Abstract
Objectives
This study was performed to evaluate retrospectively the clinical signs, complications and postoperative outcomes of feline intracranial meningioma (IM) with concurrent cingulate, transtentorial and foramen magnum herniations.
Methods
The medical records and MRI scans of cats with IM and cerebral herniation were reviewed. Cases involving concurrent cingulate, transtentorial and foramen magnum herniations were included. Owners were contacted to obtain long-term follow-up information.
Results
Seven cats (four castrated males and three spayed females) met the inclusion criteria. Median age was 13.0 years (range 9.9–16.1 years) and median duration of clinical signs was 35 days (range 21–163 days). The clinical signs of cats with cerebral herniation included visual impairment (n = 5 [71.4%]), ataxia (n = 4 [57.1%]), impaired consciousness (n = 2 [28.6%]), head pressing (n = 2 [28.6%]), paresis (n = 1 [14.3%]), torticollis (n = 1 [14.3%]) and personality changes (n = 1 [14.3%]). Median tumour volume, cranial cavity volume and tumour volume:intracranial volume ratio before surgery were 3.37 cm3 (range 3.23–11.5 cm3), 32.6 cm3 (range 29.8–78.3 cm3) and 10.4% (range 5.3–35.3%), respectively. Median overall tumour excision rate was 90.6%. Preoperative intracranial pressure (ICP) ranged from 15 to 32 mmHg (median 29 mmHg). In all cases, the ICP dropped to 0 mmHg immediately after tumour removal. No adjuvant therapy was required after surgery. The median survival period was 612 days (range 55–1453 days).
Conclusions and relevance
The results of this study indicate that surgical treatment of rostrotentorial IM is effective and allows prolonged survival, even in cats with concurrent cingulate, transtentorial and foramen magnum herniations.
Keywords
Introduction
Intracranial tumours are frequently encountered in small animal neurosurgery; the reported prevalence of intracranial tumours in cats is 2.2%. 1 Of these, 71% are primary tumours. 2 Intracranial meningioma (IM) is the most common primary intracranial tumour in cats, accounting for about 60% of cases. 1
IM originates from arachnoid epithelial cells present in the mucosal arachnoid granulations. IMs are composed of cells with both epithelial and mesenchymal morphology that form various tissue types. In veterinary medicine, IM is classified according to its tissue type and biological behaviour. 3 IM in cats shows benign behaviour and is characterised by solitary, distinctively borderless masses that do not invade the brain parenchyma and grow slowly. 4 Additionally, transtentorial herniation generally occurs in feline IM. 1 Because of its biological features, complete surgical removal of IM is possible and is the first-choice treatment; the long-term prognosis after surgery is good. 4
In cats, intracranial tumours generally grow to a huge size before clinical signs develop; cases of cerebral herniation at the time of diagnosis have also been reported.1,2 Cerebral herniation is defined as displacement and deviation of brain tissue resulting from breakdown of the tissue pressure equilibrium across regions of the cranial cavity. When an imbalance in tissue pressure exists between the left and right cerebral hemispheres, cingulate herniation may occur, in which one cerebral hemisphere deviates to the opposite side. Intracranial tumours can cause a tissue pressure imbalance between the forebrain compartment and the posterior cranial fossa, resulting in transtentorial herniation. When the volume of the posterior cranial fossa exceeds the available space as a result of transtentorial herniation caused by pressure imbalance, the cerebellum can herniate through the foramen magnum into the spinal canal. 5
Among the various types of cerebral herniations, foramen magnum herniation is generally recognised as having a poor prognosis because of constriction and impairment of the brainstem. Despite many reports on the results of surgical treatment for cats with IM and the considerable prevalence of concomitant cerebral herniation,1,4,6,7 no studies have described the characteristic features and prognosis in these cats. Therefore, we retrospectively investigated cases of surgically treated IM in cats. In particular, we evaluated cerebral herniation in these cats and assessed the effect of surgical treatment especially in cases with concurrent cingulate, transtentorial and foramen magnum herniations.
