Abstract
Objectives
The aim of the study was to evaluate whether any admission vitals correlated with the presence of brain herniation diagnosed via MRI in cats presenting with neurologic signs.
Methods
Medical records at two veterinary university referral centers were reviewed to identify cats that underwent brain MRI between 2010 and 2019. A control group of cats with intracranial lesions without concurrent brain herniation was analyzed for comparison. Data relating to signalment, vitals on admission, abnormalities observed on initial neurologic examination, underlying etiology, advanced imaging findings and outcome were reviewed. A Modified Glasgow Coma Scale (MGCS) score was determined retrospectively based on initial neurologic examination. Logistic regressions were performed to investigate the relationship between each risk factor and the odds of brain herniation as diagnosed on MRI.
Results
Thirty-two cats with brain herniation and 44 cats with abnormal brain MRI without evidence of herniation (as a control group) based on MRI findings were included. Cats with intracranial neoplasia vs other diagnoses were found to be at increased risk of herniation (odds ratio [OR] 4.8, 95% confidence interval [CI] 1.8–13.8; P = 0.001). The odds of herniation increased with age (OR 1.1, 95% CI 1.01–1.2; P = 0.031). Cats with herniation had a significantly lower level of consciousness in their MGCS score (P <0.0001) than cats without herniation. There was no significant difference in either motor activity or brainstem reflexes between the groups (P >0.05).
Conclusions and relevance
Admission heart rate and blood pressure were not associated with brain herniation. Cats with herniation were presented with a significantly lower level of consciousness in their MGCS score; however, this clinical feature cannot be directly attributable to and predictive of herniation. Older cats with intracranial neoplasia are more likely to have brain herniation.
Keywords
Introduction
Brain herniation is caused by the shifting of brain structures across anatomical boundaries within the skull as a result of rising intracranial pressure (ICP).1,2 If left untreated, it may lead to a life-threatening neurologic emergency, permanent brain injury or death. Brain herniation occurs secondarily to various intracranial lesions such as trauma, hemorrhage, edema, encephalitis and neoplasia. In some neoplastic diseases (mainly slow-growing tumors such as meningioma), the rise in the ICP may occur over months to even a year. 3 The diagnosis of brain herniation is made with advanced imaging such as MRI or CT;2,4 however, in the emergency setting brain herniation is often treated empirically.1,5,6 Cats with brain herniation may require stabilization prior to anesthesia or transport to a referral practice with neuroimaging. Therefore, initial resuscitative measures aiming for meaningful recovery, avoidance of irreversible damage and stabilization of the patient are prioritized over confirmation of herniation or diagnosis of underlying etiologies. 6
Clinical signs classically associated with elevated ICP include pupillary changes, reduction in consciousness and altered cardiorespiratory function.6,7 Among those, the Cushing reflex is a warning sign of brain herniation. 8 This reflex is an attempt to maintain appropriate cerebral perfusion pressure (CPP) in response to profoundly increased ICP. 9 As CPP is determined by the gradient between mean arterial blood pressure (MAP) and ICP, increased ICP will lead to a reduction in the gradient that jeopardizes cerebral perfusion. 4 To restore the CPP, the vasomotor center of the brain drives sympathetic activity, causing peripheral vasoconstriction, which increases MAP. 4 Once MAP has risen significantly, the carotid and aortic baroreceptors are stimulated and slow the heart rate to the point of bradycardia. 9 In people, evidence of the Cushing reflex in combination with low Glasgow Coma Scale (GCS) score serves as a triage tool for elevated ICP in patients with traumatic brain injury (TBI).6,10 A recent retrospective study demonstrated the clinical association between the Cushing reflex and brain herniation in dogs. In the study, dogs with brain herniation presented with higher systolic blood pressure (SBP), lower heart rate (HR), and lower Modified GCS (MGCS) score at triage. 11 To date, this association has not been investigated in cats with naturally occurring brain herniation.
The purpose of this retrospective case series was to evaluate admission vitals in cats with naturally occurring brain herniation vs those with intracranial lesions without brain herniation and to assess whether any specific findings had a predictive value. We hypothesized that cats with evidence of brain herniation on MRI are more likely to exhibit the Cushing reflex upon admission and have a lower MGCS score than those without brain herniation. The secondary aim of this study was to investigate a cut-off value of SBP and HR that would be indicative of the Cushing reflex in cats.
