Abstract
Objectives
Morphologic anomalies of the feline gallbladder (GB) have been previously reported in the literature. These morphologic variants are frequently encountered on routine abdominal ultrasound examination. The aim of this study was to provide an ultrasonographic classification system of these variants and document the overall incidence in the feline population.
Methods
A prospective, descriptive study was undertaken; cats that had an abdominal ultrasound examination that included at least one sagittal and transverse plane image of the GB were included. GB shape was evaluated and categorized based on a classification scheme of morphologic variants modified from the human literature. Septated (S), bilobed (B1, B2, B3), duplex (D) and complex (C) categories were described.
Results
Of 516 cats included in the study, 389 had normal GB morphology, while 127 had anomalous GB morphology. The overall incidence rate of anomalous GB morphology was 24.61%. When examined by morphologic type, the septated (S) morphology had an incidence of 9.69%. A bilobed (B) morphology was the most commonly observed classification; incidence was 14.35% within our population; incidence of B1, B2 and B3 subtypes were 2.91%, 6.98% and 4.46%, respectively. Duplex GBs only made up 0.39% of the total population. The incidence of complex (C) morphologies was 0.19%.
Conclusions and relevance
The incidence of GB morphologic anomalies was higher in our population than previously reported. Identification of these anomalies on routine ultrasound evaluation is common; numerous different morphologies can be identified and a standardized classification scheme is proposed. Complete evaluation of morphology can be challenging, particularly with regard to cystic duct anatomy. Clinical significance is uncertain and future studies are warranted to determine the relationship between morphologic variants and hepatobiliary disease.
Introduction
The feline gallbladder (GB) is typically an ovoid-to-pear shaped anechoic fluid-filled structure with a thin echogenic wall ultrasonographically identified to the right of midline surrounded by hepatic parenchyma, within a fossa formed by the right medial and quadrate liver lobes. The GB is subdivided into the fundic region (apex), body and neck, which tapers into the cystic duct; the cystic duct merges with tributaries of extrahepatic bile ducts to form the common bile duct. The ultrasonographic appearance and normal features of the feline GB have been previously described.1 –5
Embryologically, hepatobiliary structures arise from the hepatic diverticulum, a bud-like out-pouching of the ventral wall of the primitive midgut, further subdivided into the larger pars hepatica and the smaller pars cystica. The GB, cystic duct and common bile duct arise from the superior bud of the pars cystica.6 –8 The GB passes through a solid phase during development and then is eventually recanalized.6,9 Derangement in this process of differentiation can cause congenital anomalies in the location, number and/or form of the GB; morphologic anomalies result from either subdivision of the primordial GB bud (duplications) or incomplete recanalization from the solid stage (septations).8,10 –12
A breadth of congenital anatomic variants in GB morphology have been recognized and are classified based upon their embryologic origin. The earliest classification scheme was described by Boyden in 1926.10 With the goal of providing a ‘comparative and embryological background against which congenital anomalies of the human GB could be interpreted and classified’, Boyden undertook a large-scale necropsy study of human cadavers and domesticated mammals. The incidence of GB anomalies in humans proved to be rare, with only five confirmed reports in 19,000 cadavers and patients (0.26%); the currently reported incidence rate in the medical literature is between 0.25% and 4%, depending on the type of anomaly.8,10 However, Boyden discovered that the cat ‘[exhibited a great] number and variety of aberrant’ GB morphologies with an incidence rate of over 12%. 10
Two major categories of malformation were distinguished: vesica fellea divisa and vesica fellea duplex. Vesica fellea divisa results from abnormal division of the GB primordium leading to two or more GB lobes sharing a single cystic duct. These ‘bilobed’ GBs are described as externally normal in shape with a longitudinal septum, or V-shaped, with lobes demonstrating a variable degree of separation/clefting and asymmetry. The second group, vesica fellea duplex, results from a redundant biliary primordium resulting in two separate accessory GBs, each with its own cystic duct, often described as a Y-shaped GB.10,13 Also reported in the human literature are congenital septated GB morphologies characterized by transverse or longitudinal septa creating two or more communicating compartments; this type of anomaly results from erroneous recanalization of the GB from its solid phase. 8
There are few published reports in the veterinary medical literature documenting GB morphologic anomalies in the cat, which have been identified via imaging, surgery or necropsy.9,11,14 –19 Anecdotally, feline GB anomalies are commonly encountered in the clinical setting on routine abdominal ultrasound, with the impression of a higher incidence than previously reported; this observation led to a prospective investigation of the ultrasonographic appearance, degree of variability and incidence of abnormal GB morphology in our feline population.