Materials and methods
Cases
This study included cats with a histopathological diagnosis of IM in the forebrain compartment treated by surgery at the Veterinary Medical Teaching Hospital of Nippon Veterinary and Life Science University, Japan, from September 2000 to February 2017. We retrospectively selected cats with concurrent cingulate, transtentorial and foramen magnum herniations or other herniations on preoperative MRI. 1 All included cats had undergone both preoperative and postoperative MRI and had complete medical records. One of the authors collected data from the medical records on the cats’ breed, sex, age, clinical signs, imaging findings, surgical treatment, tissue diagnosis and outcomes. For cats in which the intracranial pressure (ICP) was measured intraoperatively, the cerebral perfusion pressure (CPP) was also included in the survey. In all cats, a neurolo-gical examination, blood count, blood biochemical examination and chest radiography were performed before MRI and surgery.
Anaesthesia and analgesia
MRI of the brain was performed under general anaesthesia before surgery and immediately after surgery in all cases. Anaesthesia was induced with midazolam (0.2 mg/kg IV) and propofol (6 mg/kg IV). After endotracheal intubation, anaesthesia was maintained with isoflurane and oxygen. Fentanyl (5–10 μg/kg/min) was administered during surgery; a fentanyl patch (Duprop MT patch; Janssen Pharmaceutical) was applied after surgery. Anaesthesia management during surgery included continuous monitoring of heart rate and body temperature, non-invasive blood pressure measurement, SpO2 and end-tidal CO2 measurement with electrocardiography and monitoring of vital signs (BSM-3592 LifeScope VS; Nihon Kohden). Respiration was controlled with a ventilator to maintain end-tidal CO2 in the range of 25–35 mmHg. During anaesthesia, lactated Ringer’s solution was administered at 1–3 ml/kg/h. In four cats, the subdural pressure was measured with an ICP Express and ICP Microsensor (Codman & Shurtleff). The CPP before tumour extirpation was determined from the obtained ICP and mean arterial pressure (MAP).
MRI
Imaging was performed with a 1.5 Tesla (T) MRI machine (GE Signa HDxt; GE Healthcare Japan) and a 3-T MRI machine (GE Signa EXCITE 3.0-T system; GE Healthcare Japan). First, we obtained T2-weighted (T2W) and T1-weighted (T1W) sagittal, transverse and coronal images. Second, T1W-intensified sagittal, transverse and coronal images were obtained after intravenous administration of 0.15 mmol gadolinium (0.2 ml/kg) (Omniscan IV injection 32%; Daiichi Sankyo). The imaging conditions were: repetition time 500 ms; echo time 10 ms; slice thickness 5 mm; and slice gap 0 mm.
Tumour volume and cranial cavity volume were measured in the axial plane by one of the authors using previously reported area measurement techniques. 8 On transverse T1W images under gadolinium contrast in each case, the tumour area was measured on each slice in the axial direction. Tumour volumes were determined by multiplying the sum of the area measurements by the sum of the slice thickness and gap in each plane. The brain parenchyma, tumour and cerebrospinal fluid were also measured on each slice; the total volume of the cranial cavity was similarly calculated. The ratio of the tumour volume: intracranial volume was calculated.
The volume of residual tumour was similarly determined on postoperative MRI, and the tumour excision ratio was evaluated. We also measured the lateral ventricular volume on transverse T2W images and compared preoperative and postoperative volumes. Horos version 2.1.1 (2018 Horos Project) was used to measure the tumour volume, cranial cavity volume and lateral ventricular volume. In the diagnosis of cerebral herniation, the transtentorial line to the rostroventral aspect of the cerebellum (TTX) and the line from the caudoventral aspect of the cerebellum to the foramen magnum (FMX) were measured according to the method of Lewis et al. 9
Surgery
All procedures were performed via the frontal sinus or the outer anterior tentorium, according to the tumour location. Bone resection craniotomy was performed in all cases because of the reported risk of tumour infiltration in the portion of the skull that borders the meningioma. 10 For skull resection, a 1 mm diameter round burr (burr for bone surgery [air type]; Zimmer Biomet Holding) was used; the skull was removed according to the position of the tumour on preoperative MRI. The lesion was removed while reducing the tumour volume with a supersonic emulsification suction apparatus (SonoSurg; Olympus) (Figure 1).