Material and methods
Case selection
Electronic medical records between January 2010 and May 2020 from two veterinary university referral centers were searched to identify cats that underwent brain MRIs for neurologic signs and were diagnosed with a brain lesion with or without brain herniation, by board-certified radiologists. MRI studies were performed using a 1.5 T (Infinion 1.5T; Philips Medical Systems) or 3.0 T (Skyra 3T; Siemens Medical Systems) superconducting magnet. The MRI protocol included T1-weighted (T1W) images, T2-weighted (T2W) images and fluid attenuated inversion recovery images in sagittal, transverse and/or dorsal planes, in addition to GRE/T2* and T1W images post-contrast with intravenous administration of gadoteridol enhancement (ProHance; Bracco Diagnostics). The MRI images were reviewed for diagnostic accuracy by board-certified neurologists (YM, AY) using the standard transverse and sagittal sequences as outlined above. MRI findings were considered abnormal if parenchymal changes or anomalies were present. Cats with non-compressive lesions such as ischemic infarcts were excluded as these cases lack space-occupying lesions and are less likely to cause elevated ICP.
Based on the reviewed MRI scans, cats were divided into two groups: cats with brain herniation as a study group and cats with abnormal brains without herniation as a control group. The type of brain herniation was also categorized. Herniation at the foramen magnum (FMH) was defined as the caudal displacement of the cerebellar vermis into or through the foramen magnum. Caudal transtentorial herniation (CTH) was defined as unilateral or bilateral herniation of the parahippocampal gyri of the cerebral cortex ventral to the tentorium cerebelli. Subfalcine herniation (SFH) was defined as herniation of the cingulate gyrus under the falx cerebri.
Clinical information
Records were reviewed for the patient’s signalment, history, presenting complaints, admission vitals, initial neurologic examination, concurrent medical conditions, MRI findings and outcome. Cats with complete medical records, including admission vital signs and initial neurologic findings, were included. The admission vitals included rectal temperature, HR, respiratory rate (RR) and SBP prior to any intervention. SBP was typically measured by the attending veterinarian or a veterinary technician using a Doppler flow detector with a 9.5 MHz probe. Cats with comorbidities or concurrent medications that could potentially alter SBP and HR were excluded (eg, congestive heart failure, acute or chronic kidney failure, history of long-term glucocorticoid use, blood pressure medication, etc). Where available, the time to identification of brain herniation, defined as the time of hospital admission to MRI, was recorded. The primary outcome was determined as cats that survived to discharge from the hospital. Additionally, anesthetic recovery was determined to be normal or abnormal, with abnormal recoveries categorized as prolonged (>25 mins from discontinuing anesthetic gas to extubation or failure to regain consciousness) or failure to regain spontaneous respiration requiring mechanical ventilation.
The initial neurologic findings of included cats were reviewed. Duration of neurologic clinical signs until admission in each patient was categorized as acute (⩽1 week) or chronic (>1 week). Neurolocalization was categorized as forebrain, brainstem, cerebellum, cervical or multifocal. Cats with profoundly altered mentation that presented in status epilepticus or in a cluster seizure event (with or without administered anticonvulsants) were classified as forebrain lesions. Tetraparesis/ataxia with cranial nerve deficits was considered to originate from the brainstem, and an altered level of consciousness without any other abnormalities was considered either forebrain or brainstem by origin. An MGCS score was assigned retrospectively by primary investigators from each institution (JH, YM), based on initial neurologic examination performed by board-certified neurologists or neurology residents on admission, to attempt to delineate the severity of signs to further validate the use of this scale in acute vs chronic presentations. The MGCS score was calculated across three categories (level of consciousness, motor activity and brainstem reflexes), each assigned a score of 1–6 (with 6 representing normal findings and 1 representing severe impairment), combining to a total score ranging from 3 and 18 (with a higher number generally associated with a more favorable outcome). 12
All data were aggregated into an Excel spreadsheet (Microsoft Office), and all analyses were performed using commercially available statistical software (SAS version 9.4; SAS Institute). Data were presented as median (interquartile range [IQR]). Simple logistic regressions were performed to test for association of each risk factor (age, HR, SBP, RR, weight, rectal temperature, MGCS score, MGCS motor activity, MGCS brainstem reflexes, MGCS level of consciousness, diagnosis) and odds of herniation, as well as evaluating herniation and odds of anesthetic recovery and survival to discharge. Sensitivity and specificity values were calculated for score cut-offs of 4, 5 and 6 for MGCS level of consciousness. Log-likelihood ratio test P values were reported. A significance threshold of 0.05 was used.