Materials and methods
A prospective, descriptive study was undertaken at the Foster Hospital for Small Animals at Cummings School of Veterinary Medicine at Tufts University with patient enrollment spanning a period of 18 months (August 2016–December 2017). All feline patients that had an abdominal ultrasound performed during this period were initially included. Ultrasonographic images were obtained by numerous operators (board-certified radiologists and radiology residents) in the Diagnostic Imaging Department on a Philips Epic 7G or a Philips iU22 ultrasound machine with curvilinear 8–5 MHz and/or linear 12–5 MHz transducers. To be included in the study, patients were required to have at least one still image of the GB in both a transverse and sagittal plane. Cineloops of the GB were reviewed when available but were not required for inclusion in the study. Studies were reviewed by two board-certified radiologists (DGP, HNS) until a consensus was reached in each case.
GB shape was recorded using a classification scheme of morphologic variants adapted from the human literature. 10 Three overarching morphologic groups were identified, including septated, bilobed and duplex GBs. A septated morphology (S) was defined as a GB with symmetric or asymmetric compartments created by a soft tissue septum originating along the wall of the GB body anywhere from the fundus to the GB neck and extending transversely into the GB lumen (Figure 1). A bilobed morphology (B) was defined as a fold of the GB arising at the apex with variable length of extension toward the GB neck ± formation of a cleft with separation of the two lobes by intervening hepatic parenchyma (Figure 1). The bilobed morphology was further divided into three subcategories. B1 was defined as a bilobed GB with an apical fold without evidence of a cleft/lobar separation. B2 was defined as a bilobed GB with an apical fold and clefting/separation of the lobes extending less than 50% of the length of the GB, creating a ‘heart-shaped’ appearance. B3 was defined as a bilobed GB with an apical fold and clefting/separation of the lobes extending more than 50% of the length of the GB. All GBs with bilobed morphology converged to a single cystic duct. A duplex GB (D) was defined as two GB bodies of symmetric or asymmetric volume with two distinct cystic ducts entering a single common bile duct (Figure 1). A fourth category of complex (C) was reserved for morphologies that demonstrated features of multiple categories (S, B, D).

Proposed simplified classification of gallbladder (GB) morphology. The top row demonstrates variations in location of the transverse septum along the GB axis (apex to neck) within the septated (S) group. The second row represents the three subtypes of bilobed (B) GBs based on variation in the presence and degree of clefting, including B1, B2 and B3 (left to right). The bottom demonstrates the duplex type (D)
Additional recorded features included the following: GB wall thickness (mm, normal ⩽1 mm); 3 presence and degree of sludge (0, none; 1, <25% of volume; 2, 25–50% of volume; 3, >50% of volume); presence of biliary lithiasis (defined as hyperechoic, strong shadowing concretions at any level of the biliary tree including the GB, cystic/common bile duct and/or intrahepatic ducts) (yes, no); and presence of hepatic cystadenoma-like lesions (defined as discrete structures comprising numerous adjacent to coalescing, thin-walled, anechoic cysts with distal enhancement artifact and variable intra/perilesional hyperechogenicity) (yes, no). Mural thickness, sludge and biliary lithiasis were evaluated as they could be indicators of inflammation and biliary cystadenoma-like lesions may be more prevalent in cats with biliary anomalies. Demographic data were limited to patient age, breed and sex. No clinical history, physical examination findings, laboratory or histopathologic data were reviewed.
Statistical analysis
Statistical evaluations were performed by a statistician at the SAS Institute (SAS 9.4). The signalment, presence of cystadenoma like lesions and presence of choleliths were compared, using Fisher’s exact test, between cats in the normal and abnormal groups. The presence and degree of sludge between normal and abnormal cats was evaluated using a χ 2 analysis. GB wall thickness was evaluated with a logistic regression model. A P value <0.05 was considered statistically significant.