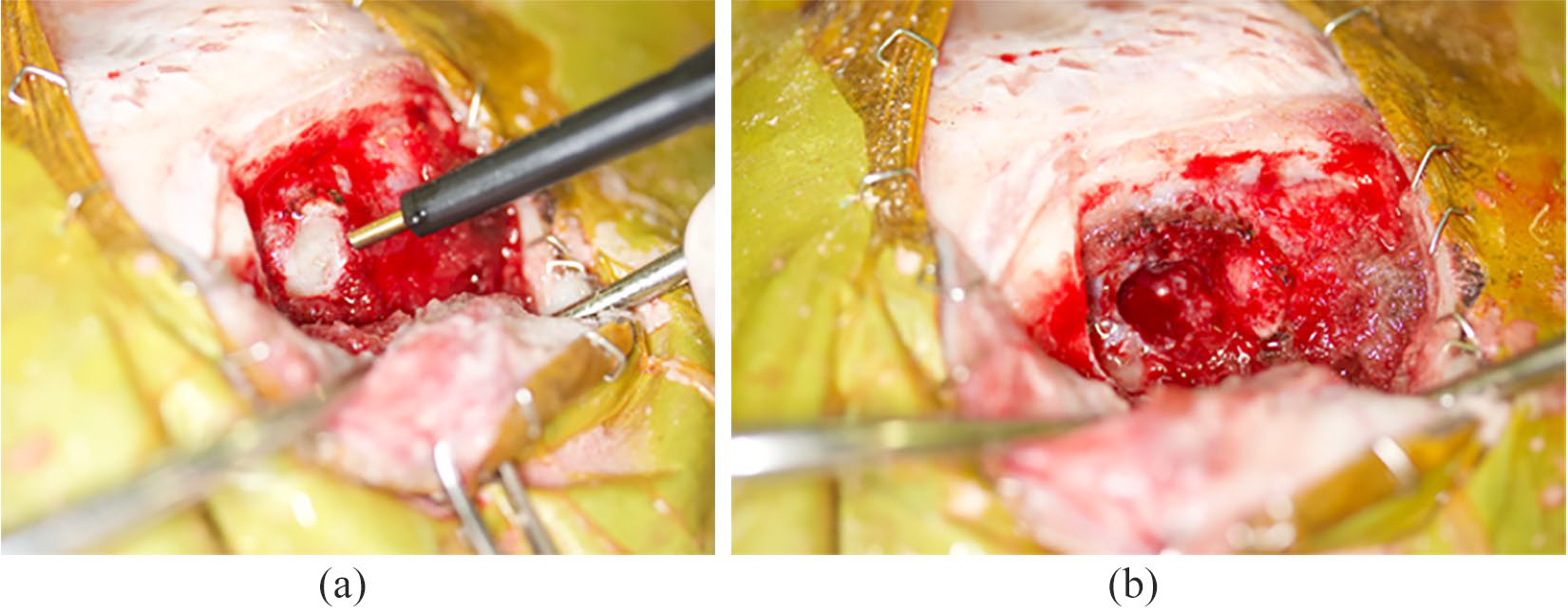
Intraoperative findings at meningiomectomy. Lesions were excised while reducing pressure within the tumour with an ultrasonic emulsification and suction device. (a) Before tumour removal; and (b) after tumour removal
For hard tumours, resection forceps (Sontec Instruments) were used to ablate the mass. A surgical microscope (Leica M525 F50; Leica Microsystems) was used for procedures in the cranial cavity. An artificial dura mater patch (Gore-Tex ePTFE patch II; WL Gore & Associates) was applied with a 5-0 polypropylene suture (PROLENE; Ethicon) in areas from which the dura was resected. At closure, the cranium was rebuilt with a titanium mesh plate (1.7 soft-type square grid; Muranaka Medical Instruments). When a titanium mesh plate was used, the plate was shaped to match the surface morphology of the cranium intraoperatively and fixed to the skull with a titanium screw (Figure 2).

Cranial reconstruction with titanium mesh plate. The plate was shaped to conform to the surface morphology of the skull and fixed to the skull with a titanium screw
To prevent cerebral oedema, 10% glycerol (0.5–1.0 g/kg q6–12h IV) was continued for 24–72 h after surgery. In addition, antibiotics were continued for 2 weeks postoperatively (enrofloxacin, 5 mg/kg q24h and chloramphenicol, 30 mg/kg q12h).