Results
A total of 79 cats that underwent brain MRIs between 2010 and 2019 were identified. Brain herniation was detected in 32/79 cats, resulting in a 40.5% prevalence. Three cats were excluded from analysis because of one or more exclusion criteria. As a result, a total of 76 cats were included for analysis. The demographic information of the included cats is summarized in Table 1. Thirty-two cats had evidence of brain herniation, and 44 cats had intracranial lesions without herniation on MRI. Among the 32 cats with herniation, 21 had both CTH and FMH, eight had FMH, two had CTH and one cat suffered from three concurrent herniations (ie, SFH, CTH and FMH). The median time between admission and MRI of the herniation group (n = 20) and control group (n = 32) was 26 h (IQR 23–53.5) and 25 h (IQR 19.1–34.7), respectively. Time lapse between admission and MRI was not significantly different (P = 0.77). Figure 1 demonstrates various etiologies diagnosed on MRI resulting in brain herniation.
Demographics for 76 cats with brain herniation (case) and intracranial lesions without herniation (control)
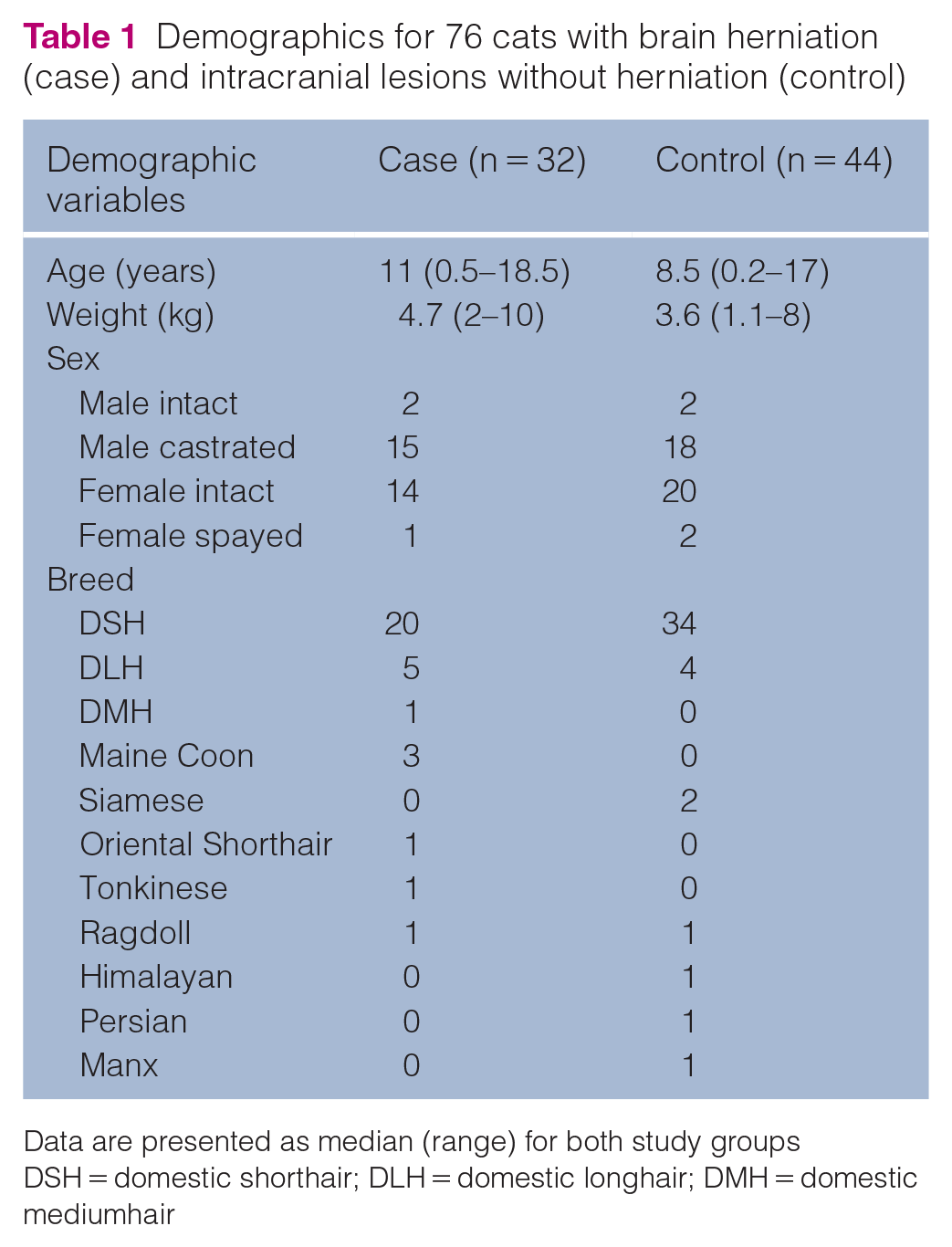
Data are presented as median (range) for both study groups
DSH = domestic shorthair; DLH = domestic longhair; DMH = domestic mediumhair
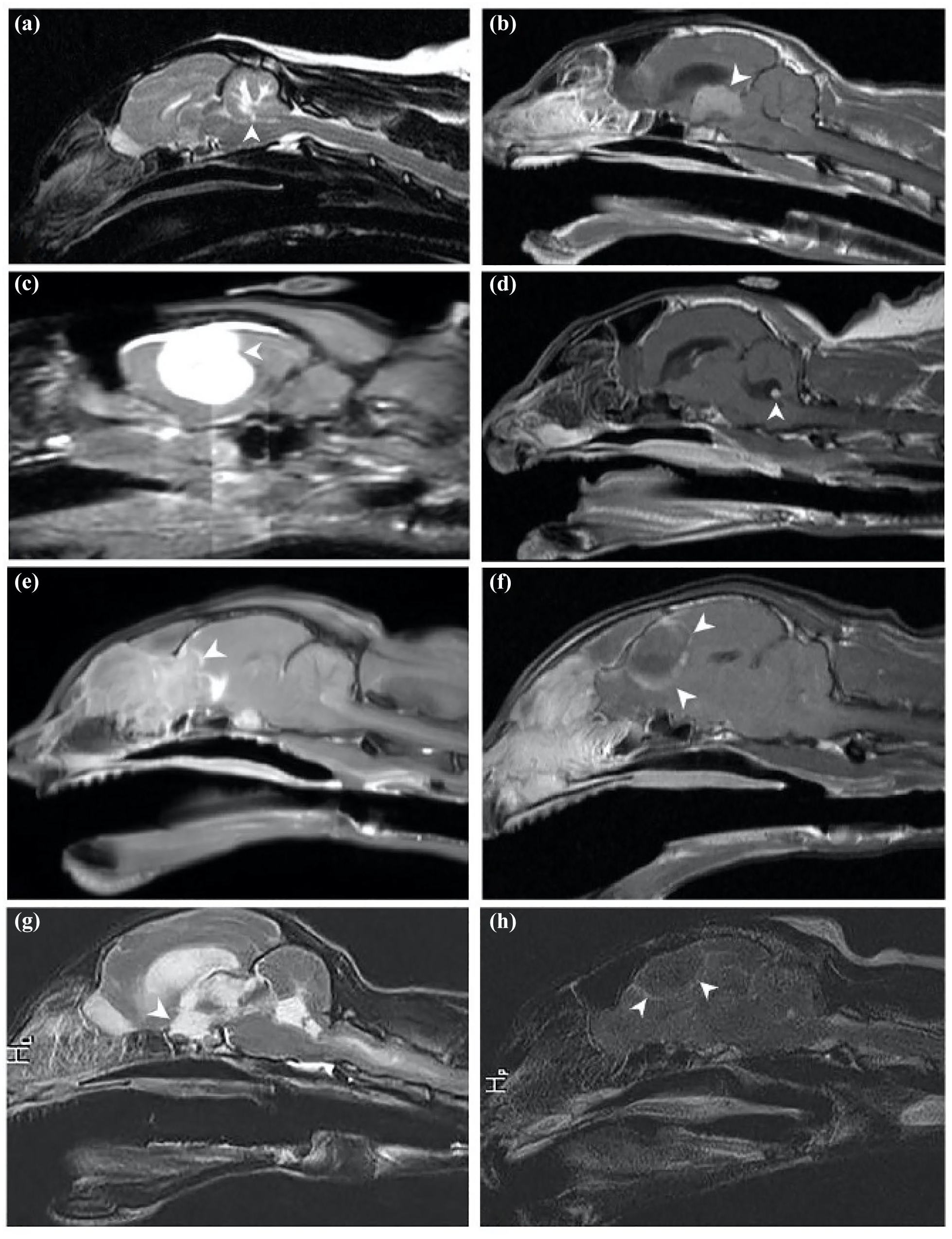
Sagittal and parasagittal T1-weighted post-gadolinium images in selected cases depicting the different etiologies causing brain herniation. (a) Infiltrative cerebellar disease; (b) choroid plexus tumor with secondary obstructive hydrocephalus; (c) meningioma; (d) meningoencephalitis of unknown etiology; (e) lymphoma penetrating from the nasal cavity; (f) an abscess suspected to originate from the nasal cavity; (g) feline infectious peritonitis; and (h) meningioma