Results
During the study period a total of 578 cats had an abdominal ultrasound performed. Of these, 61 cats were excluded. In the majority of cases (n = 53) patients were excluded for inadequate imaging of the GB (only one image plane or incomplete visualization of the GB); in three cases the GB could not be distinguished from other biliary structures; in four cases the GB was of very small volume/collapsed and morphology could not be determined; and in one case the patient had a prior chole-cystoduodenostomy, which altered the GB shape. In total, 516 cats met the inclusion criteria. Of the included patients, videos of the GB were available in 295 cases.
Following review, 389 (67.3%) cats were determined to have normal GB morphology. Within the normal group there were 207 (53.2%) males and 182 (46.8%) females. The mean age for the normal morphology group was 10.0 years (range 5 months–19 years). The majority of cats (n = 309 [79.4%]) were domestic shorthair (DSH; n = 233 [75.4%]), mediumhair (DMH; n = 28 [9.1%]) or longhair (DLH; n = 48 [15.5%]). The most common pure breeds were Maine Coon (n = 24) and Siamese (n = 16). The remainder of the normal morphology group included the following breeds: Persian (n = 4), Devon Rex (n = 4), Abyssinian (n = 3), Himalayan (n = 3), American Shorthair (n = 3), British Shorthair (n = 2), Siberian Longhair (n = 2), Korat (n = 2), Chartreux (n = 2), Ragdoll (n = 2), Russian Blue (n = 2) and Norwegian Forest Cat (n = 2). Each of the following breeds was represented by a single case: Occicat, Manx, Balinese, Burmese, Bombay, Tonkinese, Turkish Angora, Scottish Fold and Exotic Shorthair. Video clips were available for review in 182/389 (46.8%) cases.
Within the normal morphology group mean GB wall thickness was 0.85 mm (range 0.3–2.8) with 324 cases measuring ⩽1 mm. One hundred and fifty-five (39.8%) cats in the normal group did not have any GB sludge accumulation (grade 0). Grade 1 sludge was identified in 155/389 (39.8%) cats; grade 2 sludge was identified in 60/389 (15.4%) cats; grade 3 sludge was identified in 19/389 (4.9%) cats. Choleliths were identified in 16/389 (4.1%) cats. Cystadenoma-like lesions were noted in 20/389 (5.1%) cats. The data are summarized in Table 1.
Additional evaluated ultrasonographic variables reported within the normal and anomalous gallbladder (GB) groups

Data are mean (range) or n (%)
Statistical significance = P <0.05
One hundred and twenty-seven cats were determined to have anomalous GB morphology. Within the anomalous morphology group there were 80 (63.0%) males and 47 (37.0%) females. Age ranged from 5 months to 18 years (mean 9.9). The majority of cats (n = 96 [75.6%]) were DSH (n = 78 [61.4%]), DMH (n = 5 [6.4%]) or DLH (n = 13 [10.2%]). Pure breeds included Siamese (n = 7), Persian (n = 6), Ragdoll (n = 5), Maine Coon (n = 3), Himalayan (n = 3), Abyssinian (n = 2) and British Shorthair (n = 2), and a single case each for the following breeds: Chartreux, Pixie Bob and Bengal. Video clips were available for review in 113/127 (89.0%) cases.
Anomalous morphology was categorized by the previously described scheme as septated (S), bilobed (B1, B2, B3), duplex (D) or complex (C). Of the 127 anomalous GBs, 50 (39.4%) were designated as septated (Figure 2), 74 (58.3%) as bilobed (Figures 3–5), two (1.6%) as duplex (Figure 6) and one (0.8%) as complex. Within the bilobed group, 15 (11.8%) cats demonstrated the B1 subtype (Figure 3), 36 (28.4%) the B2 subtype (Figure 4) and 23 (18.1%) the B3 subtype (Figure 5). Data are summarized in Table 2.

Sagittal plane image of a septated (S) gallbladder (GB) morphology. Note the echogenic band crossing the middle of the GB

Sagittal plane image of a bilobed (B1) gallbladder (GB) morphology. A discrete hyperechoic band is dividing the GB at the level of the apex. There is an apical fold without evidence of lobar separation

Sagittal plane image of a bilobed (B2) gallbladder (GB) morphology. There is an apical fold and separation of the lobes extending less than 50% of the length of the GB, giving a heart shaped appearance to the GB

Sagittal plane image of a bilobed (B3) gallbladder (GB) morphology. There is an apical fold and clefting/separation of the lobes extending more than 50% of the length of the GB

Sagittal plane image of a duplex (D) gallbladder (GB) morphology. There are two GB bodies of symmetric size with two distinct cystic ducts (*) entering a single common bile duct
Within the anomalous morphology group mean GB wall thickness was 0.79 mm (range 0.4–2.5) with 113 cases measuring ⩽1 mm. Twenty-nine cats (22.8%) in the anomalous group did not have any GB sludge accumulation (grade 0). Grade 1 sludge was identified in 66/127 (52.0%) cats; grade 2 sludge was identified in 25/127 (19.7%) cats; grade 3 sludge was identified in 7/127 (5.5%) cats. Choleliths were identified in 7/127 (5.5%) cats. Cystadenoma-like lesions were noted in 13/127 (10.2%) cats. Data are summarized in Table 1.