Follow-up
Medical records and telephone interviews were used to determine the date and whether the cat was still alive and, if not, the cause of death.
Statistical analysis
Statistical analysis was performed with R 3.4.4 GUI 1.70 software, and a P value of <0.05 was considered statistically significant. The normality of the data was determined with the Shapiro–Wilk normality test. Because the lateral ventricular volume did not show normality (P = 0.7834), the Wilcoxon signed rank test was performed. Pre- and postoperative tumour volumes showed normality (P <0.0001); therefore, a corresponding t-test was performed.
Results
Pre- and postoperative MRI data were available for 10/14 cats with IM. There were seven cases of combined cingulate, transtentorial and foramen magnum herniations. All of these seven cats were mixed breed; three were castrated males and four were spayed females. Median age was 13.0 years (range 9.9–16.1 years). Clinical signs of the seven cats with cerebral herniation included visual impairment (n = 5 [71.4%]), ataxia (n = 4; [57.1%]), impaired consciousness (n = 2; [28.6%]), head pressing (n = 2; [28.6%]), paresis (n = 1; [14.3%]), torticollis (n = 1; [14.3%]) and personality changes (n = 1 [14.3%]). The median interval from the onset of clinical signs to diagnosis was 35 days (range 8–146 days), and the median interval from the onset of clinical signs to surgery was 35 days (range 21–163 days) (Table 1).
Characteristics of cats with rostrotentorial meningioma with concurrent cingulate, transtentorial and foramen magnum herniations

In this study, IM occurred in three olfactory bulbs, five cerebral fornices and two falces cerebri. Of the seven cats with IM and cerebral herniation, all had cingulate, transtentorial and foramen magnum hernations simultaneously present. In cases 2 and 5, IM was confirmed at two or more sites. Median tumour volume, cranial cavity volume and tumour volume: intracranial volume ratio before surgery were 3.37 cm3 (range 3.24–11.5 cm3), 32.6 cm3 (range 29.8–78.3 cm3) and 10.4% (range 5.27–35.3%), respectively. The median tumour volume after surgery was 0.30 cm3 (range 0.00–1.40 cm3), which was significantly smaller than that before surgery (P = 0.00308). In 3/7 cats, no residual tumour was found on T1W images after gadolinium administration, indicating that complete resection was achieved (Table 2).
Relative tumour volume (tumour volume:intracranial volume) and tumour excision rate

Pre = preoperatively; post = postoperatively; NA = not available
Median overall tumour excision rate was 90.6%. In the four cats in which ICP was measured during surgery, the ICP was 15–32 mmHg (median 29 mmHg) before surgery. ICP was not subsequently monitored after tumour removal because the ICP probe was removed at the closing stage of surgery. MAP was 62–85 mmHg (median 67.5 mmHg) before tumour removal and 63–112 mmHg (median: 67.5 mmHg) after surgery. The median CPP was 47 mmHg before surgery and 70 mmHg after surgery (Table 3). In all cats, the ICP dropped to 0 mmHg immediately after tumour removal.
Heart rate and mean blood pressure in cats with intraoperative monitoring of intracranial pressure (ICP)

Vital signs suggesting Cushing phenomenon were not observed during surgery
CPP = cerebral perfusion pressure; MAP = mean arterial pressure; pre = preoperatively; post = postoperatively
Median volume of the lateral ventricle was 1.63 cm3 (range 0.154–2.38 cm3) before surgery and 1.34 cm3 (range 0.288–2.14 cm3) after surgery. This change was not significant (P = 0.3125). Three of seven cats underwent follow-up MRI 15–42 days after surgery; none of the three showed tumour recurrence, and all showed resolution of the herniations. Preoperative TTX and FMX were 4.12–10.10 mm and 0.745–8.77 mm, respectively; postoperative TTX and FMX were 0.00–5.14 mm and −0.201 to 1.86 mm, respectively. At the final MRI scan, the median volume of the lateral ventricle was 1.72 cm3 (range 2.53–3.56 cm3), which was larger than that after surgery, especially in cases 2, 4 and 7 (Table 4).