The clinical findings of cats with and without herniation are summarized in Table 2. Outcome was evaluated for the herniation group (n = 32); of these, five cats were euthanized under anesthesia due to expected poor prognosis based on MRI findings, with no attempt made to recover from general anesthesia. Among the 27 cats with herniation that were allowed to recover, only one cat failed to regain consciousness and spontaneous respiration upon discontinuation of inhalant anesthesia. The remaining 26 cats recovered from anesthesia uneventfully and survived to discharge (81%); of these, long-term survival (>24 h) data were available for nine cats in the herniation group; long-term survival ranged from 3 weeks to 2.5 years. Outcome data were available on 34/44 cats in the control group; of these, five cats were euthanized under anesthesia owing to expected poor prognosis based on MRI findings. Of the 29 cats that were allowed to recover, only one cat had a prolonged recovery from anesthesia due to generalized seizure activity and hypothermia. All 29 cats recovered from anesthesia and survived to discharge (85%). The odds of abnormal anesthetic recovery and survival to discharge were not significantly affected by the presence of herniation (P = 0.253).
Clinical findings in 76 cats with brain herniation (case) and intracranial lesions without herniation (control)

Data are n (%)
Statistical findings on risk factors of the study groups are provided in Table 3. Evaluation of admission vitals revealed no significant difference in admission HR (P = 0.208), RR (P = 0.148), SBP (P = 0.507) or rectal temperature (P = 0.138) between the herniation and control groups. None of the cats in this study showed abnormal breathing patterns at admission. Cats in the herniation group were older (P = 0.031) and heavier (P = 0.035) than those in the control group. The odds of herniation increased by 10% for each year of age (odds ratio [OR] 1.1, 95% confidence interval [CI] 1.01–1.2). Cats with brain herniation had a significantly lower MGCS score (P = 0.002) compared with the control group. In