The overall incidence rate of anomalous GB morphology in our study population was 24.6%. When examined by morphologic type, the septated (S) morphology had an incidence of 9.7% in the total population. A bilobed (B) morphology was the most commonly observed classification; incidence was 14.3% within the population. When broken down into the previously described subcategories, the incidence of B1 was 2.9%, the incidence of B2 was 7.0% and the incidence of B3 was 4.5%. Duplex GB only made up 0.4% of the total population. The incidence of complex (C) morphologies was 0.2%. Data are summarized in Table 2.
Total number and incidence rate of each anomalous gallbladder morphology classification within the anomalous group and within the total study population
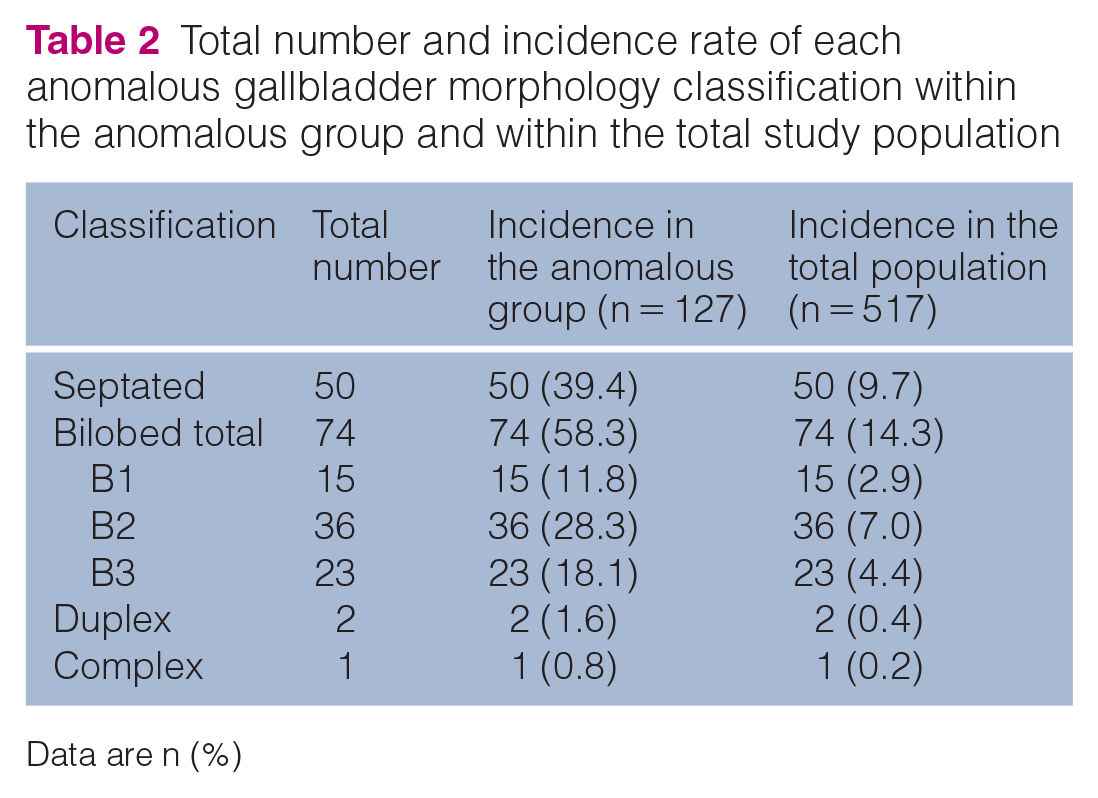
Data are n (%)
Within the total population of cats, 33 (5.7%) had cystadenoma-like lesions. Within the population of normal GBs, 20 (5.1%) patients had cystadenoma-like lesions. Within the anomalous GB group, 13 (10.2%) had cystadenoma-like lesions. In comparing the two groups, the incidence of cystadenoma was statistically significantly higher in the anomalous GB group (P = 0.038). The anomalous group also had a statistically significantly higher amount of sludge compared with the normal GB group (P = 0.007). Overall, cats with cystadenoma-like lesions were older (mean age 13.2 years) with only 4/33 cats younger than 10 years of age. The other evaluated parameters of GB wall thickness and presence of choleliths were not statistically significantly different between the two groups (P = 0.078 and P = 0.328, respectively). No statistically significant difference in sex distribution was detected between the normal and anomalous morphology groups (P = 0.064).