Changes in lateral ventricular volume and cerebral herniation

Increased ventricular volume was observed 2 weeks postoperatively. Foramen magnum herniation resolved after surgery
Pre = preoperatively; post = postoperatively; TTX = transtentorial line to rostroventral aspect of cerebellum; FMX = line from caudoventral aspect of cerebellum to foramen magnum; NA = not available
Histopathological examination of the excised tumour revealed five transitional meningiomas, two psammomatous (sand-grain type) meningiomas and one fibroblastic meningioma. All seven cats recovered well from anaesthesia and no death occurred within 48 h after surgery. In case 5, the state of consciousness declined on the first day after surgery. MRI revealed haematoma formation; therefore, craniotomy was performed again to eliminate the haematoma and obtain haemostasis. The cat’s state of consciousness recovered within 1 day after reoperation. In the other cats, the state of consciousness and visual recovery were confirmed within 3 days of surgery. In all seven cats, no adjuvant therapy was required after surgery. The median survival period was 612 days (range 55–1453 days), and the mean survival period was 586 days (Table 5).
Tumour localisation, pathological type and survival

CKD = chronic kidney disease
Case 2 had normal consciousness with visual impairment at diagnosis. MRI at 30 days after surgery showed resolution of the cingulate, transtentorial and foramen magnum herniations with lateral ventricle enlargement. The pathological diagnosis in that case was fibroblastic meningioma, and the survival time was 661 days (Figure 3). In case 4, somnolence was observed at the time of diagnosis, and the ICP before tumour resection was as high as 27 mmHg. Postoperative MRI revealed resolution of the cingulate, transtentorial and foramen magnum herniations with lateral ventricle enlargement. The pathological diagnosis was psammomatous meningioma, and the survival time was 612 days. In case 7, ataxia and torticollis were observed at diagnosis. Postoperative MRI revealed resolution of the cingulate, transtentorial and foramen magnum herniations with lateral ventricle enlargement. The pathological diagnosis was transitional meningioma, and the cat was still alive at the time of writing (463 days to date). The MRI findings for case 4 and 7 is available in the supplementary material.
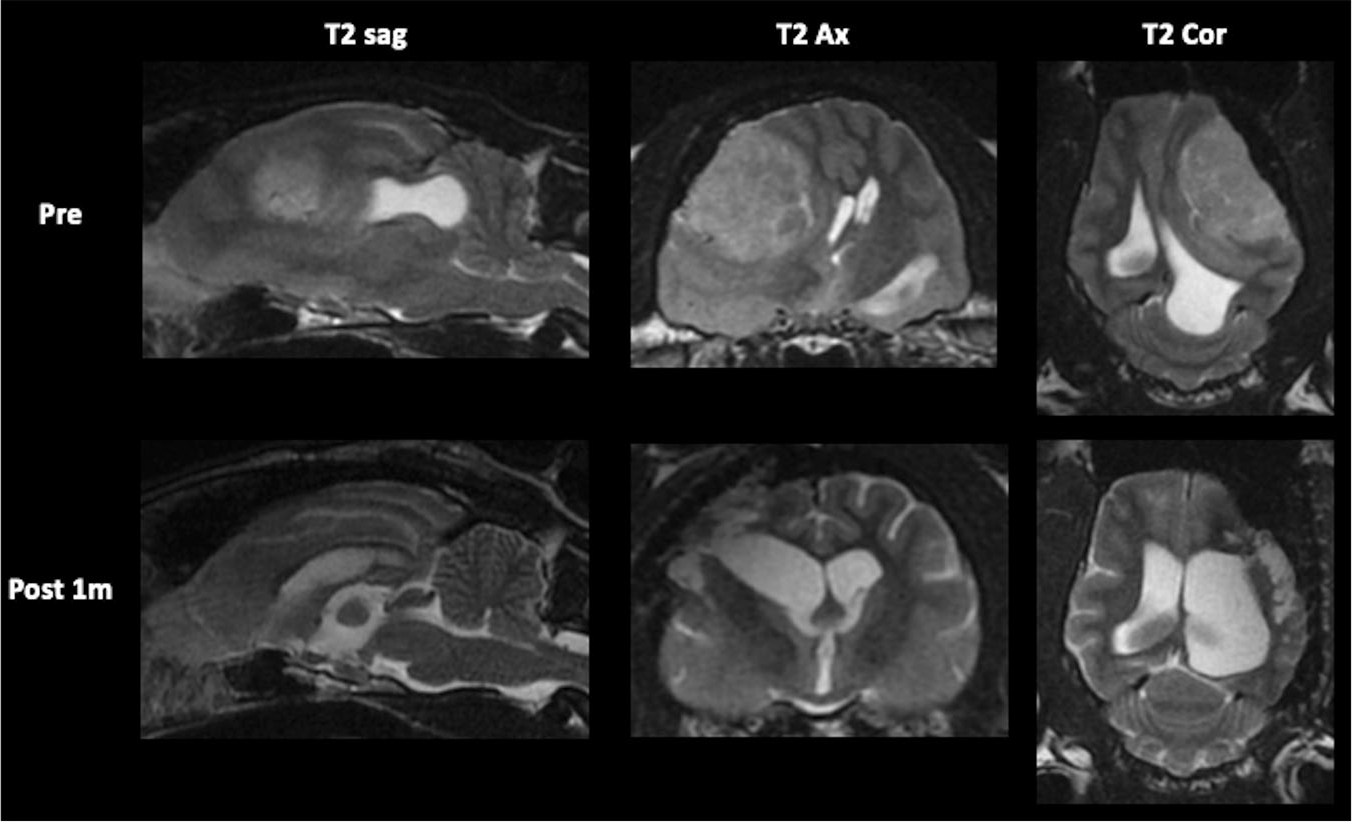
Preoperative (Pre) and postoperative (Post) MRI findings in case 2. Visual impairment was observed at the time of diagnosis, but the state of consciousness was normal. Preoperative MRI showed cingulate, transtentorial and foramen magnum herniations. On MRI 30 days postoperatively, the herniations had resolved and the lateral ventricle was larger
Discussion