cases with herniation, the MGCS level-of-consciousness score was significantly lower (P <0.001) compared with the control group. The odds of herniation decreased with increasing MGCS level-of-consciousness score (OR 0.15, 95% CI 0.05–0.38). The sensitivity of using <5 for the MGCS level-of-consciousness score cut-off was 75% and specificity was 77% to diagnose herniation. Sensitivity for a score of 4 was 12.5% (specificity 98%) and specificity for a score of 6 was 0% (sensitivity 100%). Hence, an MGCS level-of-consciousness score <5 was found as the cut-off to best balance the tradeoff between sensitivity and specificity. Cats with herniation were more likely to be diagnosed with neoplasia (P >0.001) than those in the control group. The odds of herniation increased by 4.8 times with neoplasia vs other diagnoses (OR 4.8, 95% CI 1.8–13.8).
Risk factors on admission in cats with brain herniation (case) compared with cats with intracranial lesions without herniation (control)

Data are presented as median (interquartile range) for both study groups
Significant difference between groups
HR = heart rate; bpm = beats per min; RR = respiratory rate; SBP = systolic blood pressure; MGCS = Modified Glasgow Coma Scale
Discussion
Previous studies have described the presence of the Cushing reflex in association with increased ICP, which can be seen with brain herniation.13–16 Huizing et al evaluated the skulls and brain parenchyma of normal cats without abnormal brain lesions, reporting up to 40% of incidental brain herniations. 17 Lewis et al concluded that neurologic signs directly attributable to brain herniation were absent in dogs and cats. 2 Lastly, Minato et al evaluated the variations in herniation types between dogs and cats with meningiomas. 18 The study found that cats had a higher incidence of FMH and/or CTH. The current study further supports these findings with concurrent FMH with CTH being the most common finding.