Owing to the small number of numerous represented breeds within the study population, too few degrees of freedom existed within the data to determine any breed association in a statistically rigorous and defensible fashion. Anecdotally, the Persian breed appeared slightly disproportionally represented in the anomalous morphology group, with six anomalous GB and four normal GBs; compared with the total incidence of 24.6% anomalous GB morphology in our population, 60% of included Persian cats had an anomalous GB morphology.
Discussion
In our prospective study, we evaluated 516 feline GBs with ultrasound and determined that nearly 25% of that population had anomalous GB conformation; this incidence rate is higher than previously reported (12.5%). 10 Based on the modified classification scheme herein, the most commonly identified anomaly was the bilobed subtype (14.3%) followed by the septated subtype (9.7%); duplex (0.4%) and complex (0.2%) morphologies were much less frequently encountered.
The complexity of GB morphologic variants in cats has been well described by Boyden;10 however, only few reports in the veterinary literature address the imaging appearance of these variants.11,15,17,18 Two of these reports are descriptive in nature and document bilobed (B subtype) GB morphology; in both cases the anomaly was considered unrelated to the presenting complaint and was determined to be an incidental finding.11,18 Only two case reports exist of anomalous GB morphology in cats in conjunction with clinical biliary disease; in both instances, a duplex GB was identified.15,17 There is one additional report of a bilobed GB identified at necropsy in a patient with concurrent cholangiohepatitis and cholelithiasis, but no clinical information was available, and a link between the GB anomaly and presence of hepatobiliary disease could not be determined. 19
Numerous reports of diseased bilobed and duplex GBs exist in the human literature. Anomalous GBs manifest similar pathologies to their single-vesicle counterparts, including cholelithiasis, cholecystitis, cholangitis, empyema, torsion, papilloma and carcinoma. Based on most recent review papers it is reported that atypical GB morphology does not predispose to pathology, and the documented incidence of GB disease does not differ between patients with anomalous vs normal GBs.20 –24 One report in humans proposed that patients with anomalous GB morphology were prone to inflammation and, in particular, formation of choleliths; 25 a 2021 paper in veterinary medicine suggested that biliary morphology may play a role in facilitating pancreatic juice regurgitation. 26 Therefore, some controversy over the clinical relevance of GB morphologic anomalies remains and must be further investigated in the veterinary literature.
In this study, the only variables that demonstrated a statistically significant difference between the anomalous and normal groups were the presence of GB sludge and the presence of concurrent cystadenoma-like lesions. There was no difference between the normal and anomalous morphology groups with regard to mural thickening or the presence of choleliths.