In this study, we evaluated the complications, clinical features and postoperative outcomes in cats with rostrotentorial IM with concurrent cingulate, transtentorial and foramen magnum herniations. First, we found that the incidence of concurrent cingulate, transtentorial and foramen magnum herniations in cats with rostrotentorial IM was 70%, and consciousness disorders were observed in 28.6% of these cats. Second, the average ICP among the cats in which ICP was measured preoperatively was 26.3 mmHg. Third, median survival time after surgical treatment alone in cats with IM and cingulate, transtentorial and foramen magnum herniations was 612 days.
We evaluated the clinical features of rostrotentorial IM with concurrent cingulate, transtentorial and foramen magnum herniations and its relevance to postoperative outcomes in cats. The site of IM was the olfactory bulb in three cases, the cerebral fornix in five cases and the falx cerebri in two cases. In cats with rostrotentrial IM, the incidence of cingulate, transtentorial and foramen magnum herniations was 70% (n = 7). Troxel et al 2 reported that concurrent transtentorial herniation was present in 14/33 (42%) cats with IM and that foramen magnum herniation was present in 7/33 cats (21%). The reason why 70% of cats in the present study had concurrent cerebral herniations is unknown, but they may have been associated with slow tumour growth. Because IMs grow over time, foramen magnum herniation is a phenomenon that can occur at the end of the clinical course in cats with rostrotentorial IM.
Clinical signs in cats with IM include decreased consciousness, circling, seizures, gait changes, behavioural abnormalities, and changes in personality and daily habits.1,4,6 The clinical manifestations in dogs with IM include aggression, circling and torticollis, but the most common sign is seizures.10,11 Of our seven cases, we observed visual impairment (71.4%), gait abnormality (57.1%), disturbance of consciousness (somnolence; 28.6%), head pressing (28.6%), paresis (14.3%), torticollis (14.3%) and personality changes (14.3%); the most common sign was visual impairment. In all cases, non-specific signs such as weight loss were initially present, followed by gait disorder, somnolence and visual impairment. In general, a progressive increase in tumour size causes neurological abnormalities ranging from local compressive injury of the cerebral cortex to consciousness disorder or visual impairment. Past studies have confirmed that the cerebral cortex and occipital lobe responsible for vision are sensitive to the effects of hypoxia.11–14 The high prevalence of visual impairment in cats with IM in this study may have been related to compression caused by the tumour or its secondary effects (eg, cerebral ischaemia caused by intracranial hypertension).