The principal aim of this retrospective study was to determine whether cats with suspected chronic increase in ICP leading to brain herniations had any admission vitals and/or initial neurologic findings that could predict their brain herniation. This study found that cats with brain herniations exhibited similar admission vitals as cats with intracranial lesions without herniations. Other important findings were as follows: (1) cats with herniations were presented with a significantly lower level of consciousness in their MGCS score; (2) older cats or cats with intracranial neoplasia were found to be at increased risk of herniation vs those with other diagnoses; and (3) the presence of herniation did not negatively impact anesthetic recovery or survival to discharge. Long-term mortality was not evaluated in this study.
Based on a similar study conducted in dogs, 11 our hypothesis was that a Cushing reflex will likely be exhibited in cats with brain herniation. Our hypothesis was not supported by the current study. Experimentally, Hoff and Reis demonstrated that an acute and diffuse increase in ICP in anesthetized, paralyzed cats resulted in systemic hypertension with bradycardia, suggesting its presence in cats. 16 There are known compliance mechanisms such as displacement of cerebrospinal fluid (CSF) and cerebral blood flow to autoregulate ICP. Furthermore, a recent study in rats reported reduction in brain parenchymal volume that was hypothesized as another mechanism to limit ICP rises with very-large-mass lesions. 19 Given that neoplasia was the most common diagnosis in this study (n = 22/32 [69%]), it is possible that many cats in the study were in the compensatory phases of elevated ICP secondary to slowly growing, space-occupying lesions.
Additionally, previous studies demonstrated that the simultaneous onset of hypertension and tachycardia is an early indicator of impaired brain perfusion, followed by the development of severe bradycardia.20,21 These findings suggest that each cat in the study population may have been in a variable time course of the Cushing reflex, which may have skewed our results. Lastly, the small number of cats evaluated at a single time at admission may have challenged the detection of vital changes indicative of the Cushing reflex. Further investigation is needed to evaluate the predictive role of the Cushing reflex with serial evaluation of vitals in cats with acute increases in ICP, particularly in TBI cases, as this could result in the dramatic increase in ICP with increased likelihood to observe the Cushing reflex.
Historically, the Cushing reflex was considered as a terminal, ‘last-ditch’ response rather than an early indicator of compromised CPP.4,9,20 In humans sustaining a TBI, the presence of the Cushing reflex predicted increased ICP with a high specificity but a low sensitivity, even when combining the reflex with other clinical criteria (eg, low GCS and fixed dilated pupils).6,10,22 A recent retrospective study in dogs also found that the Cushing reflex predicts brain herniation with high specificity and low sensitivity. 11 While the presence of the Cushing reflex in cats in the clinical setting appears to be limited, it is important to note that the Cushing reflex may be a late sign of increased ICP.4,6,11,20 Therefore, relying on the Cushing reflex to make treatment decisions may lead to a worse outcome.
Cats with brain herniation were more likely to be diagnosed with neoplasia than those in the control group, being the most common diagnosis in this study (n = 22/32 [69%]). This is similar to a previous report in which neoplasia (n = 79/113 [69.9%]) was the most common MRI-based diagnosis in cats with brain herniation. 23 Meningioma is the most common brain neoplasia in cats.3,24 Prior research described the tumor in cats as a firm, well-circumscribed mass, usually not invading the neural parenchyma and therefore more clearly demarcated from normal brain tissue. 25 Further support for this finding was noted in a recent study describing specific MRI findings of the intratumoral region, trying to predict the consistency of a meningeal tumor being solid (thus easier to remove) vs being a more gelatinous tumor. 24 Cat meningiomas were found to have a firmer consistency than those of dogs. 24 Postoperative mortality associated with meningioma removal in cats has been reported to be 19%; survival rates of 71% at 6 months, 66% at 1 year and 50% at 2 years have been reported. 26 These findings, along with the findings of the current study, in which more than a third of cats suffered from non-neoplastic diseases, should prompt veterinarians to advocate for advanced imaging in cats, and in the case of a diagnosed meningioma, consider tumor resection for a favorable outcome (Figure 1).
