantly higher in cats that received MMA vs MA or MD. Acepromazine and dexmedetomidine are common sedatives used in cats in clinical practice.10,16 Acepromazine is a phenothiazine derivative that has mild sedative effects in veterinary species. Acepromazine in combination with methadone resulted in mild sedation in the cats in this study, which is similar to findings of other studies when acepromazine is administered IM alone to cats 17 or in combination with methadone. 10
Dexmedetomidine is a sedative, analgesic and muscle relaxant that has been reported to result in dose-dependent sedation in cats. 18 In one study, dexmedetomidine (5 µg/kg) in combination with methadone (0.5 mg/kg) IM resulted in a higher level of sedation than in the present study; however, the dose of methadone in that study was higher. 16 The dose of methadone used in this study was at the lower end of the dose range and was chosen to mimic clinical conditions for this center. Even at high doses, IM methadone alone in cats reportedly results in no detectable sedation. 8
Alfaxalone is a synthetic, neuroactive steroid molecule that modulates GABAA and can be administered IM to produce sedation. When administered as the sole agent IM in cats, alfaxalone results in dose-dependent sedation; however, tremors and opisthotonus are common adverse effects. 15 To alleviate some adverse effects of IM alfaxalone, it is often combined with other agents. In combination with methadone, alfaxalone produced only mild sedation. 14 Alfaxalone IM in combination with methadone and midazolam has not been studied in cats; however, in dogs alfaxalone + methadone + midazolam IM resulted in poorer quality sedation than alfaxalone + midazolam alone. 19 In this study, MMA resulted in superior (highest level) sedation compared with the other two groups and prove useful as a sedative/premedication combination in cats.
In this study, all three sedation protocols resulted in a significant decrease in PCV and Hb. This has been previously reported in cats sedated with acepromazine, 3 dexmedetomidine 3 and combinations of ketamine + midazolam + buprenorphine, 2 midazolam + butorphanol,1,3 midazolam + butorphanol + ketamine, 1 midazolam + butorphanol + dexmedetomidine, 1 dexmedetomidine + ketamine + butorphanol 3 and ketamine + dexmedetomidine. 1 Contrary to those previous studies,2,3 this study also found a significant decrease in TP when cats were sedated with MA and MMA.
There are several mechanisms to explain a decrease in PCV and Hb in the perianesthetic period. These include the administration of fluids, a decrease in vascular resistance and the ensuing vasodilation, and pooling of red cells in different reservoirs due to alterations in hemodynamic function.
The main mechanisms hypothesized to result in the decrease in PCV and Hb as a result of the administration of sedative drugs include the combination of a decrease in sympathetic activity that results in vasodilation and then the sequestration of red blood cells in vascular reservoirs.1,3 Sedation in cats with acepromazine, dexmedetomidine, midazolam + butorphanol and dexmedetomidine + ketamine + butorphanol all resulted in increased splenic size; however, no association with decrease in PCV and size of the spleen was found, confirming that red blood cell sequestration likely occurs in other vascular beds or tissues.3,20
In humans it has been demonstrated that hypotension from vasodilation at anesthetic induction decreases capillary hydrostatic pressure and results in trans-capillary fluid absorption, thereby increasing plasma volume and causing hemodilution. 21 This phenomenon has also been demonstrated in pentobarbital-anesthetized dogs, in which a 15% increase in extracellular fluid occurred. 22 Similar findings are expected from sedative combinations that reduce vascular resistance and by increasing plasma volume would decrease PCV and Hb. 23 However, the effects of preanesthetic drugs on TP have been less thoroughly investigated, which typically decreases due to dilutional effects of fluid administration.20,24 In this study, TP decreased in the MA and MMA groups associated with the vasodilation caused by the drugs included in these sedative combinations. Conversely, TP did not change in the MD group, which may be the result of a higher systemic vascular resistance from the effects of dexmedetomidine, 25 contrary to the vasodilation of the other two groups.
In cats that received MD, a significant increase in glucose was identified. This is a known effect of α2-agonists because of postsynaptic inhibition of insulin release from the pancreas via α2A and α2C receptor agonism.26,27 While the changes in electrolytes were statistically significant, they were clinically irrelevant.
In this study, the effects on preanesthetic sedatives on PCV, Hb and TP were not assessed beyond 25 mins post-administration. Significant changes in this time period were identified in all cats and may be present at longer intervals or exacerbated by the effects of induction and maintenance anesthetic drugs; however, the objective of this study was to evaluate changes produced by sedation and the cats were sedated at this time frame.
This study had some limitations that are often associated with clinical trials. The sedative combinations chosen were not equipotent based on sedative effects; however, they are commonly used clinical doses and therefore the results are applicable in a practice setting. Most animals have a stress response when presented to a veterinary practice which can impact PCV; however, an attempt was made to minimize this effect through the study design by allowing cats to acclimatize to the environment and only enrolling cats that had a friendly temperament and were amenable to handling.
Conclusions
In this study, MMA resulted in better sedation than MA or MD; however, in friendly cats either protocol was sufficient for handling and catheter placement. All three sedation protocols resulted in a decrease in PCV and Hb and both MA and MMA resulted in a decrease in TP, which should be taken into consideration when interpreting these variables following sedation in cats.
Footnotes
Acknowledgements
The authors would like to thank surgery technician Magdalene Jargstorf and anesthesia technician Heather Webster for their assistance with this project.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals and procedures that differed from established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient. The study therefore had prior ethical approval from an established (or ad hoc) committee as stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