The reported frequency of consciousness disorder in cats with IM is 0–7%.4,6 In the present study, of the 28.5% of cats with impaired consciousness (somnolence), there was no association with ICP, tumour volume:brain volume ratio or survival time. Therefore, cats with rostrotentorial IM and concurrent cingulate, transtentorial and foramen magnum herniations do not necessarily have impaired consciousness.
In the current study, the average ICP among the cats in which ICP was measured preoperatively was 26.3 mmHg, confirming the presence of intracranial hypertension, and the CPP ranged from 47 to 58 mmHg. The CPP is calculated as the difference between the MAP and ICP. In general, physiological abnormalities are created within the brain when the CPP falls below 60–70 mmHg.15,16 When the CPP is severely reduced, the Cushing reflex occurs to prevent cerebral ischaemia and the CPP is maintained. 17 Based on the mean blood pressure and heart rate before tumour resection, four cats in this study did not appear to have the Cushing reflex. These findings suggest that the minimum cerebral blood flow rate was maintained during tumour removal, which may explain why the Cushing phenomenon did not develop. As findings suggesting the Cushing phenomenon were not observed during surgery, the decrease in CPP measured during this time was likely caused by transient mild hypotension accompanying anaesthesia.
In this study of cats with IM and cingulate, transtentorial and foramen magnum herniations, the median survival time after surgery was 612 days. In past studies of cats with IM, the median survival period has ranged from 21 to 37 months.1,4,6,7 However, no studies have evaluated the results of surgical treatment with a focus on cerebral herniation. There are only two reports of cerebral herniation in cats with IM to date, and the association between cerebral herniation and clinical features has not been evaluated.1,2 It can be assumed that when foramen magnum herniation is present, the ICP is extremely elevated, and this generally indicates a poor prognosis.18,19 However, in this study, no complications or perioperative death occurred; even with foramen magnum herniation, the cerebral hernation resolved after surgery. Clinical signs improved and long-term survival was achieved with surgery alone. Therefore, as in previous reports,1,4,6,7,20 surgical resection was effective and offered the possibility of good survival prospects for feline IM, even in cats with rostrotentorial IM with concurrent cingulate, transtentorial and foramen magnum herniations.
This study has some limitations. First, the number of cases was small and the relationships among cerebral herniation, ICP, CCP, cranial cavity volume, tumour volume:intracranial volume ratio, perioperative mortality and prognosis could not be evaluated. In cats with meningiomas that develop in the forebrain compartment, progression is expected to cause cingulate, transtentorial and/or foramen magnum herniations. As we evaluated these three types of herniation together in this study, we could not evaluate the prognosis of each type of herniation individually after surgical treatment. Second, the postoperative change in the lateral ventricular volume in cats with IM is unknown. Although reconstruction of the cerebral hemispheres after ventriculoperitoneal shunting in dogs occurs only in the white matter, 16 the lateral ventricle appeared to expand 1 week postoperatively in the present study; however, the clinical significance of this is unclear because of the low number of samples. Future studies with higher numbers of cases are necessary to clarify this phenomenon. Third, why the median survival time in the present study was slightly shorter than that in previous studies is unknown because postmortem histological examination of the brain was not performed. In cats with IM and cerebral herniation, surgical treatment resolves the herniation and improves clinical signs, but these cats may have shorter survival times than those without cerebral herniation.
Conclusions
Even in cats with IM causing cingulate, transtentorial and foramen magnum herniations, surgical treatment resolved the herniations and allowed prolonged survival. Early surgical resection may prolong life expectancy beyond the shorter survival times in cats with large concomitant herniations in this study.
Supplemental Material
Supplementary Material
Pre- and postoperative MRI findings in cases 4 and 7
Footnotes
Acknowledgements
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (including owned animals and data from retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Written informed consent was obtained from the owners of all animals described in this work for the procedures undertaken. For any animals individually identifiable within this publication, written informed consent for their use in the publication was obtained from the people involved.
Supplementary material
The following file is available online:
Pre- and postoperative MRI findings in cases 4 and 7.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