Human patients presenting with symptoms suggestive of biliary disease (eg, right upper abdominal quadrant pain, nausea, vomiting and icterus) often undergo preliminary abdominal ultrasound, which may identify anomalous GB morphology; when this occurs, further imaging is recommended as other processes can mimic the appearance of a bilobed GB, such as GB folding, choledochal cyst, pericholecystic fluid accumulation, GB diverticulum, vascular bands and adenomyomatosis. 27 Furthermore, ultrasonographic interrogation of the cystic and common bile duct can be challenging, hampering discrimination between bilobed and duplex GB morphologies. 20 Advanced hepatobiliary imaging with CT, endoscopic retrograde cholangiopancreatography (ERCP) or magnetic resonance cholangiopancreatography (MRCP), which is currently considered the gold standard,20,22 is routinely advocated in human patients who require surgical intervention to confirm the type of morphologic variant and investigate specific biliary anatomy (eg, relationship of the GBs to the cystic, common and extrahepatic ducts); this is particularly important when a laparoscopic surgical approach has been chosen.21,22 In these patients, complete cholecystectomy of both lobes is generally pursued. When an anomalous GB morphology is identified in an asymptomatic individual prophylactic cholecystectomy is not recommended.22 –24
Both clinical cases presented in the veterinary literature went to surgery. In the case reported by Moores and Gregory, 15 the patient suffered septic peritonitis secondary to rupture of one lobe of a duplex GB, which was identified ultrasonographically. At surgery the right lobe was described as grossly normal with normal luminal bile; the left lobe was abnormal with mural thickening, an apical rupture and multiple choleliths within the left cystic duct. Intraoperatively, it was not possible to determine where the respective cystic ducts joined the common bile duct, or the exact number and location of extrahepatic ducts. Only on post-mortem examination was it clear that the left cystic duct entered the distal common bile duct near the duodenal papilla (H-type duplex GB morphology). Woods et al described a case of GB mucocele associated with one lobe of a duplex GB. 17 Intraoperatively, the right lobe was mildly distended but contained grossly normal bile and could be easily expressed; the left lobe was abnormal in appearance with white intramural infiltrates, viscous bile and a cholelith in the proximal left cystic duct. 17 A partial cholecystectomy of the left lobe was performed; however, the left cystic duct could not be entirely removed owing to its close proximity to the right cystic duct.
As in humans, ultrasound evaluation of the feline biliary tree can be a challenge; there are inherent limitations of the modality, including the presence of artifacts (eg, rib shadowing, reverberation from overlying gastrointestinal gas), operator-dependence and the small size and often tortuous path of small ductular structures. Both MRCP and ERCP have been described in cats.28 –30 ERCP requires specialized equipment, is technically demanding and has only been investigated in healthy cats up to this point. MRCP is non-invasive and provides excellent visualization of the hepatobiliary and pancreatic ductal systems; it does, however, require general anesthesia. In each of these clinical cases additional imaging with MRCP would likely have provided valuable information regarding biliary anatomy to inform surgical planning.
Interestingly, multiple case reports in humans also document a single lobe of a bilobed/duplex GB being diseased.23,24,31 This finding calls into question whether the presence of an anomaly in conjunction with biliary disease may lead to asymmetric exacerbation due to segmentally reduced ejection fraction or trapping of debris/stones.
There are several limitations of this study. Despite the prospective enrolment of feline patients in our study, ultrasound examinations were performed by multiple ultrasonographers to investigate a variety of clinical concerns across the study period without standardized requirements for GB evaluation, which resulted in innate variability in the quality of images and videos available for retrospective review. Shadowing from overlying ribs was frequently encountered on review; this artifact was less common in transverse images but may have masked more subtle lesions, particularly transverse septations in the apical region. Still images in both planes would often look normal and may have been documented as such in the imaging report, but on close scrutiny of available videos septations were identified. Even with the availability of videos on many of our patients, assessment of the ductal anatomy (cystic, common, extrahepatic) was challenging. As a result, it is possible that the number of duplex conformations was underestimated in this population. Similarly, it was not considered feasible to accurately discriminate between Y-type and H-type (or ductular/accessory) cystic conformations; therefore, all GBs with two cystic ducts were categorized within the umbrella of ‘duplex’ malformation. In addition to anomalies of GB shape, Boyden documented variations in GB position, which was also not specifically addressed in this study.10 Clinicopathologic data and patient history were not reviewed in conjunction with the ultrasonographic findings, and, as such, no inference can be made based on our data regarding correlation of hepatobiliary disease with the presence or absence of GB anomalies.
Conclusions
Ultrasonography can readily identify morphologic anomalies of the feline GB. The incidence of such anomalies was nearly 25% in our population, with a bilobed morphology most commonly identified. Patients with anomalous GB morphology were more likely to have GB sludge and concurrent cystadenoma-like lesions; however, the presence of choleliths and mural thickening was not statistically different between the normal and anomalous GB morphology groups. Advanced imaging such as MRCP may be considered as a complementary study to screening ultrasound in cats to definitively determine the type of anomaly and ductal anatomy in cases requiring surgical intervention. The clinical relevance of GB anomalies at this point is unclear; the possibility that they may exacerbate underlying hepatobiliary disease is considered and further prospective studies are warranted.
Footnotes
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case series.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