In the current study, the age and body weight differences between the two groups were significant, with cats with herniation appearing to have been older and heavier, on average, than cats in the control group. The authors hypothesized that this is likely due to an increase in the incidence of neoplasia with increasing age and does not likely reflect a true difference between the groups. With regard to the weight differences, owing to the retrospective nature of the study, body condition score was not collected, thus conclusions on whether the weight differences have any clinical significance cannot be made.
This study attempted to investigate the utility of the MGCS score in cats with brain herniation. The MGCS score is often used in combination with other clinical signs such as pupillary changes and/or the Cushing reflex, which improves triage quality for patients with head trauma.6,22,27 Since its utility as a monitoring and outcome prediction tool has been proven in dogs with TBI,28,29 the MGCS has also been used for non-traumatic neurologic conditions such as post-cardiac arrest and brain tumors.30,31 However, the utility of the MGCS score in chronic progressive disease has not yet been validated. In this study, cats with herniation were presented with a significantly lower level of consciousness in their MGCS score, while the other two categories (motor activity and brainstem reflexes) did not approach statistical significance. Interestingly, it has previously been reported that an obtunded mentation was one of the most common neurologic abnormalities (n = 72/113 [63.7%]) found in cats with herniation, although that study did not specifically evaluate the MGSC score nor did it have a control group. 23 Additionally, 50% (n = 16/32) of the cats in the current study were neurolocalized to the forebrain. Therefore, it is unknown whether the altered mentation was due to primary intracranial lesions, secondary to increased ICP or both. Moreover, it is important to note the discrepancy between the MGCS score and the degree of neurologic abnormalities based on presenting complaint in this study. While the presenting complaints suggested significant neurologic signs, the MGCS score in both groups was >15, which indicates a favorable prognosis. These findings may suggest that the MGCS score is a more appropriate prognostic tool in the acute setting but less reliable in identifying patients with brain herniation owing to chronic intracranial changes. Further prospective studies with a higher case load are needed to elucidate this finding. Lastly, rapid, non-invasive modalities such as transcranial Doppler or transpalpebral ultrasound may provide an indirect surrogate for evaluating ICP in both acute and chronic intracranial pathologies, yet those have not been validated in cats, and are not currently considered bedside practice.32,33
The present study did not find an association between herniation and anesthetic recovery or survival to discharge. While it is generally accepted that brain herniation carries a poor prognosis, the majority of cats with herniation in this study (n = 26/27) recovered uneventfully from discontinuation of inhalant anesthetic, and, besides those euthanized, all survived until discharge. The presence of herniation did not significantly impact the likelihood of recovery from anesthesia and survival to discharge. This finding suggests that pursuing advanced diagnostic imaging under full anesthesia should not be dreaded. Furthermore, it is possible that CSF collection conducted under the same anesthesia for MRI may influence the survival rate to discharge and CSF collection should be exercised with caution (if at all) in cases of concerned increased ICP. In the present study, fewer cats from the herniation group (n = 9/32 [28%]) underwent CSF collection vs the control group (n = 27/44 [61%]). Those cats recovered uneventfully from anesthesia. Owing to the small number of cases that underwent CSF collection, it was not possible to evaluate whether CSF collection was correlated with negative impact on survival to discharge. Data regarding long-term outcomes remain to be investigated.
There are several limitations to the current study. The retrospective nature of the study could lead to biases, including differences in the management of cases and the confounding factor of euthanasia. Cats that were euthanized shortly after presentation prior to imaging may represent cases that were more severely affected and could have changed the overall outcome of cats in this study. Despite combining data from two institutions, the total number of cats that met the inclusion criteria is still small, limiting the ability to detect certain differences that may exist across larger cat populations. The timing used for calculating each patient’s MGCS score could not be standardized more accurately due to the retrospective nature of the study. Ideally, a prospective study should be performed to allow serial assessment of vitals for predicting brain herniation in cats.
Conclusions
Admission vitals could not predict brain herniation in cats with increased ICP. Future investigations are warranted to determine whether the Cushing reflex is associated with ICP in acute vs chronic disease states and whether it can be used as a non-invasive tool to predict brain herniation in a larger cat population. Older cats with intracranial neoplasia are more likely to have brain herniation. Cats with herniation were presented with a significantly lower level of consciousness in their MGCS score; however, this clinical feature cannot be directly attributable to and predictive of herniation without further investigation as it could be related to the underlying pathology alone or a summation effect.
Footnotes
Acknowledgements
The authors are grateful to Dr Deborah Keys for assistance with the statistical analysis.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
